Abstract
High human density and land use intensity often coincide with biodiversity hotspots making peri-urban reserves a keystone for conserving natural remnants in a highly anthropogenic matrix. Particularly, intense propagule pressure by alien plant species can pose a threat to native biodiversity. However, little is known about the factors that determine invasibility and the role of roads and other human disturbances for such small protected areas. Within a peri-urban reserve close to the city of Concepción, south-central Chile, we investigated the influence of different site and landscape characteristics on the richness of native and alien plant species across different habitat (ruderal, road and forest sites) and forest types (native and alien dominated forests). Compared to other protected areas, alien species were frequently found in this reserve indicating the importance of urban areas as source for alien species. Aliens concentrate around disturbed ruderal and road sites, facilitating their spread into the reserve. Natural forest areas are less invaded until now. Within forests alien plant species richness was, however, positively associated to the proximity to disturbed landscapes. Forests dominated by alien tree species within the reserve are not the source for alien species within natural forests, but they negatively affect native species richness by replacing adequate seed sources. In order to prevent a further spread of alien species into peri-urban reserves, large scale anthropogenic disturbances should be minimized, already invaded sites must be monitored and buffer-zones protecting reserves from surrounding land use activities should be established.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Peri-urban reserves are a keystone for conservation of highly endangered species and ecosystems in an otherwise human dominated landscape (Miller and Hobbs 2002; Snep et al. 2006; Hedblom and Söderström 2010; Elmqvist et al. 2013). Biodiversity hotspots usually coincide with areas of high population density under increasing urbanization pressure (Myers 1990; Cincotta and Engelman 2000; Sanderson et al. 2002). Therefore, peri-urban reserves provide the only remnant habitat in a highly disturbed matrix where they are confronted to the percolation of negative impacts from the matrix. Fire, pollution and alien plant and animal invasions are some of the most reported threats to biodiversity in peri-urban areas (McKinney 2002; Pauchard et al. 2006; Jarošík et al. 2011; Carmona et al. 2012; Spear et al. 2013; Golivets 2014). Understanding how these threats interact with the biodiversity in these environments may help to reduce their effects and elaborate adequate mitigation plans (McKinney 2002; Miller and Hobbs 2002; Kowarik 2011).
Alien plant invasions have been extensively studied in association to urban environments and road networks. Cities act as major sources of alien plant invasions increasing the diversity and density of propagules in the landscape, while roads contribute to their dispersal (Kowarik 2011; Aikio et al. 2012; Golivets 2014; Vicente et al. 2014). Therefore, peri-urban reserves are subject to intense propagule pressure by its boundaries and through the network of roads and trails, which are not only used for visitation but for inter-urban transportation. If peri-urban reserves aim to maintain their biodiversity in the long run, they must resist the continuous invasions of plants from the highly disturbed matrix. Unfortunately, little is known in how these small reserves cope with the high invasion and what factors determine invasibility at the landscape scale. Studies have found, for all types of protected areas, that there is a positive correlation in the number of alien and native plant species, meaning that areas rich in native biodiversity can also harbor more alien species (Allen et al. 2009). It is, however, not clear if this relationship prevails in heavily invaded peri-urban areas or if alien diversity is a detriment to native diversity here.
Landscape ecology principles may provide interesting insights in how native and alien plant species interact and how threatened native biodiversity is in these highly anthropogenic environments (González-Moreno et al. 2015). Understanding how plant diversity is distributed at the landscape scale within peri-urban areas, as well as where major sources of plant invasions occur is of vital importance for conservation planning (Gordon et al. 2009; McDonald et al. 2009; Borgström et al. 2012; Hulme et al. 2014; Moreira-Arce et al. 2015). Recognizing the hotspots of native biodiversity and plant invasions may further help to tackle specific pathways for invasion, through active control, and by prioritizing the protection of the more rich environments.
Mediterranean Chile, a biodiversity hotspot (Myers 1990; Myers et al. 2000), may be an excellent study system to understand the interaction between native and alien plant species at the landscape scale. This region has experienced major landscape changes in the past decades due to the expansion of alien forest plantations, agricultural activities, and urban and industrial sprawl leading to a substantial loss of natural ecosystems (Sala et al. 2000; Aguayo et al. 2009) and to a high number of alien species compared to other Latin American countries (Fuentes et al. 2013). Particularly, the highly productive and diverse coastal ranges characterized by a high endemism correspond with areas of high human density and land use and are merely represented in the Chilean National System of Protected Wild Areas (SNASPE; Simonetti 1994; Armesto et al. 1998; Smith-Ramírez 2004; Pliscoff and Fuentes-Castillo 2011).
The area around the city of Concepción used to be a highly diverse transition zone between the Mediterranean and temperate biomes of Chile (Amigo and Ramírez 1998; Fuentes et al. 2013), but is now characterized by forest plantations, industrial complexes and uncontrolled urban sprawl with satellite neighborhoods emerging in large numbers. As a consequence, there is a loss in native ecosystems and a gain in alien species (Pauchard et al. 2006; Figueroa et al. 2011), which can potentially decrease native species numbers and lead to local extinctions (Gaertner et al. 2009). Despite such a highly disturbed matrix around Concepción, a remnant of the potential natural forest ecosystem could prevail due to its function as drinking water reservoir. The now protected National Reserve Nonguén represents the last major remnant of the deciduous forest of Concepción (Gajardo 1994), which once covered the coastal mountain range in this region and has been replaced almost entirely by agricultural crops and forest plantations. It protects several endangered species according to IUCN such as the deer Pudu puda (Silva-Rodríguez et al. 2010) or the frogs Rhinoderma darwinii and R. rufum (Soto-Azat et al. 2013), which all suffer from habitat loss and human-induced disturbance. Among plant species the endangered Citronella mucronata and Gormotega keule occur. The reserve further represents one of two known locations of the endangered Berberis negeriana (Hechenleitner et al. 2005). For visitors three main hiking trails have been established in order to experience the native flora and fauna. Due to its proximity (ca. 4 km) to the rapidly expanding city of Concepción it is, however, questionable, if the natural forest vegetation can be preserved particularly regarding the substantial threat by the diverse and abundant alien flora in this area (Pauchard et al. 2006; Fuentes et al. 2013).
To better understand the current state of native plant diversity and its susceptibility to invasion by alien species, we evaluated the influence of different habitat types present within the reserve on the distribution of native and alien species. These habitat types include (1) ruderal sites that lack a tree cover and experienced frequent disturbance e.g. by dumping construction material in the past, and that are now dominated by alien grasses, (2) road sites and (3) forest interiors dominated either by native or alien tree species. We evaluated the impact of broader environmental variables and the landscape context, especially in the role of urban (Botham et al. 2009) and riparian areas (Parendes and Jones 2000) as sources and corridors for alien species invasion, on both species groups. We hypothesize higher alien plant species richness at open disturbed sites (Maskell et al. 2006) and along roads (Parendes and Jones 2000; Pauchard and Alaback 2004; Jiménez et al. 2013), whereas native species are more diverse in forest interiors. In addition we expect alien but not native species to be positively associated with alien forest plantations and the proximity to human settlements and streams.
Methods
Study area
The study was conducted in the National Reserve Nonguén, approximately 4 km south-east of the peripheries of Concepción, south-central Chile, Biobío-region (36°54′S, 72°58′W). The climate is temperate-Mediterranean, with a mean annual precipitation sum of 1100 mm (1970–2000) with 60 % of the precipitation falling from May to July. The mean annual temperature is 12.9 °C with highest temperatures in January (16.9 °C) and lowest in July (9.2 °C; Castillo Fontannaz 2001). The predominant soil type is a fertile, well drained alfisol on granite rock with a medium to fine texture and a high base saturation (Casanova et al. 2013).
The reserve was established in December 2009 on 3037 ha in order to protect the last remnants of the temperate-Mediterranean coastal deciduous forest that once covered the whole coastal area of this region, but is widely replaced by plantations of Pinus radiata and Eucalyptus spec. today (Gajardo 1994; Luebert and Pliscoff 2006). The natural vegetation of the study area was conserved under local protection because this forest represented an important reservoir of drinking water for the city of Concepción (Habit et al. 2003). Almost three quarters of the reserve are covered with natural forests dominated by Nothofagus obliqua. In the north-eastern part of the reserve plantations of Pinus radiata and Eucalyptus spec. prevail. In this area, a wildfire ca.12 year ago, destroyed the vegetation and promoted the establishment of Australian Acacia-species (A. melanoxylon, A. dealbata) which form dense stands. In the reserve center old stands of Pinus radiata with a high proportion of native trees in the lower tree layer connect between natural forests and Pinus radiata-plantations. Few open areas close to recreational sites in the western part of the reserve are characterized by alien grasses and ruderal species. Although most parts of the natural forest have limited access, roads crossing the reserve have long existed and since 2009 are implemented as hiking trails. The matrix surrounding the reserve is highly disturbed dominated by industrial forest plantations of Pinus radiata and Eucalytus spec., urban and suburban neighborhoods and some small-scale agricultural use. Roads along the reserve border connect the urban areas of Concepción, Hualqui and Chiguayante with distances of the urban edges to the reserve border of 2 to 4 km (Fig. 1a). Among all provinces of the Biobío-region Concepción was most affected by forest fires during the last 30 years with a total burnt area of 142,000 ha (CONAF 2013).
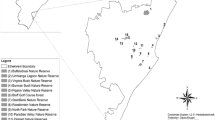
a Location of the National reserve Nonguén (framed) close to the city of Concepción in south-central Chile, region Biobío and b position of the sampled road, ruderal and forest transects established along the main forest roads and hiking trails. There was no spatial autocorrelation for species numbers and composition according to Mantel-tests. Forest transects were placed at least 25 m from the road or trail edge. The maps were obtained from Google Earth. For detailed location of transects see Online Resource 1
Data collection
We used 2 by 50 m transects divided in 5 subplots (2 by 10 m) to record native and alien plant species in the Nonguén Reserve. Following the existing roads and trails we established a total of 75 transects on ruderal sites (n = 6), along forest roads (n = 34) and inside the forest (n = 35, Fig. 1b, Online Resource 1). Road and forest transects were placed in all mentioned forest types (natural forest, old pine forest, pine plantation, eucalyptus plantation, Acacia forest, Online Resource 2). While the position of the ruderal transects was chosen randomly in the available sites, the transect position along roads and in forests was selected according to forest type (overstorey tree species >5 m) and accessibility of the forests. The side of the road sampled was chosen randomly. All roads were unpaved. Each road plot started with 0.5 m distance to the pavement (Online Resource 3).
In each 2 by 10 m subplot, we recorded presence and cover of all vascular plants in November 2012. Each transect was checked for new species in February 2013. Species cover was estimated using the methodology of Braun-Blanquet (1964). All vegetation surveys were conducted by the same observer at both survey dates. Woody species <5 m in height were assigned to the shrub (>0.5 m <5 m in height) and herb layer (<0.5 m in height). For the present study we concentrated on herb layer data.
We distinguished between native and alien species and between different life cycles (short-lived; perennial; woody) according to Zuloaga et al. (2008). Short-lived species generally characterize disturbed sites, and comprise most of Chile’s alien flora (Fuentes et al. 2013), whereas woody aliens have the greater effect on native species diversity (Gaertner et al. 2009). Furthermore, species were assigned to different light categories: shade-tolerant, intermediate, light-demanding. For alien species we used light indicator values (LIV) by Ellenberg et al. (2001). Plants with LIV 1–3 were classified as shade-tolerant, with LIV 4–6 as intermediate and plants with LIV 7–9 as light-demanding. All native and alien species (if not listed in Ellenberg et al. 2001) listed by Matthei (1995) were classified as light-demanding. Further, species part of the characteristic floristic composition of closed forest communities (including sclerophyllous, evergreen and deciduous forests) according to Luebert and Pliscoff (2006) were classified as shade-tolerant, species characteristic both in closed forests and in open vegetation types (including the Chilean Matorral and herbaceous communities) were regarded as intermediate; species only characteristic in open vegetation types were classified as light-demanding (see Online Resource 4 for the light and life cycle categories of all recorded species). We concentrated on the light demand of species as this trait is seen as the most important regarding the invasion of forest understories. Although most alien species are light-demanding, those that fully or partially tolerate shade can have the most severe impact on forest ecosystems (Martin et al. 2009).
We recorded several environmental, topographic and landscape variables measured either on subplot or transect level (Table 1). The light intensity is expressed by the indirect site factor obtained from a hemispheric photograph taken in the center of each subplot at 2 m height in February when trees were fully foliated (Pryor 2010). We took soil samples from the upper 5 cm of each subplot and pooled the samples per transect. The pooled samples were analyzed for pH and C/N ratio. On road sites the distance of each transect to the forest edge and the road width (both measured from the transect center; Online Resource 3) were additionally measured. Roads were assigned to different use categories based on a subjective evaluation of the frequency of vehicle traffic (Table 1).We recorded elevation, aspect and slope on each subplot. By using the geographic latitude, the aspect and the slope we calculated the potential direct annual radiation reaching the ground using equations introduced by McCune and Dylan (2002). Landscape variables include the distance of each transect center to the nearest stream and the distance to the nearest settlement. Further the percentage of natural forests, alien-dominated forests, open vegetation and area outside the reserve within a 500-m circle around each transect center was calculated (Table 1). Landscape calculations were based on geographic data layers provided by the Lab of Landscape Ecology of the University of Concepción using ArcGIS 10.2. These layers contained the hydrology and forest types across the reserve.
Data analysis
Total species numbers and numbers of native and alien species were calculated transect- and subplot-based for each habitat and forest type. The percentage of species assigned to different light and life cycle categories was calculated per habitat and forest type. Comparisons between habitat and forest types were conducted using transect-based one way ANOVA for normal distributed or Kruskal–Wallis-H-test for non-normal distributed data followed by the adequate post hoc tests. The same methods were applied comparing the recorded environmental, topographic and landscape variables among the three habitat types. In order to indentify indicator species for forest, ruderal and road transects, we performed an indicator species analysis (ISA) according to Dufrêne and Legendre (1997). This analysis calculates an indicator value (IV) for each species per habitat type as the proportional abundance of a species within a group relative to the abundance in all groups multiplied by the proportional frequency of this species in each group. The calculated IV ranges between 0 (no indication) and 1 (perfect indication) with a perfect indicator being always and exclusively present in a certain habitat. The significance of each indicator value is tested by Monte Carlo simulation using 1000 iterations (McCune and Grace 2002).
To analyze the influence of the different variables on the number of native and alien species in forest and road transects, we used generalized linear mixed effect models (GLMM) with Poisson error distribution based on subplot data: the number of native and alien species either on road or forest transects were analyzed as response variable. Subplot nested in transect was analyzed as random effect. We fitted three models containing either the environmental, topographic or landscape variables and the interaction with the forest habitat (differentiating between natural and alien forest) or in terms of road transects with the road use type as fixed effects. We fitted a fourth model using the number of alien species and its interaction with forest habitat or road use type as predictor variable for native species numbers or vice versa. By gradually removing insignificant terms from the models we achieved the most parsimonious model describing native and alien species richness. We pooled all alien-dominated forest types as alien forest as we were mainly interested in the difference between natural and alien-dominated stands. We excluded ruderal transects from further analyses as they were restricted to the entrance area of the reserve.
Principal component analyses (PCA) of all significant variables on forest and road transects were performed in order to derive an independent gradient represented by the first principle component. For each species occurring at least twice in forests or on road transects we determined the distribution range by extracting the minimum and maximum score of transects the species occurred in. By differentiating native and alien species we were able to indentify an overlapping of the distribution ranges. The packages lme4 (GLMM), labdsv (ISA) and MASS (PCA) of the R software (version 3.0.2; R Core Team) were used. Statistical significance was assumed at p < 0.05 if not stated otherwise.
Results
Environmental, topographic and landscape variables
Ruderal transects were characterized by the highest light availability, the lowest elevation and the highest proportion of open area in the surrounding compared to roads and forests (Table 2). Light availability was higher and C/N ratio lower along roads compared to the forest interior (Table 2). Ruderal and road transects differed in pH-values and in mean distance to water bodies with ruderal transects being less acidic and closer to streams. Forest transects were characterized by the thickest litter layer and the steepest slopes (Table 2).
Native and alien species across habitat types
We recorded 112 native and 69 alien species on all transects. 37 native species are endemic to Chile (Online Resource 4). Most species were found on ruderal transects containing more aliens than native species (Fig. 2). Forest and road transects were richer in native than in alien species, with significantly more aliens on road compared to forest transects. Except for two tree species (Cupressus macrocarpa, Pinus pinaster) that only occurred in forest transects with constancies below 10 %, all alien species sampled in forests were more frequent in road transects.
Mean number of all species, alien species and native species sampled in forest, road and ruderal transects. Different lower case letters (a, b) indicate significant differences in either total, alien or native species numbers among habitat types, different upper case letters (A, B) between alien and native species numbers within one habitat type
Forest type had no significant influence on alien species richness on road transects, but within forests with lowest numbers in natural forests (Fig. 3). The highest native species richness among forest types was recorded in natural forests, the lowest in forests dominated by Acacia species. Roads next to natural stands were significantly richer in native species compared to roads associated with all other forest types.
Mean number of all species, alien species and native species sampled in the different forest types in forest (left diagram) and road (right diagram) transects. Different lower case letters (a, b) indicate significant differences in total, alien or native species numbers among forest types within forest and road transects
Forests contained the highest percentage of shade-tolerant and lowest percentage of short-lived species (Online Resource 5). Short-lived plants occur especially on ruderal transects. Woody species are dominant on forest and road transects. This pattern is preserved when regarding native and alien species separately. The majority of alien species across all habitats is light-demanding.
According to indicator species analysis most species indicative of conditions on ruderal and road transects are aliens, whereas only native species could be identified as indicators for forest conditions (Table 3).
Response of native and alien species richness to abiotic variables
We could identify several variables, which significantly affect native and alien plant species richness in forest (Online Resource 6) and road transects (Online Resource 7). Alien species numbers in forests benefit from an alien canopy, elevation in natural stands, slope and the proximity to the reserve border. A high abundance of alien-dominated stands in the surrounding of forest transects though and high radiation decrease alien species richness. Native species are more diverse in natural stands and promoted by light and C/N-ratio. In alien stands the number of native species is negatively affected by an increasing percentage of alien forests in the surrounding (Online Resource 6).
Road transects adjacent to natural forests contain more alien species than road transects adjacent to alien stands. Road use, light, and the C/N-ratio in natural forest roads are positively correlated with alien species numbers. They decrease with distance to settlements, but significantly only on low-use-roads. Native species numbers increase with transect’s distance to the forest edge, with slope, with the proportion of natural stands in the surrounding, and with pH on natural forest roads. They are, however, negatively associated with the percentage of open sites in the surrounding and with the C/N ratio on low use roads. Wide roads promote native plant species richness if the road use is at low to medium level. Light promotes native species richness on low use roads, but not on medium use roads (Online Resource 7). There was no significant interaction between native and alien species richness on forest or road transects.
We combined all significant variables into one independent gradient (=1. PCA axis) for forest and road transects that each explained one third of the variation among transects. The first principle for forests explains 29.4 % of the variation among transects and had the highest loadings for elevation (−0.52), % area outside the reserve in a 500 m-circle around transects (−0.44), slope (0.50), light availability (−0.35) and vegetation type (native vs. alien; −0.35). It represents a naturalness gradient with Eucalyptus-plantations scoring the lowest and natural stands the highest on the 1. PCA-axis. For road transects, the gradient explains 30.5 % of the variation and has the highest loadings for road type from low to high use roads (−0.46), distance to settlements (−0.45), % of natural forest in a 500 m-circle around transects (0.43), road width (−0.37), and litter layer thickness (0.31). Thus, this principle represents a decreasing disturbance gradient from high-use to low-use-roads. Rubus ulmifolius is the only alien species occurring over the whole gradient on forest transects, all other aliens are restricted to more disturbed sites (Fig. 4). In general aliens are characterized by lower PCA scores than native species (T-Test, t = 2.27, p = 0.028) for forest transects. Along roads, mainly alien species (with the exception for Cryptocarya alba, Viola portalesia and Lobelia tupa) occur at the lowest range of the gradient representing high disturbance (Fig. 5). Species occurring at low disturbance rates are mainly native. Thus, alien species have significantly lower scores than native species (T-Test, t = 3.97, p < 0.001).
Distribution ranges (lines) of species surveyed in forest transects along an independent gradient determined by principal component analysis of environmental, topographic and landscape variables that significantly explained native and alien plant species richness. Species are grouped according to their native or alien status and their light preferences
Distribution ranges (lines) of species surveyed in road transects along an independent gradient determined by principal component analysis of environmental, topographic and landscape variables that significantly explained native and alien plant species richness. Species are grouped according to their native or alien status and their light preferences
Discussion
Our results demonstrate the susceptibility of the peri-urban reserve Nonguén to alien plant invasion. We showed that alien plant species benefit from the proximity to urban areas as major source for alien species and utilize a variety of dispersal corridors into the reserve. Although native forest interiors, which are the hotspots of native diversity and the main conservation concern in Nonguén, sampled along the major roads and hiking trails are still less invaded within the herb layer compared to disturbed sites, native diversity is threatened by a large number of alien species with the potential to invade these forest interiors.
The percentage of alien species in Nonguén (38 %) is much higher compared to other Chilean protected areas (11–25 %; Muñoz Schick 1980; Pauchard et al. 2000; Pauchard and Alaback 2004; Jiménez et al. 2013), and to Chile in general (ca.14 %; Arroyo et al. 2000; Zuloaga et al. 2008; Fuentes et al. 2013). The nearby urban areas offer highly disturbed and fertile habitats and contain parks and gardens, where alien species were planted for ornamental reasons (Chytrý et al. 2008; Botham et al. 2009; Kowarik 2011). Further, the previous agricultural land use, starting with the Spanish colonization 500 years ago, and the intensive recreational use of remaining natural habitats close to cities are highly important factors for the large number of alien species in the peri-urban area of Concepción (Pauchard et al. 2006). High human density in the boundaries of protected areas often facilitates the visitor access and increases the alien plant propagule supply (Spear et al. 2013). Our results indicate that roads act as conduits for alien species, and that vehicles and visitors may function as one important dispersal agent for aliens into the reserve. At smaller scales, there is also percolation of alien species from heavily invaded patches to areas dominated by native species through seed dispersal and vegetative reproduction. This complex landscape context (González-Moreno et al. 2015), where sources and sinks of alien species are connected through the dense road and trail network in the reserve, creates a challenging scenario for conservation efforts.
We could show that a high diversity of alien species uses the ruderal, frequently disturbed sites, located close to the reserve entrance, for an initial establishment. Particularly weeds with early successional traits such as poor shade-tolerance, short life span, small seed size or long seed viability colonized these sites and benefitted from disturbance (Bazzaz 1979; Ricklefs et al. 2008). As the majority of weeds in Chile are alien species, the high propagule pressure explains the higher number of alien compared to native species at ruderal sites (Arroyo et al. 2000; Rejmánek et al. 2005). These disturbed sites were closer to streams compared to the sampled road and forest transects. Thus, water ways represent an important corridor for alien species into the reserve from far away sources (Merritt and Wohl 2006). Examples are Plantago lanceolata, Poa trivialis or Senecio aquaticus, species we identified as indicators of ruderal sites in Nonguén, which use hydrochory as the main dispersal mode and which could establish in suitable sites close to streams (Fuentes et al. 2013). In addition wetlands attract birds which contribute to seed dispersal through droppings or the adherence of small seeds to feet and plumes (Ricklefs et al. 2008).
Forest roads, however, represented the main dispersal way of alien species into the reserve with ruderal sites acting as source populations for most species (Parendes and Jones 2000; Watkins et al. 2002; Pauchard and Alaback 2004; Jiménez et al. 2013). A high road use enhanced alien species richness assuming intense propagule transport by vehicles and a facilitating effect by high light availability and frequent disturbance (Chytrý et al. 2008). With decreasing light and disturbance intensity as seen on low-use roads, the proximity to settlements as source for alien species becomes most important.
High alien species diversity was not restricted to roads crossing forests dominated by alien species such as Eucalyptus spec., Pinus radiata or Acacia melanoxylon. Roads next to natural forests harbor almost the complete set of alien species recorded along roads and therefore act as propagule sources for further invasion into natural forest habitats. The species richness next to natural forests might be explained by the diversity of tree species in the adjacent overstorey ameliorating soil conditions based on a better decomposition of some native tree species particularly compared to Pinus radiata (Lusk et al. 2001; Rivas et al. 2009; Furey et al. 2014). Native species showed the same preferences for roads next to natural stands leading to a low, but significant positive correlation between alien and native species numbers on road transects (rSpearman = 0.381, p = 0.026) indicating no competitive exclusion, but a similar response to environmental conditions (Maskell et al. 2006; Tomasetto et al. 2013). GLMM models revealed, however, no significant interaction between alien and native species numbers neither in forests nor along roads.
The number of alien species along roads is independent of the surrounding land use. The opposite was true for native species. Native species richness is promoted by a high percentage of natural stands in the vicinity representing available seed sources, but decreases with a high percentage of open area in the surrounding mainly dominated by alien seed sources. A positive influence of distance to the forest edge for native species numbers might be a combined effect of light availability increasing and litter layer decreasing with distance to the forest edge.
Forest stands dominated by alien trees have reduced native species richness (Bremer and Farley 2010). Particularly Acacia-stands showed low species numbers confirming results by other authors and underlining the significance of woody alien species for native biodiversity (Fuentes-Ramírez et al. 2010; González-Muñoz et al. 2012). Australian Acacia species belong to the top invaders and have already altered community composition and ecosystem services in different regions of the world (Gaertner et al. 2009; Le Maitre et al. 2011). These species can produce many seeds that are able to germinate particularly after fire. They can form very dense and dark stands out-competing other species. Native species richness in alien forests in Nonguén further decreased when potential native seed sources in the surrounding, represented by natural forests, were missing. Native plant populations are often small and isolated in peri-urban areas and therefore face the risk of extinction. Our results underline the importance of preserving as much native remnants as possible in an urbanized landscape and the key role of peri-urban reserves for this task (McKinney 2002).
As alien species richness in natural stands decreases with the percentage of alien-dominated forests in the surrounding, alien forests are not the sources for further alien species spread within these native remnants inside the reserve. The proximity to the matrix outside, however, promoted alien species richness both in natural and in alien forests and illustrates the high propagule pressure. Within the matrix prescribed burns after forest harvests are regularly used for land management and often get beyond control particularly in the region around Concepción (CONAF 2013). These fires have also entered the reserve at the edges leaving fire traces on several trees in transects close to the border (personal observation). Gómez-Gonzáles and Cavieres (2009) demonstrated the promotion of short-lived alien plant species over annual, perennial and woody native species in the Chilean matorral after litter burning of varying intensities and concluded a stimulation of alien seed germination either by heat or by chemical compounds from ash or smoke. A heat-stimulated germination is, however, not restricted to short-lived herbaceous species. Alien tree and shrub species that originate from regions with a frequent occurrence of natural induced fires, in contrast to Chile where fires are human-induced, also have an advantage over native species after fire (e.g. Acacia species, Genista monspessulana). Particularly G. monspessulana, originating from Mediterranean Europe, was observed in forest transects that were once affected by fire (García et al. 2010). The spread of this species from the boundaries may also change the fire regime within native forests in the long-run facilitating its further spread (García et al. 2015). Until now, both G. monspessulana and Acacia melanoxylon, are only sparsely present in forests. They were, however, frequent and abundant along roads using them as dispersal corridors from the borders into the reserve. Their partial shade-tolerance makes them, and other shade-tolerant aliens detected along roads, potential colonizers of the light-limited forest interior in the future particularly after small-scale canopy opening. Moreover, plant species that are mostly classified as light-demanding might be able to adapt to shady conditions as shown for Prunella vulgaris in temperate evergreen rainforests in Chile (Godoy et al. 2011). A higher plasticity of alien species in the invaded range compared to their native range was often observed in different plant species allowing an adaptation to novel environments such as light limited forest understories (García et al. 2010; Ross et al. 2009; Bossdorf et al. 2005). Until now, only Rubus ulmifolius was able to colonize the whole gradient of forests surveyed in Nonguén, as this alien species can spread vegetatively and does not have to rely on sufficient light for flowering.
Although the degree of invasion into forest interiors was still low, ruderal and road sites in Nonguén can act as a source for alien species in particular after natural or anthropogenic canopy openings (Spence et al. 2011). Although there is not yet an official management plan for the Nonguén reserve, a substitution of alien-dominated plantations by native species is planned (CONAF personal communication). In some areas such measures already started by removing Eucalyptus and Acacia species on large areas. These measures will, however, promote the establishment of aliens (e.g. Acacia spec.,G. monspessulana) in the short term and facilitate their spread into the reserve center. A similar pattern was observed on clear cuts adjacent to roads in North America (Parendes and Jones 2000; Watkins et al. 2002) and can already be observed in Nonguén. On different research sites, native tree species were planted after a total or partial overstorey removal. These sites are largely invaded by Acacia melanoxylon. Thus, a restoration of natural forests should rather be achieved by establishing shade-tolerant tree species (e.g. Cryptocarya alba, Persea lingue, Aextoxicon punctatum) into existing forest plantations. Although the regeneration of native plants is not always successful under alien-dominated forests (Mascaro et al. 2008), there are also results indicating a facilitation of native tree regeneration by alien forests (Yang et al. 2009; Abelleira-Martínez 2010; Fuentes-Ramírez et al. 2011; Becerra and Montenegro 2013). 70 % of the species recorded in pine plantations were regenerating native woody species that might form the next tree regeneration in the long term in Nonguén.
Invasive alien species present along roads and in open areas should be intensively monitored and manually removed if spreading into more natural areas. Fire as a tool to reduce undesired plants as a first measure must be avoided. By this the impact of very invasive alien species might be minimized. To limit the impact of human land use in the surroundings of the reserve a buffer zone should be established.
Our results underline the continuous pressure peri-urban reserves are facing from a surrounding highly human impacted landscape and from landscape corridors facilitating the spread of alien species, which are able to colonize a wide range of site conditions. By regarding different habitat types, we could, however, also demonstrate the resistance of forest interiors to invasion until now and the importance of conserving natural forest remnants for native biodiversity under high alien propagule pressure (Hulme et al. 2014). The high number of aliens at disturbed sites and along roads in the investigated peri-urban reserve also highlights the necessity of regular monitoring programs for nature reserves in such anthropogenic landscapes around the world. A comprehensive understanding of the landscape may help to better identify the sources of potential new invasions and to prioritize control in those areas that serve as conduits for invasions (e.g. roads, trails). Native biodiversity hotspots are able to withstand invasions even under intense propagule pressure, thus restoration efforts should continue to maintain and expand these conservation areas. Only by combining adequate monitoring and management efforts (e.g. early removal of spreading aliens, buffer zone establishment, prevention of large scale disturbances, restoration) a further spread of very frequent alien species into the last remnants of natural ecosystems in an urbanized landscape can be prevented.
References
Abelleira-Martínez OJ (2010) Invasion by native tree species prevents biotic homogenization in novel forests of Puerto Rico. Plant Ecol 211:49–64
Aguayo M, Pauchard A, Azócar G, Parra O (2009) Cambio del uso del suelo en el centro sur de Chile a fines del siglo XX. Entendiendo la dinámica especial y temporal del paisaje. Rev Chil Hist Nat 82:361–374
Aikio S, Duncan RP, Hulme PE (2012) The vulnerability of habitats to plant invasion: disentangling the roles of propagule pressure, time and sampling effort. Global Ecol Biogeogr 21:778–786
Allen JA, Brown CS, Stohlgren TJ (2009) Non-native plant invasions of United States National Parks. Biol Invasions 11:2195–2207
Amigo J, Ramírez C (1998) A bioclimatic classification of Chile: woodland communities in the temperate zone. Plant Ecol 136:9–26
Armesto JJ, Rozzi R, Smith-Ramírez C, Arroyo MTK (1998) Conservation targets in South American temperate forests. Science 282:1271–1272
Arroyo MTK, Marticorena C, Matthei O, Cavieres L (2000) Plant invasions in Chile: present patterns and future predictions. In: Mooney HA, Hobbs RJ (eds) Invasive species in a changing world. Island Press, Washington, DC, pp 385–421
Bazzaz FA (1979) The physiological ecology of plant succession. Annu Rev Ecol Syst 10:351–371
Becerra PI, Montenegro G (2013) The widely invasive tree Pinus radiata facilitates regeneration of native woody species in semi-arid ecosystems. Appl Veg Sci 16:173–183
Borgström S, Cousins SAO, Lindborg R (2012) Outside the boundary—land use changes in the surroundings of urban nature reserves. Appl Geogr 32:350–359
Bossdorf O, Auge H, Lafuma L, Rogers WE, Sieman E, Prati D (2005) Phenotypic and genetic differentiation between native and introduced plant populations. Oecologia 144:1–11
Botham MS, Rothery P, Hulme PE, Hill MO, Preston CD, Roy DB (2009) Do urban areas act as foci for the spread of alien plant species? An assessment of temporal trends in the UK. Divers Distrib 15:338–345
Braun-Blanquet J (1964) Pflanzensoziologie: Grundzüge der Vegetationskunde, 2nd edn. Springer, Wien
Bremer LL, Farley KA (2010) Does plantation forestry restore biodiversity or create green deserts? A synthesis of the effects of land-use transitions on plant species richness. Biodivers Conserv 19:3893–3915
Carmona A, González ME, Nahuelhual L, Silva J (2012) Spatio-temporal effects of human drivers on fire danger in Mediterranean Chile. Bosque 33:321–328
Casanova M, Salazar O, Seguel O, Luzio W (2013) The soils of Chile. Springer, Dordrecht
Castillo Fontannaz C (2001) Estadísticas climatologia. Dirección meterologica de Chile. http://164.77.222.61/climatologia/. Accessed 11 May 2015
Chytrý M, Maskell LC, Pino J, Pyšek P, Vilà M, Font X, Smart SM (2008) Habitat invasions by alien plants: a quantitative comparison among Mediterranean, subcontinental and oceanic regions of Europe. J Appl Ecol 45:448–458
Cincotta RP, Engelman R (2000) Nature’s place—human population and the future of biological diversity. Population Action International, Washington, DC
CONAF (2013) Estadísticas históricas incendios forestales. Corporación Nacional Forestal Chile. http://www.conaf.cl/incendios-forestales/incendios-forestales-en-chile/estadisticas-historicas/. Accessed 11 May 2015
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366
Ellenberg H, Weber HE, Düll R, With V, Werner W (2001) Zeigerwerte von Pflanzen in Mitteleuropa. Scr Geobot 18:1–262
Elmqvist T, Fragkias M, Goodness J, Güneralp B, Marcotullio PJ, McDonald RI, Parnell S, Sendstad M, Schewenius M, Seto KC, Wilkinson C (eds) (2013) Urbanization, biodiversity and ecosystem services: challenges and opportunities. Springer, Dordrecht
Figueroa JA, Teillier S, Castro SA (2011) Diversity patterns and composition of native and exotic floras in central Chile. Acta Oecol 37:103–109
Fuentes N, Pauchard A, Sánchez P, Esquivel J, Marticorena A (2013) A new comprehensive database of alien plant species in Chile based on herbarium records. Biol Invasions 15:847–858
Fuentes-Ramírez A, Pauchard A, Marticorena A, Sánchez P (2010) Relación entre la invasion de Acacia dealbata Link (Fabaceae: Mimosoideae) y la riqueza de species vegetales en el centro-sur de Chile. Gayana Bot 67:188–197
Fuentes-Ramírez A, Pauchard A, Cavieres LA, Carcía RA (2011) Survival and growth of Acacia dealbata vs. native trees across an invasion front in south-central Chile. For Ecol Manag 261:1003–1009
Furey C, Tecco PA, Perez-Harguindeguy N, Giorgis MA, Grossi M (2014) The importance of native and exotic plant identity and dominance on decomposition patterns in mountain woodlands of central Argentina. Acta Oecol 54:13–20
Gaertner M, Breeyen AD, Hui C, Richardson DM (2009) Impacts of alien plant invasions on species richness in Mediterranean-type ecosystems: a meta-analysis. Prog Phys Geog 33:319–338
Gajardo R (1994) La vegetación natural de Chile. Clasificación y distribución geográfica. Editorial Universitaria, Santiago
García RA, Pauchard A, Cavieres LA, Peña E, Rodriguez MF (2010) El fuego favorece la invasión de Teline monspessulana (Fabacea) al aumentar su germinación. Rev Chil Hist Nat 83:443–452
García RA, Engler ML, Peña E, Pollnac FW, Pauschard A (2015) Fuel characteristics of the invasive shrub Teline monspessulana (L.) K. Koch. Int J Wildland Fire. doi:10.1071/WF13078
Godoy O, Saldaña A, Fuentes N, Valladares F, Gianoli E (2011) Forests are not immune to plant invasions: phenotypic plasticity and local adaptation allow Prunella vulgaris to colonize a temperate evergreen forest. Biol Invasions 13:1615–1625
Golivets M (2014) Ecological and biological determination of invasion success of non-native plant species in urban woodlands with special regard to short-lived monocarps. Urban Ecosyst 17:291–303
Gómez-González S, Cavieres LA (2009) Litter burning does not equally affect seedling emergence of native and alien species of the Mediterranean-type Chilean matorral. Int J Wildland Fire 18:213–221
González-Moreno P, Delgado JD, Vilà M (2015) Una vision a escala de paisaje de las invasiones biológicas. Ecosistemas 24:84–92
González-Muñoz N, Costa-Tenorio M, Espigares T (2012) Invasion of alien Acacia dealbata on Spanish Quercus robur forests: impact on soils and vegetation. For Ecol Manag 269:214–221
Gordon A, Simondson D, White M, Moilanen A, Dekessy SA (2009) Integrating conservation planning and landuse planning in urban landscapes. Landsc Urban Plan 91:183–194
Habit E, Victoriano P, Rodríguez-Ruiz A (2003) Variaciones espacio-temporales del ensamble de peces de un sistema fluvial de bajo orden del centro-sur de Chile. Rev Chil Hist Nat 76:3–14
Hechenleitner P, Gardner MF, Thomas PI, Echeverría C, Escobar B, Brownless P, Martínez C (2005) Plantas Amenazadas del Centro-Sur de Chile. Distribución, Conservación y Propagación. 1st ed. Universidad Austral de Chile y Real Jardín Botánico de Edimburgo
Hedblom M, Söderström B (2010) Landscape effects on birds in urban woodlands: an analysis of 34 Swedish cities. J Biogeogr 37:1302–1316
Hulme PE, Pyšek P, Pergl J, Jarošik V, Schaffner U, Vilà M (2014) Greater focus needed on alien plant impacts in protected areas. Conserv Lett 7:459–466
Jarošík V, Pyšek P, Kadlec T (2011) Alien plants in urban nature reserves: from red-list species to future invaders? NeoBiota 10:27–46
Jiménez A, Pauchard A, Marticorena A, Bustamante RO (2013) Patrones de distribución de plantas introducidas en areas silvestres protegidas y sus areas adyacentes del centro-sur de Chile. Gayana Bot 70:110–120
Kowarik I (2011) Novel urban ecosystems, biodiversity, and conservation. Environ Pollut 159:1974–1983
Le Maitre DC, Gaertner M, Marchante E, Ens E-J, Holmes PM, Pauchard A, O’Farrell PJ, Rogers AM, Blanchard R, Blignaut J, Richardson DM (2011) Impacts of invasive Australian acacias: implications for management and restoration. Divers Distrib 17:1015–1029
Luebert F, Pliscoff P (2006) Sinopsis bioclimática y vegetacional de Chile. Editorial Universitaria, Santiago
Lusk CH, Donoso C, Jiménez M, Moya C, Oyarce G, Reinso R, Saldaña A, Villegas P, Matus F (2001) Decomposición de hojarasca de Pinus radiata y tres species arbóreas nativas. Rev Chil Hist Nat 74:705–710
Martin PH, Canham CD, Marks PL (2009) Why forests appear resistant to exotic plant invasions: intentional introductions, stand dynamics, and the role of shade tolerance. Front Ecol Environ 7:142–149
Mascaro J, Becklund KK, Hughes RF, Schnitzer SA (2008) Limited native plant regeneration in novel, exotic-dominated forests on Hawaii. For Ecol Manag 256:593–606
Maskell LC, Bullock JM, Smart SM, Thompson K, Hulme PE (2006) The distribution and habitat associations of non-native plant species in urban riparian habitats. J Veg Sci 17:499–508
Matthei O (1995) Manual de las malezas que crecen en Chile. Alfabeta Impresores, Santiago
McCune B, Dylan K (2002) Equations for potential annual direct incident radiation and heat load. J Veg Sci 13:603–606
McCune B, Grace JB (2002) Analysis of ecological communities. MjM Software Design, Gleneden Beach
McDonald RI, Forman RTT, Kareiva P, Neugarten R, Salzer D, Fisher J (2009) Urban effects, distance, and protected areas in an urbanizing world. Landsc Urban Plan 93:63–75
McKinney ML (2002) Urbanization, biodiversity, and conservation. Bioscience 15:883–890
Merritt DM, Wohl EE (2006) Plant dispersal along rivers fragmented by dams. River Res Appl 22:1–26
Miller JR, Hobbs RJ (2002) Conservation where people live and work. Conserv Biol 16:330–337
Moreira-Arce D, de la Barrera F, Bustamante RO (2015) Distance to suburban/wildland border interacts with habitat type for structuring exotic plant communities in a natural area surrounding a metropolitan area in central Chile. Plant Ecol Divers 8:363–370. doi:10.1080/17550874.2014.983201
Muñoz Schick M (1980) Flora del Parque National Puyehue. Editorial Universitaria, Santiago
Myers N (1990) The biodiversity challenge: expanded Hot-Spots analysis. Environmentalist 10:243–256
Myers N, Mittermeier RA, Mittermeier CG, da Fonseca GAB, Kent J (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Parendes LA, Jones JA (2000) Role of light availability and dispersal in exotic plant invasion along roads and streams in H.J. Andrews Experimental Forest, Oregon. Conserv Biol 14:64–75
Pauchard A, Alaback PB (2004) Influence of elevation, land use, and landscape context on patterns of alien plant invasions along roadsides in protected areas of south-central Chile. Conserv Biol 18:238–248
Pauchard A, Ugarte E, Millán J (2000) A multiscale method for assessing vegetation baseline of environmental impact assessment (EIA) in protected areas of Chile. In: McCool SF, Cole DN, Borrie WT, O’Loughlin J (eds) Conference on wilderness science in a time of change. 3. Wilderness as a place for scientific inquiry. Proceedings RMRS-P-15-Vol 3. U.S. Forest Service, Rocky Mountain Research Station, Ogden
Pauchard A, Aguayo M, Peña E, Urrutia R (2006) Multiple effects of urbanization on the biodiversity of developing countries: the case of a fast-growing metropolitan area (Concepción, Chile). Biol Conserv 127:272–281
Pliscoff P, Fuentes-Castillo T (2011) Representativeness of terrestrial ecosystems in Chile’s protected area system. Environ Conserv 38:303–311
Pryor DD (2010) Analysis of light environments under forest canopies using an integrated digital hemispherical image system. Dissertation, Staffordshire University
Rejmánek M, Richardson DM, Pyšek P (2005) Plant invasions and invasibility of plant communities. In: van der Maarel E (ed) Vegetation ecology. Blackwell, Oxford, pp 332–355
Ricklefs RE, Guo Q, Qian H (2008) Growth form and distribution of introduced plants in their native and non-native ranges in Eastern Asia and North America. Divers Distrib 14:381–386
Rivas Y, Oyarzún C, Godoy R, Valenzuela E (2009) Mineralización del nitrógeno, carbon y actividad enzimática del suelo en un bosque de Nothofagus obliqua (Mirb) Oerst y una plantación de Pinus radiata D. Don del centro-sur de Chile. Rev Chil Hist Nat 82:119–134
Ross CA, Faust D, Auge H (2009) Mahonia invasions in different habitats: local adaptation or general –purpose genotypes? Biol Invasions 11:441–452
Sala OE, Chapin FS III, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774
Sanderson EW, Jaiteh M, Levy MA, Redford KH, Wannebo AV, Woolmer G (2002) The human footprint and the last of the wild. Bioscience 52:891–904
Silva-Rodríguez EA, Verdugo C, Aleuy OA, Sanderson JG, Ortega-Solís GR, Osorio-Zúñiga F, González-Acuña D (2010) Evaluating mortality sources for the vulnerable pudu Pudu puda in Chile: implication for the conservation of a threatened deer. Oryx 44:97–103
Simonetti JA (1994) Threatened biodiversity as an environmental problem in Chile. Rev Chil Hist Nat 67:315–319
Smith-Ramírez C (2004) The Chilean coastal range: a vanishing center of biodiversity and endemism in South American temperate rainforests. Biodivers Conserv 13:373–393
Snep RPH, Opdam PFM, Baveco JM, Wallis DeVries MF, Timmermans W, Kawak RGM, Kuypers V (2006) How peri-urban areas can strengthen animal populations within cities: a modeling approach. Biol Conserv 127:345–355
Soto-Azat C, Valenzuela-Sánchez A, Collen B, Rowcliffe JM, Veloso A, Cunningham AA (2013) The population decline and extinction of Darwin’s Frogs. PLoS One 8:e66957. doi:10.1371/journal.pone.0066957
Spear D, Foxcroft LC, Bezuidenhout H, McGeoch MM (2013) Human population density explains alien species richness in protected areas. Biol Conserv 159:137–147
Spence LA, Ross JV, Wiser SK, Allen RB, Coomes DA (2011) Disturbance affects short-term facilitation, but not long-term saturation, of exotic plant invasion in New Zealand forest. Proc R Soc B 278:1457–1466
Tomasetto F, Duncan RP, Hulme PE (2013) Environmental gradients shift the direction of the relationship between native and alien plant species richness. Divers Distrib 19:49–59
Vicente JR, Pereira HM, Randin CF, Gonҫalves J, Llomba A, Alves P, Metzger J, Cezar M, Guisan A, Honrado J (2014) Environment and dispersal paths override life strategies and residence time in determining regional patterns of invasion by alien plants. Perspect Plant Ecol 16:1–10
Watkins RZ, Chen J, Pickens J, Brosofske KD (2002) Effects of forest roads on understory plants in a managed hardwood landscape. Conserv Biol 17:411–419
Yang L, Liu N, Rain H, Wang J (2009) Facilitation by exotic Acacia: Acacia auriculiformis, Acacia mangium as nurse plants in South China. For Ecol Manag 257:1786–1793
Zuloaga FO, Morrone O, Belgrano M (2008) Catálogo de las plantas vasculares del Cono Sur (Argentina, Sur de Brasil, Chile, Paraguay y Uruguay) Volumen I, II, III. Monogr Syst Bot 107, Mo Bot Gard Press, St. Louis
Acknowledgments
We thank the Bauer Foundation (Bauer Stiftung zur Förderung von Wissenschaft und Forschung) within the Stifterverband für die Deutsche Wissenschaft for funding our research. A. Pauchard was funded by the Ministry of Economy grant ICM P05-002 and the CONICYT grant PFB-23. We are further grateful to Cristian Echeverría and his Lab of Landscape Ecology of the University of Concepción for providing the GIS layers of the Nonguén reserve, to Burkhard Müller-Using and the administration of the reserve for help with logistics, to Alejandra Jiminéz and Víctor Finot for help with species identification, and to Peter White and two anonymous reviewers for helpful comments to improve the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by David Hawksworth.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Heinrichs, S., Pauchard, A. Struggling to maintain native plant diversity in a peri-urban reserve surrounded by a highly anthropogenic matrix. Biodivers Conserv 24, 2769–2788 (2015). https://doi.org/10.1007/s10531-015-0964-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-015-0964-y