Abstract
Developing nations in tropical regions harbour rich biological resources on which humans depend for food, income and employment, yet data to aid their management is often lacking. In West Africa, the diversity and fisheries of elasmobranchs are poorly documented, despite them being known to be economically important and vulnerable to overexploitation. Rapid qualitative surveys of fish processing and landing sites in The Gambia from 2010-2018 revealed valuable new data on species composition, biology, relative abundance, fisheries and utilisation by humans. Diversity and abundance was dominated by batoids, with a major component comprising a large guitarfish (Glaucostegus cemiculus) that was apparently targeted, and a small whipray (Fontitrygon margaritella). Nearly all taxa recorded are classified by the IUCN Red List as Critically Endangered (angel sharks Squatina spp.), Endangered, Data Deficient, or Not Evaluated; several were endemic, of exceptional evolutionary distinctness, cryptic, possibly undescribed, and rare (including stingray Hypanus rudis not apparently recorded since description in 1870). Significant threats to biodiversity, coastal livelihoods and possibly food security are identified based on the apparent importance of elasmobranch fisheries and processing; the known inability of key taxa to withstand intensive fisheries; ‘fishing down the food web’ by intensive utilisation of F. margaritella; and the absence or rarity of previously common elasmobranch species that may be severely depleted in the region. This study provides data that may act as a starting point to aid sustainability accreditation of local fin-fisheries, and demonstrates the value of inexpensive and low-resolution data collection in developing countries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Much tropical biodiversity is located in developing nations, where the threat of overexploitation is greatest (Myers et al. 2000). Least Developed Countries (LDCs) are ‘low-income countries confronting severe structural impediments to sustainable development’ (UNDESA 2018). In West Africa, the adjoining LDCs of Mauritania, Senegal, The Gambia, Guinea Bissau, Guinea, Sierra Leone, and Liberia span a vast area and over 2000 km of continuous coastline. Unsustainable resource exploitation in the region is reflected in marked declines of terrestrial and freshwater biodiversity, while overfishing of marine species threatens the food security and coastal livelihoods of up to 400 million people (Mallon et al. 2015; Polidoro et al. 2016). Artisanal marine fisheries in the region are recognised as a priority in terms of overexploitation, dependency of human communities, and need for better data and management (Belhabib et al. 2018).
The Gambia is the smallest country on mainland Africa, entirely surrounded by Senegal except for its coast on the eastern Atlantic. The country has a human development index ranking of 160 (out of 179 nations), and food security is a major issue confronting the country’s population, which is highly dependent on rice imports and therefore vulnerable to fluctuations in global markets (Moseley et al. 2010; FAO 2018a). Fish is the main supplier of animal protein in the diets of most Gambians, while fisheries and related activities are the main source of income for coastal fishing communities and support the livelihoods of an estimated 200,000 people (United Nations 2014).
Chondrichthyan fishes comprising sharks, rays (batoids) and chimaeras have been identified as having a higher extinction risk than most other vertebrates (Dulvy et al. 2014). Limiting life characters such as relatively large size, late maturity and few young are unable to withstand intensive targeted and bycatch fisheries, and five of the seven most threatened families are batoids found in warm shallow coastal waters (Dulvy et al. 2014). Regional extinctions of sawfishes on a global scale are now relatively well documented (Thorson 1982; Robillard and Séret 2006; Moore 2015; Leeney and Downing 2016), while severe declines of other batoid groups such as guitarfishes have occurred but are less well known (Moore 2017).
The West Africa region is home to a specialised elasmobranch industry of fisheries, processing and trade, which was started in The Gambia by Ghanaian immigrants (Walker et al. 2005; Diop and Dossa 2011). Furthermore, West Africa also has been identified as one of five global hotspots to prioritize for conservation of chondrichthyans based on species richness, endemism and evolutionary distinctness, and as a priority region for the conservation of Critically Endangered angel sharks (Gordon et al. 2017; Stein et al. 2018). Despite this importance, little is known about elasmobranch biodiversity and fisheries in West Africa, although there has been some work focusing on reproductive biology of a few species (Capapé et al. 2002; Seck et al. 2004; Valadou et al. 2006). Prompted by the 2001 launch of a Plan of Action for the conservation and management of shark populations by a regional fishery organisation (the Sub-Regional Fisheries Commission (SFRC/CSRP) elasmobranch surveys were undertaken at markets and landing sites in SFRC member states countries (Mauritania, Senegal, The Gambia, Guinea Bissau, Guinea, Sierra Leone and Cape Verde). These provide the only regional overview of elasmobranch fisheries (Diop and Dossa 2011). Although these authors provided relative abundance of elasmobranch species by country, they did not provide detailed country-specific data, and data derived from fisheries observers and officers in the report may be of questionable reliability (BS, pers. obs.). An accurate assessment of trends is also made difficult by landings data that are erratic and likely to be unreliable. Between 1990 and 2016 Gambia reported an average of 911 t of sharks and rays annually (3.1% of total marine fish production, MFP), but this varied wildly from 194 to 4,022 t (and 1 to 13.2% of MFP) (FAO 2018b) and was notably different to other reports (Saine 2011). Studies of artisanal fish processing in Gambia suggest that smoked and dried elasmobranchs are of economic importance to coastal communities, mostly for export and particularly to Ghana, but also for some local consumption (Mbenga 1996; Njai 2000; Saine 2011). Very limited useful data is available on elasmobranch population trends in the Gambia, but declines in shark fisheries between 2001 and 2011 have been noted (Saine 2011), as has the severe decline of sawfishes since the mid-1970s (Leeney and Downing 2016). West Africa has recently been recognised as a global priority in terms of data collection and management of chondrichthyan fisheries (Dulvy et al. 2017).
Given the apparent importance of the region to elasmobranch biodiversity, fisheries and the people reliant on them, data on aspects such as species composition, relative abundance, and utilisation are essential in developing strategies for biodiversity conservation, fisheries management and food security. While a suite of high-resolution data would be ideal, obtaining these are not feasible in developing countries or LDCs with no, or very limited, local resources. In these situations, inexpensive techniques such as interviews and community-based monitoring can provide essential data on poorly-known marine vertebrate groups of economic and/or conservation importance, in Africa and elsewhere (Aylesworth et al. 2017; Humber et al. 2017; Braulik et al. 2018). For elasmobranchs in the tropics and subtropics, surveys of fish markets, landing and processing sites have provided a relatively inexpensive source of invaluable data on biodiversity and fisheries (e.g. White and Dharmadi 2007), although surveys in sub-Saharan Africa are, to date, very limited (Diop and Dossa 2011; Barrowclift et al. 2017). With the aim of addressing the significant current data gaps on biodiversity, relative abundance and utilisation, here we provide results of rapid, low-resolution surveys of elasmobranch landing and processing sites in The Gambia.
Methods and materials
Study area
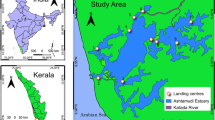
The Gambia’s physical environment is dominated by the River Gambia and its estuary, which joins the exposed coast of the tropical eastern Atlantic (Fig. 1). A long dry season extends from November to May, with a rainy season from June/July to October. The study was undertaken at fisheries landing sites and associated processing communities on the coast (Fig. 1). Artisanal fisheries are the most important of the sector in the country, and these are conducted by traditional open wooden pirogues (approximately 10–15 m length) with outboard motors, launched from the beach. Major fisheries are based on the use of encircling nets for small pelagics (e.g. bonga shad Ethmalosa fimbriata and Sardinella spp.), and set bottom gillnets for a range of demersal taxa such as sole (Soleidae) and tonguefishes (Cynoglossidae) (FAO 2007; UN 2014, unpublished data). Landing sites often have adjacent areas for processing of fish for consumption or export, such as air-drying racks and smokehouses (FAO 2007; United Nations 2014; unpublished data). Initial surveys identified Ghanatown and Gunjur as having a significant elasmobranch component in landings and/or processing areas, so later surveys focused on these locations.
Surveys
Surveys were conducted annually in the dry season (March/April) of 2011–2018, and also in the wet season (July) of 2010. Surveys typically comprised small groups of undergraduate students visiting sites once every few days over a two-week period, photographing all elasmobranchs for later collation and review. A more detailed survey was undertaken in the dry season of 2014 (30th March–11th April) when the students were joined by one of the authors (ABMM) to verify identifications and collect further information. From 2014 onwards, the surveys were assisted by a local interpreter from the Ministry of Fisheries.
Identification, enumeration and measurement
Field identifications were initially made with Séret (2006), supplemented by Compagno and Roberts (1984) to differentiate similar-sized Fontitrygon margarita and F. margaritella; all batoid identifications and nomenclature were later confirmed against Last et al. (2016a). Two groups of batoids, cownose rays (Rhinopteridae) and butterfly rays (Gymnuridae) were usually only identified to genus level because of their cryptic nature combined with dessication and damage.
Detailed enumeration, measurement and sexing of a large proportion of elasmobranchs seen was not feasible, as they were often present as large stacked piles of dried specimens ready for sale or export (e.g. at Gunjur). Qualitative and semi-quantitative observations were recorded, and subsamples of more accessible individuals (e.g. while on drying racks, or landed fresh) were measured. For intact sharks, guitarfishes, skates and electric rays total length (TL) was measured, with disk width (DW) measured for other batoids. Measurement of intact blackchin guitarfish Glaucostegus cemiculus was usually not possible as these were butchered into four or more sections by fishers immediately upon landing for drying purposes. To estimate size, intact head sections allowed measurement of pre-orbital length (POL; snout tip to perpendicular line drawn between anterior margin of orbits) in the field. The TL was then estimated (ETL) by multiplying POL by a factor (6) based on POL averaging about 16.6% of TL in published studies (Ben Souissi et al. 2007 (inc. Figure 4a); Séret et al. 2016a) and 25 individuals where both TL and POL was measured in the Gambia in March 2018.
Results and discussion
Overview
At least 27 elasmobranch species were recorded, comprising 9 sharks and 18 batoids; the most important family were stingrays (Dasyatidae) with 7 species (Table 1). Batoids also dominated abundance and biomass. Of the species regularly occurring in significant quantity the most abundant was the pearl whipray Fontitrygon margaritella (Dasyatidae) and the blackchin guitarfish Glaucostegus cemiculus (Glaucostegidae). Common guitarfish Rhinobatos rhinobatos (Rhinobatidae) occurred regularly throughout surveys but in smaller numbers, and cownose ray Rhinoptera sp. (Rhinopteridae) occurred in significant quantity once. Most other species occurred occasionally or rarely.
Key commercial species
Glaucostegus cemiculus was commonly recorded at the landing and processing site of Ghanatown in all years, including the wet season survey of July 2010. It appeared that a targeted fishery operated for this species at this site, where it was often observed as the dominant elasmobranch being landed fresh and butchered into sections, or drying on nearby racks (Fig. 2a). Dorsal and upper caudal fins were always removed by fishers, apparently before or just after landing (Fig. 2b; also see Moore 2017 Fig. 1); the rostral cartilage of individual fish was also cut by fishers with a distinctive mark, presumably to identify ownership on drying racks. A total of 314 head sections measured at Ghanatown on two separate occasions in March and April 2014 provided ETL ranging from 102–204 cm, with 141–150 cm ETL the most frequently occurring size class (Fig. 3). A total of 26 whole fresh individuals at Ghanatown on 27th March 2018 comprised twenty females (128.6–240.1 cm TL, mean 181.4 cm TL) and five males (153.1–195.6 cm TL, mean 167.6 cm TL) (Fig. 2b).
Size-frequency (Estimated Total Length; see “Methods”) distribution of blackchin guitarfish Glaucostegus cemiculus on 31st March and 11th April 2014 at Ghanatown, The Gambia. Arrows indicate approximate size by which most or all males and females are mature, based on other studies of this species in West Africa (Seck et al. 2004; Valadou et al. 2006; Séret et al. 2016a)
The maximum TL recorded in the present study (240.1 cm) was slightly smaller than the maximum TL of 245 cm recorded from a smaller sample size (n = 79) of landings into Senegal in 1994–2000 (Seck et al. 2004) and smaller than the maximum reported TL of 265 cm (Séret et al. 2016a). A significant decline in the average size of this species in Mauritania was reported between 1998 and 2007 (Diop and Dossa 2011). The size at maturity for G. cemiculus has been reported as ranging between 153 and 174 cm TL for females and 138 and 154 cm TL for males (Seck et al. 2004; Valadou et al. 2006; Séret 2016a). Based on this, many of the (unsexed) individuals in the present study were likely to be immature (Fig. 3), consistent with findings from a larger study in Mauritania (Valadou et al. 2006).
Glaucostegus cemiculus has previously been reported as frequent or abundant in much of the region (Diop and Dossa 2011). The Red List assessment for G. cemiculus is Endangered based on biological vulnerability (e.g. low fecundity), exposure to intensive fisheries throughout its range, high value, and evidence of declines (Notarbartolo di Sciara et al. 2016; based on a 2007 assessment). These declines have included extinction in the northern Mediterranean, a decrease in abundance in Guinea-Bissau, and an observed decrease in the size of landed individuals from adults to juveniles in a targeted fishery in Mauritania between 1998 and 2003 (Notarbartolo di Sciara et al. 2016).
The pearl whipray Fontitrygon margaritella was commonly recorded in all years and sometimes extremely abundant at processing sites, indicating intensive utilisation. As a typical example, at Gunjur on 1–4 April 2014 several piles of hundreds of dried elasmobranchs consisting mostly of F. margaritella (Fig. 4a) were recorded, along with several drying racks totalling an estimated 2–3000 elasmobranchs, almost entirely F. margaritella (Fig. 4b); the latter included numerous small (ca. 10 cm DW) individuals. A measured subsample of 119 drying F. margaritella on a different occasion (Ghanatown, 7th April 2014) all but one of which were female, showed a modal and maximum DW of 31 and 34 cm respectively (Fig. 5), expanding the known size of this species (30 cm, Séret 2016b). In marked contrast to our findings, this species (as D. margaritella) was not recorded at all in previous surveys of the region except in Guinea where it was ‘very rare’ (Diop and Dossa 2011). The current Red List assessment for F. margaritella is Data Deficient (Marshall and Cronin 2016, accessed in 2007) as knowledge of fisheries interactions at the time was uncertain, and it was thought that its small size may minimise targeted capture. Our data suggest that, targeted or otherwise, F. margaritella is subject to intensive and sustained fisheries mortality at all life history stages in The Gambia and in urgent need of re-assessment.
Critically endangered species
A single individual each of two Critically Endangered angel shark species were recorded. In April 2017 a smoothback angel shark Squatina oculata of an estimated 100 cm TL was found split and dried with pectoral, dorsal and caudal fins removed (Table 1, Fig. 6a); in March 2018, a single whole female of the sawback angelshark Squatina aculeata of an estimated 150 cm TL was recorded, which also had dorsal and caudal fins removed (Table 1, Fig. 6b). All three species of Squatina from the eastern Atlantic are Critically Endangered based on former abundance, severe fishery-driven declines and rarity of recent records, and quantifying landings in West Africa is a conservation priority given minimal current knowledge (Morey et al. 2007a, b; Gordon et al. 2017). Diop and Dossa (2011) recorded S. oculata and S. aculeata as ‘frequent’ and ‘quite frequent’ respectively in mainland SFRC countries, but this is in marked contrast to the narrative from other data sources (e.g. from Russian trawl surveys and Senegalese artisanal fishers) which reported both species as being common around the 1970 s and 1980 s, but as extremely rare now. Regional research surveys recorded very sparse occurrence, with the last Gambian records for S. oculata consisting of six individuals between 1986 to 2000, and of a single S. aculeata in 1998 (Morey et al. 2007a, b). The removal of fins recorded in our surveys is of interest given that the shark fin trade has not previously been identified as a specific threat to Squatina species (Clarke et al. 2007; Morey et al. 2007a, b; Gordon et al. 2017).
Endangered batoid species
The common guitarfish Rhinobatos rhinobatos (Rhinobatidae) was never recorded in abundance, although single individuals or small numbers were frequently recorded at landing and processing sites in all years, either as fresh landings or on drying racks (Table 1). While fins were removed by fishers on some fresh specimens (including small individuals of ca. 50 cm TL) this practice did not appear to be consistent (or as consistent as that observed for G. cemiculus) as intact fins were sometimes observed on drying whole specimens. Rhinobatos rhinobatos was previously described as abundant and one of the most common elasmobranchs in the region, frequently or very frequently captured in the Gambia and all other mainland SFRC countries (Notarbartolo di Sciara et al. 2007; Diop and Dossa 2011; Séret 2016a). Analysis of trawl data from Mauritania revealed strong declines in R. rhinobatos abundance over the period 1990–2010 (Meissa and Gascuel 2015). The global assessment for Rhinobatos rhinobatos is Endangered based on past and suspected future declines, such as evidence of regional extinction in the northern Mediterranean (Notarbartolo di Sciara et al. 2007).
The medium-sized daisy whipray Fontitrygon margarita was only recorded in small numbers on two occasions (Table 1) at Ghanatown amongst fresh landings dominated by G. cemiculus. Regional surveys by Diop and Dossa (2011) only recorded this species in Guinea where it was ‘very rare’, and it is currently considered Endangered (Compagno and Marshall 2016, based on a 2004 assessment) given larger size, low fecundity and apparent declines in abundance.
Species of exceptional evolutionary distinctness
Individuals of the striped panray Zanobatus schoenleinii (Zanobatidae) were frequently observed (at least in 2011, 2014, 2015 and 2018) at landing sites such as Gunjur, often in some abundance, consistent with surveys of the wider region (Diop and Dossa 2011, Séret 2016c). Whole fresh or dessicated individuals were always found discarded on the ground around fishing communities indicating it is not consumed or used in The Gambia, which contrasts with reported utilization in Guinea (Valenti 2009). This species has recently been identified as the single most evolutionary distinct species of all chondrichthyan fishes (Stein et al. 2018), and is currently assessed as Data Deficient (Valenti 2009). A second species of the genus Zanobatus, Z. maculatus, was recently described from the Gulf of Guinea (Séret 2016d), although its true distribution in West Africa needs to be determined.
Rarely recorded endemic species
Two electric rays (Torpedinidae) endemic to West Africa were found discarded whole on the beach, comprising one individual each of the rosette torpedo Torpedo bauchotae and West African torpedo T. mackayana (Table 1). Both species are Data Deficient and rarely recorded, with T. bauchotae known from only a few specimens (Pheeha and Cronin 2009; Séret et al. 2009; Dossa and Diop 2011; de Carvalho et al. 2016). In addition, two individuals of a scyliorhinid shark assumed to be the regionally endemic West African catshark Scyliorhinus cervigoni (Table 1) had been skinned and had fins drying on nearby racks. This species is very poorly known and was either absent or very rare in previous surveys (Burgess 2006; Diop and Dossa 2011).
Data Deficient Species
A suite of at least four Data Deficient stingrays (Dasyatidae) recorded occasionally or rarely (whole and fresh or dried) comprised brown stingray Bathytoshia lata, smalltooth stingray Hypanus rudis, marbled stingray Dasyatis marmorata (Notarbartolo di Sciara et al. 2009), common stingray D. pastinaca (Serena et al. 2009a) and round stingray Taeniurops grabatus (Serena et al. 2009b) (Table 1).
While B. lata is currently considered Least Concern, the 2007 assessment predates taxonomic revision and therefore requires updating; Eastern Atlantic populations of B. lata were known as Dasyatis centroura (Ebert et al. 2016; Last et al. 2016b). Previous surveys of the region only recorded D. centroura in Guinea, as ‘very rare’ (Diop and Dossa, 2011), possibly due to difficulties discriminating this species. The taxonomic status of Hypanus rudis is uncertain, and our records are of note as this species is known only from the original description from the Nigerian coast, with the holotype probably lost and no records since (Last et al. 2016b; Séret et al. 2016b). Separate photographic records of very large stingrays (>200 cm DW) obtained locally may be H. rudis (Ruth H. Leeney, unpublished data). Only two individuals of the distinctive D. marmorata were recorded which is consistent with rarity in surveys of the region by Diop and Dossa (2011), yet it was apparently not uncommon off Mauritania in 1998–2002 where over 1000 individuals were recorded in surveys of fish landing sites (Valadou et al. 2006). Dasyatis pastinaca is difficult to discriminate from the reportedly sympatric D. tortonesei (Last et al. 2016b), and there may be further cryptic diversity within D. pastinaca (BS pers. obs.). In previous surveys of the region D. pastinaca was reported as uncommon, and not recorded from Senegal or Gambia (Diop and Dossa 2011). While the global Red List Assessment for D. pastinaca is Data Deficient (Serena et al. 2009a), a more recent assessment for the Mediterranean was Vulnerable, based on suspected declines (Serena et al. 2016). A single Taeniurops grabatus recorded in our survey reflects an absence of mainland records in previous regional surveys (Diop and Dossa 2011).
Only two individuals of the bullray Aetomylaeus bovinus were recorded (Table 1). In 1989 this species (reported as Pteromylaeus bovinus) was one of the most common elasmobranchs in research fishing off Guinea-Bissau by a Portuguese institute, and in 2002 it was common off Senegal (Diop and Dossa 2011; Wintner 2016). However it was not recorded at all in the region in more recent surveys, excepting in Guinea where it was ‘very rare’. The current global assessment for this species is Data Deficient, but is considered Critically Endangered in the Mediterranean where severe declines are suspected (Walls and Buscher 2016; Wintner 2016).
Not Evaluated species
At least four taxa were recorded that have not been evaluated by the IUCN Red List, consisting of species that have only recently been described or are undergoing taxonomic treatment. Dogfish sharks (Squalidae) were recorded on one occasion in 2011 (Table 1) but could not be confidently identified to species level as most individuals had pectoral, dorsal, and lower and upper caudal fin lobes removed. The taxonomic status of Squalus in the eastern Atlantic is highly complex (Verissimo et al. 2016) with revision ongoing; several species could potentially occur off West Africa, including the newly described Smith’s dogfish shark Squalus margaretsmithae (Viana et al. 2017) and S. probatovi, a valid species known from Angola that has recently been resurrected (Viana and Carvalho 2018).
Two species within the cryptic brown skate Raja miraletus species complex were occasionally recorded: the newly described R. parva, and the undescribed R. cf. miraletus (Last and Séret 2016) (Table 1). The apparent rarity of species in this complex in our surveys is notably different to it (as R. miraletus) being reported as ‘very frequent’ and one of the commonest elasmobranchs of the region (Diop and Dossa 2011).
As noted, most processed specimens of butterfly rays (Gymnuridae) were only identified to genus (Gymnura sp.), although some fresh individuals were identified as the newly described G. sereti (Yokota and de Carvalho 2017). A larger species, G. altavela is also known to occur on the coast of West Africa (Yokota et al. 2016) and larger individuals we recorded are likely to have been this species (Table 1). While our surveys recorded Gymnura in most years they were only ever present as one, or a few (<10), individuals fresh or on drying racks and assumed to be consumed for food. In previous surveys of the region, Diop and Dossa (2011) only recorded Gymnura in Guinea where they were ‘very rare’. While the global assessment for G. altavela is Vulnerable (Vooren et al. 2007), Mediterranean populations are Critically Endangered based on large size, disappearance from historic range and suspected significant declines in abundance (Walls et al. 2016).
Cownose rays (Rhinopteridae) were also only identified to genus (Rhinoptera) as cryptic diversity exists in West Africa, with at least one undescribed species possibly present (Last et al. 2016c). Rhinoptera were only recorded occasionally as single or a few individuals in most years, but in 2015 a total of 961 individuals were recorded on drying racks at Gunjur and Ghanatown (Fig. 7). Based on size at maturity (LT50) of 77.5 cm and 80.2 cm DW for male and female R. marginata respectively from Mauritania (Valadou et al. 2006) only 1% or less of this catch was of a size likely to be mature. Significant conservation concern has been raised for Rhinoptera species due to exceptionally low fecundity of one pup and susceptibility to intensive fisheries from aggregation in large schools. A congener in the western Atlantic has reportedly been extirpated from southern Brazil due to fisheries (Vooren and Lamónaca 2004).
Disk width-frequency distribution of 961 cownose ray Rhinoptera sp., Ghanatown and Gunjur, The Gambia, April 2015. Arrows indicate approximate size at 50% maturity for males and females of Rhinoptera bonasus from Mauritania (Valadou et al. 2006)
Other shark species
Milk shark Rhizoprionodon acutus were occasionally recorded as single or a few individuals in all years, with a single photograph of numerous individuals on drying racks from the 2010 survey possibly indicating greater abundance in the wet season. It was recorded in abundance in The Gambia in 2010–2011 (Bojang 2011); this species is considered Least Concern (Simpfendorfer 2003). The remaining shark species were recorded occasionally or rarely (Table 1) and included young scalloped hammerhead Sphyrna lewini, which is Endangered (Baum et al. 2007). Other shark taxa recorded were smoothhound Mustelus mustelus (Vulnerable, Serena et al. 2009c), and barbelled houndshark Leptocharias smithii and bull shark Carchahinus leucas, both of which are Near Threatened (Compagno 2005; Simpfendorfer and Burgess 2009).
Value of the study approach
The core of this survey was essentially the collation of many hundreds of photographs by undergraduate students with no previous experience of elasmobranchs, yet it has provided important insights into several poorly known aspects of their diversity and fisheries in West Africa. These include (1) expanding knowledge on diversity, biology and species composition, including new data on size (F. margaritella) and notable records of Critically Endangered, rare and poorly known species (e.g. Squatina spp., Hypanus rudis); (2) documenting biological indicators of the fishery such as relative abundance and, most notably, immature juveniles forming a significant component of catches of key commercial species (G. cemiculus, F. margaritella, Rhinoptera sp.); (3) documenting fisheries and utilisation, including apparently targeted fisheries for high-value species (G. cemiculus), significant and previously undocumented fisheries mortality for small species, both for consumption (F. margaritella) and as discards (Z. schoenleinii), and processing of shark fins in taxa not generally considered important in the fin trade (Squalus spp., Squatina spp., Scyliorhinus cervigoni); and (4) providing evidence of the apparent importance of specialised elasmobranch industries to coastal livelihoods in The Gambia. Despite its largely qualitative and low-resolution nature, the study has clearly demonstrated a valuable and cost-effective approach to elasmobranch data collection in the developing world, and more broadly helped aid the call for more data on artisanal fisheries in the region (Belhabib et al. 2018).
Rarity and absence
Our surveys sampled an intensive shallow water demersal fishery, which in some cases targeted shark-like batoids (guitarfishes), and was dominated by batoids. Several taxa were recorded in our surveys only rarely, and several species that might have been expected to occur did not. This rarity is likely due to reflect the influence of many factors including bias from sampling and fisheries practices, and, in some cases, true abundance. The most significant influence is likely to be due to nearly all our surveys being limited to March and April, under-sampling taxa present in other months. Further, as the fishery appeared to operate mostly in shallow waters it is unlikely to effectively sample taxa generally found in deeper, cooler water in the tropics (e.g. Squalus and Raja), and at-sea discarding practices of undesirable species (e.g. Torpedo) may also have contributed to rarity of records. Some rarity might also be explained by our study location being towards the edge of a species’ known distribution (e.g. T. bauchotae, F. margarita).
Even taking these factors into account, a number of demersal elasmobranch species that we recorded only rarely have been (1) previously reported as abundant in the region (2) have shown evidence of fishery-driven declines elsewhere in their range and (3) would be expected to be caught in the fishery; examples include angel sharks (Squatina sp.) and bullray Aetomylaeus bovinus. Similarly, it was notable that over 7 years of survey we failed to record evidence of a single individual of a number of highly threatened species of large sharks and shark-like batoids whose distribution includes The Gambia that are known or suspected to have experienced severe declines (e.g. great hammerhead Sphyrna mokarran, sawfishes Pristis sp., African wedgefish Rhynchobatus luebberti)(Robillard and Séret 2006; Denham et al. 2007; Diop and Dossa 2011; Leeney and Downing 2016; Moore 2017).
Risks to biodiversity, coastal livelihoods and food security
In addition to those may have already been severely depleted, it is highly likely that the trajectory of relatively abundant species we recorded will follow a similar path in the near future due to their limiting life history characters and intensive artisanal fisheries. As noted above the large-bodied G. cemiculus is known to have declined elsewhere in its range, reflecting reported declines of larger guitarfishes (Moore 2017). The intensive utilisation of the small and apparently previously unused stingray F. margaritella (for which little or no biological information exists on which to base extinction risk assessment) may represent a case of fishing down marine food webs, and indicate that exploitation is unsustainable (Pauly et al. 1998). Indeed, some fishers we spoke with indicated the batoid fishery had developed in response to a decline of the shark fishery. As well as a depletion of elasmobranch abundance, the fishery represents a risk to taxa that are almost entirely unknown, including potentially undescribed species. Collapse of regional elasmobranch fisheries could have far-reaching biodiversity consequences; Brashares et al. (2004) demonstrated increased hunting of terrestrial wildlife for bushmeat in years of poor fish supply in West Africa.
Our study identified fisheries and industries that appear to be wholly or largely reliant on elasmobranchs, although further research on socio-economic aspects is clearly needed. Given the known importance of fishing to dietary protein, income and employment in The Gambia and its limited and vulnerable food production options (Moseley et al. 2010; United Nations 2014; FAO 2018a), further depletions or collapse of elasmobranch populations could have far-reaching consequences for humans. Overexploitation and declines of sharks have been highlighted as one of the factors in a potential food security crisis in Madagascar (Le Manach et al. 2012).
Challenges and solutions
West African fisheries in general face multiple significant challenges, including strong declines in artisanal catch per unit effort (Belhabib et al. 2018), intensive and often unreported overfishing by foreign interests including the EU and China (Pala 2013; Ramos and Grémillet 2013), open access waters intensively exploited by migrant fishers (Binet et al. 2012) and predicted significant decline in fisheries production from climate change (Lam et al. 2012). In this context, solutions to unsustainable elasmobranch fisheries may not seem attainable. However, in The Gambia elasmobranch fisheries may be being supported by a cycle of loans and debt, with fishers apparently willing to seek alternative livelihoods if capital to start other businesses was available (Mendy 2011); this may present opportunities for conservation intervention. The smoking of small pelagic fishes for export has also been proposed as an alternative livelihood (Diop and Dossa 2011); but this comes with its own conservation and sustainability issues including use of mangrove wood and the overexploited status of small pelagics (Diop and Dossa 2011; Polidoro 2016).
One possible solution to help alleviate risks to both elasmobranchs and human communities is the development of accredited fisheries. The current bycatch of guitarfishes and other threatened elasmobranchs may represent a constraint to social and economic development in The Gambia by preventing access to high-value eco-labelled export markets. The bottom gillnet sole fishery is of major interest to EU markets and retailers, but has previously failed Marine Stewardship Council sustainability accreditation pre-assessment on aspects including a lack of catch-specific information on bycatch species (including ‘high risk’ guitarfish) (Gabis et al. 2012; Marine Stewardship Council 2013; Coastal Resources Centre 2014). The current paper goes some way to providing these missing data.
References
Aylesworth L, Phoonsawat R, Suvanachai P, Vincent ACJ (2017) Generating spatial data for marine conservation and management. Biodivers Conserv 26:383–399. https://doi.org/10.1007/s10531-016-1248-x
Barrowclift E, Temple AJ, Stead S, Jiddawi NS, Berggren P (2017) Social, economic and trade characteristics of the elasmobranch fishery on Unguja Island, Zanzibar, East Africa. Mar Policy 83:128–136. https://doi.org/10.1016/j.marpol.2017.06.002
Baum J, Clarke S, Domingo A, Ducrocq M, Lamónaca AF, Gaibor N, Graham R, Jorgensen S, Kotas JE, Medina E, Martinez-Ortiz J, Monzini Taccone di Sitizano J, Morales MR, Navarro SS, Pérez-Jiménez JC, Ruiz C, Smith W, Valenti SV, Vooren CM (2007) Sphyrna lewini. The IUCN Red List of Threatened Species 2007: e.T39385A10190088. http://dx.doi.org/10.2305/IUCN.UK.2007.RLTS.T39385A10190088.en Accessed 13 Feb 2018
Belhabib D, Greer K, Pauly D (2018) Trends in Industrial and Artisanal Catch Per Effort in West African Fisheries. Conserv Lett 11:1–10. https://doi.org/10.1111/conl.12360
Ben Souissi J, Golani D, Mejri H, Ben Salem M, Capape C (2007) First confirmed record of the Halave’s Guitarfish, Rhinobatos halavi (Forsskål, 1775) (Chondrichthyes: Rhinobatidae) in the Mediterranean Sea with a description of a case of albinism in elasmobranchs. Cah Biol Mar 48:67–75
Binet T, Failler P, Thorpe A (2012) Migration of Senegalese fishers: a case for regional approach to management. Maritime Studies 11:1. https://doi.org/10.1186/2212-9790-11-1
Bojang F (2011) Reproduction of sharks and rays in Gambian sea waters. Abstract page 28, in: Books of abstracts, Colloque international sur la conservation et la gestion durable des populations de requins en Afrique de l’Ouest: bilan et perspectives, Dakar (Senegal), 25–27 July 2011, Sub-Regional Fisheries Commission
Brashares JS, Arcese P, Sam MK, Coppolillo PB, Sinclair ARE, Balmford A (2004) Bushmeat Hunting, Wildlife Declines, and Fish Supply in West Africa. Science 306:1180–1183. https://doi.org/10.1126/science.1102425
Braulik GT, Kasuga M, Wittich A, Kiszka JJ, MacCaulay J, Gillespie D, Gordon J, Said SS, Hammond PS (2018) Cetacean rapid assessment: an approach to fill knowledge gaps and target conservation across large data deficient areas. Aquat Conserv 28:216–230. https://doi.org/10.1002/aqc.2833
Burgess GH (2006) Scyliorhinus cervigoni. The IUCN Red List of Threatened Species 2006: e.T60234A12331467. http://dx.doi.org/10.2305/IUCN.UK.2006.RLTS.T60234A12331467.en. Accessed 05 Feb 2018
Capapé C, Seck AA, Gueye-Ndiaye A, Diatta Y, Diop M (2002) Reproductive biology of the smoothback angel shark, Squatina oculata (Elasmobranchii: Squatinidae), from the coast of Senegal (eastern tropical Atlantic). J Mar Biol Assoc UK 82:635–640. https://doi.org/10.1017/S0025315402005994
Clarke SC, Milner-Gulland EJ, Cemare TB (2007) Social, economic, and regulatory drivers of the shark fin trade. Mar Resour Econ 22:305–327
Coastal Resource Centre (2014) The Gambia Sole Fishery: Towards MSC Certification and Use of an Eco-label —An FIP-like Process. http://www.crc.uri.edu/download/RKF13_GambiaSoleFisheryMSCCert.pdf Accessed 02 Feb 2018
Compagno LJV (2005) Leptocharias smithii. The IUCN Red List of Threatened Species 2005: e.T39350A10212293. http://dx.doi.org/10.2305/IUCN.UK.2005.RLTS.T39350A10212293.en. Accessed 02 Feb 2018
Compagno LJV, Marshall LJ (2016) Fontitrygon margarita. The IUCN Red List of Threatened Species 2016: e.T161495A104172339. http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T161495A104172339.en. Accessed 10 Aug 2017
Compagno LJV, Roberts T (1984) Marine and freshwater stingrays (Dasyatidae) of west Africa, with description of a new species. Proceedings of the California Academy of Sciences 43:283–300
de Carvalho MR, Last PR, Séret B (2016) Torpedo rays, family Torpedinidae. In: Last PR, White WT, de Carvalho MR, Séret B, Stehmann MF, Naylor GJP (eds) Rays of the world. CSIRO Publishing, Clayton South
Denham J, Stevens JD, Simpfendorfer C, Heupel MR, Cliff G, Morgan A, Graham R, Ducrocq M, Dulvy NK, Seisay M, Asber M, Valenti SV, Litvinov F, Martins P, Lemine Ould Sidi M, Tous P, Bucal D (2007) Sphyrna mokarran. The IUCN Red List of Threatened Species 2007: e.T39386A10191938. http://dx.doi.org/10.2305/IUCN.UK.2007.RLTS.T39386A10191938.en. Accessed 29 May 2018
Diop M, Dossa J (2011) 30 years of shark fishing in West Africa. Development of fisheries, catch trends, and their conservation status in Sub-Regional Fishing Commission member countries. FIBA Publications
Dulvy NK, Fowler SL, Musick JA, Cavanagh RD and others. (2014) Extinction risk and conservation of the world’s sharks and rays. eLife 3: e00590. https://doi.org/10.7554/elife.00590
Dulvy NK, Simpfendorfer CA, Davidson NK, Fordham SV, Brautigam A, Sant G, Welch DJ (2017) Challenges and priorities in shark and ray conservation. Curr Biol 27:R565–R572
Ebert DA, Vidthayanon DA, Samiengo B (2016) Bathytoshia lata. The IUCN Red List of Threatened Species 2016: e.T161386A104066775. http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T161386A104066775.en. Accessed 24 Aug 2017
FAO (2007) Fishery country profile: The Gambia. Available at: http://www.fao.org/fishery/docs/DOCUMENT/fcp/en/FI_CP_GM.pdf. Accessed 1 Feb 2018
FAO (2018a) Gambia at a glance. http://www.fao.org/gambia/fao-in-gambia/gambia-at-a-glance/en/. Accessed 31 July 2018
FAO (2018b) Fisheries Global Information System (FIGIS) query. https://www.fao.org/fishery/figis/en. Accessed 1 June 2018
Gabis G, Drammeh O, Nichols E, Kelpsite L, Castro K, Parkins C, Mendy A, Ceesay S, Joof J (2012) Bycatch Assessment in the Gambian Sole Bottom Gillnet Fishery. Coastal Resources Center, University of Rhode Island, Gambia-Senegal Sustainable Fisheries Project (USAID/Ba Nafaa), p 20
Gordon CA, Hood AR, Barker J, Bartolí À, Dulvy NK, Jiménez Alvarado D, Lawson JM, Meyers EKM (2017) Eastern Atlantic and Mediterranean Angel Shark Conservation Strategy. The Shark Trust. https://www.zsl.org/sites/default/files/media/2017-07/Angel%20Shark%20Conservation%20Strategy_FINAL.PDF. Accessed 6 June 2018
Humber F, Andriamahainoa ET, Berizinya T, Botosoamanantoa R, Godley BJ, Gough C, Pedrona S, Ramaherya V, Broderick AC (2017) Assessing the small-scale shark fishery of Madagascar through community-based monitoring and knowledge. Fish Res 186:131–143. https://doi.org/10.1016/j.fishres.2016.08.012
Lam VWY, Cheung WWL, Swartz W, Sumaila UR (2012) Climate change impacts on fisheries in West Africa: implications for economic, food and nutritional security. Afr J Mar Sci 34:103–117. https://doi.org/10.2989/1814232X.2012.673294
Last PR, Séret B (2016) A new Eastern Central Atlantic skate Raja parva sp. nov. (Rajoidei: rajidae) belonging to the Raja miraletus species complex. Zootaxa 4147:477–489
Last PR, White WT, de Carvalho MR, Séret B, Stehmann MF, Naylor GJP (2016a) Rays of the world. CSIRO Publishing, Clayton South
Last PR, Manjaji-Matsumoto BM, Naylor GJP, White WT (2016b) Stingrays, Family Dasyatidae. In: Last PR, White WT, de Carvalho MR, Séret B, Stehmann MF, Naylor GJP (eds) Rays of the world. CSIRO Publishing, Clayton South, pp 522–618
Last PR, White WT, Jones CM (2016c) Rhinopteridae. In: Last PR, White WT, de Carvalho MR, Séret B, Stehmann MF, Naylor GJP (eds) Rays of the world. CSIRO Publishing, Clayton South, pp 733–740
Le Manach F, Gough C, Harris A, Humber F, Harper S, Zeller D (2012) Unreported fishing, hungry people and political turmoil: the recipe for a food security crisis in Madagascar? Mar Policy 36:218–225. https://doi.org/10.1016/j.marpol.2011.05.007
Leeney RH, Downing N (2016) Sawfishes in The Gambia and Senegal – shifting baselines over 40 years. Aquat Conserv 26:65–278. https://doi.org/10.1002/aqc.2545
Mallon, D.P., Hoffmann, M., Grainger, M.J., Hibert, F., van Vliet, N. and McGowan, P.J.K. (2015). An IUCN situation analysis of terrestrial and freshwater fauna in West and Central Africa. Occasional Paper of the IUCN Species Survival Commission No. 54. Gland, Switzerland and Cambridge, UK: IUCN. x + 162 pp
Marine Stewardship Council (2013) German retailer Kaufland donates EUR 100,000 to the Gambian artisanal tonguesole fishery for social projects and upcoming MSC assessment. https://www.msc.org/newsroom/news/german-retailer-kaufland-donates-eur-100-000-to-the-gambian-artisanal-tonguesole-fishery-for-social-projects-and-upcoming-msc-assessment/%3Fsearchterm%3Dkaufland Accessed 7 Sept 2017
Marshall LJ, Cronin ES (2016) Fontitrygon margaritella. The IUCN Red List of Threatened Species 2016: e.T161520A104173198. http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T161520A104173198.en. Accessed 24 Aug 2017
Mbenga A (1996) Marketing and distribution of artisanal fisheries in the Gambia: women as fish protein suppliers in The Gambia. Master’s thesis, University of Tromso
Meissa B, Gascuel D (2015) Overfishing of marine resources: some lessons from the assessment of demersal stocks off Mauritania. ICES J Mar Sci 72:414–427
Mendy AN (2011) Progress and impact of Gambia National Plan of Action for sharks. Abstract page 29, in: Books of abstracts, Colloque international sur la conservation et la gestion durable des populations de requins en Afrique de l’Ouest: bilan et perspectives, Dakar (Senegal), 25-27 July 2011, Sub-Regional Fisheries Commission
Moore ABM (2015) A review of sawfishes (Pristidae) in the Arabian region: diversity, distribution, and functional extinction of large and historically abundant marine vertebrates. Aq Conserv 25:656–677. https://doi.org/10.1002/aqc.2441
Moore ABM (2017) Are guitarfishes the next sawfishes? Extinction risk and an urgent call for conservation action. Endanger Species Res 34:75–88. https://doi.org/10.3354/esr00830
Morey G, Serena F, Mancusi C, Coelho R, Seisay M, Litvinov F, Dulvy N (2007a) Squatina aculeata. The IUCN Red List of Threatened Species 2007: e.T61417A12477164. http://dx.doi.org/10.2305/IUCN.UK.2007.RLTS.T61417A12477164.en. Accessed 24 May 2018
Morey G, Serena F, Mancusi C, Coelho R, Seisay M, Litvinov F, Dulvy N (2007b) Squatina oculata. The IUCN Red List of Threatened Species 2007: e.T61418A12477553. http://dx.doi.org/10.2305/IUCN.UK.2007.RLTS.T61418A12477553.en. Accessed 24 May 2018
Moseley WG, Carney J, Becker L (2010) Neoliberal policy, rural livelihoods, and urban food security in West Africa: a comparative study of The Gambia, Côte d’Ivoire, and Mali. Proc Nat Acad Sci USA 107:5774–5779. https://doi.org/10.1073/pnas.0905717107
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858. https://doi.org/10.1038/35002501
Njai SE (2000) Traditional fish processing and marketing of the Gambia. The United Nations University Fisheries Training Programme, Final project
Notarbartolo di Sciara G, Bradai MN, Morey G, Marshall AD, Compagno LJV, Mouni A, Hicham M, Bucal D, Dulvy N, Heenan A, Coelho R (2007) Rhinobatos rhinobatos. The IUCN Red List of Threatened Species 2007: e.T63131A12620901. http://dx.doi.org/10.2305/IUCN.UK.2007.RLTS.T63131A12620901.en. Accessed 07 Sept 2017
Notarbartolo di Sciara GM, Bradai N, Serena F, Mancusi CM (2009) Dasyatis marmorata. The IUCN Red List of Threatened Species 2009: e.T161748A5494614. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T161748A5494614.en. Accessed 06 July 2017
Notarbartolo di Sciara G, Bradai MN, Morey G, Brahim K, Camara L, Litvinov F, Dulvy N, Doumbouya F, Ducrocq M, Heenan A, Sidi N (2016) Glaucostegus cemiculus. The IUCN Red List of Threatened Species 2016: e.T63132A104009894. http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T63132A104009894.en. Accessed 07 Sept 2017
Pala C (2013) Detective work uncovers under-reported overfishing. Nature 496:18
Pauly D, Christensen V, Dalsgaard J, Froese R, Torres F Jr (1998) Fishing down marine food webs. Science 279:860–863
Pheeha S, Cronin ES (2009) Torpedo mackayana. The IUCN Red List of Threatened Species 2009: e.T161411A5418113. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T161411A5418113.en. Accessed 06 July 2017
Polidoro B, Ralph GM, Strongin K, Harvey M, Carpenter KE, Adeofe TA, Arnold R, Bannerman P, Bibang Bi Nguema JN, Buchanan JR, Camara KMA, Camara YH, Cissoko K, Collette BC, Comeros-Raynal MT, De Bruyne G, Diouf M, Djiman R, Ducrocq M, Gon O, Harold AS, Harwell H, Hilton-Taylor C, Hines A, Hulley PA, Iwamoto T, Knudsen S, Lewembe JDD, Linardich C, Lindeman K, Mbye EM, Mikolo JE, Monteiro V, Mougoussi JB, Munroe T, Mve Beh JH, Nunoo FKE, Pollock C, Poss S, Quartey R, Russell B, Sagna A, Sayer C, Sidibe A, Smith-Vaniz W, Stump E, Sylla M, Tito De Morais L, Vié JC, Williams A (2016) Red List of Marine Bony Fishes of the Eastern Central Atlantic. Gland, Switzerland: IUCN. viii + 80 pp
Ramos R, Grémillet D (2013) Overfishing in west Africa by EU vessels. Nature 496:300. https://doi.org/10.1038/496300a
Robillard M, Séret B (2006) Cultural importance and decline of sawfish (Pristidae) populations in West Africa. Cybium 30(suppl):23–30
Saine A (2011) Evaluation of the socio-economic importance of the shark fishery and the marketing channels in the Gambia. Abstract page 52, in: Books of abstracts, Colloque international sur la conservation et la gestion durable des populations de requins en Afrique de l’Ouest: bilan et perspectives, Dakar (Senegal), 25-27 July 2011, Sub-Regional Fisheries Commission
Seck AA, Diatta Y, Diop M, Guélorget O, Reynaud C, Capapé C (2004) Observations on the reproductive biology of the blackchin guitarfish, Rhinobatos cemiculus E. Geoffroy Saint-Hilaire, 1817(Chondrichthyes, Rhinobatidae) from the coast of Senegal (Eastern Tropical Atlantic) Scientia Gerudensis 27: 19–30
Serena F, Mancusi C, Morey G, Ellis JR (2009a) Dasyatis pastinaca. The IUCN Red List of Threatened Species 2009: e.T161453A5427586. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T161453A5427586.en. Accessed 24 Aug 2017
Serena F, Notarbartolo di Sciara G, Mancusi C (2009b) Taeniurops grabata. The IUCN Red List of Threatened Species 2009: e.T161513A5440298. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T161513A5440298.en. Accessed 29 May 2018
Serena F, Mancusi C, Clò S, Ellis J, Valenti SV (2009c) Mustelus mustelus. The IUCN Red List of Threatened Species 2009: e.T39358A10214694. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T39358A10214694.en. Accessed 31 May 2018
Serena F, Mancusi C, Morey G, Ellis JR (2016) Dasyatis pastinaca. The IUCN Red List of Threatened Species 2016: e.T161453A97841681. Accessed 31 July 2018
Séret B (2006) Guide d’identification des principales espèces de requins et de raies de l’Atlantique tropical oriental, à l’usage des enquêteurs et des biologistes des pêches. Identification guide of the main shark and ray species of the eastern tropical Atlantic for the purpose of the fishery observers and biologists. Guia de identificaçao das principais espécies de raias e tubarões do Atlântico oriental tropical para observadores de pesca e biólogos (traduction portugaise par R. Coelho). FIBA, IUCN & PRCM, 76 p
Séret B (2016a) Rhinobatidae. Guitarfishes. Pp. 1357-1364. In: Carpenter K. & N. de Angelis (ed), The living marine resources of the Eastern Central Atlantic. Volume 2. Bivalves, gastropods, hagfishes, sharks, batoid fishes and chimaeras. FAO Species Identification Guide for Fishery Purposes. Rome: FAO. Pp. 665–1509
Séret B (2016b) Zanobatus maculatus, a new species of panray from the Gulf of Guinea, eastern central Atlantic (Elasmobranchii: batoidea: Zanobatidae). Zootaxa 4161:509–522
Séret B (2016b) Dasyatidae. Stingrays. Pp. 1404-1419. In: Carpenter K. & N. de Angelis (ed), The living marine resources of the Eastern Central Atlantic. Volume 2. Bivalves, gastropods, hagfishes, sharks, batoid fishes and chimaeras. FAO Species Identification Guide for Fishery Purposes. Rome: FAO. Pp. 665–1509
Séret B (2016c) Zanobatidae. Panrays. Pp. 1365-1369. In: Carpenter K. & N. de Angelis (ed), The living marine resources of the Eastern Central Atlantic. Volume 2. Bivalves, gastropods, hagfishes, sharks, batoid fishes and chimaeras. FAO Species Identification Guide for Fishery Purposes. Rome: FAO. Pp. 665–1509
Séret B, Cronin ES, Valenti SV (2009) Torpedo bauchotae. The IUCN Red List of Threatened Species 2009: e.T161562A5451911. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T161562A5451911.en. Accessed 06 July 2017
Séret B, Last PR, Naylor GJP (2016a) Glaucostegidae. In: Last PR, White WT, de Carvalho MR, Séret B, Stehmann MF, Naylor GJP (eds) Rays of the world. CSIRO Publishing, Clayton South, pp 110–116
Séret B, Couzens G, Valenti SV (2016b) Hypanus rudis. The IUCN Red List of Threatened Species 2016: e.T161620A104133548. http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T161620A104133548.en. Accessed 16 Aug 2017
Simpfendorfer CA (2003) Rhizoprionodon acutus. The IUCN Red List of Threatened Species 2003: e.T41850A10579779. http://dx.doi.org/10.2305/IUCN.UK.2003.RLTS.T41850A10579779.en. Accessed 31 May 2018
Simpfendorfer C, Burgess GH (2009) Carcharhinus leucas. The IUCN Red List of Threatened Species 2009: e.T39372A10187195. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T39372A10187195.en. Accessed 31 May 2018
Stein RW, Mull CG, Kuhn TS, Aschliman NC, Davidson LNK, Joy JB, Smith GJ, Dulvy NK, Mooers AO (2018) Global priorities for conserving the evolutionary history of sharks, rays and chimaeras. Nat Ecol Evol 2:288–298. https://doi.org/10.1038/s41559-017-0448-4
Thorson TB (1982) The impact of commercial exploitation on sawfish and shark populations in Lake Nicaragua. Fisheries 7:2–10. https://doi.org/10.1577/1548-8446(1982)007%3C0002:TIOCEO%3E2.0.CO;2
UNDESA (2018) Least Developed Countries. United Nations Development Policy and Analysis Division. www. un.org/development/desa/dpad/least-developed-country-Category. Accessed 6 June 2018
United Nations (2014) The fisheries sector in the Gambia: trade, value addition and social inclusiveness, with a focus on women. UN conference on trade and development enhanced integrated framework. Report UNCTAD/DITC/2013/4
Valadou B, Brêthes J-C, Inejih CAO (2006) Observations biologiques sur cinq espèces d’Élasmobranches du Parc national du Banc d’Arguin (Mauritanie). Cybium 30:313–322
Valenti SV (2009) Zanobatus schoenleinii. The IUCN Red List of Threatened Species 2009: e.T161519A5441827. http://dx.doi.org/10.2305/IUCN.UK.2009-2.RLTS.T161519A5441827.en. Accessed 06 July 2017
Verissimo A, Zaera-Perez D, Leslie R, Iglesias S, Séret B, Grigoriou P, Sterioti A, Gubili C, Barria C, Duffy C, Hernandez S, Batjakas I, Griffiths A (2016) Molecular diversity and distribution of the eastern Atlantic and Mediterranean dogfishes Squalus highlight taxonomic issues in the genus. Zoolog Scr 46:414–428
Viana ST de F, Lisher MW, de Carvalho MR (2017) Two new species of short-snouted dogfish sharks of the genus Squalus Linnaeus, 1758, from southern Africa (Chondrichthyes: Squaliformes: Squalidae). Mar Biodivers. https://doi.org/10.1007/s12526-017-0673-8
Viana, ST de F, de Carvalho MR. (2018). Resurrection and redescription of the Southern dogfish Squalus probatovi (Squalidae), a valid species from Angola. J Ichthyol. https://springerlink.bibliotecabuap.elogim.com/article/10.1134/S003294521805020X
Vooren CM, Lamónaca AF (2004) Rhinoptera brasiliensis. IUCN Red List of Threatened Species Available at http://www.iucnredlist.org [e.T44595A10912274. http://dx.doi.org/10.2305/IUCN.UK.2004.RLTS.T44595A10912274.en. Accessed 12 Aug 2017
Vooren CM, Piercy AN, Snelson Jr. FF, Grubbs RD, Notarbartolo di Sciara G, Serena S (2007) Gymnura altavela. The IUCN Red List of Threatened Species 2007: e.T63153A12624290. http://dx.doi.org/10.2305/IUCN.UK.2007.RLTS.T63153A12624290.en. Accessed 30 June 2018
Walker P, Cavanagh RD, Ducrocq M, Fowler SL (2005) Northeast Atlantic (including Mediterranean and Black Sea). 71-95 In: Fowler, SL, Cavanagh RD, Camhi M, Burgess GH, Cailliet GM, Fordham SV, Simpfendorfer CA, Musick JA (eds.). Sharks, Rays and Chimaeras: The Status of the Chondrichthyan Fishes. Status Survey. IUCN/SSC Shark Specialist Group. IUCN, Gland, Switzerland and Cambridge, UK. x + 461 pp
Walls RHL, Buscher E (2016) Aetomylaeus bovinus. The IUCN Red List of Threatened Species 2016: e.T60127A81163810. Accessed July 2017
Walls RHL, Vacchi M, Notarbartolo di Sciara G, Serena F, Dulvy NK (2016) Gymnura altavela. The IUCN Red List of Threatened Species 2016: e.T63153A16527909. Accessed 10 Aug 2017
White WT, Dharmadi (2007) Species and size composition and reproductive biology of rays (Chondrichthyes, Batoidea) caught in target and non-target fisheries in eastern Indonesia. J Fish Biol 70:1809–1837. https://doi.org/10.1111/j/1095-8649.2007.01458.x
Wintner S (2016) Aetomylaeus bovinus. The IUCN Red List of Threatened Species 2016: e.T60127A104022824. http://dx.doi.org/10.2305/IUCN.UK.2016-3.RLTS.T60127A104022824.en. Accessed 06 July 2017
Yokota L, de Carvalho MR (2017) Taxonomic and morphological revision of butterfly rays of the Gymnura micrura (Bloch & Schneider 1801) species complex; with the description of two new species (Myliobatiformes: gymnuridae). Zootaxa 4332:1–74
Yokota L, White WT, deCarvalho MR (2016) Butterfly rays, family Gymnuridae. In: Last PR, White WT, de Carvalho MR, Séret B, Stehmann MF, Naylor GJP (eds) Rays of the world. CSIRO Publishing, Clayton South, pp 511–521
Acknowledgements
We would like to thank the following for their kind help: University of Cumbria (UoC) staff Billy Sinclair and Volker Deecke for providing support in the field and photographs, and Claire Holt for providing the map; UoC undergraduate students who collected photographs and data between 2011 and 2018 (Roger Atkins, Callum Atkinson, Curtis Blank, Curnow Bascombe, Sophie Cornfoot. Adam Crompton, Leanne Edwards, Emma Hampsey, Rachael Hardcastle, Ryan Harvey, Alexandra Hesse, Jess Holdsworth, Jake Horwood, Sara Iversen, Kayley Knight, Nicole Mallett, Rahul Mehrotra, Suzannah Miller, Nathan Murray, Sally O’Neil, Oissene Purser, Sam Sewell, Jennifer Stollery, Philip Stone, Sam Thompson, Annabel Walker-Evans); Marcelo de Carvalho and Sarah Viana for providing identification advice; Ebrahima Ceesay (Fisheries Department) for guidance and translation; and Abou Saidy for organising local transport. The opinions expressed in this paper do not necessarily represent the views of the employers of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Angus Jackson.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the Topical Collection: Coastal and marine biodiversity.
Rights and permissions
About this article
Cite this article
Moore, A.B.M., Séret, B. & Armstrong, R. Risks to biodiversity and coastal livelihoods from artisanal elasmobranch fisheries in a Least Developed Country: The Gambia (West Africa). Biodivers Conserv 28, 1431–1450 (2019). https://doi.org/10.1007/s10531-019-01732-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-019-01732-9