Abstract
Wetlands have an inordinate influence on the global greenhouse gas budget, but how global changes may alter wetland contribution to future greenhouse gas fluxes is poorly understood. We determined the greenhouse gas balance of a tidal marsh exposed to nine years of experimental carbon dioxide (CO2) and nitrogen (N) manipulation. We estimated net carbon (C) gain rates by measuring changes in plant and soil C pools over nine years. In wetland soils that accrete primarily through organic matter inputs, long-term measurements of soil elevation, along with soil C density, provide a robust estimate of net soil C gain. We used net soil C gain along with methane and nitrous oxide fluxes to determine the radiative forcing of the marsh under elevated CO2 and N addition. Nearly all plots exhibited a net gain of C over the study period (up to 203 g C m−2 year−1), and C gain rates were greater with N and CO2 addition. Treatment effects on C gain and methane emissions dominated trends in radiative forcing while nitrous oxide fluxes in all treatments were negligible. Though these soils experience salinities that typically suppress methane emissions, our results suggest that elevated CO2 can stimulate methane emissions, overcoming positive effects of elevated CO2 on C gain, converting brackish marshes that are typically net greenhouse gas sinks into sources. Adding resources, either CO2 or N, will likely increase “blue carbon” accumulation rates in tidal marshes, but importantly, each resource can have distinct influences on the direction of total greenhouse forcing.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Wetlands exert inordinate leverage on greenhouse gas budgets owing to the capacity to sequester large amounts of carbon (C) per area (McLeod et al. 2011). Yet, these ecosystems are frequently converted for development and agriculture, limiting the potential of coastal ecosystems to mitigate future climate change. Wetland conversion releases “blue C” previously stored by these ecosystems over long timescales and costs the global economy $6–42 billion annually (Pendleton et al. 2012). Tidal marshes are generally net C sinks, as the accumulation of soil volume prevents them from being converted to open water as sea level rises. Across 154 sites that span a latitudinal range of 22.4°S–55.5°N, tidal salt marshes gained 210 g C m−2 year−1 on average (Chmura et al. 2003), an amount similar to that of brackish marshes (Smith et al. 1983). However, global changes like elevated carbon dioxide (CO2) and nitrogen (N) pollution can influence the uptake and release of C from ecosystems and may alter C storage in marshes.
The effects of elevated CO2 and N pollution on net CO2 balance are poorly understood in tidal marshes. Elevated CO2 can enhance short-term rates of soil accumulation (Langley et al. 2009a) but can also stimulate soil organic C decomposition (Wolf et al. 2007), so the long-term balance remains uncertain. Similarly, some studies suggest N can stimulate soil elevation gain (Morris et al. 2014) and others show that N addition may interrupt the mechanisms that drive elevation gain (Deegan et al. 2012). Even if N addition strengthens C sequestration, the greater CO2 sink may be offset owing to the stimulation of methane (CH4) and nitrous oxide (N2O) emissions (Liu and Greaver 2009). Beyond C gain, accounting for the radiative forcing of wetlands requires quantifying feedbacks through emissions of these other important greenhouse gases. These gases are regulated by biogeochemical processes sensitive to global changes, such as N pollution and feedbacks propagated by rising greenhouse gas concentrations.
Wetlands contribute 55% to annual global CH4 emissions (Le Mer and Roger 2001), yet few studies have assessed the effects of elevated CO2 and N addition on CH4 flux in these ecosystems. Average emissions of CH4 increased in response to elevated CO2 in a brackish marsh dominated by the C3 sedge Schoenoplectus americanus, although the effect was sporadic, likely due to temporal changes in sulfate reduction, which can inhibit methanogenesis (Marsh et al. 2005). In an earlier study, CH4 emissions were 80% higher under elevated CO2 than control plots at this same site and were associated with higher peak plant biomass (Dacey et al. 1994). Stimulation of CH4 emissions under elevated CO2 has also been documented in a tidal freshwater swamp mesocosm and was associated with a simultaneous decrease in transpiration, which may have promoted methanogenesis through increased soil water content (Megonigal and Schlesinger 1997). N addition can increase CH4 emissions by inhibiting CH4 consumption and enhancing CH4 production. Though N addition has been shown to either stimulate or inhibit CH4 oxidation (Bodelier and Laanbroek 2004), most evidence suggests N fertilizer inhibits CH4 oxidation (Alam and Jia 2012; Conrad and Rothfuss 1991; Le Mer and Roger 2001; Smith et al. 2000). Ammonium (NH4 +) fertilizers can inhibit CH4 oxidation through enzymatic substrate competition (Gulledge and Schimel 1998). N addition stimulated CH4 emissions in the tidal Minjiang River estuary of southeast China by 124% in a marsh dominated by Cyperus malaccensis and 64% in a marsh dominated by Spartina alterniflora (Mou et al. 2014). Similarly, N addition stimulated CH4 emissions in three southern California salt marshes (Irvine et al. 2012) and a brackish marsh mesocosm experiment in east China, possibly by enhancing biomass, stem density, and transport of photosynthate belowground (Zhang et al. 2010). N availability could yield important interactions with elevated CO2 in regards to affecting CH4 emissions, but N effects have not been directly compared to CO2 effects in the same marsh ecosystem.
Marshes and other wetlands emit 30–650 kg N ha−1 year−1 as volatile compounds, several-fold greater than emission rates from any other ecosystem per area (Bowden 1986). A portion (up to 10 kg N ha−1 year−1) of these emissions is in the form of N2O (Bowden 1986), which offsets 263–270 times an equivalent mass of sequestered CO2 over a 100-year horizon (Neubauer and Megonigal 2015). Studies on the effect of elevated CO2 on N2O flux have largely been conducted in upland, grassland, and agricultural ecosystems, and report conflicting trends (Barnard et al. 2005; Brown et al. 2012; Niboyet et al. 2011). To our knowledge, no studies have examined this effect in marsh and other unmanaged wetland ecosystems despite their favorable environmental conditions for N2O production. N addition commonly increases N2O emissions in marshes and other ecosystems (Barnard et al. 2005; Hamersley and Howes 2005; Kinney and Valiela 2013; Reinhardt et al. 2006; White and Reddy 1999), particularly if added as nitrate (NO -3 ). In summary, how global change drivers, such as chronic N pollution and rising atmospheric CO2, affect greenhouse gas fluxes in tidal marsh ecosystems remains unclear.
Although atmospheric concentration of CO2 has increased by 40%, N2O by 20%, and CH4 by 150% since pre-industrial times (Hartmann et al. 2013), emissions of these radiatively important gases are poorly constrained in wetlands, and many studies focus solely on one of these gases. Understanding how chronic global change drivers like N pollution and elevated CO2 affect blue C and greenhouse gas emissions from tidal wetlands is essential for predicting the radiative forcing of wetlands in the future and advancing the valuation of coastal ecosystems. We sought to determine how N pollution and elevated CO2 affect C sequestration and greenhouse gas fluxes in a tidal brackish marsh. We used a combination of in situ greenhouse gas flux measurements and rates of C sequestration to calculate annual radiative forcing under factorial CO2 and N treatments that have been applied for nine years. To estimate the average rate of net belowground ecosystem C gain, we measured soil elevation change over eight years, along with detailed profiles of soil C density. To assess net CH4 and N2O fluxes, we performed in situ flux measurements using static chambers. We calculated the effects of CO2 and N treatments on radiative forcing by converting rates of soil C sequestration, CH4 emissions, and N2O emissions in each treatment combination into CO2-equivalent (CO2eq) units, which we then summed to give the total CO2eq flux of the marsh under each scenario. This calculation allowed us to determine whether elevated CO2 and N caused a net decrease in CO2eq flux (i.e. negative radiative forcing) or a net increase in CO2eq flux (i.e. positive radiative forcing).
We tested two main hypotheses: (1) N addition causes positive radiative forcing (i.e. increases net emissions of greenhouse gases in CO2eq units) by reducing belowground C sequestration and enhancing greenhouse gas emissions and (2) Elevated CO2 causes positive radiative forcing by enhancing greenhouse gas emissions enough to override enhanced belowground C sequestration. The knowledge that N addition tended to diminish soil elevation gain early on in this experiment (Langley and Megonigal 2010) combined with past studies supporting N enhancement of CH4 and N2O emissions led us to hypothesize that N addition would cause positive radiative forcing. Elevated CO2 has increased belowground root productivity and stimulated marsh elevation gain at this site (Langley et al. 2009a). However, elevated CO2 was shown to stimulate CH4 emissions in a companion experiment to this site (Dacey et al. 1994; Marsh et al. 2005) and has been shown to enhance N2O emissions, as described previously. We expected elevated CO2 to enhance N2O and CH4 emissions to a magnitude that would outweigh an increase in C gain, thus causing positive radiative forcing.
Materials and methods
Site description
The study took place at the Global Change Research Wetland (GCREW), a relatively unpolluted brackish marsh located along the Rhode River (38°52′26″N, 76°32′58″W) at the Smithsonian Environmental Research Center in Edgewater, MD. The C3 sedge, Schoenoplectus americanus, along with two C4 grass species, Spartina patens and Distichlis spicata, compose >99% of plant biomass in the study plots. Because of relative functional similarity, S. patens and D. spicata are treated as a single functional group for this study, referred to as “grasses”. The soil is >85% organic to a depth of 5 m. Mean tidal range is 40 cm and the high marsh zone is 40–60 cm above mean low water level. Salinity ranges from 4 to 20. Mean low temperature is −4 °C in January and mean high temperature is 31 °C in July. Soil temperature at 10 cm depth and water level were logged at the GCREW site during the study period.
Twenty open-top chambers were constructed over octagonal 3.3-m2 plots in the summer of 2005 (Langley et al. 2009b). These plots were factorially exposed to two levels of atmospheric CO2 (ambient or ambient + 340 ppm) and two levels of N addition (0 or 25 g N m−2 year−1) beginning in May of 2006 (n = 5). The additional CO2 delivered into +CO2 and N + CO2 plots is depleted in δ13C. The mixture of added and ambient CO2 results in atmospheric δ13C–CO2 of roughly −28‰ in elevated CO2 compared to −8‰ in ambient chambers. Treatment abbreviations used are as follows: control = ambient N and ambient CO2; +N = N fertilization and ambient CO2; +CO 2 = ambient N and elevated CO2; N + CO 2 = N fertilization and elevated CO2. The concentration of CO2 added simulates moderate projections of atmospheric [CO2] for the year 2080 (Collins et al. 2013) and N fertilization simulates soil N availability in sites more heavily polluted by N in runoff. Atmospheric deposition alone is unlikely to lead to the rates of NH4 + input used in this study. For comparison, average N loads to the Chesapeake Bay are 14 g N m−2 year−1 although much higher levels up to 100 g m−2 year−1 occur in more polluted tributaries (Kemp et al. 2005). NH4Cl was applied to the N-fertilized chambers at five times approximately monthly from May to September, in aliquots of 5 g N m−2 as NH4Cl dissolved in water taken from the Rhode River, the source of water that floods the marsh during natural tidal cycles. Fertilizer solution (5 L) was applied by backpack sprayer followed by a 5 L rinse with unamended river water. Unfertilized plots received 10 L of river water without added N, applied the same way at the same times. Pure CO2 was mixed with ambient air and delivered through manifolds into elevated CO2 chambers during the growing season to achieve target concentration (ambient + 340 ppm) during daylight hours (see Langley et al. 2009b for further technical details).
Net ecosystem C gain
We estimated net C gain using a mass balance approach that incorporated soil [C], soil bulk density, and net soil elevation change (for net belowground C gain), and short-term (annual) aboveground C gain using annual aboveground biomass, over the course of the experiment, from 2006 to 2013. Annual aboveground C mass was determined as the product of aboveground biomass and tissue [C]. Biomass was determined for each species using either allometry or biomass clippings (Langley and Megonigal 2010). Subsamples of clipped biomass from each plot each year were analyzed for C composition using a combustion elemental analyzer coupled to a mass spectrometer (Smithsonian Stable Isotope Laboratory, Suitland, MD). The average rate of change in aboveground C mass was estimated by linear trends in annual C mass in each plot.
Surface elevation tables (SETs) were established in each plot in 2005 to allow an acclimation period before treatments were initiated in 2006. A detailed description of SET design is given elsewhere (Langley et al. 2009b). Surface elevation has been measured at least once in spring, summer, and fall each year since elevation tables were installed (Langley et al. 2009a). Surface elevation provides a robust measure of soil mass gain in the highly organic soils of this marsh, representing the difference between C inputs and losses. To avoid reliance on individual time points, which are subject to unexplained short-term variation, we estimated 9-year linear trends in elevation change as the slope of total elevation change by time. Elevation change reflects the soil profile from surface to the bottom of a 7 m-deep survey rod; however, we assume that most of the change occurred between the soil surface and contact of the peat horizon with a mineral horizon at 4.5 m depth.
Soil C density and δ13C
Soil cores were taken in 2014 to a depth of 60 cm from each plot using a gouge auger (6.1 cm diameter, AMS, Inc., American Falls, ID). The auger was 1 m long and had an open-face, semi-cylindrical chamber designed to remove an intact soil cylinder while preserving bulk density. Each core was sliced vertically along the cutting edges of the barrel and sectioned into 2 cm depth intervals from 0 to 10 cm, 5 cm intervals from 10 to 30 cm, and 10 cm intervals from 30 to 60 cm. Samples were dried at 60 °C for two weeks. Bulk density was estimated, and subsamples were analyzed for C composition in the same manner as described above (UC Davis Stable Isotope Facility, Davis, CA). Total belowground C to 60 cm was calculated as the product of soil [C] and soil bulk density. Belowground C gain was estimated as the product of soil elevation change over 9 years and soil C density of the top 10 cm of soil. This approach is supported by δ13C evidence showing that new C inputs over the duration of this study reside predominantly in the upper 10 cm.
In-situ N2O and CH4 Flux
To estimate in situ N2O and CH4 fluxes from soil to the atmosphere, air-tight chambers (1 L) were attached to two gas flux collars (10 cm diameter PVC pipe segments, implanted to a depth of 30 cm since 2006) within each plot. Plant stems were folded to fit inside the chambers when necessary. Overlying water partially filled each chamber, leaving ~0.3 to 0.7 L headspace, which was accounted for in gas flux calculations. In July 2014, gas fluxes were measured in four plots balanced across treatments (control, +N, +CO2, and N + CO2) each day for four days for a total of 16 plots. All 20 plots were sampled over three days in October 2014 and July 2015, and over two days in April 2015. Gas samples were collected in syringes (20 mL) from each chamber after pre-injection with ambient air (4–6 samples per time series) over the course of ~2 h. Headspace was mixed 10 times before samples were taken. Samples were taken from an elevated walkway to avoid changes in pressure that are known to cause ebullition. Gas samples were immediately transported back to the lab and transferred into 12-mL vials with screw-caps and septa (Exetainer Brand, Labco Ltd.) that were previously flushed with N2 and evacuated with a vacuum pump (Alcatel 2004a). Samples were analyzed for N2O and CH4 concentrations on a gas chromatograph and autosampler (Varian 450 GC with a CTC Analytics CombiPAL autosampler with an ECD detector for N2O detection, FID detector for CH4 detection, and TCD detector for CO2 detection). Individual chamber height above ground, corresponding water level, and the number of stems within respiration collars were measured for each plot at the time of gas sampling. Water, soil, and air temperature were also recorded.
Calculation of carbon dioxide-equivalent fluxes
The net effects of our treatments on rates of soil C sequestration, CH4 emissions, and N2O emissions on radiative forcing (i.e. climate forcing) were calculated using traditional global warming potentials (GWP) and the alternative sustained global warming potential (SGWP). The GWP is the most commonly used metric for comparing the radiative impacts of different gases that vary in atmospheric lifetimes, radiative characteristics, and cycling. The GWP is modeled as a single pulse emission of the gas of interest, which is then tracked over one hundred years or any other arbitrary period of time. The GWP represents the cumulative radiative effect over the time period of the gas pulse compared to a pulse emission of an equal mass of CO2 (Sect. 8.7.1.2 of Myhre et al. 2013). The SGWP is calculated precisely the same way except the gas is released as a sustained emission rather than a pulse (Neubauer and Megonigal 2015), which yields different conversion factors. We present GWP because this is the traditional approach, and SGWP because we expect the treatment effects to be sustained over time.
Annual GWP-carbon dioxide-equivalent (GWP-CO2eq) fluxes were calculated for each gas using a GWP of 1 for CO2, 32 for CH4, and 263 for N2O on a one hundred year horizon (see Online Resource 2, Neubauer and Megonigal 2015). Annual SGWP-carbon dioxide-equivalent (SGWP-CO2eq) fluxes were calculated for each gas using a SGWP of 1 for CO2, 45 for CH4, and 270 for N2O on a one hundred year horizon (Neubauer and Megonigal 2015). For comparison, C gain, calculated as the average annual change in soil C stock over 9 years of experimental treatment, needed to be converted into CO2 flux to be incorporated into GWP and SGWP calculations. To do so, C gain was multiplied by 3.667 (molar mass of CO2/molar mass of C) to convert to CO2 flux, and -1 to express net CO2 sequestration as a sink relative to the atmosphere. SGWP-CO2eq flux was calculated for each greenhouse gas using the equation:
where SGWP-CO2eq flux is carbon dioxide equivalent flux (in units of g m−2 year−1), GHG flux is the net flux of the greenhouse gas being converted into SGWP-CO2eq flux (positive values for CH4 and N2O sources and negative values for CO2 sinks), and SGWPGHG is the sustained global warming potential of the greenhouse gas being converted into SGWP-CO2eq flux.
To scale to annual SGWP-CO2eq fluxes, we averaged July fluxes in 2014 and 2015 and assumed that July flux data are representative of the four-month growing season, October flux data are representative of two months each fall, and April flux data are representative of two months each spring (see Online Resource 3). During the remaining four months of the year, soils at Kirkpatrick Marsh are typically frozen and emissions are likely negligible (Megonigal and Schlesinger 2002). Our extrapolation may miss a potential pulse in emissions directly upon ice melting but otherwise approximates annual SGWP-CO2eq flux. While gas fluxes vary over time, we expect that scaling our rates across months with similar climatic conditions leads to reasonable estimates of SGWP-CO2eq fluxes because soil temperature, water table, and salinity are the primary drivers of CH4 emissions in coastal wetlands (Olsson et al. 2015) and conditions during sampling were near the respective extrapolation period average (Online Resource 3). We expected CH4 emissions to hold more importance for total SGWP-CO2eq flux than N2O flux (due to low rates of N2O emissions during preliminary trials, results not shown).
We acknowledge that the timeframe of our greenhouse gas measurements does not overlap with that of our C gain measurements. We could restrict the C flux analysis to the interval for which we have CH4 and N2O fluxes, but that would not be nearly as robust as the full time series, as short-term fluctuation in elevation could mask long-term trends.
Statistical analyses
Treatment effects were tested using two-way factorial ANOVAs for C gain, gas fluxes, and SGWP-CO2eq fluxes with CO2 and N as main factors. Three-way factorial ANOVAs were used for C density and δ13C, with CO2, N, and depth as main factors. Normality was assessed using the Shapiro–Wilk test and homogeneity of variances was assessed using Bartlett’s test. Data were transformed with the natural log as needed. We followed the two-way factorial ANOVA on belowground C gain with a T Test in which control plots were compared to a pooled group of all treated plots. Two-way repeated measures MANOVAs were used for aboveground C mass through time. Tukey’s HSD post hoc test was performed to determine pairwise differences. Significance levels were set at p < 0.05 for plant responses and p < 0.10 for soil responses following the convention of Marsh et al. (2005) because soil responses are one step removed from the direct effects of elevated CO2, which introduces additional variation. Statistical analyses were performed using Rstudio version 0.98.490 (R Core Team 2013) and JMP Pro 12.0.1 (SAS Institute Inc., Cary, NC). One CH4 flux measurement from a N + CO2 plot was excluded due to an unreasonably high rate, the time series of which suggested disturbance that may have caused unnatural ebullition.
Results
Ecosystem C sequestration
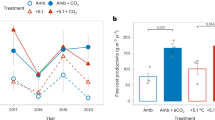
Elevated CO2 and N addition did not change belowground net C gain significantly when evaluated separately (Online Resource 1); however a comparison of control plots to a pooled group of all treated plots showed that addition of either N or CO2 increased belowground C gain significantly (T-Test, F1,18 = 6.545, p = 0.020, Fig. 1). Both factors also increased soil C density at several depths, particularly in combination (Fig. 2b, Online Resource 4). Furthermore, soil δ13C in the N + CO2 treatment was more negative compared to soil δ13C under elevated CO2 alone below 10 cm, indicating that N enhanced belowground inputs of newly fixed C (Fig. 2a, Online Resource 5). On average, belowground net C gain was 20.0 ± 12.1 g C m−2 year−1 in control plots. N addition increased belowground net C gain by 60.5 g C m−2 year−1 and elevated CO2 similarly increased belowground net C gain by 59.5 g C m−2 year−1 on average. Likewise, the combined treatment led to an increase of 60.8 g C m−2 year−1 on average. There were no significant treatment effects on aboveground net C gain, but elevated CO2 tended to increase and N addition tended to decrease aboveground net C gain (Online Resource 1, Fig. 1).
Mean annual net C gain in belowground plant matter and soil (BG) and aboveground plant matter (AG). N = 20. Error bars represent standard error. Addition of resources, CO2 or N, significantly increased net C gain (T-Test, F1,18 = 6.545, p = 0.020). AG net C gain did not significantly differ by treatment
In most years, S. americanus aboveground C mass increased significantly under the combination of elevated CO2 and N addition (two-way repeated measures MANOVA, CO2 × N: F1,16 = 8.327, p = 0.011, Table 1; Fig. 3a). N alone generally decreased C mass and elevated CO2 alone had either no effect or led to a slight increase in C mass for S. americanus (Fig. 3a). N addition increased C mass compared to the control in grasses, however the effect was reduced when combined with elevated CO2 (two-way repeated measures MANOVA, CO2 × N: F1,16 = 7.316, p = 0.016, Fig. 3b). In grasses, net aboveground C mass tended to slightly decrease under elevated CO2 alone (Fig. 3b). With all species together, N addition significantly increased C mass compared to the control (two-way repeated measures MANOVA, F1,16 = 29.400, p = 0.0001), but N + CO2 plots tended to exhibit the highest aboveground C mass since 2010 ( Fig. 3c).
Mean aboveground (AG) C mass from years 2006 to 2012 in S. americanus (a), grasses (b), and total aboveground biomass (c). Error bars represent standard error and are slightly offset to improve clarity. Results of statistical analyses are shown in Table 1. Note that Y-axes are different to also improve clarity
Greenhouse gas and CO2eq fluxes
N addition had a significant impact on in situ N2O emissions in July of 2014, leading to rates triple those measured in control plots and the highest average rates of any month or treatment (average of 8.55 ± 1.29 ug N2O-N m−2 h−1, Fig. 4). However this effect of N addition was reduced through an interaction with elevated CO2 (two-way ANOVA, CO2 × N: F1,12 = 7.329, p = 0.019), so that rates in the combined treatment only increased by an average of 49% compared to the control (Table 2; Fig. 4). No significant differences were found in October, April, or July of 2015 across treatments (Table 2), but the average N2O emissions in April were similar to those in July of 2014 (Fig. 4).
Mean N2O flux in situ across treatments in July and October of 2014 and April and July of 2015. N = 20, except for July 2014, in which N = 16. Error bars represent standard error. For July 2014, a significant interaction of CO2 × N was found by two-way ANOVA (F1,12 = 7.329, p = 0.019). Pairwise comparisons are indicated. Columns that do not share a letter are significantly different from one another for July 2014 data. For the other three time points, no significant differences were found by two-way ANOVA
Elevated CO2 increased CH4 emissions significantly to an average of 3.1 mg CH4–C m−2 h−1 (over 13 times the rate of average CH4 emissions in control plots) in July of 2014 (F1,11 = 3.462, p = 0.090) and tended to increase CH4 emissions on all other sampling dates except for April (Table 2; Fig. 5). This large increase in CH4 emissions was due to a subset of plots both with and without N addition, leading to high variability (Fig. 5). N addition also increased CH4 emissions significantly (triple the rate of average CH4 emissions in control plots) in July of 2014 (two-way ANOVA, F1,11 = 3.462, p = 0.0897, Fig. 5). CH4 flux was negligible in April relative to the other months (Fig. 5).
Mean CH4 flux in situ across treatments in July and October of 2014 and April and July of 2015. N = 20, except for July 2014, in which N = 16. Error bars represent standard error. Both elevated CO2 and N addition significantly increased CH4 flux in July of 2014 (Table 2). Note that CH4 emissions in April were low but positive
There were no significant differences in total SGWP-CO2eq fluxes across treatments (Online Resource 1). Despite the variability of SGWP-CO2eq fluxes in some treatments, the overall trends are still ecologically relevant. Control and +CO2 plots were greenhouse sources (caused positive radiative forcing) on average, while +N and N + CO2 plots were greenhouse sinks (caused negative radiative forcing) on average (Fig. 6). Comparisons between SGWP- and GWP-CO2eq fluxes are shown in Online Resource 2. Changes in the balance between C gain and CH4 emissions primarily drove trends in total radiative forcing despite the higher SGWP of N2O (Fig. 7b). N2O emissions had no bearing on the direction of total radiative forcing.
Mean annual SGWP-CO2 equivalent (CO2eq) fluxes total (a) and split by greenhouse gas (b). N = 19. Error bars represent standard error. Note that CO2eq flux of N2O is difficult to see, as values for N2O were negligible relative to those of CH4 and CO2. Positive values represent greenhouse gas sources and negative values represent sinks. No significant differences were found in total SGWP-CO2eq flux across treatments by two-way ANOVA
Discussion
We show that, contrary to our predictions, addition of N, CO2, or both resources enhanced C sink activity by similar amounts in a tidal brackish marsh primarily due to enhanced C storage belowground (Fig. 1). This response mitigated some (in +CO2 plots) or all (in control and N + CO2 plots) of the increase in radiative forcing caused by CH4 emissions (Fig. 7). Enhanced CH4 emissions in +CO2 plots rendered +CO2 plots net greenhouse sources (positive radiative forcing) on average despite the increase in C storage, though this effect was variable among plots (Figs. 1, 7). N2O emissions had negligible bearing on total radiative forcing, despite the high SGWP of this greenhouse gas, because emission rates were so low (Figs. 4, 7). Below, we explore the interesting finding that N addition increased belowground C gain in the context of our hypothesis regarding the effect of N on radiative forcing, followed by a discussion of C gain and radiative forcing under elevated CO2. We then examine the implications of aboveground C mass in response to global change.
Long-term ecosystem C gain with N addition
Our findings indicate that high inputs of N to brackish marshes may increase their capacity to act as net C sinks (Fig. 1), tending to reduce their contribution to global warming (Figs. 6, 7). These findings contradict results showing that an oligotrophic salt marsh became a net C source with N addition (Morris and Bradley 1999). While N loading could stimulate decomposition in marshes, N addition can also increase biomass and lead to net C gain, as highlighted by this study.
The increase in belowground C sequestration with N addition (Fig. 1) countered our predictions based on findings elsewhere. We hypothesized that N addition would diminish soil elevation gain and increase fluxes of N2O and CH4, thereby increasing total radiative forcing. The observed increase in C gain under N addition cannot be explained by greater root production, at least at shallow depths, as fine root production declined with N addition between 0 and 30 cm depth (Langley and Megonigal 2010). Decomposition does not respond to N addition at some marsh sites (Anisfeld and Hill 2012; Graham and Mendelssohn 2014) and is also unlikely to explain the observed increase in C accumulation. We hypothesize that increased root production below a depth of 30 cm in +N plots (unpublished data) may have increased marsh elevation and net C gain, overriding reduced fine root biomass at shallower depths, consequently reducing radiative forcing (Figs. 1, 6). Furthermore, N addition may stimulate deposition of newly fixed C deep in the soil profile. Because the additional CO2 delivered into both the +CO2 and N + CO2 plots is depleted in 13C, a comparison of the 13C signatures in these two treatments indicates how N addition affected deposition of newly fixed C. Soil δ13C in the N + CO2 treatment was indeed more negative compared to δ13C under elevated CO2 alone below 10 cm, suggesting that N addition may increase deposition of newly fixed C at greater soil depth (Fig. 2a, Online Resource 5). Furthermore, N addition did significantly increase C density at several depths (Fig. 2b, Online Resource 4). Overall, N addition tended to decrease total radiative forcing by significantly enhancing C gain, similar to other recent studies (Graham and Mendelssohn 2014), despite higher CH4 and N2O emissions compared to controls.
Our hypothesis that N addition increases N2O and CH4 fluxes was correct during certain seasons (Figs. 4, 5), but emissions were not high enough to counteract the effect of increased belowground C sequestration on total radiative forcing (Fig. 7). N addition stimulated N2O flux significantly in July of 2014 and tended to stimulate N2O flux in October and April, although rates were trivial in the overall greenhouse gas budget (Figs. 4, 7). By adding NH4 +, we may have directly stimulated nitrification or indirectly stimulated denitrification, both of which can produce N2O. Rates of N2O emissions in situ (Fig. 4) were low compared to those observed in other marshes and much lower than potential rates at this site as measured in laboratory incubations (Pastore et al. 2016; VanZomeren et al. 2012; White and Howes 1994). N2O flux does vary spatially and over time (Orr et al. 2014; Shrestha et al. 2012), however our measurements over three seasons indicate that N2O emissions are unlikely to matter to the total radiative forcing of this marsh (Fig. 7). N stimulated CH4 flux significantly in July of 2014 (312% higher than the control, Fig. 5), but the increase was not high enough relative to belowground C gain to cause positive radiative forcing (Figs. 6, 7). This response is consistent with other studies in brackish and salt marshes and may be due to increased biomass and soil C inputs (Fig. 3c; Irvine et al. 2012; Mou et al. 2014; Zhang et al. 2010). Overall, CH4 emission rates (Fig. 5) fell within the ranges reported for a variety of wetlands worldwide (Whalen 2005) and a C3-dominated community adjacent to the site of the present study (Marsh et al. 2005). To offset the effect of C gain on total radiative forcing in +N plots, CH4 emissions would need to average 0.93 mg CH4–C m−2 h−1 over the course of a year. Instantaneous rates rarely surpassed this value during any season, and therefore it is likely that greater C storage with N addition overrode positive effects of N addition on CH4 emissions, increasing the greenhouse sink strength of this marsh (Figs. 6, 7).
C gain and variability in radiative forcing under elevated CO2
As atmospheric CO2 concentration continues to rise, marshes may have variable impacts on climatic change due to differences in factors that affect C gain and greenhouse gas emissions, such as salinity and plant composition. In this brackish marsh, elevated CO2 tended to increase total radiative forcing, but responses were highly variable across plots, with some plots acting as net greenhouse gas sources while others acted as sinks (Fig. 6). The increase in belowground C gain under elevated CO2 was similar in magnitude to that of +N plots (Fig. 1) and was perhaps due to a stimulation of fine root productivity (Langley et al. 2009a). Enhanced CH4 emissions (Fig. 5) countered this increase in C gain and led to high average total SGWP-CO2eq flux (Fig. 7a). Similarly, a meta-analysis of studies investigating the effects of CO2 on N2O and CH4 fluxes showed that elevated CO2 stimulated CH4 emissions by 13.2% in wetlands, reducing the greenhouse mitigation effect of greater soil C storage under elevated CO2 (van Groenigen et al. 2011). The high variability in CH4 emissions in the present study (Fig. 5) may have been partially due to ebullition, which was evidenced by large jumps in CH4 concentration in some accumulation curves. However, elevated CO2 stimulation of CH4 emissions (Dacey et al. 1994; Marsh et al. 2005) and porewater CH4 (Keller et al. 2009; Marsh et al. 2005) in the S. americanus-dominated community of a companion experiment at the same site provide further support that elevated CO2 increases CH4 production in this brackish marsh. This response may be due to increased soil C inputs from shoot or root biomass at this site (Langley et al. 2009a), and therefore an increase in organic C to support fermentation, sulfate reduction and depletion (Keller et al. 2009), and ultimately methanogenesis.
CH4 emissions from sites with salinities above the threshold of 18 are typically low at ambient CO2 (<1 mg CH4 m−2 h−1 in gas units or <0.75 mg CH4–C m−2 h−1 in C units) (Olsson et al. 2015; Poffenbarger et al. 2011), but our findings indicate that elevated CO2 may raise this salinity threshold. Salinity averages 10 (range from 4 to 20) at this site, and CH4 emissions averaged 0.31 mg CH4–C m−2 h−1 under ambient conditions across sampling dates (Fig. 6). In a climate change context, the direction of the greenhouse balance under ambient conditions is not relevant to the issue of radiative forcing because this site presumably had the same radiative balance in past centuries when the climate was more stable. However, changes in the radiative balance due to our treatments are relevant because they are caused by anthropogenic activity. Despite relatively high pore water salinity, elevated CO2 increased the likelihood of a plot becoming a net greenhouse source (Fig. 6). This occurred through enhanced CH4 emissions under elevated CO2 (by an average of 1.42 mg CH4–C m−2 h−1 across sampling dates) likely due to increased soil C inputs. Importantly for wetland creation and restoration activities, these results suggest that elevated CO2 will raise the salinity threshold at which the radiative effects of C sequestration exceed those of CH4 emissions, such that restoration efforts are less likely to have the desired net neutral or cooling effect on climate with CO2 rise.
In +N plots, elevated CO2 did not increase the radiative balance to the extent it did in unfertilized plots (Fig. 6) because CH4 emissions were not stimulated as strongly (Figs. 5, 7a). Variability was less than under elevated CO2 alone (Fig. 6). The combination of elevated CO2 and N addition did not increase net belowground C gain in an additive manner as expected, but rather led to an increase similar to observed increases when either resource was added alone (Fig. 1); however, the increase was still enough to offset the effect of CH4 emissions on total radiative forcing (Figs. 1, 7).
Aboveground ecosystem C uptake
The magnitude of aboveground C gain was higher than one might expect after nine years of treatment application, reaching from about 10–50% as high as belowground net C gain in the various treatment combinations (Fig. 1). The step-change in treatment conditions when treatment application began contributed to these large effects. Plants in wetlands are extremely dynamic, and communities may adjust rapidly to novel conditions, including via changes in productivity and aboveground biomass (Langley and Megonigal 2010; Langley et al. 2013). However, over nearly a decade, increased biomass was surpassed in magnitude by increases in soil organic C, which will continue accumulating as long as soil elevation gain can keep up with rising seas (Kirwan and Guntenspergen 2012).
The trends of increasing peak biomass indicate that annual patterns of C exchange may be gaining in amplitude. However, owing to its annual growth, senescence, and relatively rapid decomposition, aboveground biomass may not contribute much to the long-term trend of radiative forcing at this site. Upon senescence, a large portion of aboveground litter C is consumed by microbes or detritivores, and so, is rapidly respired back to the atmosphere. Alternatively, litter is exported with tides or resides in the litter layer. Aboveground litter C that does ultimately enter the soil would be captured by our soil C assessment. Therefore, we do not include trends in aboveground biomass C in our calculations of SGWP-CO2eq fluxes because in this herbaceous ecosystem, aboveground biomass does not represent a durable C pool.
Conclusions
We show that C sink activity in a brackish marsh is similarly enhanced by addition of N, CO2, or both resources together, primarily due to enhanced C storage belowground. These results also suggest that changes in net C gain and CH4 emissions primarily drive the potential for tidal brackish marshes to impact global climate change. Overall, this marsh acts as a strong net greenhouse gas sink (negative radiative forcing) with N addition. Conversely, elevated CO2 stimulated CH4 emissions, rendering many plots net greenhouse sources (positive radiative forcing) compared to ambient conditions. Aboveground net C uptake was substantial, corroborating the recent finding that aboveground vegetation responses can dominate sub-decadal changes in coastal wetland C mass (Doughty et al. 2015), although this mechanism is most important to long-term ecosystem C balance in ecosystems with perennial plant communities such as shrub-dominated marshes or mangrove forests. In the longer term, vegetation pools will reach equilibrium while soil organic C will continue to accumulate as long as soil elevation gain keeps up with rising seas (Kirwan and Guntenspergen 2012).
Our objective was to investigate CO2 and N treatment effects on the radiative forcing of a natural marsh rather than to scale our measurements, and we therefore caution against generalizing our results to all marsh ecosystems. Differences in salinity, temperature, vegetation, and other factors that vary across marshes strongly affect greenhouse gas emissions. Salinity, in particular, has strong effects on CH4 flux and can shift the balance between a marsh acting as a net source or sink of C (Weston et al. 2014). Though this marsh is characterized by salinities that typically suppress CH4 emissions, our results suggest that elevated CO2 could transform net greenhouse sinks into sources, thus potentially increasing radiative forcing. While N2O flux is highly variable across sites and has a high SGWP, even the highest measured in situ rates in the present study had negligible effects on total radiative forcing, similar to findings of Wang et al. (2015). We demonstrate that N addition, elevated CO2, and associated vegetation change in wetlands can have sustained effects on ecosystem C status on a decadal time scale, and that the magnitude of the effect of CH4 emissions on ecosystem radiative forcing is similar to that of C gain rates.
References
Alam MS, Jia Z (2012) Inhibition of methane oxidation by nitrogenous fertilizers in a paddy soil. Front Microbiol 3(246):1–13
Anisfeld SC, Hill TD (2012) Fertilization effects on elevation change and belowground carbon balance in a Long Island Sound tidal marsh. Estuaries Coasts 35(1):201–211
Barnard R, Leadley PW, Hungate BA (2005) Global change, nitrification, and denitrification: a review. Glob Biogeochem Cycles 19(1):1–13
Bodelier PLE, Laanbroek HJ (2004) Nitrogen as a regulatory factor of methane oxidation in soils and sediments. FEMS Microbiol Ecol 47(3):265–277
Bowden WB (1986) Gaseous nitrogen emmissions from undisturbed terrestrial ecosystems: an assessment of their impacts on local and global nitrogen budgets. Biogeochemistry 2(3):249–279
Brown JR, Blankinship JC, Niboyet A, van Groenigen KJ, Dijkstra P, Le Roux X, Leadley PW, Hungate BA (2012) Effects of multiple global change treatments on soil N2O fluxes. Biogeochemistry 109(1–3):85–100
Chmura GL, Anisfeld SC, Cahoon DR, Lynch JC (2003) Global carbon sequestration in tidal, saline wetland soils. Glob Biogeochem Cycles 17(4):1–12
Collins M, Knutti R, Arblaster JM, Dufresne JL, Fichefet T, Friedlingstein P, Gao X, Gutowski WJ, Johns T, Krinner G (2013) Long-term climate change: projections, commitments and irreversibility. In: Climate change 2013: the physical science basis. contribution of working group i to the fifth assessment report of the intergovernmental panel on climate change [Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
Conrad R, Rothfuss F (1991) Methane oxidation in the soil surface layer of a flooded rice field and the effect of ammonium. Biol Fertil Soils 12(1):28–32
Dacey JWH, Drake BG, Klug MJ (1994) Stimulation of methane emission by carbon dioxide enrichment of marsh vegetation. Nature 340(6484):47–49
Deegan LA, Johnson DS, Warren RS, Peterson BJ, Fleeger JW, Fagherazzi S, Wollheim WM (2012) Coastal eutrophication as a driver of salt marsh loss. Nature 490(7420):388–392
Doughty CL, Langley JA, Walker WS, Feller IC, Schaub R, Chapman SK (2015) Mangrove range expansion rapidly increases coastal wetland carbon storage. Estuaries Coasts 39(2):385–396
Graham SA, Mendelssohn IA (2014) Coastal wetland stability maintained through counterbalancing accretionary responses to chronic nutrient enrichment. Ecology 95(12):3271–3283
Gulledge J, Schimel JP (1998) Low-concentration kinetics of atmospheric CH4 oxidation in soil and mechanism of NH4 + inhibition. Appl Environ Microbiol 64(11):4291–4298
Hamersley MR, Howes BL (2005) Coupled nitrification-denitrification measured in situ in a Spartina alterniflora marsh with a 15NH4 + tracer. Mar Ecol Prog Ser 299:123–135
Hartmann, DL, Klein Tank AMG, Rusticucci M, Alexander LV, Brönnimann S, Charabi Y, Dentener FJ, Dlugokencky EJ, Easterling DR, Kaplan A, Soden BJ, Thorne PW, Wild M, Zhai PM (2013) Observations: atmosphere and surface. In: Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change [Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
Irvine IC, Vivanco L, Bentley PN, Martiny JBH (2012) The effect of nitrogen enrichment on C1-cycling microorganisms and methane flux in salt marsh sediments. Front Microbiol 3(90):142–151
Keller JK, Wolf AA, Weisenhorn PB, Drake BG, Megonigal JP (2009) Elevated CO2 affects porewater chemistry in a brackish marsh. Biogeochemistry 96(1):101–117
Kemp WM, Boynton WR, Adolf JE, Boesch DF, Boicourt WC, Brush G, Cornwell JC, Fisher TR, Glibert PM, Hagy JD (2005) Eutrophication of Chesapeake Bay: historical trends and ecological interactions. Mar Ecol Prog Ser 303(21):1–29
Kinney EL, Valiela I (2013) Changes in δ15N in salt marsh sediments in a long-term fertilization study. Mar Ecol Prog Ser 477:41–52
Kirwan ML, Guntenspergen GR (2012) Feedbacks between inundation, root production, and shoot growth in a rapidly submerging brackish marsh. J Ecol 100(3):764–770
Langley JA, Megonigal JP (2010) Ecosystem response to elevated CO2 levels limited by nitrogen-induced plant species shift. Nature 466(7302):96–99
Langley JA, McKee KL, Cahoon DR, Cherry JA, Megonigal JP (2009a) Elevated CO2 stimulates marsh elevation gain, counterbalancing sea-level rise. Proc Natl Acad Sci 106(15):6182–6186
Langley JA, Sigrist MV, Duls J, Cahoon DR, Lynch JC, Megonigal JP (2009b) Global change and marsh elevation dynamics: experimenting where land meets sea and biology meets geology. Smithson Contrib Mar Sci 38:391–400
Langley JA, Mozdzer TJ, Shepard KA, Hagerty SB, Patrick Megonigal J (2013) Tidal marsh plant responses to elevated CO2, nitrogen fertilization, and sea level rise. Glob Change Biol 19(5):1495–1503
Le Mer J, Roger P (2001) Production, oxidation, emission and consumption of methane by soils: a review. Eur J Soil Biol 37(1):25–50
Liu L, Greaver TL (2009) A review of nitrogen enrichment effects on three biogenic GHGs: the CO2 sink may be largely offset by stimulated N2O and CH4 emission. Ecol Lett 12(10):1103–1117
Marsh AS, Rasse DP, Drake BG, Megonigal JP (2005) Effect of elevated CO2 on carbon pools and fluxes in a brackish marsh. Estuaries 28(5):694–704
McLeod E, Chmura GL, Bouillon S, Salm R, Björk M, Duarte CM, Lovelock CE, Schlesinger WH, Silliman BR (2011) A blueprint for blue carbon: toward an improved understanding of the role of vegetated coastal habitats in sequestering CO2. Front Ecol Environ 9(10):552–560
Megonigal JP, Schlesinger WH (1997) Enhanced CH4 emission from a wetland soil exposed to elevated CO2. Biogeochemistry 37(1):77–88
Megonigal JP, Schlesinger W (2002) Methane-limited methanotrophy in tidal freshwater swamps. Glob Biogeochem Cycles 16(4):1–10
Morris JT, Bradley PM (1999) Effects of nutrient loading on the carbon balance of coastal wetland sediments. Limnol Oceanogr 44(3):699–702
Morris JT, Nyman JA, Shaffer GP (2014) The influence of nutrients on the coastal wetlands of the Mississippi delta. Perspectives on the restoration of the Mississippi Delta. Springer, Netherlands, pp 111–123
Mou X, Liu X, Tong C, Sun Z (2014) Responses of CH4 emissions to nitrogen addition and Spartina alterniflora invasion in Minjiang River estuary, southeast of China. Chin Geogr Sci 24(5):562–574
Myhre G, Shindell D, Bréon F-M, Collins W, Fuglestvedt JS, Huang J, Koch D, Lamarque J-F, Lee D, Mendoza B, Nakajima T, Robock A, Stephens G, Takemura T, Zhang H (2013) Anthropogenic and natural radiative forcing. In: Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Cambridge [Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA. pp 659–740
Neubauer SC, Megonigal JP (2015) Moving beyond global warming potentials to quantify the climatic role of ecosystems. Ecosystems 18(6):1000–1013
Niboyet A, Le Roux X, Dijkstra P, Hungate BA, Barthes L, Blankinship JC, Brown JR, Field CB, Leadley PW (2011) Testing interactive effects of global environmental changes on soil nitrogen cycling. Ecosphere 2(5): art56
Olsson L, Ye S, Yu X, Wei M, Krauss KW, Brix H (2015) Factors influencing CO2 and CH4 emissions from coastal wetlands in the Liaohe Delta, Northeast China. Biogeosci Discuss 12(4):3469–3503
Orr CH, Predick KI, Stanley EH, Rogers KL (2014) Spatial autocorrelation of denitrification in a restored and a natural floodplain. Wetlands 34(1):89–100
Pastore MA, Megonigal JP, Langley JA (2016) Elevated CO2 promotes long-term nitrogen accumulation only in combination with nitrogen addition. Glob Change Biol 22(1):391–403
Pendleton L, Donato DC, Murray BC, Crooks S, Jenkins WA, Sifleet S, Craft C, Fourqurean JW, Kauffman JB, Marbà N (2012) Estimating global “blue carbon” emissions from conversion and degradation of vegetated coastal ecosystems. PLoS ONE 7(9):e43542
Poffenbarger HJ, Needelman BA, Megonigal JP (2011) Salinity influence on methane emissions from tidal marshes. Wetlands 31(5):831–842
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/
Reinhardt M, Müller B, Gächter R, Wehrli B (2006) Nitrogen removal in a small constructed wetland: an isotope mass balance approach. Environ Sci Technol 40(10):3313–3319
Shrestha J, Niklaus PA, Frossard E, Samaritani E, Huber B, Barnard RL, Schleppi P, Tockner K, Luster J (2012) Soil nitrogen dynamics in a river floodplain mosaic. J Environ Qual 41(6):2033–2045
Smith CJ, DeLaune RD, Patrick WH (1983) Carbon dioxide emission and carbon accumulation in coastal wetlands. Estuar Coast Shelf Sci 17(1):21–29
Smith KA, Dobbie KE, Ball BC, Bakken LR, Sitaula BK, Hansen S, Brumme R, Borken W, Christensen S, Priemé A (2000) Oxidation of atmospheric methane in Northern European soils, comparison with other ecosystems, and uncertainties in the global terrestrial sink. Glob Change Biol 6(7):791–803
van Groenigen KJ, Osenberg CW, Hungate BA (2011) Increased soil emissions of potent greenhouse gases under increased atmospheric CO2. Nature 475(7355):214–216
VanZomeren CM, White JR, DeLaune RD (2012) Fate of nitrate in vegetated brackish coastal marsh. Soil Sci Soc Am J 76(5):1919–1927
Wang H, Liao G, D’Souza M, Yu X, Yang J, Yang X, Zheng T (2015) Temporal and spatial variations of greenhouse gas fluxes from a tidal mangrove wetland in Southeast China. Environ Sci Pollut Res 23(2):1873–1885
Weston NB, Neubauer SC, Velinsky DJ, Vile MA (2014) Net ecosystem carbon exchange and the greenhouse gas balance of tidal marshes along an estuarine salinity gradient. Biogeochemistry 120(1–3):163–189
Whalen SC (2005) Biogeochemistry of methane exchange between natural wetlands and the atmosphere. Environ Eng Sci 22(1):73–94
White DS, Howes BL (1994) Long-term 15N-nitrogen retention in the vegetated sediments of a New England salt marsh. Limnol Oceanogr 39(8):1878–1892
White JR, Reddy KR (1999) Influence of nitrate and phosphorus loading on denitrifying enzyme activity in Everglades wetland soils. Soil Sci Soc Am J 63(6):1945–1954
Wolf AA, Drake BG, Erickson JE, Megonigal JP (2007) An oxygen-mediated positive feedback between elevated carbon dioxide and soil organic matter decomposition in a simulated anaerobic wetland. Glob Change Biol 13(9):2036–2044
Zhang Y, Ding W, Cai Z, Valerie P, Han F (2010) Response of methane emission to invasion of Spartina alterniflora and exogenous N deposition in the coastal salt marsh. Atmos Environ 44(36):4588–4594
Acknowledgements
The authors thank J. Duls, G. Peresta, and A. Peresta for assistance with data collection and maintenance of the experiment and treatments at the Smithsonian Global Change Research Wetland. We also thank M. Vile for technical insight, as well as two anonymous reviewers for the helpful insights. This work was supported by National Science Foundation-LTREB Program Grants DEB-0950080 and DEB-1457100, the Smithsonian Institution, and Villanova University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Melany Fisk.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pastore, M.A., Megonigal, J.P. & Langley, J.A. Elevated CO2 and nitrogen addition accelerate net carbon gain in a brackish marsh. Biogeochemistry 133, 73–87 (2017). https://doi.org/10.1007/s10533-017-0312-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-017-0312-2