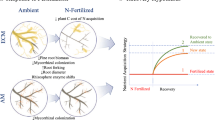
Abstract
Enhanced nitrogen (N) availability in temperate forests has altered ecosystem carbon (C) and N cycling. Recent research has shown that these alterations lead to reductions in belowground C allocation by trees and that the consequences of these reductions on soil C and nutrient cycling may vary by mycorrhizal type. We hypothesized that trees that associate with ectomycorrhizal fungi (ECM) would reduce C allocation towards roots and mycorrhizal fungi to a greater extent than trees that associate with arbuscular mycorrhizal fungi (AM) in response to > 25 years of N fertilization. We further hypothesized that N induced decoupling of roots and microbes in ECM trees would be evidenced by greater declines in extracellular enzyme activities. We measured belowground C allocation to fine root biomass and mycorrhizal colonization in 6 AM and 6 ECM dominated plots in the N fertilized and reference watersheds at the Fernow Experimental Forest in West Virginia, USA. We compared these to measurements of simple-C, complex-C, nitrogen, and phosphorus acquiring enzyme activities in organic horizon, bulk mineral, and rhizosphere soil fractions. N fertilization reduced fine root biomass and mycorrhizal colonization in both AM and ECM stands. We found more consistent reductions in enzyme activities in ECM soils than AM soils under N fertilization which may have been driven by greater declines in root-C transfer to soil microbes. This mechanism helps to explain variability in soil C cycling responses across N gradient and fertilization experiments and may prove useful in predicting the fate of soil C stocks in response to N deposition.





Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Data availability
The data supporting these findings is available in the Environmental Data Initiative database at https://doi.org/10.6073/pasta/51e2c55d42a3c68b1a8c7efab2050c69.
References
Adams M, Angradi T (1996) Decomposition and nutrient dynamics of hardwood leaf litter in the fernow whole-watershed acidification experiment. For Ecol Manage 83(1–2):61–69
Adams M, Edwards P, Wood F, Kochenderfer J (1993) Artificial watershed acidification on the Fernow Experimental Forest, USA. J Hydrol 150(2–4):505–519
Argiroff WA, Zak DR, Pellitier PT, Upchurch RA, Belke JP (2022) Decay by ectomycorrhizal fungi couples soil organic matter to nitrogen availability. Ecol Lett 25(2):391–404
Averill C, Dietze MC, Bhatnagar JM (2018) Continental-scale nitrogen pollution is shifting forest mycorrhizal associations and soil carbon stocks. Glob Change Biol 24(10):4544–4553
Bedison JE, McNeil BE (2009) Is the growth of temperate forest trees enhanced along an ambient nitrogen deposition gradient? Ecology 90(7):1736–1742
Brzostek ER, Greco A, Drake JE, Finzi AC (2013) Root carbon inputs to the rhizosphere stimulate extracellular enzyme activity and increase nitrogen availability in temperate forest soils. Biogeochemistry 115(1):65–76
Brzostek ER, Dragoni D, Brown ZA, Phillips RP (2015) Mycorrhizal type determines the magnitude and direction of root-induced changes in decomposition in a temperate forest. New Phytol 206(4):1274–1282
Bzdyk RM, Olchowik J, Studnicki M, Nowakowska JA, Oszako T, Urban A, Hilszczańska D (2019) Ectomycorrhizal colonisation in declining oak stands on the Krotoszyn Plateau. Pol for 10(1):30
Carrara JE, Walter CA, Hawkins JS, Peterjohn WT, Averill C, Brzostek ER (2018) Interactions among plants, bacteria, and fungi reduce extracellular enzyme activities under long-term N fertilization. Glob Change Biol 24(6):2721–2734
Carrara JE, Walter CA, Freedman ZB, Hostetler AN, Hawkins JS, Fernandez IJ, Brzostek ER (2021) Differences in microbial community response to nitrogen fertilization result in unique enzyme shifts between arbuscular and ectomycorrhizal-dominated soils. Glob Change Biol 27(10):2049–2060
Carrara JE, Fernandez IJ, Brzostek ER (2022) Mycorrhizal type determines root–microbial responses to nitrogen fertilization and recovery. Biogeochemistry 157(2):245–258
Comas LH, Callahan HS, Midford PE (2014) Patterns in root traits of woody species hosting arbuscular and ectomycorrhizas: implications for the evolution of belowground strategies. Ecol Evol 4(15):2979–2990
Eastman BA, Adams MB, Brzostek ER, Burnham MB, Carrara JE, Kelly C, McNeil BE, Walter CA, Peterjohn WT (2021) Altered plant carbon partitioning enhanced forest ecosystem carbon storage after 25 years of nitrogen additions. New Phytol 230(4):1435–1448
Edwards PJ, Wood F (2011) Fernow experimental forest daily precipitation. U.S. Department of Agriculture, Forest Service, Northern Research Station, Newtown Square, PA. Updated 27 August 2021. https://doi.org/10.2737/RDS-2011-0014
Fog K (1988) The effect of added nitrogen on the rate of decomposition of organic matter. Biol Rev 63(3):433–462
Frey SD, Ollinger S, Nadelhoffer KE, Bowden R, Brzostek E, Burton A, Caldwell B, Crow S, Goodale CL, Grandy AS (2014) Chronic nitrogen additions suppress decomposition and sequester soil carbon in temperate forests. Biogeochemistry 121(2):305–316
Gilliam FS, Turrill NL, Aulick SD, Evans DK, Adams MB (1994) Herbaceous layer and soil response to experimental acidification in a central Appalachian hardwood forest. J of Env Qual. https://doi.org/10.2134/jeq1994.00472425002300040032x
Gilliam FS, Welch NT, Phillips AH, Billmyer JH, Peterjohn WT, Fowler ZK, Walter CA, Burnham MB, May JD, Adams MB (2016) Twenty-five-year response of the herbaceous layer of a temperate hardwood forest to elevated nitrogen deposition. Ecosphere 7(4):e01250
Giovannetti M, Mosse B (1980) An evaluation of techniques for measuring vesicular arbuscular mycorrhizal infection in roots. New Phytol 84:489–500
Hicks Pries CE, Lankau R, Ingham GA, Legge E, Krol O, Forrester J, Fitch A, Wurzburger N (2023) Differences in soil organic matter between EcM-and AM-dominated forests depend on tree and fungal identity. Ecology 104(3):e3929
Hurlbert SH (1984) Pseudoreplication and the design of ecological field experiments. Ecol Monogr 54(2):187–211
Janssens I, Dieleman W, Luyssaert S, Subke J-A, Reichstein M, Ceulemans R, Ciais P, Dolman AJ, Grace J, Matteucci G (2010) Reduction of forest soil respiration in response to nitrogen deposition. Nat Geosci 3(5):315–322
Kaiser K, Wemheuer B, Korolkow V, Wemheuer F, Nacke H, Schöning I, Schrumpf M, Daniel R (2016) Driving forces of soil bacterial community structure, diversity, and function in temperate grasslands and forests. Sci Rep 6(1):1–12
Kochenderfer JN (2006) Fernow and the Appalachian hardwood region. In: Adams MB, DeWalle DR, Hom JL (eds) The Fernow watershed acidification study. Springer, Dordrecht, pp 17–39
Lauber CL, Hamady M, Knight R, Fierer N (2009) Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl Environ Microbiol 75(15):5111–5120
Lin G, McCormack ML, Ma C, Guo D (2017) Similar below-ground carbon cycling dynamics but contrasting modes of nitrogen cycling between arbuscular mycorrhizal and ectomycorrhizal forests. New Phytol 213(3):1440–1451
Lindahl BD, Kyaschenko J, Varenius K, Clemmensen KE, Dahlberg A, Karltun E, Stendahl J (2021) A group of ectomycorrhizal fungi restricts organic matter accumulation in boreal forest. Ecol Lett 24(7):1341–1351
Lovett GM, Arthur MA, Weathers KC, Fitzhugh RD, Templer PH (2013) Nitrogen addition increases carbon storage in soils, but not in trees, in an eastern US deciduous forest. Ecosystems 16:980–1001
Midgley MG, Phillips RP (2014) Mycorrhizal associations of dominant trees influence nitrate leaching responses to N deposition. Biogeochemistry 117(2):241–253
Midgley MG, Phillips RP (2016) Resource stoichiometry and the biogeochemical consequences of nitrogen deposition in a mixed deciduous forest. Ecology 97(12):3369–3378
Midgley MG, Brzostek E, Phillips RP (2015) Decay rates of leaf litters from arbuscular mycorrhizal trees are more sensitive to soil effects than litters from ectomycorrhizal trees. J Ecol 103(6):1454–1463
Moore JA, Anthony MA, Pec GJ, Trocha LK, Trzebny A, Geyer KM, van Diepen LT, Frey SD (2021) Fungal community structure and function shifts with atmospheric nitrogen deposition. Glob Change Biol 27(7):1349–1364
Morrison EW, Frey SD, Sadowsky JJ, van Diepen LT, Thomas WK, Pringle A (2016) Chronic nitrogen additions fundamentally restructure the soil fungal community in a temperate forest. Fungal Ecol 23:48–57
Morrison EW, Pringle A, van Diepen LT, Frey SD (2018) Simulated nitrogen deposition favors stress-tolerant fungi with low potential for decomposition. Soil Biol Biochem 125:75–85
Oksanen J, Blanchet F, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin P, O’Hara R, Simpson G, Solymos P et al (2022) Vegan: Community Ecology Package, 2.6–2. 2022.
Pellitier PT, Zak DR (2018) Ectomycorrhizal fungi and the enzymatic liberation of nitrogen from soil organic matter: why evolutionary history matters. New Phytol 217(1):68–73
Pellitier PT, Zak DR (2021) Ectomycorrhizal fungal decay traits along a soil nitrogen gradient. New Phytol 232(5):2152–2164
Phillips RP, Fahey TJ (2005) Patterns of rhizosphere carbon flux in sugar maple (Acer saccharum) and yellow birch (Betula allegheniensis) saplings. Glob Change Biol 11(6):983–995
Phillips RP, Fahey TJ (2006) Tree species and mycorrhizal associations influence the magnitude of rhizosphere effects. Ecology 87(5):1302–1313
Phillips RP, Brzostek E, Midgley MG (2013) The mycorrhizal-associated nutrient economy: a new framework for predicting carbon–nutrient couplings in temperate forests. New Phytol 199(1):41–51
Pregitzer KS, Burton AJ, Zak DR, Talhelm AF (2008) Simulated chronic nitrogen deposition increases carbon storage in Northern Temperate forests. Glob Change Biol 14(1):142–153
Pritsch K, Garbaye J (2011) Enzyme secretion by ECM fungi and exploitation of mineral nutrients from soil organic matter. Ann for Sci 68(1):25–32
Quinn TR, Canham CD, Weathers KC, Goodale CL (2010) Increased tree carbon storage in response to nitrogen deposition in the US. Nat Geosci 3(1):13–17
Raczka NC, Carrara JE, Brzostek ER (2022) Plant–microbial responses to reduced precipitation depend on tree species in a temperate forest. Glob Change Biol. https://doi.org/10.1111/gcb.16340
Read D, Perez-Moreno J (2003) Mycorrhizas and nutrient cycling in ecosystems–a journey towards relevance? New Phytol 157(3):475–492
Rousk J, Bååth E, Brookes PC, Lauber CL, Lozupone C, Caporaso JG, Knight R, Fierer N (2010) Soil bacterial and fungal communities across a pH gradient in an arable soil. ISME J 4(10):1340–1351
Saiya-Cork K, Sinsabaugh R, Zak D (2002) The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol Biochem 34(9):1309–1315
Sinsabaugh RL (2010) Phenol oxidase, peroxidase and organic matter dynamics of soil. Soil Biol Biochem 42(3):391–404
Terrer C, Vicca S, Hungate BA, Phillips RP, Prentice IC (2016) Mycorrhizal association as a primary control of the CO2 fertilization effect. Science 353(6294):72–74
Yin H, Wheeler E, Phillips RP (2014) Root-induced changes in nutrient cycling in forests depend on exudation rates. Soil Biol Biochem 78:213–221
Zak DR, Holmes WE, Burton AJ, Pregitzer KS, Talhelm AF (2008) Simulated atmospheric NO3− deposition increases soil organic matter by slowing decomposition. Ecol Appl 18(8):2016–2027
Zak DR, Argiroff WA, Freedman ZB, Upchurch RA, Entwistle EM, Romanowicz KJ (2019) Anthropogenic N deposition, fungal gene expression, and an increasing soil carbon sink in the Northern Hemisphere. Ecology 100(10):e02804
Acknowledgements
We acknowledge Mary Beth Adams, Tom Schuler, and the US Forest Service staff at the Fernow Experimental Forest for logistical assistance and access to the experimental watersheds. We thank Christopher Walter, Mark Burnham, and Justin Mathias for technical assistance. We thank the undergraduate researchers who helped collect the data presented in this study. This work was supported by the National Science Foundation Graduate Research Fellowship to Joseph Carrara under Grant No. DGE-1102689.
Funding
This work was supported by the National Science Foundation Graduate Research Fellowship to Joseph Carrara under Grant No. DGE-1102689.
Author information
Authors and Affiliations
Contributions
JEC and ERB designed the study. Material preparation, data collection and analysis were performed by all authors. The first draft of the manuscript was written by JEC and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor :Caitlin Hicks Pries.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Carrara, J.E., Raczka, N.C. & Brzostek, E.R. Long-term nitrogen fertilization impacts plant-microbial interactions differently in arbuscular and ectomycorrhizal trees. Biogeochemistry 166, 109–122 (2023). https://doi.org/10.1007/s10533-023-01099-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-023-01099-8