Abstract
There is newly invigorated interest in intensifying agriculture in northern Australia, and research has identified up to 17 M ha of suitable soils, much of which are floodplains. Given that 88% of these best soils for agriculture occur outside the current protected area network, and floodplains are a minority habitat in northern Australia, developing northern Australia whilst maintaining its biodiversity will be a great challenge. Ants are a particularly prominent and diverse faunal group within northern Australia, but almost nothing is known of the floodplain fauna. We sampled 20 pairs of plots in floodplains and surrounding savannas in three locations in the high rainfall zone of the Northern Territory to investigate the ant faunal composition of the floodplains, to determine to what extent they are comprised of species unique to floodplains or just a subset of the broader savanna fauna. We collected 114 species from 29 genera. Eighteen species were found only in floodplain plots, but only three were definitively floodplain specialists and another four had a propensity for clay soils. For all locations combined, savanna plots contained approximately 1.7 times more species than floodplain plots. Multivariate analysis found that the two habitats supported distinct ant faunas and the three locations also had distinct faunas. Nine species in this study were new to science, suggesting that there are many other species yet to be discovered. The results indicate that floodplains have conservation value for ant biodiversity, as is the case for other components of the biota. Efforts to intensify agriculture on north Australian floodplains therefore need to have effective conservation considerations if we are to achieve ecologically sustainable development.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Northern Australia is exceptional in global standards because it contains a relatively low human population living within the largest intact savannas in the world, covering approximately 2 million square kilometres (Woinarski et al. 2007). The greatest land-use throughout northern Australia is pastoralism, whereby livestock are stocked at low density throughout large areas of predominantly untransformed environments. But the development of northern Australia using more intensive forms of agriculture has been the policy ambition for many governments for over a century. This political aspiration is driven by perceived opportunities for Australia to trade more with Asia in food commodities, as well as broader national policy goals surrounding regional development (Australian Government 2015). As a result there have been numerous attempts to establish intensive agriculture in northern Australia, most of which have failed (Bauer 1977; Ash 2013). Regardless, political will to develop the north has recently been reinvigorated (Joint Select Committee on Northern Australia 2014; Australian Government 2015), and there is now much scientific scrutiny into what capacity the landscape holds and what opportunities can be pursued (CSIRO 2009; Petheram et al. 2018). Given that habitat transformation or destruction is the greatest cause of biodiversity loss globally (Brooks et al. 2002; Travis 2003), developing northern Australia whilst maintaining its biodiversity will be a great challenge (Morán-Ordóñez et al. 2017), requiring a balance between conservation and agricultural interests (Wilson et al. 2010; Shackelford et al. 2015).
Research to date has identified up to 17 M ha (14%) of soils in northern Australia suitable for intensive, irrigated agriculture (CSIRO 2009; Webster et al. 2009; Wilson et al. 2009). Much of these soils include those of the relatively fertile floodplains associated with the major river systems that drain the high-rainfall, subcoastal regions. Of the non-savanna habitats within northern Australia, seasonally-inundated floodplains are among the least studied for their biota, with conservation attention to date focusing instead on the region’s sandstone country and rainforest patches. A predominate finding of rainforest studies has been high levels of endemism and biotic composition that is highly disjunct with that in the broader landscape, and species that often have very localised distributions (Russel-Smith 1991; Gambold and Woinarski 1993; Woinarski 1993; Andersen et al. 2007). Such characteristics make these habitats and their biota vulnerable to disturbance. Floodplain plants, and to a lesser extent vertebrates, are also comprised largely of species restricted to this habitat, although predominantly with broad geographic distributions (Woinarski and Braithwaite 1991; Cowie et al. 2000; Halford and Fensham 2014). However, practically nothing is known about the largest faunal group, the invertebrates. Given that 88% of the best soils for agriculture occur outside the current protected area network (Morán-Ordóñez et al. 2017) greater knowledge of the floodplain invertebrate fauna is required if development is to be achieved without the loss of species.
Ants are a particularly prominent faunal group within Australia, and have been found to be important drivers of soil-based ecological processes in both natural and agricultural systems (Cammeraat and Risch 2008; Evans et al. 2011; Del Toro et al. 2012). Ants are also notably diverse within Australia, having potentially around 10,000 species (Andersen 2016; Oberprierler et al. 2018). Much of this species diversity occurs within northern Australia, with more than 900 species being recorded from the Top End of the Northern Territory alone (Andersen et al. 2018), which encompasses only 10% of northern Australia within a single climatic zone. Yet very little of northern Australia has been sampled for ants, and there are many species that remain to be discovered especially from within under-sampled habitats. To date there has been no formal ant sampling in floodplains in northern Australia, and very limited sampling of the most similar habitat—seasonally-waterlogged (black/clay) soils (Andersen 1993; Hoffmann 2000, 2003; Andersen et al. 2015). General findings from the studies on black soils is that these habitats support lower ant diversity than surrounding well-drained soils, that there are numerous species that are likely to be specialists of these habitats, and that the species composition of the communities as displayed by functional group profiles (Andersen 1995) are markedly different, with savanna ant communities containing more Generalised Myrmicinae, Hot-climate Specialists and other minor functional groups. Here, we provide the first survey of floodplain ant communities in northern Australia, to determine to what extent they are comprised of species unique to floodplains or just a subset of the broader savanna fauna, and to consider the conservation implications of proposed land-use intensification within these environments.
Methods
Study sites
The study was conducted throughout the upper latitudes of Australia’s Northern Territory (Fig. 1), where the rainfall is highest in the Northern Territory. We focused on the higher rainfall region because this is where most of the floodplains occur. The region has a seasonal monsoonal climate, with high temperatures (17–33 °C) throughout the year and an annual rainfall of approximately 1200–1800 mm falling predominantly during the summer wet season. Ant communities were sampled in three locations: Darwin, Melville Island (hereafter referred to as Melville) and North East Arnhem Land (hereafter referred to as Arnhem) to cover the breadth of the northern-most latitudes of the Northern Territory. All sites had little prior history of anthropogenic disturbance. Sites around Darwin were vacant government land that had been de-stocked of cattle more than 50 years prior, and those at Melville and Arnhem were Aboriginal Freehold tenure with no history of utilisation. At most, all locations have been subject to some utilisation by feral pigs and Asian water buffalo.
Sampling
At each of the three locations, ants were sampled in paired plots on a floodplain having clay soil and an adjacent savanna having well-drained loam soil (Darwin: 10 pairs; Melville: 8 pairs; and Arnhem: 2 pairs) giving a total of 40 plots. Only two pairs were possible in Arnhem due to the scarcity of floodplains in the region where we could access. Floodplains ranged in size from 2 to 30 ha, most often being oblong-shaped following a shallow drainage line, with a distance of < 200 m across the width. As such, some floodplain plots were only 50 m away from the savanna edge, but this distance is deemed far greater than the foraging distance of most species so edge effects were not considered an issue. In all cases, paired plots were spaced 100–260 m apart, and were closer to each other than to other plots which were always farther than 340 m apart. At Darwin and Melville, two pairs of plots were used per discrete floodplain, whereas only one pair of plots was used per floodplain in Arnhem due to space constraints. Ants were sampled at each plot using an array of 15 pitfall traps (4.2 cm diameter specimen containers), partly filled with ethylene glycol as a preservative. Pitfall traps were set in a 3 × 5 grid with 10 m spacing and operated for 48 h. Darwin plots were sampled in June 2011, Melville in July 2011 and Arnhem in September 2013. Ants were sorted to species-level, and undescribed species were matched with species numbers (e.g. sp. 4) in the CSIRO Darwin laboratory ant collection. Where a species couldn’t be matched it was given a letter code that applies to this study only.
Analysis
To assess sampling completeness of the two habitats at each of the three locations, we created individual-based rarefaction curves using EstimateS (Colwell 2013). Plot-level species richness of the habitats by location and of the two habitats for all locations combined was compared using a one-way ANOVA and a non-parametric Mann–Whitney U-test respectively in Statistica 11. A Cochran’s test was used to confirm data homogeneity for the one-way ANOVA.
Each species was classified as a floodplain specialist or not based on the collection data found here, coupled with the collection data of specimens of the same species held at the CSIRO Darwin laboratory. The CSIRO Darwin laboratory holds by far the most comprehensive collection of northern Australian ants, containing specimens from extensive hand collections made by numerous researchers, and vouchers from all published ant surveys conducted in northern Australia over the past 30 years. This collection is now increasingly being used to quantify the biogeography of Australia’s ants (e.g. Andersen 2016; Andersen et al. 2015, 2018).
The species composition of the two habitats was compared using non-metric multidimensional scaling of species presence/absence data in Primer 6 (Clarke and Gorley 2003). The similarity matrix was constructed using a Bray–Curtis association index. ANOSIM was used to test for clustering of plots within the two habitats.
Species were assigned to functional groups (Online Appendix 1) based on ant responses to stress and disturbance (Andersen 1995). Eight functional groups are relevant to this study: Dominant Dolichoderinae (primarily species of Iridomyrmex), Generalised Myrmicinae (Monomorium, Crematogaster and Pheidole), Opportunists (predominantly Rhytidoponera, Nylanderia and Tetramorium), Subordinate Camponotini (predominantly Camponotus and Polyrhachis), Hot-climate Specialists (Melophorus, Meranoplus and some Monomorium), Tropical-climate Specialists (Oecophylla smaragdina), Cryptic Species (predominantly Solenopsis, Hypoponera and Strumigenys) and Specialised Predators (Pseudoneoponera, Leptogenys and Ectomomyrmex). Because only environmental stress is applicable in this study (i.e. all sites were free from anthropogenic disturbance), and floodplains present greater stress than savanna for ant habitat, we anticipated a greater proportion of Opportunitsts to Generalised Myrmicinae in floodplains, lower presence of minor specialist groups in floodplains, but a less predictable dynamic for Dominant Dolichoderinae because both habitats are open from an ant’s perspective with the ground receiving high levels of solar insolation. Tropical-climate Specialists, Cryptic Species and Specialist Predators were collected too infrequently for meaningful analysis individually, so they were grouped together as Others. The mean plot-level contribution of each functional group to species-richness and abundance was compared between the two habitats using the non-parametric Mann–Whitney U-test in Statistica 11.
Results
Overall we sampled 14,199 ants comprising 114 species from 29 genera (Online Appendix 1); 71 species from Darwin, 73 from Melville and 30 from Arnhem. The most speciose genera were Pheidole (15 species), Monomorium (12), Iridomyrmex (10) and Rhytidoponera (10). Rarefaction analysis showed that the ant faunas of the three locations and two habitats were sufficiently well sampled to make meaningful comparisons between them (Fig. 2). Five species were found in both habitats in all three locations: Monomorium sp. 24, Pheidole sp. 3, Iridomyrmex sp. 1, Iridomyrmex pallidus and Camponotus crozieri. Twelve species were found in savanna in all three locations and an additional 53 species were found only in savanna plots. Seven species, Rhytidoponera sp. 2, Cardiocondyla atalanta, Monomorium sp. 24, Pheidole sp. 3, Iridomyrmex pallidus, Iridomyrmex sp. 1 and Camponotus crozieri were found in floodplain at all three locations, and an additional 18 species were found only in floodplain. Of these 18, based on additional specimens within the CSIRO Darwin ant collection, only Pheidole sp. B and Iridomyrmex obscurus) are clearly floodplain specialists, and the new Melophorus from Melville belongs to a complex of floodplain specialists, but on clay soils in lower rainfall areas that merely waterlog, not flood. Four other species, Nylanderia sp. C, Pseudoneoponera sp. A and sp. 11, and Rhytidoponera haeckeli have been predominantly collected on clay/waterlogged soils. All others have previously been collected in non-floodplain habitats with the exception of the new Pheidole species whose habitat affiliation cannot be confidently determined. Notably, no exotic ant species were collected in floodplain plots (Online Appendix 1).
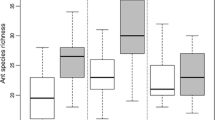
Species richness was greater in savanna compared with floodplain plots at all three locations, especially at Darwin (Fig. 3), but only statistically so in Darwin (One-way ANOVA: F5 = 13.7, P < 0.0001). For all locations combined, savanna plots contained approximately 1.7 times more species than floodplain plots (10 vs. 17), with this difference being statstically significant (Mann–Whitney U-test: Richness, U = 43, z = 4.2, P < 0.0001).
Multivariate analysis found that the two habitats savanna and floodplain plots supported distinct ant faunas (Fig. 4; ANOSIM: R = 0.348, P = 0.001). Additionally, the two well-sampled locations of Darwin and Melville also supported distinct faunas (ANOSIM: R = 0.411, P = 0.001), indicating regional as well as habitat distinctions.
Functional group profiles of the two habitats were very similar for both species composition and abundance (Fig. 5). Only the relative contribution of the abundance of Opportunists and Generalised Myrmicinae differed significantly between the two habitats (Table 1), with there being more Opportunists in floodplain and more Generalised Myrmicinae in savanna.
Discussion
The floodplain ant fauna
This study provides the first assessment of ant species diversity and uniqueness within floodplains of northern Australia, and is one of only a few assessments of ant communities in flood-prone environments anywhere in Australia (Meeson et al. 2002; Ballinger et al. 2007; Horrocks et al. 2012). Approximately 7% of the floodplain ant fauna were either floodplain specialists or are predominantly associated with clay soils, and the compositional mix of the floodplain ant fauna was distinctly different to the savanna fauna. This compositional distinctiveness is consistent with all other studies comparing ant community composition on waterlogged vs well-drained soils (Andersen 1993; Hoffmann 2000, 2003; Andersen et al. 2015), for other taxa in the same environments (Woinarski and Braithwaite 1991; Cowie et al. 2000; Halford and Fensham 2014) as well as for taxa inhabiting other minority habitats throuhgout northern Australia (Russel-Smith 1991; Gambold and Woinarski 1993; Woinarski 1993; Andersen et al. 2007).
The biological significance of the regional fauna was further highlighted by nine species being collected for the first time, two being found only within floodplain. Notably all nine new species were found within Melville and Arnhem, both of which are regions that have not been well surveyed for ants and no doubt contain many more as-yet unknown species. Notably there were no exotic ants present in floodplain and very few in savanna, characteristic of most of northern Australia where there has been very little land transformation from anthropogenic utilization.
Unsurprisingly, floodplain contained both lower plot-level and total ant diversity than savanna, as has been found in studies contrasting the ant faunas of clay versus well drained soils in northern Australia (Andersen 1993; Hoffmann 2000, 2003; Andersen et al. 2015). Surviving even short-term inundation is challenging for ants, requiring adaptive behaviours that either allow persistence under water, or flexibility to move colonies to dry locations (Nielsen 2011). The strategies used by ants in the seasonally-inundated floodplains of northern Australia are completely unknown. Additional challenges would also be faced for underground nest integrity when the water recedes, and the soil contorts, forming deep cracks. Such dynamic soil movement and reduced soil volume would require species to be highly flexible in nest spatial configuration. Finally, floodplains support few, if any, trees, which results in fewer carbohydrate and niche resources for ants, both of which are important drivers of localised ant diversity (Blüthgen et al. 2000, 2004; Klimes et al. 2012).
That the functional group profiles of the savanna versus floodplain faunas were remarkably similar was not expected. But in line with the predictable dynamics of the Australian functional group scheme (Andersen 1995), Opportunist abundance and species richness contributed relatively more in floodplain than savanna, with the reverse being so for Generalised Myrmicinae.
Not surprisingly, the combined findings of floodplain having lower species diversity, a greater proportion of Opportunistic species, and compositionally distinct fauna comprised of both habitat specialists and broader generalists from surrounding habitats is consistent with global patterns of ant communities in flooded environments (Majer and Delabie 1994; Milford 1999; Glaser 2007; Nielsen 2011). One exception though is the central South American floodplain systems, especially along the Río Paraná and the Río Paraguay, which is the native range of three of the world’s worst invasive ant species: the red imported fire ant Solenopsis invicta, the Argentine ant Linepithema humile and the little fire ant Wasmannia auropunctata, as well as others that have become problematic outside of their native range: S. richteri, Pheidole obscurithorax, Pseudomyrmex gracilis and Nylanderia fulva. The ant communities in these systems appear to be speciose and highly competitive (LeBrun et al. 2007; Calcattera et al. 2010), potentially with a greater number of aggressive species relative to most other flooded ant communities. It is postulated that the combination of high interspecific competition with the environmental challenge of flooding has selected for traits that make these, and potentially other, species successful invasive species (LeBrun et al. 2007).
Conservation implications
So what can be said of the potential impacts of land utilisation for commercial purposes in floodplains? Ultimately any impacts will be dependent upon the type of land use that is implemented, and such impacts are predictable from what is known of the responses of ants to disturbance globally. Following we present the current and likely future land uses in order of increasing disturbance and ecological impacts. Notably, this disturbance gradient and the ecological consequences would apply to flooplain systems globally, not just in northern Australia.
The current usage of floodplain systems in northern Australia is predominantly cattle grazing on otherwise unmodified land, or at most with the addition of “improved” pastures (exotic fodder species) such as olive hymenachne Hymenachne amplexicaulis and para grass Urochloa mutica. Nothing is known of the effects of cattle grazing on seasonally-inundated floodplain ant communities in Australia’s wet tropics, nor of the interaction with introduced pasture species. However, research of grazing impacts on ant communities within natural grasslands in semi-arid Northern Territory on seasonally-waterlogged soils has found that grazing induces species compositional changes, but does not necessarily affect total species richness (Hoffmann 2000); patterns that hold globally for grazing on all soil types (Hoffmann 2010; Arcoverde et al. 2017). The major caveat though being that such research into the effects of grazing globally has been conducted in systems that have been grazed for many decades, often historically at higher intensities. Therefore there are extremely few truly ungrazed areas to measure as a real control (most often none within any study), so realistically grazing-sensitive species would have undergone their significant negative dynamics long ago, leaving behind a fauna to be measured that is much more grazing tolerant.
Should cattle grazing intensify in floodplains, there are two conservation considerations. First is the lack of refugia for habitat specialists that may be grazing-sensitive. Such species in semi-arid and arid regions would possibly be able to persist in waterlogged locations most distant from water sources where grazing pressure is lowest, but no such refugia exist on a seasonally-inundated floodplains without physical ungulate exclusion (i.e. fencing). Second is that increasing cattle production is likely to come from the introduction and promotion of exotic fodder species. Notably the two pasture species favoured in floodplain systems, and potentially others, are also serious conservation threats through their habit of forming monocultures and modifying environmental dynamics such as fire regimes, shading and hydrology, all which reduce coexistence of native biota (Holm et al. 1977; Ferdinands et al. 2005; Godfree et al. 2017).
The greatest environmental impacts would occur from land modification for intensive agriculture, whereby all native vegetation would be removed and the soil tilled. Such habitat change induces greatest species loss for all taxa, particularly of specialised and arboreal species, leaving only a few disturbance-tolerant and generalist species persisting (Kondoh 1978; Gómez et al. 2003; House et al. 2012; Botha et al. 2015; Solar et al. 2016). This level of habitat and resultant faunistic change is very comparable to the effects of urbanisation (McKinney 2008; Heterick et al. 2000; Buczkowski and Richmond 2012). More native species are able to be conserved when tillage does not occur (Fernandez et al. 2018), and when structural diversity and plant diversity is relatively greater (i.e., monoculture crop vs. mixed-species cropping, tree crops or crops with grassy inter-crop spaces) (Perfecto et al. 2003; Brühl and Eltz 2010; Fayle et al. 2010; Gaigher and Samways 2010; Chong et al. 2011). Maximal species diversity is retained when long-term crops (e.g. coffee) are embedded within minimally transformed natural vegetation (Perfecto et al. 1997, 2003; De la Mora et al. 2013).
Historically, there have been multiple attempts to establish rice and cotton industries on seasonally-inundated floodplains throughout northern Australia, but most have failed, including all within the Northern Territory (Ash 2013). There were many reasons why these operations failed, including high pest load, difficult market access, high production costs, and poor management (Bauer 1977), all challenges that still persist today. But new technologies, new varieties of plants better adapted to local conditions, new markets, improved supply-chain infrastructure, and greater understanding of the requirements of agricultural production in northern Australia are likely to result in future attempts to grow these and other crops at commercial scales. All of these operations will have the greatest impact on floodplain biota.
In summary, north Australian floodplains contain high ant diversity, with some unique ant species, and faunal compositions that are both distinct from surrounding habitats as well as distinct regionally. This faunal distinctiveness, coupled with the lack of exotic species, suggests that floodplains have definite conservation value for ant biodiversity. Given that this study found nine species new to science in a relatively small sampling effort, no doubt there are numerous other floodplain specialist species yet to be discovered in floodplains across the greater breadth of northern Australia. These outcomes reflect those of other biota in floodplains, and strongly suggest that efforts to intensify agriculture on north Australian floodplains need to also have effective conservation considerations if we are to achieve ecologically sustainable development.
References
Andersen AN (1993) Ants as indicators of restoration success at a uranium mine in tropical Australia. Restor Ecol 1:156–167
Andersen AN (1995) A classification of Australian ant communities, based on functional groups which parallel plant life-forms in relation to stress and disturbance. J Biogeogr 22:15–29
Andersen AN (2016) Ant megadiversity and its origins in arid Australia. Austral Entomol 55:132–137
Andersen AN, van Ingen LT, Campos RI (2007) Contrasting rainforest and savanna ant faunas in monsoonal northern Australia: a rainforest patch in a tropical savanna landscape. Aust J Zool 55:363–369
Andersen AN, Del Toro I, Parr CL (2015) Savanna ant species richness is maintained along a bioclimatic gradient of increasing latitude and decreasing rainfall in northern Australia. J Biogeogr 42:2313–2322
Andersen AN, Hoffmann BD, Oberprieler S (2018) Diversity and biogeography of a species-rich ant fauna of the Australian seasonal tropics. Insect Sci 25:519–526
Arcoverde GB, Andersen AN, Setterfield SA (2017) Is livestock grazing compatible with biodiversity conservation? Impacts on savanna ant communities in the Australian seasonal tropics. Biodivers Conserv 26:883–897
Ash AJ (2013) Factors driving the viability of major cropping investments in northern Australia—an historical analysis. CSIRO, Australia
Australian Government (2015) Our north, our future: white paper on developing northern Australia. ACT, Canberra
Ballinger A, Lake PS, Mac Nally R (2007) Do terrestrial invertebrates experience floodplains as landscape mosaics? Immediate and longer-term effects of flooding on ant assemblages in a floodplain forest. Oecologia 152:227–238
Bauer FH (1977) Cropping in north Australia: anatomy of success and failure. North Australia Research Unit, Australian National University, Darwin, p 267
Blüthgen N, Verhaagh M, Goitía W, Jaffé K, Morawetz W, Barthlott W (2000) How plants shape the ant community in the Amazonian rainforest canopy: the key role of extrafloral nectaries and homopteran honeydew. Oecologia 125:229–240
Blüthgen N, Stork NE, Fiedler K (2004) Bottom-up control and co-occurrence in complex communities: honeydew and nectar determine a rainforest ant mosaic. Oikos 106:344–358
Botha M, Siebert SJ, van den Berg J, Maliba BG, Ellis SM (2015) Plant and arthropod diversity patterns of maize agro-ecosystems in two grassy biomes of South Africa. Biodivers Conserv 24:1797–1824
Brooks TM, Mittermeier RA, Mittermeier CG, Da Fonseca GA, Rylands AB, Konstant WR, Flick P, Pilgrim J, Oldfield S, Magin G, Hilton-Taylor C (2002) Habitat loss and extinction in the hotspots of biodiversity. Conserv Biol 16:909–923
Brühl CA, Eltz T (2010) Fuelling the biodiversity crisis: species loss of ground-dwelling forest ants in oil palm plantations in Sabah, Malaysia (Borneo). Biodivers Conserv 19:519–529
Buczkowski G, Richmond DS (2012) The effect of urbanization on ant abundance and diversity: a temporal examination of factors affecting biodiversity. PLoS ONE 7(8):e41729
Calcattera LA, Cuiezzo F, Carera SM, Briano JA (2010) Ground ant diversity (Hymenoptera: Formicidae) in the Iberá nature reserve, the largest wetland of Argentina. Ann Entomol Soc Am 103:71–83
Cammeraat ELH, Risch AC (2008) The impact of ants on mineral soil properties and processes at different spatial scales. J Appl Entomol 132:285–294
Chong CS, Thomson LJ, Hoffmann AA (2011) High diversity of ants in Australian vineyards. Aust J Entomol 50:7–21
Clarke KR, Gorley RN (2003) Primer v6: user manual tutorial. Primer-e, Plymouth marine Laboratory, Plymouth
Colwell RK (2013) EstimateS-statistical estimation of species richness and shared species from samples. http://www.purl.oclc.org/estimates. Accessed 15 Aug 2017
Cowie ID, Short PS, Osterkamp-Madsen M (2000) Floodplain Flora: a Flora of the Coastal Floodplains of the Northern Territory, Australia. Flora of Australia supplementary series number 10. (ABRS: Canberra; PWCNT: Darwin.)
CSIRO (2009) Northern Australia land and water science review. CSIRO, Canberra
De la Mora A, Murnen CJ, Philpott SM (2013) Local and landscape drivers of biodiversity of four groups of ants in coffee landscapes. Biodivers Conserv 22:871–888
Del Toro I, Ribbons RR, Pelini SL (2012) The little things that run the world revisited: a review of ant-mediated ecosystem services and disservices (Hymenoptera: Formicidae). Myrmecol News 17:133–146
Evans TA, Dawes TZ, Ward PR, Lo N (2011) Ants and termites increase crop yield in a dry climate. Nat Commun 2:262
Fayle TM, Turner EC, Snaddon JL, Chey VK, Chung AYC, Eggleton P, Foster WA (2010) Oil palm expansion into rain forest greatly reduces ant biodiversity in canopy, epiphytes and leaf-litter. Basic Appl Ecol 11:337–345
Ferdinands K, Beggs K, Whitehead P (2005) Biodiversity and invasive grass species: multiple-use or monoculture? Wildl Res 32:447–457
Fernandez WD, Lange D, Pereira JM, Raizer J (2018) Ant community in neotropical agrosystems: a four-year study in conventional and no-tillage systems. Sociobiology 65:130–137
Gaigher R, Samways MJ (2010) Surface-active arthropods in organic vineyards, integreated vineyards and natural habitat in the Cape floristic region. J Insect Conserv 14:595–605
Gambold N, Woinarski JCZ (1993) Distributional patterns of herpetofauna in monsoon rain forests of the Northern Territory, Australia. Aust J Ecol 18:431–449
Glaser F (2007) Ants (Hymenoptera, Formicidae) in alpine floodplains—ecological notes and conservation aspects. Internationales LIFE-symposium, riverine landscapes, restoration—flood protection—conservation, Natur in Tirol 13—Naturkundliche Beiträge der Abteilung Umweltschutz: 147–163
Godfree R, Firn J, Johnson S, Knerr N, Stol J, Doerr V (2017) Why non-native grasses pose a critical emerging threat to biodiversity conservation, habitat connectivity and agricultural production in multifunctional rural landscapes. Landsc Ecol 32:1219–1242
Gómez C, Casellas D, Oliveras J, Bas JM (2003) Structure of ground-foraging ant assemblages in relation to land-use change in the northwestern Mediterranean region. Biodivers Conserv 12:2135–2146
Halford JJ, Fensham RJ (2014) Vegetation and environmental relationship of ephemeral subtropical wetlands in central Queensland, Australia. Aust J Bot 62:499–510
Heterick BE, Casella J, Majer JD (2000) Influence of Argentine and coastal brown ant (Hymenoptera: Formicidae) invasions on ant communities in Perth gardens, Western Australia. Urban Ecosyst 4:277–292
Hoffmann BD (2000) Changes in ant species composition and community organisation along grazing gradients in semi-arid rangelands of the Northern Territory. Rangel J 22:171–189
Hoffmann BD (2003) Responses of ant communities to experimental fire regimes on rangelands in the Victoria River district of the northern territory. Austral Ecol 28:182–195
Hoffmann BD (2010) Using ants for rangeland monitoring: global patterns in the responses of ant communities to grazing. Ecol Indic 10:105–111
Holm LG, Plunknett DL, Pancho JV, Herberger JP (1977) The world’s worst weeds. University Press of Hawaii, Honolulu
Horrocks GFB, Cunningham SC, O’Dowd DJ, Thomson JR, Mac Nally R (2012) Floodplain ants show a stronger response to an extensive flood than to variations in fallen-timber load. Austral Ecol 37:518–528
House APN, Burwell CJ, Brown SD, Walters BJ (2012) Agricultural matrix provides modest habitat value for ants on mixed farms in eastern Australia. J Insect Conserv 16:1–12
Joint Select Committee on Northern Australia (2014) Pivot north. Inquiry into the development of northern Australia—final report. The Parliament of the Commonwealth of Australia, Canberra
Klimes P, Idigel C, Rimandai M, Fayle TM, Janda M, Weiblen GD, Novotny V (2012) Why are there more arboreal ant species in primary than in secondary tropical forests? J Anim Ecol 81:1103–1112
Kondoh M (1978) A comparison among ant communities in the anthropogenic environment. Memorab Zool 29:79–92
LeBrun EG, Tillberg CV, Suarez AV, Folarait PJ, Smith CR, Holway DA (2007) An experimental study of competition between fire ants and Argentine ants in their native range. Ecology 88:63–75
Majer JD, Delabie JHC (1994) Comparison of the ant communities of annually inundated and terra firme forests at Trombetas in the Brazilian Amazon. Inst Soc 41:343–359
McKinney ML (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11:161–176
Meeson N, Robertson AI, Jansen A (2002) The effects of flooding and livestock on post-dispersal seed predation in river red gum habitats. J Appl Ecol 39:247–258
Milford ER (1999) Ant communities in flooded and unflooded riparian forest of the middle Rio Grande. Southwest Nat 44:278–286
Morán-Ordóñez A, Whitehead AL, Luck GW, Cook GD, Maggini R, Fitzsimons JA, Wintle BA (2017) Analysis of trade-offs between biodiversity, carbon farming and agricultural development in northern Australia reveals the benefits of strategic planning. Conserv Lett 10:94–104
Nielsen MG (2011) Ants (Hymenoptera: Formicidae) of mangrove and other regularly inundated habitats: life in physiological extreme. Myrmecol News 14:113–121
Oberprierler SK, Andersen AN, Moritz CC (2018) Ants in Australia’s monsoonal tropics: CO1 barcoding reveals extensive unrecognised diversity. Diversity 10:36
Perfecto I, Vandermeer J, Hanson P, Cartín V (1997) Arthropod biodiversity loss and the transformation of a tropical agro-ecosystem. Biodivers Conserv 6:935–945
Perfecto I, Mas A, Dietsch T, Vandermeer J (2003) Conservation of biodiversity in coffee agroecosystems: a tri-taxa comparison in southern Mexico. Biodivers Conserv 12:1239–1252
Petheram C, Chilcott C, Watson I, Bruce C (eds) (2018) Water resource assessment for the Darwin catchments. A report to the Australian government from the CSIRO northern Australia water resource assessment, part of the national water infrastructure development fund: water resource assessments. CSIRO, Australia
Russel-Smith J (1991) Classification, species richness, and environmental relations of monsoon rainforest vegetation in the Northern Territory. J Veg Sci 2:259–278
Shackelford GE, Steward PR, German RN, Sait SM, Benton TG (2015) Conservation planning in agricultural landscapes: hotspots of conflict between agriculture and nature. Divers Distrib 21:357–367
Solar RRC, Barlow J, Schoereder JH, Berenguer E, Ferreira JN, Gardner TA (2016) Biodiversity consequences of land-use change and forest disturbance in the Amazon: a multi-scale assessment using ant communities. Biol Conserv 197:98–107
Travis JMJ (2003) Climate change and habitat destruction: a deadly anthropogenic cocktail. Proc R Soc Lond B Biol Sci 270:467–473
Webster T, Rippin L, Morison J, Herr A, Abel N, Taylor B, Clark E, Stone P (2009) Chapter 10: irrigated agriculture: development opportunities and implications for northern Australia. In: Stone P (ed) Northern Australian land and water science review. Northern Australia Land and Water Taskforce and CSIRO, Canberra
Wilson P, Ringrose-Voase AJ, Jacquier DW, Gregory L, Webb M, Wong MTF, Powell B, Brough D, Hill J, Lynch B, Schoknecht N, Griffin EA (2009) Land and soil resources in northern Australia. In: Stone P (ed) Northern Australian land and water science review 2009. Northern Australia Land and Water Taskforce and CSIRO, Canberra, pp 1–47
Wilson KA, Meijaard E, Drummond S, Grantham HS, Boitani L, Catullo G, Christie L, Dennis R, Dutton I, Falcucci A, Maiorano L (2010) Conserving biodiversity in production landscapes. Ecol Appl 20:1721–1732
Woinarski JCZ (1993) A cut and paste community: birds of monsoon rainforests in Kakadu National Park, Northern Territory. Emu 93:100–120
Woinarski JCZ, Braithwaite RW (1991) Wildlife of Kakadu Stage III: a synthesis. Report to ANPWS. CSIRO, Darwin
Woinarski J, Mackey B, Nix H, Traill B (2007) The nature of northern Australia: its natural values, ecological processes and future prospects. Australian National University Press, Canberra
Acknowledgements
We thank Magen Pettit for overseeing the laboratory work, and Alan Andersen, Chris Chilcott and two anonymous reviewers for commenting on the draft manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David Hawksworth.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hoffmann, B.D., Ivaldi, F., Perez, A. et al. Are north Australian floodplain ant communities comprised of a distinct fauna, or a subset of the broader savanna fauna? Implications for conservation. Biodivers Conserv 28, 3465–3477 (2019). https://doi.org/10.1007/s10531-019-01832-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-019-01832-6