Abstract
Unprecedented anthropogenic changes to biodiversity and biogeography demand a greater understanding of the consequences of altered faunal composition for ecosystem functioning. Selective predation has important, yet poorly understood effects on ecosystem stability, and can be strongly influenced by the relative frequencies of different prey types in the environment. Yet, how predators adjust their selection for prey according to their environmental frequency is often overlooked. Here, we assessed frequency dependent selection of prey by dingoes and foxes in the Australian desert, biannually, across a nine-year period (2007–2016). Both predators exhibited potentially destabilizing, negative frequency dependent selection for prey. Foxes persisted to preferentially consume a threatened, native rodent (Notomys fuscus) when it was environmentally scarce. Bolstered by the observation that N.fuscus occurs at low densities in areas where foxes are common, our results suggest that N.fuscus is particularly vulnerable to predation by this predator; possibly because it is naïve and/or lacks adaptations to avoid or escape predation by the relatively recently introduced fox. Dingoes tended to consume reptiles when they were scarce; potentially constituting a conservation concern if selected reptilian taxa are threatened. Foxes avoided, thus were unlikely to control populations of overabundant kangaroos, while both foxes and dingoes showed a preference for, and may therefore control populations of invasive rabbits. The integration of our results into the relative suites of (de)stabilizing influences exerted by dingoes and foxes is important to provide a more dynamic insight into how each predator impacts their naturally fluctuating ecosystems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Unprecedented anthropogenic changes to biodiversity and biogeography demand a greater understanding of the consequences of altered faunal composition for ecosystem functioning. Selective predation- the utilization of a nonrandom subset of available prey types –has strong, yet poorly understood influences on community level dynamics and ecosystem stability (Hoy et al. 2021). A prey type is considered ‘selected for’ when its relative frequency in the predator’s diet exceeds its frequency in the environment, and ‘avoided’ when its dietary frequency is lower than that in the environment. Selective foraging is a dynamic process that can be strongly influenced by changes in the relative frequency of prey types in the environment (Murdoch 1969; Oaten and Murdoch 1975). Yet, the majority of studies on this topic quantify selective foraging at a single point in time (Paltridge, 2002; Cupples et al., 2011). While such quantifications may reveal a predator’s preference of one prey type over the suite of others available at the time of sampling, they are unlikely to capture the true predation risk to a prey type, should a preferred type become unavailable (Allen et al., 2012).
How predators adjust their prey preferences according to the relative frequency of prey types in the environment has important, yet often overlooked implications for ecosystem stability. For example, positive, frequency-dependent predation (when the strength of selection for a prey type increases with its relative availability in the environment) may regulate the abundance of common prey while providing refuge for rare prey types, and often promotes ecosystem stability (Oaten and Murdoch 1975). Positive frequency dependent selection is also associated with another stabilizing foraging behaviour - prey switching (when a predator’s selection for abundant, unpreferred prey increases when preferred prey types are relatively rare) (Murdoch 1969). Negative frequency dependent predation (when predators persist in eating a preferred prey type even when it is scarce) on the other hand, can disfavor the coexistence of different prey types if the selection of rare prey makes them more likely to become (locally) extinct. Negative frequency dependent predation consequently tends to be associated with destabilizing ecosystem effects (O’Donoghue et al., 1998).
The Strzelecki and Simpson deserts in central Australia provide excellent arenas to examine how selective predation can be influenced by fluctuations in the frequency of prey types. The region experiences wildly fluctuating, non-cyclic variation in prey frequencies, which are largely driven by irregular rain events (Pavey et al., 2008; Spencer et al., 2017, Figs. 4 and 7). The region also contains two canid predators; the dingo (Canis dingo: 12 − 20 kg) and the recently introduced red fox (Vulpes vulpes; 3–14 kg). The foraging behaviours and interactions between these predators are of great ecological and economic significance due to their heavily debated impacts on vulnerable native mammal populations and farming practices (Moseby et al. 2012; Cooke and Soriguer 2017). The dingo is Australia’s largest terrestrial predator and has coexisted with extant native prey species since its arrival on the continent ~ 5000 years ago (Fleming et al. 2001; Savolainen et al. 2004). Since European colonization in 1788, dingoes have been exterminated from much of the mainland Australia because they attack livestock (Fleming et al. 2001; Philip 2021). Along with numerous other placental mammals, Europeans introduced the red fox; an efficient mesopredator considered a significant threat to the long-term survival of many endemic Australian mammals, especially small, desert-dwelling mammals weighing between 50 and 5500 g (Letnic and Dickman 2006; McKenzie et al. 2007)).
The mesopredator release hypothesis posits that the presence of dingoes in Australia reduces predation pressures on small mammals in Australia, by suppressing and/or changing the foraging behaviours of foxes (and other mesopredators); however, this is debated (reviewed in: Letnic et al., 2012; Allen et al., 2013; Cooke and Soriguer, 2017). The composition of dingo and fox diets is well documented (Vernes et al. 2001; Brooke and Kutt, 2011; Cupples et al., 2011; Doherty et al., 2019), yet the dietary responses of dingoes and foxes to fluctuating prey remain largely unexplored (however, see Spencer et al., 2017; Corbett and Newsome, 1987). Dingoes exhibit considerable dietary overlap with foxes; thus, the presence of dingoes would only yield biodiversity benefits if their direct predation risk to vulnerable small mammals is less than their indirect ability to reduce fox predation on the same prey (Holt and Lawton 1994). Developing research into frequency dependent prey selection is important to provide a more comprehensive and dynamic understanding of the predation risks imposed by dingoes and foxes in their naturally fluctuating environments.
The ‘dingo fence’ (a 5500 km structure excluding dingoes from the south-east part of Australia) runs through the Strzelecki and Simpson deserts, and provides an excellent opportunity to compare the large-scale effects of prey frequency on fox and dingo foraging preferences in their respective ecosystems (McKnight 1969). This is because the relative abundance of dingoes and foxes is reversed on each side of the fence due to the suppression of dingoes by pastoralists and the subsequent competitive release of foxes on the NSW side of the fence (Letnic and Dworjanyn 2011). In addition, the suppression of dingoes in NSW has been accompanied by a shift in the Strzelecki Desert ecosystem, evidenced by the irruption of kangaroo and emu populations, an increase in woody shrub cover, decrease in grass cover and suppression of small prey species that are frequently consumed by foxes (Gordon et al. 2017; Morris and Letnic 2017; Wijas and Letnic 2021).
In the present study, we quantified frequency dependent selection of six important prey species of dingoes and foxes (outside and inside the dingo fence, respectively), biannually, across a nine-year period (2007–2016). We quantified the strength of selection for different prey using the Manly Chesson selection index (α), which reflects the relative frequency of prey in the environment and the predator’s diet, and examined whether values of α were dependent on the frequency of prey in the environment. We also assessed whether values of α covaried with predator density (as predators may become less selective as their density, and hence competition for food, increases (Barnard and Brown 1981)).
Methods
Study area
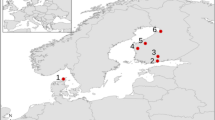
To counter the threat posed to livestock by dingoes and prevent the incursion of dingoes into sheep grazing lands in arid Australia, an existing rabbit fence was heightened and converted into a ‘dingo fence’ during the early to mid-20th century. To date, the dingo fence extends over 5,500 km between New South Wales (NSW), Queensland (QLD) and South Australia (SA), (McKnight 1969) (Fig. 1). We conducted our study between May 2007 and July 2016 on each side of a section of the dingo fence that delineates the borders of the states of New South Wales and South Australia (following the meridian 141°E through the sand dunes of the Strzelecki Desert, Fig. 1). Dingoes were common and foxes were rare at two of our study sites; Quinyambie and Strzelecki Regional Reserve, that were located outside (on the SA side of) the dingo fence (Fig. 1). Foxes are thought to be rare at sites where dingoes are common due to top-down regulation of their populations by dingoes (Letnic et al. 2011). Due to intensive population control, dingoes were rare at our other two study sites; Sturt National Park and Winnathee, located inside (on the NSW side of) the dingo fence. Foxes are abundant at these sites due to release from top-down regulation of their populations by dingoes (Letnic et al. 2011). To account for livestock grazing, one site on either side of the fence was grazed (Quinyambie and Winnathee) and one site on either side of the fence was in a conservation area with no grazing (Strzelecki Regional Reserve and Sturt National Park) (Fig. 1).
Map of Australia showing the study sites (grey points) with the dingo fence (bold black line). SRR stands for Strzelecki regional reserve, NP stands for national park, SA stands for South Australia, NSW stands for New South Wales, QLD stands for Queensland (Wijas and Letnic 2021)
Species abundance
We indexed the abundance of foxes, dingoes and six prey types categorized as; rabbits (Oryctolagus cuniculus), hopping mice (Notomys fuscus), long-haired rats (Rattus villosissimus), reptiles, kangaroos, and small rodents (< 30 g, excluding hopping mice)). These prey types were included in our analyses as they were present in > 5% of scats over our nine-year sampling period. Foxes, dingoes, kangaroos and rabbits are crepuscular; thus, we indexed the abundance of these mammals through spotlight surveys conducted after sunset. Spotlight surveys were conducted in March, June/July, and November of each year on unpaved, private roads which all have infrequent traffic. An observer sat on the roof of a four-wheel-drive vehicle moving at 15 km/h and a 50 W spotlight was used to equally scan the two sides of the vehicle. We conducted 2–3 spotlight surveys during each trip. On each sampling occasion the abundance of foxes, dingoes, kangaroos and rabbits was indexed over 30 km of road at each of the four study sites (on the same roads at each site on each trip). The detectability of kangaroos was limited by the range of the spotlight beam to 100 m on either side of the vehicle. Rabbits were more difficult to detect than the larger mammals because of their smaller body size. Overall, 95% of the rabbits we observed were within a 60 m belt on either side of the vehicle (Mills et al. 2020). Consequently, for rabbit density estimates we truncated the transect belt width to 60 m either side of the vehicle. The abundance of each prey was expressed as the number of individuals sighted per hectare. This number was then multiplied by 100, giving the number of individuals spotted per km2.
We conducted trapping to index the abundance of hopping mice, long-haired rats, other small mammals, and reptiles. Hopping mice, long-haired rats, and other small mammals, were routinely caught in both Sherman traps and pitfall traps while reptiles were captured exclusively in pitfall traps. At each site on each survey occasion, we deployed 160 Sherman box traps (H. B. Sherman Traps, Inc., 7.5 × 9 × 23 cm) and 72 pitfall traps (PVC pipe 16 × 60 cm) across eight one-hectare trapping grids located on sand dunes. Trapping grids were at least 1 km apart. Twenty Sherman box traps were placed on each of the eight one-hectare trapping grids and baited using a mixture of oats, peanut butter, and sugar syrup. Each of the eight trapping grids also had nine pitfall traps, fitted with a 10 m drift fence extending out on either side of the trap. On each sampling occasion trapping was carried out for two to three consecutive nights and the bait was replenished each day. The abundances of hopping mice, long-haired rats and other small mammals were expressed as number per hectare, reptile abundance was expressed as the number of captures per km2.
Dietary frequency
Dingo and fox scats were collected by observers walking during the daytime along the same vehicle tracks that the spotlight surveys were conducted on. Collected scats were air-dried in paper bags before being oven-dried in the laboratory for 12 h at 100 °C. After drying, individual scats were placed in nylon bags and then washed in a washing machine (Cupples et al. 2011). After washing, remains in the scats were examined under a microscope and identified by comparing diagnostic material (that is, hair cross-sections, teeth, claws) against known reference specimens (Cupples et al. 2011). The percentage frequency of occurrence of dietary items (that is, species of prey) in the scats was calculated as the number of scats in which the dietary item was identified, divided by the total number of scats sorted.
Statistical analysis
We estimated fox and dingo selection for each prey type using the Manly Chesson selection index (α), for each prey type i. α quantifies dietary frequency in relationship to environmental frequency (Cuthbert et al. 2018; Hoy et al. 2021; Chesson 1978; Manly 1974). Specifically, α is a relative measure of selection calculated as:
where ri is the proportion of prey item i in the diet, ei is the proportion of prey item i in the environment and m represents the number of prey types in the environment (m = 6 in our analyses). Following Hoy et al. (2021, 2019), the response variable αi was logit-transformed. A logit-transformation was necessary because α is constrained between 0 and 1, thus has a binomial distribution where the variance is a function of the mean - reaching a maximum value at a proportion of 0.5 and declining to zero at proportions of zero (total avoidance) and one (total selection). The logit transformation is used to linearize the sigmoid relationship between the binary response variable (α) and explanatory variables by ‘expanding the ends of the scale towards zero and one’, making its distribution more suitable for a linear mixed model (Armitage and Berry, 1994). Logit-transformed αi values ranged from − 3.2 (complete avoidance, untransformed value = 0) to 3.2 (strongest possible selection, untransformed value = 1). If αi = 1/m, the predator is feeding randomly on this prey type, and the prey is consumed in proportion to its abundance in the environment (because m = 6 in our analyses this equates to: αi = 0.17, or -0.6 when logit transformed). This approach requires the probability of detecting different prey in traps to be consistent among prey types. In our system, prior work has indicated that the relative ‘trap-ability’ of the prey species in our study accurately reflects their abundances and is not an artefact of changes in animal behaviour or susceptibility to capture (Mills et al. 2021; Letnic and Koch 2010; Gordon et al. 2017).
Linear mixed models were conducted in R v.4.0.1 (R Core Development Team, 2021) to assess the association between selection for a prey type (αi) (included as a dependent variable), the environmental abundances of each prey type and the environmental abundance of the respective predator (i.e., dingo or fox) (included as fixed effects). Prey type was included as a random factor to explore whether frequency dependent selection by predators varied among prey. When ‘site’ (grazed or ungrazed) was included into the linear mixed models as an additional random effect, it did not change the effect size or statistical significance of any fixed effects. ‘Site’ was therefore removed from our final models to reduce model complexity and the risk of overfitting the model (i.e., describing the random error in the data rather than the relationships between variables).
Results
In total, 995 scats were collected between May 2007 and July 2016 (632 dingo scats and 363 fox scats).
Dingo foraging behaviours
Kangaroos were omitted from the dietary analyses of dingoes since, due to the relative scarcity of this prey in the environment (Fig. 2), we were unable to calculate values of α for kangaroos by dingoes. Nevertheless, while kangaroos were extremely rare outside the dingo fence (making up an average of only 0.13% of the prey in the environment over our seven-year sampling period) they were found in 3.16% of scats, possibly suggesting that kangaroos are highly selected for by dingoes, which may contribute to why kangaroos are so rare outside the dingo fence. Hopping mice and rabbits were the most abundant taxa outside the dingo fence (where dingoes are common) (Fig. 2), and these taxa also made up the majority of the dingoes’ diet (Fig. 2). Across our study sample, dingoes had a neutral preference for long-haired rats, a preference for rabbits and reptiles and avoided hopping mice and small rodents (Figs. 3 and 4). In 2007, small rodents made up more than 60% of the available prey in the environment compared to an average of 7% across all following years (Fig. 4). This ‘outlier’ did not affect our estimations of average dingo selection (α) for small rodents, as this metric could not be calculated in 2007 as we did not detect any small rodents in dingoes’ diet.
Differences in the relative abundance and consumption of different prey for dingoes (outside the fence) and foxes (inside the dingo fence) between 2007–2016. Grey bars indicate the proportion of dingo/fox-consumed prey types, averaged across years. Black bars indicate the proportion of prey types in the environment, averaged across years
Dingo preference for different prey types using the logit transformed values of the Manly Chesson selection index (αi). The dashed, red horizonal line (at -0.6 on the y-axis) indicates that the dingo is feeding randomly, αi > -0.6 indicates a preference for the prey and αi < -0.6 indicates avoidance. Vertical lines indicate 25th, 50th and 75th percentiles, blue ‘x’ represent mean values
Temporal changes in environmental prey abundance and occurrence in dingo diet. Dashed, vertical grey lines indicate sampling periods where rainfall was greater than the previous year. Note, for illustrative purposes x-axis ticks are not scaled proportionately to the length of time between observations
Across all prey types, dingoes tended to exhibit negative frequency dependent selection, i.e., dingoes’ preference for a given prey was generally negatively associated with the abundance of that prey type in the environment (Table 1; Figs. 4 and 5). This trend was primarily driven by dingoes’ negative frequency dependent selection for hopping mice (estimate =-0.027 (SE = 0.010), t= -2.608 p = 0.023 *), reptiles (estimate =-0.071 (SE = 0.026), t= -2.647, p = 0.023 *), and rats (estimate=-0.074 (SE = 0.033), t= -2.221, p = 0.051) (Fig. 5). Dingoes showed no frequency dependent selection for rabbits (estimate= -0.013 (SE = 0.017), t= -0.789, p = 0.447) or small rodents (estimate= -0.053 (SE = 0.034), t= -1.548, p = 0.1561). Selective foraging of different prey types was not related to dingo density (Table 1).
Frequency dependent selection of foxes (left) and dingoes (right) for different prey types. Generally, both predators tended to exhibit negative frequency dependent selection, i.e., as a prey type becomes more abundant in the environment (x-axis) the predator’s preference for that prey type decreases (y-axis). Asterisk’s (*) indicate prey types for which the trend of negative frequency dependent selection approached the 5% level of statistical significance
Fox foraging behaviours
Long-haired rats were omitted from the dietary analyses for foxes due to the relative scarcity this prey type in the predators’ diet and environment (Fig. 2). Kangaroos were the most abundant taxa inside the dingo fence (where dingoes were rare and foxes were common) (Figs. 2 and 7), while hopping mice made up the majority of the foxes’ diet (Fig. 2). Rabbits, reptiles, kangaroos and small rodents made up between 17 and 25% of the fox’s diet, while long-haired rats made up < 5% of the foxes’ diet (Fig. 2). Foxes tended to avoid kangaroos, reptiles and small rodents, show a neutral preference for hopping mice, and a preference for rabbits (Fig. 6).
Fox preference for different prey types using the logit transformed values of the Manly Chesson selection index (αi). Dashed, red horizonal line (at -0.6 on the y-axis) indicates that the fox is feeding randomly, αi > -0.6 indicates a preference for the prey and αi < -0.6 indicates avoidance. Vertical lines indicate 25th, 50th and 75th percentiles, blue ‘x’ represent mean values
Temporal changes of prey taxa abundance in the environment and occurrence in fox diet. Dashed, vertical grey lines indicate sampling periods where rainfall was greater than the previous year. Note, for illustrative purposes x-axis ticks are not proportionately scaled to the length of time between observations
Across all prey types, foxes tended to exhibit negative frequency dependent selection (i.e., foxes’ preference for its prey was generally negatively associated with the abundance of that prey type in the environment) (Table 2; Fig. 5). This trend was primarily driven by foxes’ negative frequency dependent selection for hopping mice (estimate =-0.037(SE = 0.009), t= -3.679 p = 0.014 *) and small rodents (estimate =-0.044 (SE = 0.019), t= -2.327, p = 0.067) (Fig. 5). Foxes showed no frequency dependent selection for kangaroos (estimate= -0.009 (SE = 0.015), t= -0.583, p = 0.573), reptiles (estimate= -0.016 (SE = 0.008), t= -1.743, p = 0.120) or rabbits (estimate=-0.004 (SE = 0.013), t= -0.329, p = 0.749). Selective foraging of different prey types was generally not related to fox density (Table 2).
Discussion
In our study, both dingoes and foxes tended to exhibit negative frequency dependent selection for prey, such that the strength of each predator’s selection for a prey type tended to decrease with that prey’s abundance in the environment. This trend was primarily driven by negative frequency dependent selection for a vulnerable species; the hopping mouse; (Letnic and Dworjanyn 2011), by both predators. Importantly, however, only foxes persisted to preferentially select hopping mice when they were scarce in the environment. Dingoes’ consumption of hopping mice was not ‘preferential’ (i.e., dingoes generally avoided hopping mice) and occurred exclusively when hopping mice were frequent in the environment (Fig. 5). Hopping mice occur at much lower densities inside the fence (where foxes are abundant and dingoes are rare), and occur within a clade of rodents that has undergone extensive declines since the introduction of foxes and cats (Letnic et al. 2012). Given that foxes continue to consume hopping mice at very low densities, our findings may suggest that hopping mice are particularly vulnerable to predation by foxes, and may be ‘easy prey’ for this predator (Smith and Quin 1996; Letnic and Dworjanyn 2011). A potential explanation for this susceptibility is that these rodents are highly vulnerable to predation by relatively recently introduced predators, such as foxes, because they are naïve (they have not co-evolved with these predators), and lack adaptions to avoid or escape them (Moseby et al. 2016; Carthey and Banks 2014).
Dingoes did exhibit negative frequency dependent selection for long-haired rats and reptiles when they were scarce, and foxes also exhibited negative frequency dependent selection for small rodents when they were scarce. It is beyond the scope of our analyses to determine why selection for many prey types tended to be negatively frequency dependent. However, one possibility is that it is beneficial for predators to continue searching for the more vulnerable and/or preferred prey, even when they are relatively rare, rather than to risk attacking the more abundant prey that may be more difficult to find, catch or consume. Alternatively, both predators may have developed a ‘search image’ allowing them to become increasingly efficient at hunting particular prey, even when those prey become scarce, potentially representing the development of type-2 functional responses. The relative frequency of prey types in the environment may also influence prey behaviour and/or condition, and thus, where and how predators search for prey. Consequently, observances of negative frequency dependent selection in our study may alternatively/additionally suggest that at lower densities some prey types are easier to catch. Selective foraging by dingoes and foxes was not related to predator density in our study indicating that predators did not become less selective when intraspecific competition for food increased. However, this interpretation depends on whether predator abundances passed a threshold that would lead to competitive interactions, which we did not assess.
Before the potential ecosystem impacts of negative frequency dependent foraging are discussed, it may first be valuable to differentiate relative predation pressures on populous invasive and/or pest species versus vulnerable and/or native species. The Mammalia species composition of arid-zone Australia is largely dominated by invasive species (e.g., rabbits), overabundant macropods (e.g., kangaroos) and relict assemblages of other native species (Letnic & Dickman 2006), many of which are threatened, such as hopping mice and other ‘small rodents’ (e.g., fawn hopping-mice (Notomys cervinus) and plains mice (Pseudomys australis) and others which are of less concern (e.g., long-haired rats) (Radford et al. 2018). Although both predators tended to exhibit negative frequency dependent prey selection in our study, the potentially destabilizing impacts of these behaviours by foxes may constitute a greater conservation concern than those imposed by dingoes, as foxes seem to be more likely to add pressure to threatened populations (hopping mice and small rodents). That said, our study, and others (Paltridge et al., 2002; Doherty et al., 2019), do indicate that dingoes are likely to exhibit selective preferences for reptiles, particularly during periods of dry climatic conditions. By showing that dingoes exhibit negative dependent frequency selection for reptiles in our sample, we highlight a need for future work on the potentially detrimental effects of dingo predation on reptile populations.
The most abundant prey type inside the dingo fence was kangaroos, an overabundant taxon which may contribute to the suppression of other taxa through overgrazing (Fisher et al. 2021)). Yet, foxes tended to avoid kangaroos (likely because adult kangaroos are too big to handle), thus are unlikely to control this populous macropod at our study sites. That said, foxes do kill juvenile kangaroos and have been shown to limit grey kangaroo (Macropus giganteus) populations in Namadgi National Park, Australia; a habitat primarily comprised of sub-alpine bushland (Banks et al., 2000). It is unclear why similar limitations were not observed in our study; however, this may be related to differences in primary productivity fluctuations between arid and more temperate habitats. While we were unable to calculate a Manly Chesson index for selection of kangaroos by dingoes (due to their relatively scarcity in the dingo’s environment), kangaroos were found in a considerably higher proportion of dingo scats, possibly suggesting that this prey are highly selected for by dingoes, and may explain why kangaroos are so rare outside the fence. Additionally, foxes and dingoes both showed a preference for introduced rabbits, highlighting the value of both predators in the regulation of this ‘pest’ prey species in the Australian desert (Newsome et al. 1989; Pech et al. 1992; Banks 2000). All in all, whether the patterns of frequency dependent selection in our study are ecologically relevant for predicting predator prey dynamics does require further work, e.g., insight into kill rates and predation rates (the proportion of the prey killed) for each predator (Vucetich et al. 2011) and/or computer simulations of the resiliency of the prey population to different foraging strategies (such as those ran in Hoy et al., 2021)).
Australia has suffered the greatest loss of native mammal species globally (Loehle and Eschenbach 2012); 10% of 271 terrestrial species endemic to Australia have become extinct in the last 200 years, and a further 21% are considered threatened (Woinarski et al. 2015; Fleming and Bateman, 2016). Our findings suggest a tendency for both foxes and dingoes to exhibit negative frequency dependent selection. However, because foxes seem to direct such behaviours towards a vulnerable, native species (hopping mice), our results may suggest that this predator has a more destabilizing effect on threatened prey communities than dingoes. This view is bolstered by the observation that foxes do not control kangaroos, whereas dingoes are likely to, and hopping mice are common on the dingo side of the fence. It is important to note that many other forces unaccounted for in our study may counterbalance these (de)stabilizing effects, including functional response dynamics, emigration/immigration, and the composition of invasive/native/economically valuable flora and fauna etc. Consequently, the integration of our results into the suite of processes that exert stabilizing or destabilizing influences on prey populations is key to understand the true ecosystem impacts of each predator.
Data Availability
The datasets generated during and analyzed during the current study are available from the corresponding author on request.
References
Allen BL, Fleming PJS (2012) Reintroducing the dingo: the risk of dingo predation to threatened vertebrates of western New South Wales. Wildl Res 39:35–50
Allen BL, Fleming PJS, Allen LR, Engeman RM, Ballard GA, Leung LKP (2013) As clear as mud: a critical review of evidence for the ecological roles of australian dingoes. Biol Conserv 159:158–174
Armitage P, Berry G Statistical Methods in Medical Research (3rd edition). Blackwell 1994
Banks PB (2000) Can foxes regulate rabbit populations?The Journal of Wildlife Management:401–406
Banks PB, Newsome AE, Dickman CR (2000) Predation by red foxes limits recruitment in populations of eastern grey kangaroos. Austral Ecol 25:283–291. https://doi.org/10.1046/j.1442-9993.2000.01039.x
Barnard CJ, Brown CAJ (1981) Prey size selection and competition in the common shrew (Sorex araneus L.). Behav Ecol Sociobiol 8:239–243. https://doi.org/10.2307/4599387
Brook LA, Kutt AS (2011) The diet of the dingo (Canis lupus dingo) in north-eastern Australia with comments on its conservation implications. Rangel J 33:79–85
Carthey AJ, Banks PB (2014) Naïveté in novel ecological interactions: lessons from theory and experimental evidence. Biol Rev 89(4):932–949
Chesson J (1978) Measuring preference in selective predation. Ecology 59(2):211–215. https://doi.org/10.2307/1936364
Cooke BD, Soriguer RC (2017) Do dingoes protect Australia’s small mammal fauna from introduced mesopredators? Time to consider history and recent events. Food Webs 12:95–106. https://doi.org/10.1016/j.fooweb.2016.04.002
Corbett LK, Newsome AE (1987) The feeding ecology of the dingo: III. Dietary relationships with widely fluctuating prey populations in arid Australia: an hypothesis of alternation of predation. Oecologia 74(2):215–227. https://doi.org/10.1007/BF00379362
Cupples JB, Crowther MS, Story GL, Letnic M (2011) Dietary overlap and prey selectivity among sympatric carnivores: could dingoes suppress foxes through competition for prey? J Mammal 92:590–600
Cuthbert RN, Dickey JWE, McMorrow C, Laverty C, Dick JTA (2018) Resistance is futile: lack of predator switching and a preference for native prey predict the success of an invasive prey species. Royal Society of Open Science. https://doi.org/10.1098/rsos.180339
Doherty TS, Davis NE, Dickman CR, Forsyth DM, Letnic M, Nimmo DG, Palmer R, Ritchie EG, Benshemesh J, Edwards G, Lawrence J, Lumsden L, Pascoe C, Sharp A, Stokeld D, Myers C, Story G, Story P, Triggs B, Venosta M, Wysong M, Newsome TM (2019) Continental patterns in the diet of a top predator: Australia’s dingo. Mammal Rev 49:31–44. https://doi.org/10.1111/mam.12139
Fisher AG, Mills CH, Lyons M, Cornwell WK, Letnic M (2021) Remote sensing of trophic cascades: multi-temporal landsat imagery reveals vegetation change driven by the removal of an apex predator. Landscape Ecol 36:1341–1358. https://doi.org/10.1007/s10980-021-01206-w
Fleming P, Corbett L, Harden R, Thomson P (2001) Managing the impacts of Dingoes and other Wild Dogs. Bureau of Rural Sciences: Canberra, ACT, Australia)
Fleming PA, Bateman PW (2016) The good, the bad, and the ugly: which australian terrestrial mammal species attract most research. Mammal Rev 46(4):241–254
Gordon CE, Eldridge DJ, Ripple WJ, Crowther MS, Moore BD, Letnic M (2017) Shrub encroachment is linked to extirpation of an apex predator. J Anim Ecol 86(1):147–157
Holt RD, Lawton JH (1994) The ecological Consequences of Shared Natural Enemies. Annu Rev Ecol Syst 25:495–520
Hoy SR, MacNulty DR, Metz MC, Smith DW, Stahler DR, Peterson RO, Vucetich JA (2021) Negative frequency-dependent prey selection by wolves and its implications on predator–prey dynamics. Anim Behav 179:247–265. https://doi.org/10.1016/j.anbehav.2021.06.025
Letnic M, Dickman CR (2006) Boom means bust: interactions between the El Niño/Southern Oscillation (ENSO), rainfall and the processes threatening mammal species in arid Australia. Biodivers Conservatopm 15:3847–3880
Letnic M, Dworjanyn SA (2011) Does a top predator reduce the predatory impact of an invasive mesopredator on an endangered rodent? Ecography 34(5):827–835
Letnic M, Greenville A, Denny E, Dickman CR, Tischler M, Gordon C, Koch F (2011) Does a top predator suppress the abundance of an invasive mesopredator at a continental scale? Glob Ecol Biogeogr 20(2):343–353
Letnic M, Koch F (2010) Are dingoes a trophic regulator in arid Australia? A comparison of mammal communities on either side of the dingo fence. Austral Ecol 35:167–175. https://doi.org/10.1111/j.1442-9993.2009.02022.x
Letnic M, Ritchie EG, Dickman CR (2012) Top predators as biodiversity regulators: the dingo Canis lupus dingo as a case study. Biol Rev 87:390–413
Loehle C, Eschenbach W (2012) Historical bird and terrestrial mammal extinction rates and causes. Divers Distrib 18:84–91. https://doi.org/10.1111/j.1472-4642.2011.00856.x
Manly BFJ (1974) A model for certain types of selection experiments. Biometrics 36(1):9–18
McKenzie NL, Burbidge AA, Baynes A, Brereton RN, Dickman CR, Gordon G, Gibson LA, Menkhorst PW, Robinson AC, Williams MR, Woinarski JCZ (2007) Analysis of factors implicated in the recent decline of Australia’s mammal fauna. J Biogeogr 34:597–611. https://doi.org/10.1111/j.1365-2699.2006.01639.x
McKnight TL (1969) Barrier fencing for vermin control in Australia. Geogr Rev 59:330–347
Mills CH, Waudby H, Finlayson G, Parker D, Cameron M, Letnic M (2020) Grazing by over-abundant native herbivores jeopardizes conservation goals in semi-arid reserves. Global Ecol Conserv 24. https://doi.org/10.1016/j.gecco.2020.e01384
Mills CH, Wijas B, Gordon CE, Lyons M, Feit A, Wilkinson A, Letnic M (2021) Two alternate states: shrub, bird and mammal assemblages differ on either side of the Dingo Barrier Fence. Australian Zoologist 41(3):534–549
Morris T, Letnic M (2017) Removal of an apex predator initiates a trophic cascade that extends from herbivores to vegetation and the soil nutrient pool. Proceedings of the Royal Society B: Biological Sciences 284(1854): 20170111
Moseby KE, Blumstein DT, Letnic M (2016) Harnessing natural selection to tackle the problem of prey naïveté. Evol Appl 9(2):334–343
Moseby KE, Neilly H, Read JL, Crisp HA (2012) Interactions between a top order predator and exotic mesopredators in the australian rangelands. Int J Ecol. https://doi.org/10.1155/2012/250352
Murdoch WW (1969) Switching in General Predators: experiments on Predator specificity and Stability of Prey populations. Ecol Monogr 39(4):335–354
Newsome AE, Parer I, Catling PC (1989) Prolonged prey suppression by carnivores—predator-removal experiments. Oecologia 78(4):458–467
O’Donoghue M, Boutin S, Krebs CJ, Zuleta G, Murray DL, Hofer EJ (1998) Functional responses of coyotes and lynx to the snowshoe hare cycle. Ecology 79:1193–1208. https://doi.org/10.2307/176736
Oaten A, Murdoch WW (1975) Switching, functional response, and stability in predator prey systems. Am Nat 109(967):299–318. https://doi.org/10.1086/282999
Paltridge Rachel (2002) The diets of cats, foxes and dingoes in relation to prey availability in the Tanami Desert, Northern Territory. Wildl Res 29:389–403
Pavey CR, Eldridge SR, Heywood M (2008) Population Dynamics and Prey Selection of native and Introduced Predators during a Rodent Outbreak in Arid Australia. J Mammalogy Volume 89 3:674–683. https://doi.org/10.1644/07-MAMM-A-168R.1
Pech RP, Sinclair ARE, Newsome AE, Catling PC (1992) Limits to predator regulation of rabbits in Australia: evidence from predator-removal experiments. Oecologia 89(1):102–112
Philip J (2021) A historical review of australian aerial vertebrate pest control, targeting dingoes and wild dogs 1946–2019. Australian Zoologist 41(3):580–592. https://doi.org/10.7882/AZ.2020.011
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/
Radford JQ, Woinarski JCZ, Legge S, Baseler M, Bentley J, Burbidge AA, Bode M, Copley P, Dexter N, Dickman CR, Gillespie G, Hill B, Johnson CN, Kanowski J, Latch P, Letnic M, Manning A, Menkhorst P, Mitchell N, Morris K, Moseby K, Page M, Ringma J (2018) Degrees of population-level susceptibility of australian terrestrial non-volant mammal species to predation by the introduced red fox (Vulpes vulpes) and feral cat (Felis catus). Wildl Res 45:645–657
Savolainen P, Leitner T, Wilton AN, Matisoo-Smith E, Lundeberg J (2004) A detailed picture of the origin of the australian dingo, obtained from the study of mitochondrial DNA. Proc Natl Acad Sci USA 101:12387–12390
Smith AP, Quin DG (1996) Patterns and causes of extinction and decline in australian conilurine rodents.“ Biological conservation. 77:243–2673
Spencer EE, Newsome TM, Dickman CR (2017) Prey selection and dietary flexibility of three species of mammalian predator during an irruption of non-cyclic prey. Royal Open Society sci.4170317170317
Vernes K, Dennis A, Winter J (2001) Mammalian Diet and Broad Hunting Strategy of the Dingo (Canis familiaris dingo) in the Wet Tropical rain forests of northeastern Australia. Biotropica 33(2):339–345
Vucetich JA, Hebblewhite M, Smith DW, Peterson RO (2011) Predicting prey population dynamics from kill rate, predation rate and predator–prey ratios in three wolf-ungulate systems. J Anim Ecol 80:1236–1245. https://doi.org/10.1111/j.1365-2656.2011.01855.x
Wijas B, Letnic M (2021) Top-down effects have primacy over bottom-up effects on the population dynamics of a flightless desert bird. J Arid Environ 195:104611
Woinarski JCZ, Burbidge AA, Harrison PL (2015) A review of the conservation status of australian mammals. Therya 6(1):155–166
Acknowledgements
Christopher Gordon, Freya Gordon, James Rees and Charlotte Mills assisted with data collection. Thanks to landholders, particularly the Ogilvy family who provided access to the study areas. Scat analysis was conducted by Georgeanna Story. Matthew Broderick helped with manuscript formatting.
Funding
Funding was provided by the Australian Research Council (Grant No. DP180101477).
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Contributions
Elizabeth L Sheldon and Mike Lentic contributed to the study conception and design. Field work and data collection was performed by all authors. Data analysis was performed by Elizabeth L Sheldon. The first draft of the manuscript was written by Elizabeth L Sheldon. Elizabeth L Sheldon, Mike Lentic and Benjamin Feit reviewed and edited previous versions of the manuscript and all authors approved the final version.
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no relevant financial or non-financial conflicts of interest to declare.
Ethics approval
This research was carried out under UNSW ethics approval 12/87A.
Additional information
Communicated by Stephen Garnett.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sheldon, E.L., Feit, B., Feit, A. et al. Negative frequency dependent prey selection by two canid predators and its implications for the conservation of a threatened rodent in arid Australia. Biodivers Conserv 32, 1671–1686 (2023). https://doi.org/10.1007/s10531-023-02570-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-023-02570-6