Abstract
Maximizing biodiversity persistence in heterogeneous human-modified landscapes is hindered by the complex interactions between habitat quality and configuration of native and non-native habitats. Here we examined these complex interactions considering avian diversity across 26 sampling sites, each of which comprised of three sampling points located across a gradient of disturbance: core native habitat fragment, fragment edge, and non-native adjacent matrix. The 78 sampling points were further nested within three neotropical biomes—Amazonia, Cerrado and Pantanal—in central-western Brazil. Matrix type consisted of cattle pastures in the Amazon and teak plantations in the Pantanal and Cerrado. We considered the interactive effects of (1) disturbance-context: fragment core, edge and adjacent matrix, (2) matrix type: tree plantation or cattle pastures, both subject to varying land-use intensity, and (3) native habitat configuration (fragment size, shape and isolation) on bird species richness, abundance and composition. Based on point-count surveys, we recorded 210 bird species. Bird species richness and abundance declined across the disturbance gradient, while genus composition only differed within the adjacent matrix, particularly cattle-pastures. The effect of native habitat area was positive but only detected at fragment edges. Overall bird diversity increased at sites characterized by higher availability of either relict trees within pasture landscapes or old-growth trees within teak plantation landscapes. The core of native fragments played a primary role in ensuring the persistence of bird diversity, regardless of fragment size. In contrast to pastures, tree plantations likely harbour a higher proportion of forest-dependent species while bird diversity can be further enhanced by reduced management intensity in both matrix types. Strategies to maximize avian persistence should not only include retaining native habitats, but also maximizing the size of core native habitats. Likewise, more structurally complex matrix types should be encouraged while maintaining low levels of land-use intensity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The rapid expansion of anthropogenic activities into once remote forest areas has led to unprecedented levels of biodiversity loss worldwide (Gibson et al. 2011; Newbold et al. 2016). Currently, roughly half of the global land surface is occupied by human-modified landscapes, consisting of heterogeneous mosaics of native and non-native land-cover of different sizes, degrees of isolation and under varying intensities of land use (Hansen et al. 2020). Maximizing biodiversity persistence while maintaining agricultural productivity is a major 21st-century challenge (Fahrig et al. 2011), and more empirical tests are required to understand the conservation value of these landscapes (Arroyo-Rodríguez et al. 2020).
Species diversity in human-modified landscapes is expected to be primarily affected by habitat quality, which is generally higher in natural than in anthropogenic habitats (e.g. cattle pastures and fast-growing tree plantations) (Barlow et al. 2007; Gibson et al. 2011; Moura et al. 2013). Within native habitat remnants of once continuous expanses of primary habitat, species diversity is expected to be reduced by both smaller habitat areas, which constrain population sizes and limit habitat diversity, and isolation from other native habitats (Stouffer and Bierregaard Jr. 1995), which further affects species colonization rates (MacArthur and Wilson 1967). Moreover, edge effects tend to modify the vegetation structure, particularly at more irregularly-shaped fragments, often depressing the availability of trophic and structural resources (Laurance et al. 1998), which negatively affects biodiversity (Laurance 2004; Harrison and Banks-Leite 2019). Within non-native matrix habitats, species diversity is further affected by their quality as suggested by the structural fragment-matrix contrast (Driscoll et al. 2013). The higher the structural similarity between native and non-native habitats, the lower that contrast and the higher the chance of safeguarding functional connectivity (Prevedello and Vieira 2010) and species persistence across the landscape (Pfeifer et al. 2017). In landscapes dominated by terrestrial matrix areas, matrix quality is particularly important (Wiens 1995), enabling some species to not only move between fragments (Biz et al. 2017; Godoi et al. 2018) but also take advantage of matrix resources, boosting the overall biodiversity sustained within human-modified landscapes (Gascon et al. 1999; Daily et al. 2001; Mendenhall et al. 2014, but see Mahood et al. 2012). Furthermore, depending on their characteristics, the nearby matrix has the potential to affect species diversity within native remnants (Hatfield et al. 2020), and vice-versa (Tubelis et al. 2004). For instance, the higher the matrix quality, the higher the species diversity at nearby native fragments (Antongiovanni and Metzger 2005; Stouffer et al. 2006). Finally, the set of species persisting in such landscapes further depends on their intrinsic ecological traits (Matuoka et al. 2020; Newbold et al. 2014), with forest specialist species rapidly vanishing from non-native habitats and small, structurally degraded native remnants (Lees and Peres 2008; Morante-Filho et al. 2016).
In the tropics, the rapid expansion of human-modified landscapes occurred mostly at the expense of native habitats (Hansen et al. 2013). In Brazil alone, ~ 29.8 million hectares of natural vegetation were lost during the last 16 years, much of which was replaced by exotic cattle pastures (IBGE 2020). This matrix type, however, is characterized by high structural contrast with remaining forest fragments, thereby likely representing an overall hostile environment (Stouffer and Bierregaard Jr. 1995). Meanwhile, fast-growing tree plantations are becoming attractive options for landowners, given their benefits in terms of both economic and carbon sink prospects (Nölte et al. 2018). In Brazil, the forestry industry has been particularly targeting previously degraded areas. Tree plantations have expanded by over 8 million ha since 1995, 93,957 ha (0.01%) of which is occupied by teak monoculture (Tectona grandis) (IBGE 2020). Plantations of this Asian timber species are rapidly expanding across tropical regions, including Central Africa and Central and South America (Tewari 1992). Given the overall higher structural complexity of tree plantations, rural landscapes dominated by a tree plantation matrix could then be expected to sustain higher levels of biodiversity (e.g. Barbosa et al. 2017; Biz et al. 2017; Hatfield et al. 2020, but see Barros et al. 2019 and Lees et al. 2015).
Here we examine complex interactions between the quality and configuration of native and non-native habitats considering bird species diversity across human-modified landscapes in Brazil, which harbours one of Earth’s richest avifauna (Marini and Garcia 2005). To do so, we surveyed 26 sampling sites, each of which comprised of three sampling plots located along a marked gradient of disturbance: core native habitat fragment, fragment edge, and adjacent matrix. Sampling sites were further nested within three adjacent biomes—Amazonia, Cerrado and the Pantanal wetlands—in the state of Mato Grosso, where ~ 5 million ha of forest cover has been lost during the last two decades (IBGE 2020), plus a further ~ 348,100 ha of deforestation over the 2019–2020 period (INPE 2021). We investigated the interactive effects of the (1) disturbance-context: fragment core, edge and adjacent matrix, (2) matrix type: teak plantations or cattle pastures, both subject to varying land-use intensity, and (3) native habitat configuration: fragment size, shape and isolation, on bird species richness, abundance and composition. Along the disturbance gradient, we hypothesized that bird diversity consistently declines from core fragment areas to the adjacent matrix. Regarding matrix type, we hypothesized that bird diversity was positively affected by the structural complexity of matrix areas, so that higher species richness and abundance were expected at landscapes comprised of tree plantations rather than pastures, further diverging in species composition. Within each of the disturbance-contexts surveyed (i.e. fragment core, edge and adjacent matrix), we expected bird assemblages to be further affected by native remnant configuration (fragment size, shape and isolation) and the intensity of matrix land use (number of relict trees within pastures, and height and age of teak plantations).
Methods
Study area
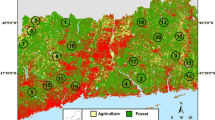
This study was carried out in the highly biodiverse state of Mato Grosso, the only sub-national unit in South America hosting Amazonian rainforests (53%), woody Cerrado scrublands (40%), and the Pantanal wetlands (7%) (IBGE 2020). We surveyed 26 sampling sites, including 10 Amazonian, six Cerrado, and 10 Pantanal sites (Fig. 1; see Table S1 in the Supplementary Material). Within each of the 26 sampling sites, we surveyed three plots: one in the fragment core, one in the fragment edge and another at the adjacent matrix area, amounting to a total of 78 surveyed sites. Anthropogenic matrix areas dating back to the 1960s are represented by cattle pastures converted from native forests, which were further converted into teak plantations in both Cerrado and Pantanal biomes. In the Amazon, cattle pastures were characterized by a variable number of relict trees (71.3 \(\pm\) 55.6 trees per plot, 6–190 trees). These hardwood trees were often left within pastures to provide shade for bovine cattle. In Cerrado and Pantanal vegetation, teak monoculture varied in terms of maturity (plantation age, 14.19 \(\pm\) 3.49 years, 10–19 years) and height (17.6 \(\pm\) 2.2 m, 13.3–22.6 m). The overall region is characterized by a tropical climate with marked wet (October to April) and dry seasons (May to September) (Golfari et al. 1978). Mean annual temperature (\(\pm\) SD) is 20 \(\pm\) 7ºC, and precipitation ranges between 1000 and 1500 mm/year (Koopen 1948). Surveys were carried out between July 2013 and March 2014, corresponding to the dry season in the Cerrado and the rainy season in the Pantanal, and between February and July 2016 in the Amazon, corresponding to both seasons. Given that 98% of all species recorded in this study are year-round residents (Somenzari et al. 2018), we do not expect our bird surveys to have been affected by the seasonal influx of migrants. Despite structural differences between adjacent biomes, all localities surveyed share similar animal and plant biotas (Marinho-Filho et al. 1994; Passos et al. 2018).
Geographic setting of sites surveyed in this study (red dots) spanning across the a Amazon (dark green), b Cerrado (light green) and c Pantanal (light orange) biomes in Mato Grosso, Brazil (upper left), showing the location of the three study regions. In the inset maps a–c, remaining native and non-native habitats are shown in green and yellow, respectively; the three different shades of green correspond to different native habitats available (from darker to lighter green: closed-canopy forest, savanna and grasslands, respectively). Surveyed sites are numbered according to Table S1
Avian surveys
Birds were surveyed using point-counts and mist-nets, both carried out during periods of very high bird activity (06:00–10:00 h) over three days by an experienced observer (OMAN) and a field assistant. Point-counts consisted of visual (8 × 42 binoculars) and acoustic detection of birds, spanning a 50-m radius, during 15-min periods. During each field campaign, we surveyed three sampling sites, except for two campaign encompassing four sites: (1) Sites 12, 13, 14 and 20, and (2) Sites 1, 3, 5 and 6. Sampling sites surveyed within the same field campaign were located closer to each other and were further surveyed every three days, so that the first site surveyed was additionally surveyed during days 4 and 7 of that campaign. Auditory detection was assisted by recordings obtained during point count surveys, using a Panasonic® RR-US551 recorder and a semi-directional microphone. These recordings allowed us to identify previously unrecognized bird records, based on available personal databases. Bird identification followed the information available in Sick (1997), Mata and Erize (2006), Ridgely and Tudor (2009) and Piacentini et al. (2015). We additionally checked for any taxonomic updates using the Handbook of the Birds of the World and BirdLife International (2018). Mist-netting was also carried out simultaneously with point-count surveys, but not within the smallest habitat fragments, due to space restrictions, nor within pasture-matrix areas, due to the frequent presence of cattle. In addition, bird detections obtained from mist-nets amounted for only 6% (N = 303) of all records and 30% (N = 76) of all species recorded. For these reasons, data obtained using mist-netting was insufficient to characterize bird diversity within each sampling site. Given the unfeasibility of using mist-netting data in separate analyses, data in this study are restricted to point-count surveys (for additional details on mist-netting data, see Table S3).
Within each sampling site, sampling plots were located in (1) core areas of fragments, between 75 and 400 m from the nearest edge according to fragment size, (2) peripheral fragment areas 50 m from the edge, and (3) adjacent matrix areas, 50 m away from the edge (Fig. S1). At core and edge plots, sampling consisted of one point-count, except for very small or irregularly shaped fragments. In those cases (Sites 14, 15, 19 and 26), where the fragment interior was never farther than 150 m from the edge, we conducted only one point-count at ‘core’ plots. However, point-counts obtained in those fragments could be distinguished into records at either the core or the edge of native fragments.
Configuration and land-use metrics
Patch and landscape configuration metrics characterized the native fragments, whereas land-use intensity characterized the adjacent non-native matrix area. As configuration metrics, we considered the fragment size, isolation and shape, and as a land-use intensity metric, the matrix complexity. Fragment size was defined as the total area of the native habitat fragment; isolation as the linear distance from the fragment edge to the nearest neighboring native fragment; and shape as the Shape Index (SI) given by the equation: [\(SI=\frac{P}{200\left[{\left(\pi A\right)}^{0.5}\right]}\)], where P is the fragment perimeter (m), and A is the fragment area (ha). In a perfect circle SI = 1.0, whereas more irregular shapes have higher values (Laurance and Yensen 1991). In sampling sites comprised of cattle pastures, matrix complexity was characterized by the number of relict trees counted within a 400-m circular buffer surrounding each focal fragment. These variables were extracted using the satellite imagery available on the Google Earth platform for the corresponding sampling years. In sampling sites comprised of teak monoculture, matrix complexity was defined in terms of plantation age and tree height, which were provided by Foresteca S/A, the company managing those plantation landholdings.
Data analysis
Bird responses across the disturbance gradient
Across the 78 sampling plots nested within 26 sites and three biomes, we first evaluated the effects of (1) disturbance-context (fragment core, edge and adjacent matrix), (2) matrix contrast and (3) native habitat area (i.e. fragment size) on bird species richness, abundance and composition. Species composition across all biomes was examined at the genus-level and therefore referred to as ‘genus composition’. This allowed us to minimize differences in species composition between biomes due to closely-related congeners and maximise differences between habitats of varying degrees of human disturbance. Genus composition was analysed using nonmetric multidimensional scaling (NMDS) ordination based on the quantitative Bray–Curtis similarity matrix of genus composition using the vegan R package (Oksanen et al. 2020). The first two NMDS axes allowed us to preserve the original dissimilarities under a reduced number of dimensions (stress = 0.233), and derive scores used in further analyses. Considering species richness and abundance as response variables, we applied Generalized Linear Mixed Models (GLMMs) with a Poisson distribution. For genus composition (i.e., the first and second NMDS axes), we applied Linear Mixed Models (LMMs) with a Gaussian distribution. To account for natural variability in ecological metrics between study regions, we considered sampling plots to be nested within sites (N = 26) and biome (Amazonia: N = 10 landscapes, Cerrado: N = 6, and Pantanal: N = 10), included in the models as random factors. As explanatory variables, we considered the categorical disturbance-context (fragment core, edge and matrix), the binary matrix contrast (1 = silviculture, 2 = pasture) and the native habitat area, corresponding to the size of native fragments. The effects of either fragment size or matrix contrast on bird diversity likely depend on disturbance-context. For example, matrix type and fragment area effects may be more evident within the matrix or within the fragment, respectively. Therefore, we added two interaction terms, one between disturbance-context and matrix contrast and another between disturbance-context and fragment size. We additionally evaluated effects of vegetation biome by repeating the modelling but including biome as a fixed term. Because these models did not support additional categorical variables for our number of plots (N = 76), matrix contrast was considered as a random effect. For each categorical variable, results are interpreted in relation to the category held in the model intercept. GLMMs and LMMs were performed using the lme4 R package (Bates et al. 2012). Additional fragmentation metrics, such as fragment shape and isolation, were not considered here to minimize model complexity, thereby maximizing model performance.
Bird responses within each disturbance-context
We then analysed predictors of avian assemblage structure (species richness, abundance and genus composition) for each disturbance-context—fragment core, edge and adjacent matrix—further considering cattle pasture and teak plantation sites also separately. Therefore, we ensured further exploration of intrinsic matrix features (i.e., number of relict trees in cattle pastures and age and height of teak plantations). Pasture sites were analyzed using Generalized Linear Models (GLM) to explain bird species richness and abundance, using a Poisson distribution, Linear Models (LMs) to explain genus composition (NMDS1) using a Gaussian distribution. As explanatory variables, we considered native fragment area (log10 x), shape (log10 x), degree of fragment isolation (log10 x), and number of relict trees in the nearby matrix. Teak plantation sites were similarly analysed with GLMs and a LM including the same explanatory variables, except for matrix complexity, which were here defined as the age and height of teak plantations. In addition, since teak plantation landscapes were distributed in both Pantanal and Cerrado biomes, biome was additionally considered as an explanatory variable. Prior to analysis, we examined a Pearson correlation matrix to control for high levels of explanatory variable inter-dependence, but no variables were highly correlated (r > 0.75). All data analyses were performed in R (R Development Core Team 2015).
Results
Based on point-count surveys, we obtained 5805 detections of 210 species, 60 (28.6%) of which were recorded either only once or twice throughout the sampling. The most prevalent recorded species were the psittacids Brotogeris chirini (7.5% of all records) and Diopsittaca nobilis (4.9%), detected across all three biomes, and the gregarious columbid Zenaida auriculata (6.4%) recorded in both core and edge sites of only one Pantanal fragment. We obtained 2760 overall records of 158 species in core plots, but this declined to 1730 records (151 species) at edges and 1315 records (119 species) at matrix plots. From those species, 13.3% (N = 28) were restricted to fragment cores, 10.0% (N = 21) to edges, and 11.0% (N = 23) to matrix plots. Yet 46.0% (N = 97) of all species were recorded both within fragments (core or edge) and matrix plots (Table S2).
Bird responses across the disturbance gradient
On average (\(\pm\) SD), 26.5 \(\pm\) 7.2 species were recorded within core fragment areas, 22.4 \(\pm\) 11.2 species along the edge, and 15.5 \(\pm\) 6.3 species in the adjacent matrix (Fig. 2). As such, when bird diversity within core fragment areas was relativized to zero (x = 0), species richness and abundance became increasingly negative across edges and matrix plots (Fig. 3a–b). However, the main changes were related to confidence intervals, particularly in terms of abundance (Fig. 3b). In terms of genus composition, there were marked differences both across sites comprised of different matrix types and across the disturbance gradient. First, genus-level composition in teak plantation sites partially overlapped those consisting of cattle pastures (Fig. 3c). Second, genus composition was similar between fragment core and edge plots, most evidently at teak plantation sites, but there was little overlap with matrix plots, particularly for pasture sites (Fig. 3c–d; an ordination plot representing the genera in Fig. S2).
Relative changes in the mean \(\pm\) SD a bird species richness and b abundance recorded within the different disturbance-contexts surveyed: fragment core, edge and adjacent matrix. This corresponded to 78 plots nested within 26 sites spanning across the Amazon, Cerrado and Pantanal biomes in Mato Grosso, Brazil. c non-metric multidimensional scaling (NMDS) ordination plot representing each sampling plot performed at the genus-level; different colours denote the different disturbance-contexts (fragment core in green, edge in orange, and matrix in yellow) and different symbols denote the different matrix types surrounding habitat fragments (cattle pasture = circles; teak plantation = triangles). d Relative changes in the mean \(\pm\) SD genus composition (NMDS1) within the different disturbance-contexts surveyed. Relative species richness, abundance and genes composition values were obtained by setting the mean values of fragment core plots to zero
Taking into account the potential interaction between disturbance-context and either native fragment area or matrix type, species richness at core plots was higher than both at the edge (\(\beta\)edge = − 1.105, P < 0.0001) or matrix plots (\(\beta\)matrix = − 0.373, P = 0.077) (Table S3). The same was observed for species abundance, which was also lower at edges (\(\beta\)edge = − 1.670, P < 0.0001) and matrix plots (\(\beta\)matrix = − 1.58, P < 0.0001) (Table S3). In terms of genus composition, matrix sites were considerably different (NMDS2: \(\beta\)matrix = 0.183, P = 0.060), and further varied according to the matrix type (NMDS1: \(\beta\)matrix contrast = 0.101, P = 0.027), especially at pasture plots (NMDS1: \(\beta\)matrix * matrix contrast = 0.196, P = 0.003) (Table S3). Species richness, abundance and genus composition were also affected by matrix type but at specific disturbance-contexts (Fig. 4). Along edges, species richness was higher whenever the matrix consisted of cattle pastures (\(\beta\)edge * matrix contrast = 0.288, P = 0.016) (Fig. 4a). Likewise, species abundance was higher in pasture sites at both edge and matrix sites (\(\beta\)edge * matrix contrast = 0.465, P < 0.0001; \(\beta\)matrix * matrix contrast = 0.413, P < 0.001) (Fig. 4b). Genus composition diverged between different matrix types but only at matrix sites (\(\beta\)matrix * matrix contrast = 0.196, P = 0.003) (Fig. 4c; Table S3). The effect of native habitat area was evident along edges, where most notably both species richness (\(\beta\)edge * area = 0.286, P < 0.001) and abundance increased (\(\beta\)edge * area = 4.98, P < 0.001) (Fig. 5; Table S3).
Mean (\(\pm\) 95% CI) bird a species richness, b abundance, and c genus composition (NMDS1) across the different disturbance-contexts surveyed—fragment core, edge and adjacent matrix—and different matrix types—pasture (light green) and teak plantations (green). Data are presented for the 78 plots nested within 26 sampled sites across the Amazon, Cerrado and Pantanal biomes
Relationship between bird a species richness, b abundance and c genus composition (NMDS1) and native fragment area (log10 x) across the disturbance gradient. Data are presented for 78 plots nested within 26 sampled sites across the Amazon, Cerrado and Pantanal vegetation biomes. Lines indicate model-adjusted fits and shaded areas represent the 95% confidence region. P values are shown for the strongest relationships (P \(\le\) 0.05) and are colour-coded according to the respective disturbance-context (i.e., core, edge or matrix)
Regional differences were also observed in bird diversity across different biomes. Although species abundance tended to be higher at Pantanal sites (\(\beta\)Pantanal = 0.390, P = 0.063), species richness was lower at edges (\(\beta\)edge * Pantanal = − 0.484, P = 0.001), and so was species abundance at both edge (\(\beta\)edge * Pantanal = − 0.740, P < 0.001) and matrix sites (\(\beta\)matrix * Pantanal = − 0.643, P < 0.001). As indicated by the NMDS analysis, genus composition diverged significantly between Amazonian core plots and both the Cerrado edge plots (\(\beta\)edge * Cerrado = − 0.231, P = 0.009) and Pantanal matrix plots (\(\beta\)matrix * Pantanal = − 0.184, P = 0.014).
Bird responses within each disturbance-context
Additional predictors of bird diversity were detected when considering each disturbance-context for each of the two different matrix settings. Across sites whose matrix consisted of cattle pastures (Table 1), species richness increased along the fragment edges with fragment area and with the number of relict trees at the adjacent matrix. Species abundance also increased with the number of relict trees but only at matrix sites. Genus composition, however, was not well predicted by any of the variables considered (Table S5).
Across sites comprised of teak plantations (Table 2), species richness at core areas was higher for less irregularly shaped remnants (negative coefficient for shape) and older surrounding teak trees. At the edges, species richness increased with fragment area but decreased with the height of surrounding teak trees. Within matrix plots, species richness was higher when cohorts of teak trees were older. Species abundance at core sites was affected by all variables considered. Species abundance along edges increased with fragment area and decreased with both distance to the nearest fragment and height of teak trees. Species abundance in the matrix increased in more mature teak plantations near less-irregularly shaped fragments. Genus composition at edge plots was only affected by fragment shape (Table S6). In addition, species richness at Pantanal sites was higher at core and matrix plots compared to Cerrado plots, but lower along the edge. The same was observed for species abundance, which was also higher at core and matrix plots.
Discussion
Our findings show that the remaining bird diversity across the highly heterogeneous human-modified landscapes of central-western Brazil is shaped by the synergistic effects of local disturbance-context (core–edge-matrix), matrix habitat quality as a function of both matrix type and land-use intensity, and native habitat configuration mostly determined by native vegetation remnant size, but also by the shape and degree of isolation of remnants. As expected, core areas of native remnants harboured higher avian diversity than either fragment edges or the agricultural matrix. Although bird species richness was relatively similar at sites with matrix areas consisting of either cattle pastures or teak silviculture, species richness was higher along fragment edges if these were surrounded by pastures. Likewise, species abundance was also higher at fragment edges and matrix areas at pasture sites. Genus composition was significantly dissimilar between native fragments (core and edge) and their adjacent matrix, as well as between matrix types at matrix sites. Moreover, regardless of matrix type, bird diversity was a function of matrix quality, with higher diversity in older and higher-statured teak plantations and pasture plots hosting a greater availability of relict live trees. Finally, the effect of native habitat area was detected only along fragment edges, where both species richness and abundance increased with fragment size.
One of the limitations of this study, however, was the unfeasibility of adequately testing the biome effect, which could not be distinguished from the effect of matrix type. Indeed, the only matrix type in Amazonian sampling sites was cattle pastures, while both Cerrado and Pantanal sites only included teak plantations. This is the reason why we controlled for the biome effect. As expected, when biome was considered as a fixed factor, genus composition differed at both Cerrado and Pantanal sites compared to the Amazonian ones, but only within matrix plots. This was more likely due to matrix type, rather than to a biome effect.
Disturbance gradient
Across all three biomes surveyed, bird diversity declined from core natural habitat patches to adjacent matrix sites. Similar findings were observed in assessments of bird responses to similar tropical forest disturbance gradients (Daily et al. 2001; Macchi et al. 2020). Small mammal assemblages within the same Cerrado and Pantanal sites surveyed here were also more diverse in core natural remnant areas (Casagrande and Santos-Filho 2019). Habitat quality is known to be higher in core areas of forest fragments, where a higher abundance and diversity of structural and trophic resources are likely to be available (Malcolm 1994). In addition, many microhabitat conditions may be restricted to core native habitats (Anjos 2006), which would explain the large proportion of bird species exclusively recorded within those plots (Lovejoy et al. 1986), including extinction-prone species (e.g., Tinamus tao and Tyranneutes stolzmanni; Stotz 1996). Along fragment edges, elevated solar radiation and temperatures drive more desiccated conditions that alter vegetation and leaf-litter microclimates, often degrading habitat conditions for most strict forest-affiliated species (Laurance 1991; Malcolm 1994). This explains the lower species richness and abundance observed along fragment edges, in addition to a divergent species composition that likely corresponds to the replacement of forest-interior species by non-forest-dependent open-habitat species, as is often the case of understorey insectivores (Lees and Peres 2008; Morante-Filho et al. 2016; Bueno et al. 2018). The number of bird species on average declined from the fragment edge to adjacent exotic pastures and teak monocultures. Therefore, although the wider countryside can potentially sustain moderate levels of species diversity (Mendenhall et al. 2014), bird diversity at the intersection of the three of the largest biomes in the central portion of South America relied primarily on areas of native habitat remnants (cf. Barlow et al. 2007; Gibson et al. 2011).
Silviculture vs pasture matrix
Genus composition differed between matrix types especially where matrix areas were comprised of cattle pastures. Indeed, compared to adjacent native fragments, the teak plantation matrix harboured fewer species but more similar genus composition, whereas cattle pastures retained fewer species but a much higher species turnover. Despite the relatively higher abundance of birds occupying cattle pastures, compared to teak plantations, the set of species in cattle pastures is likely to correspond to habitat generalists and open-habitat specialists (Bueno and Peres 2018). Compared to pastures, tree plantations likely sustained effective habitat use or transient movements of a higher proportion of forest-dependent species. In the Brazilian Atlantic forest, bird diversity also benefited from more structurally complex matrix types (Barbosa et al. 2017; Biz et al. 2017; Hatfield et al. 2020). Moreover, the structural contrast between native habitat remnants and exotic matrix areas was clearly greater for pastures than for tree plantations, which reflects the higher tree stature and closed-canopy forest of the Amazon biome, compared to the relatively open Cerrado and Pantanal environments.
Land-use intensity within matrix areas
When considering each disturbance-context for each of the two matrix settings separately, additional predictors of bird diversity were detected. In terms of matrix complexity, isolated relict trees left in Amazonian cattle pastures ensured an increase in number of species within the matrix and the number of individuals along edges. Relict trees boosts matrix habitat quality, mimicking some features of old-growth habitats, enough to increase bird abundance within nearby native fragments (cf. Hatfield et al. 2020). It is possible that relict trees isolated in the middle of cattle pastures further contributed to soften the fragment-matrix structural contrast. Indeed, under the extreme habitat conditions imposed by cattle pastures, even single isolated trees can provide minimal increments in limiting trophic and structural resources, and are further used as stepping stones to maintain functional connectivity of bird assemblages in fragmented landscapes (Tremblay and St. Clair 2011; Godoi et al. 2018), particularly for gap-crossing forest-dependent species (Lindenmayer et al. 2008).
Furthermore, we found that teak plantation age was the most important positive determinant of avian diversity in both fragment core and matrix plots surrounding the relatively open Cerrado and Pantanal vegetation. Older teak plantations are characterized by higher complexity in vertical stratification, including the presence of shrubs in the understorey, providing higher availability of both structural and trophic resources (Hatfield et al. 2020). For instance, in Tanzanian dry woodlands, many forest-dependent species thrived in older teak plantation, while young plantations were primarily used by open-habitat species (Jenkins et al. 2003). Moreover, the higher bird abundance we observed along the edges of native fragments surrounded by low-statured teak plantations may be due to the structural similarity between the relatively open biomes and the adjacent plantation, provided that critical microhabitats and resources are retained. Overall, our findings are in agreement with previous studies on human-modified landscapes showing that structurally complex matrix areas containing elements of forest habitat are more likely to promote biodiversity persistence (Mendenhall et al. 2014; Frishkoff and Karp 2019).
Configuration of native habitat fragments
We expected higher species diversity in larger native habitat fragments, as predicted by the Island Biogeography Theory (MacArthur and Wilson 1967) or, at forest plots surrounded by greater habitat amounts, according to the Habitat Amount Hypothesis (Fahrig 2013). In our surveyed sites, however, the effect of habitat area was only perceived at edge plots, which harboured higher species diversity at larger fragments. Within core remnant areas, habitat quality may be less affected by changes in fragment size (Malcolm 1994), thereby maintaining relatively high species diversity regardless of area effects. Habitat conditions at the fragment edge, however, are expected to be more variable, further depending on fragment shape (Laurance and Yensen 1991). The species-area relationship we observed may in part result from the intensity of edge effects, which may be lower in larger fragments, thereby supporting higher species diversity (Ewers et al. 2007; Fletcher et al. 2007). In fact, fragment shape was important for genus composition along edge plots, but only in teak plantation sites. Similar findings were reported for Atlantic Forest birds, where edge-sensitive species that avoid forest margins were only present at the core of large fragments where the most intensive edge effects did not penetrate, thereby explaining the increasing strength of edge effects with fragment size (Banks-Leite et al. 2010).
Conservation implications
Our study highlights that understanding the drivers of biodiversity persistence in human-modified landscapes requires integrating information on habitat quality and configuration, in addition to land-use intensity, and their respective interaction terms. In terms of conservation implications, as native habitats generally harbour higher levels of bird diversity, maintaining primary habitat cover even in natural grasslands and seasonally-flooded savannahs should remain the highest conservation priority (Peres 2005; Barlow et al. 2007; Gibson et al. 2011). Given the detrimental role of edge effects shaping bird assemblages within tropical forest fragments, preserving sufficiently large core areas is particularly important across fragmented forest landscapes (Banks-Leite et al. 2010). In addition, maximizing the size of native fragments while avoiding irregularly-shaped fragment would contribute to maintain the diversity of forest-affiliated species. More structurally complex matrix types, such as tree plantations, are likely to sustain a higher proportion of forest-dependent species (Barbosa et al. 2017; Biz et al. 2017; Hatfield et al. 2020). In that case, it is particularly important that native regeneration in the silvicultural undergrowth is spared from chemical herbicides (Louzada et al. 2010). In addition, fast-growing tree plantations promote carbon sequestration, further contributing to mitigate climate change as already examined for teak plantations (Nölte et al. 2018). Moreover, regardless of matrix type, adequate management of matrix areas that maximizes structural complexity can further increase bird diversity. In the case of cattle pastures, structural complexity can be achieved by retaining relict old-growth trees and, in the case of teak plantations, by delaying clear-cutting until tree stands are at least 18 years-old, while retaining the native plantation understorey, and ensuring that some large native trees are spared following systematic felling. Our findings contribute to the notion that human-modified landscapes require critical planning and habitat management to maximize biodiversity persistence across tropical environments.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Anjos L (2006) Bird species sensitivity in a fragmented landscape of the Atlantic Forest in Southern Brazil. Biotropica 38:229–234
Antongiovanni M, Metzger JP (2005) Influence of matrix habitats on the occurrence of insectivorous bird species in Amazonian forest fragments. Biol Conserv 122:441–451
Arroyo-Rodríguez V, Fahrig L, Tabarelli M, Watling JI, Tischendorf L, Benchimol M, Cazetta E, Faria D, Leal IR, Melo FPL, Morante-Filho JC, Santos BA, Arasa-Gisbert R, Arce-Peña N, Cervantes-López MJ, Cudney-Valenzuela S, Galán-Acedo C, San-José M, Vieira ICG, Slik JWF, Nowakowski AJ, Tscharntke T (2020) Designing optimal human-modified landscapes for forest biodiversity conservation. Ecol Lett 23:1404–1420
Banks-Leite C, Ewers RM, Metzger JP (2010) Edge effects as the principal cause of area effects on birds in fragmented secondary forest. Oikos 119:918–926
Barbosa KVC, Knogge C, Develey PF, Jenkins CN, Uezu A (2017) Use of small Atlantic Forest fragments by birds in Southeast Brazil. Perspect Ecol Conserv 15:42–46
Barlow J, Gardner TA, Araujo IS, Ávila-Pires TC, Bonaldo AB, Costa JE, Esposito MC, Ferreira LV, Hawes J, Hernandez MIM, Hoogmoed MS, Leite RN, Lo-Man-Hung NF, Malcolm JR, Martins MB, Mestre LAM, Miranda-Santos R, Nunes-Gutjahr AL, Overal WL, Parry L, Peters SL, Ribeiro-Junior MA, da Silva MNF, Motta CS, Peres CA (2007) Quantifying the biodiversity value of tropical primary, secondary, and plantation forests. Proc Natl Acad Sci 104:18555–18560
Barros FM, Martello F, Peres CA, Pizo MA, Ribeiro MC (2019) Matrix type and landscape attributes modulate avian taxonomic and functional spillover across habitat boundaries in the Brazilian Atlantic Forest. Oikos 128:1600–1612
Bates D, Maechler M, Bolker B, Walker S, Christensen RHB, Singmann H, Dai B, Scheipl F, Grothendieck G, Green P, Fox J, Bauser A, Krivitsky PN (2012) Package ‘lme4’. CRAN. R Foundation for Statistical Computing, Vienna
Biz M, Cornelius C, Metzger JPW (2017) Matrix type affects movement behavior of a Neotropical understory forest bird. Perspect Ecol Conserv 15:10–17
Bueno AS, Dantas SM, Henriques LMP, Peres CA (2018) Ecological traits modulate bird species responses to forest fragmentation in an Amazonian anthropogenic archipelago. Divers Distrib 24:387–402
Casagrande AF, Santos-Filho MD (2019) Use of forest remnants and teak (Tectona grandis) plantations by small mammals in Mato Grosso, Brazil. Stud Neotrop Fauna Environ 54:181–190
Daily GC, Ehrlich PR, Sánchez-Azofeifa GA (2001) Countryside biogeography: use of human-dominated habitats by the avifauna of southern Costa Rica. Ecol Appl 11:1–13
Driscoll DA, Banks SC, Barton PS, Lindenmayer DB, Smith AL (2013) Conceptual domain of the matrix in fragmented landscapes. Trends Ecol Evol 28:605–613
Ewers RM, Thorpe S, Didham RK (2007) Synergistic interactions between edge and area effects in a heavily fragmented landscape. Ecology 88:96–106
Fahrig L (2013) Rethinking patch size and isolation effects: the habitat amount hypothesis. J Biogeogr 40:1649–1663
Fahrig L, Baudry J, Brotons L, Burel FG, Crist TO, Fuller RJ, Sirami C, Siriwardena GM, Martin JL (2011) Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol Lett 14:101–112
Fletcher RJ Jr, Ries L, Battin J, Chalfoun AD (2007) The role of habitat area and edge in fragmented landscapes: definitively distinct or inevitably intertwined? Can J Zool 85:1017–1030
Frishkoff LO, Karp DS (2019) Species-specific responses to habitat conversion across scales synergistically restructure Neotropical bird communities. Ecol Appl 29:e01910
Gascon C, Lovejoy TE, Bierregaard RO Jr, Malcolm JR, Stouffer PC, Vasconcelos HL, Laurance WF, Zimmerman B, Tocher M, Borges S (1999) Matrix habitat and species richness in tropical forest remnants. Biol Conserv 91:223–229
Gibson L, Lee TM, Koh LP, Brook BW, Gardner TA, Barlow J, Peres CA, Bradshaw CJA, Laurance WF, Lovejoy TE, Sodhi NS (2011) Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 478:378–381
Godoi MN, Laps RR, Ribeiro DB, Aoki C, de Souza FL (2018) Bird species richness, composition and abundance in pastures are affected by vegetation structure and distance from natural habitats: a single tree in pastures matters. Emu 118:201–211
Golfari L, Caser RL, Moura VPG (1978) Zoneamento ecológico esquemático para reflorestamento no Brasil (2a aproximacão). PRODEPEF, Belo Horizonte
Handbook of the Birds of the World and BirdLife International (2018) Handbook of the birds of the world and birdLife international digital checklist of the birds of the world. Version 3. http://datazone.birdlife.org/userfiles/file/Species/Taxonomy/HBW-BirdLife_Checklist_v3_Nov18.zip. Accessed 20 Oct 2020
Hansen MC, Potapov PV, Moore R, Hancher M, Turubanova SA, Tyukavina A, Thau D, Stehman SV, Goetz SJ, Loveland TR, Kommareddy A, Egorov A, Chini L, Justice CO, Townshend J (2013) High-resolution global maps of 21st-century forest cover change. Science 342:850–853
Hansen MC, Wang L, Song XP, Tyukavina A, Turubanova S, Potapov PV, Stehman SV (2020) The fate of tropical forest fragments. Sci Adv 6:eaax8574
Harrison MLK, Banks-Leite C (2019) Edge effects on trophic cascades in tropical rainforests. Conserv Biol 34:977–987
Hatfield JH, Barlow J, Joly CA, Lees AC, Parruco CHDF, Tobias JA, Orme CDL, Banks-Leite C (2020) Mediation of area and edge effects in forest fragments by adjacent land use. Conserv Biol 34:395–404
IBGE (2020) Instituto Brasileiro de Geografia e Estatística. Monitoramento da cobertura do uso da terra do Brasil 2016–2018. http://ibge.gov.br/. Accessed 20 Oct 2020
INPE (2021) Instituto Nacional de Pesquisas Espaciais: a taxa consolidada de desmatamento por corte raso para os nove estados da Amazônia Legal (AC, AM, AP, MA, MT, PA, RO, RR e TO) em 2019 é de 10.129 km2. http://www.inpe.br/noticias/noticia.php?Cod_Noticia=5465. Accessed 07 June 2021
Jenkins RKB, Roettcher K, Cort G (2003) The influence of stand age on wildlife habitat use in exotic Teak tree Tectona grandis plantations. Biodivers Conserv 12:975–990
Koopen W (1948) Climatologia, com um estúdio de los clima de la Tierra. FCE, México
Laurance WF (1991) Edge effects in tropical forest fragments: application of a model for the design of nature reserves. Biol Conserv 57:205–219
Laurance SG (2004) Responses of understory rain forest birds to road edges in central Amazonia. Ecol Appl 14:1344–1357
Laurance WF, Yensen E (1991) Predicting the impacts of edge effects in fragmented habitats. Biol Conserv 55:77–92
Laurance WF, Ferreira LV, Rankin-de Merona JM, Laurance SG (1998) Rain forest fragmentation and the dynamics of Amazonian tree communities. Ecology 79:2032–2040
Lees AC, Peres CA (2008) Avian life-history determinants of local extinction risk in a hyper-fragmented neotropical forest landscape. Anim Conserv 11:128–137
Lees AC, Moura NG, de Almeida AS, Vieira IC (2015) Poor prospects for avian biodiversity in Amazonian oil palm. PLoS ONE 10:e0122432
Lindenmayer DB, Cunningham RB, MacGregor C, Crane M, Michael D, Fischer J, Montague-Drake R, Felton A, Manning A (2008) Temporal changes in vertebrates during landscape transformation: a large-scale “natural experiment.” Ecol Monogr 78:567–590
Louzada J, Gardner T, Peres CA, Barlow J (2010) A multi-taxa assessment of nestedness patterns across a multiple-use Amazonian forest landscape. Biol Conserv 143:1102–1109
Lovejoy TE, Bierregaard RO Jr., Bierregaard RO, Rylands AB, Malcolm JR, Quintela CE, Harper LH, Brown KS Jr, Powell AH, Powell GVN, Schubart HOR, Hays MB (1986) Edge and other effects of isolation on Amazon forest fragments. In: Soule ME (ed) Conservation biology: the science of scarcity and diversity. Sinauer Associates, Sunderland
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Press Princeton, Princeton
Macchi L, Decarre J, Goijman AP, Mastrangelo M, Blendinger PG, Gavier-Pizarro GI, Murray F, Piquer-Rodriguez M, Semper-Pascual A, Kuemmerle T (2020) Trade-offs between biodiversity and agriculture are moving targets in dynamic landscapes. J Appl Ecol 57:2054–2063
Mahood SP, Lees AC, Peres CA (2012) Amazonian countryside habitats provide limited avian conservation value. Biodivers Conserv 21:385–405. https://doi.org/10.1007/s10531-011-0188-8
Malcolm JR (1994) Edge effects in central Amazonian forest fragments. Ecology 75:2438–2445
Marinho-Filho J, Reis ML, Oliveira PS, Vieira EM, Paes MN (1994) Diversity standards and small mammal numbers: conservation of the cerrado biodiversity forests in the zoogeography of the cerrado’s non-volant mammalian fauna. An Acad Bras Cienc 66:149–156
Marini MA, Garcia FI (2005) Bird conservation in Brazil. Conserv Biol 19:665–671
Mata JRR, Erize F (2006) Rumboll, M. Birds of South America—non passerines: Rheas to woodpeckers. Princeton University Press, Princeton
Matuoka MA, Benchimol M, de Almeida-Rocha JM, Morante-Filho JC (2020) Effects of anthropogenic disturbances on bird functional diversity: A global meta-analysis. Ecol Indic 116:106471
Mendenhall CD, Karp DS, Meyer CF, Hadly EA, Daily GC (2014) Predicting biodiversity change and averting collapse in agricultural landscapes. Nature 509:213–217
Morante-Filho JC, Arroyo-Rodríguez V, Faria D (2016) Patterns and predictors of β-diversity in the fragmented Brazilian Atlantic forest: a multiscale analysis of forest specialist and generalist birds. J Anim Ecol 85:240–250
Moura NG, Lees AC, Andretti CB, Davis BJ, Solar RR, Aleixo A, Barlow J, Ferreira J, Gardner TA (2013) Avian biodiversity in multiple-use landscapes of the Brazilian Amazon. Biol Conserv 167:339–348
Newbold T, Hudson LN, Phillips HRP, Hill SLL, Contu S, Lysenko I, Blandon A, Butchart SHM, Booth HL, Day J, Palma A, Harrison MLK, Kirkpatrick L, Pynegar E, Robinson A, Simpson J, Mace GM, Scharlemann JPW, Purvis A (2014) A global model of the response of tropical and sub-tropical forest biodiversity to anthropogenic pressures. Proc R Soc B 281:20141371
Newbold T, Hudson LN, Hill SL, Contu S, Gray CL, Scharlemann JP, Börger L, Phillips HRP, Sheil D, Lysenko I, Purvis A (2016) Global patterns of terrestrial assemblage turnover within and among land uses. Ecography 39:1151–1163
Nölte A, Meilby H, Yousefpour R (2018) Multi-purpose forest management in the tropics: Incorporating values of carbon, biodiversity and timber in managing Tectona grandis (teak) plantations in Costa Rica. For Ecol Manag 422:345–357
Oksanen J, Blanchet FG, Friendly M, Kindt R, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2020) Package ‘vegan’. Community ecology package, version 2.5-7
Passos FB, Marimon BS, Phillips OL, Morandi PS, das Neves EC, Elias F, Reis SM, de Oliveira B, Feldpausch BR, Júnior BHM (2018) Savanna turning into forest: concerted vegetation change at the ecotone between the Amazon and “Cerrado” biomes. Braz J Bot 41:611–619
Peres CA (2005) Why we need megareserves in Amazonia. Conserv Biol 19:728–733
Pfeifer M, Lefebvre V, Peres CA, Banks-Leite C, Wearn OR, Marsh CJ, Butchart SHM, Arroyo-Rodríguez V, Barlow J, Cerezo A, Cisneros L, D’Cruze N, Faria D, Hadley A, Harris SM, Klingbeil BT, Kormann U, Lens U, Medina-Rangel GF, Morante-Filho JC, Olivier P, Peters SL, Pidgeon A, Ribeiro DB, Scherber C, Schneider-Maunoury L, Struebig M, Urbina-Cardona N, Watling JI, Willig MR, Wood EM, Ewers RM (2017) Creation of forest edges has a global impact on forest vertebrates. Nature 551:187–191
Piacentini VQ, Aleixo A, Agne CE, Mauricio GN, Pacheco JF, Bravo GA, Brito GRR, Naka LN, Olmos F, Posso S, Silveira LF, Betini GS, Carrano E, Franz I, Lees AC, Lima LM, Pioli D, Schunck F, Amaral FR, Bencke GA, Cohn-Haft M, Figueiredo LFA, Straube FC, Cesari E (2015) Annotated checklist of the birds of Brazil by the Brazilian Ornithological Records Committee. Rev Bras Ornitol 23:91–298
Prevedello JA, Vieira MV (2010) Does the type of matrix matter? A quantitative review of the evidence. Biodivers Conserv 19:1205–1223
R Development Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing. http://www.R-project.org/
Ridgely RS, Tudor G (2009) Field guide to the songbirds of South America: the passerines, 1st edn. University of Texas Press, Austin
Sick H (1997) Ornitologia Brasileira. Nova Fronteira, Rio de Janeiro
Somenzari M, Amaral PPD, Cueto VR, Guaraldo ADC, Jahn AE, Lima DM, Lima PC, Lugarini C, Machado CG, Martinez J, do Nascimento JLX, Pacheco JF, Paludo D, Prestes NP, Serafini PP, Silveira F, de Sousa AEBA, de Sousa NA, de Souza MA, Telino-Júnior WR, Whitney BM (2018) An overview of migratory birds in Brazil. Papéis Avulsos de Zoologia. https://doi.org/10.11606/1807-0205/2018.58.03
Stotz DF (1996) Neotropical birds: ecology and conservation. University of Chicago Press, Chicago
Stouffer PC, Bierregaard RO Jr (1995) Use of Amazonian forest fragments by understory insectivorous birds. Ecology 76:2429–2445
Stouffer PC, BierregaardStrongLovejoy ROCTE Jr (2006) Long-term landscape change and bird abundance in Amazonian rainforest fragments. Cons Biol 20:1212–1223
Tewari DN (1992) A monograph on teak (Tectona grandis Linn. f.). International book distributors
Tremblay MA, St. Clair CC (2011) Permeability of a heterogeneous urban landscape to the movements of forest songbirds. J Appl Ecol 48:679–688
Tubelis DP, Cowling A, Donnelly C (2004) Landscape supplementation in adjacent savannas and its implications for the design of corridors for forest birds in the central Cerrado, Brazil. Biol Conserv 118:353–364
Wiens JA (1995) Habitat fragmentation: island v landscape perspectives on bird conservation. Ibis 137:97–104
Acknowledgements
We thank Floresteca S/A and landowners for permission to work within their landholdings, and all personnel involved during the fieldwork for their cooperation and hospitality. We also thank Breno Dias Vitorino for reviewing species migratory behaviour.
Funding
This study was funded by the State of Mato Grosso Research Foundation (FAPEMAT) and the Brazilian Ministry of Education (CAPES). AFP was funded by the European Union’s Horizon 2020 research and innovation program under the Marie Skłodowska-Curie grant agreement No. 792678.
Author information
Authors and Affiliations
Contributions
OMAN: investigation, writing—reviewing and editing; AFP: formal analysis, visualization, writing—original draft; CAP: conceptualization, writing—reviewing and editing; MdS-F: conceptualization, writing—reviewing and editing, funding acquisition, supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Consent to participate
All authors agree have reviewed and agree with the submission of this version of the manuscript.
Consent to publish
All authors accept the responsibility for releasing the material included in this manuscript.
Ethical approval
The manuscript contains original data and is not under consideration in another journal.
Additional information
Communicated by Eckehard G. Brockerhoff.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nogueira, O.M.A., Palmeirim, A.F., Peres, C.A. et al. Synergistic effects of habitat configuration and land-use intensity shape the structure of bird assemblages in human-modified landscapes across three major neotropical biomes. Biodivers Conserv 30, 3793–3811 (2021). https://doi.org/10.1007/s10531-021-02275-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-021-02275-8