Abstract
Loss of habitat and biological invasion are the main threats to biodiversity. In intensive agricultural or urban landscapes, forest fragments, even if they are small and isolated represent biodiversity refugia. Environmental variables such as landscape structure, abiotic conditions and anthropogenic disturbance affect the biodiversity of the fragments. In this study, we explored plant species richness in 48 forest fragments embedded in predominantly agricultural landscapes on the alluvial plains of the Mura and Drava rivers in NE Slovenia. We determined several forest fragment characteristics such as perimeter, area, shape complexity, length and proportion of edge shared with adjacent land-cover types and anthropogenic disturbance indicated by the presence of footpaths and waste disposed in fragments. The abiotic condition of these fragments was assessed by Ellenberg indicator values. We built generalized linear models and ordination analyses to assess the importance of environmental variables for the richness and composition of alien plant species and other ecologically meaningful plant groups. Shape complexity had a consistent positive effect on the richness of native and alien plants species. Major drivers of alien plant composition in forest fragments included adjacent land-cover and urbanization level. An increasing proportion of arable land along the forest fragment perimeter negatively affected the richness of alien plants, while the effect of urban areas was positive. Our results confirmed that forest fragments in the rural–urban matrix represent biodiversity refugia and support native plant species; however, they are not resistant to invasions; instead invasion depends on the land-cover type in the surroundings of a given forest fragment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
One of the main threats causing biodiversity loss is forest area destruction resulting in isolated forest fragments (Botkin et al. 2007; Harsch et al. 2017; Haddad et al. 2015). Forest fragments that persist in a biotically depleted matrix of intensive agricultural land or urban areas, represent refugia for biodiversity and form local biodiversity hotspots (Becerra and Simonetti 2020; Chapman et al. 2015; Decocq et al. 2016). However, in the agricultural or urban matrix, forest fragments are predominantly small, isolated, with increased edge area, and under threat of disappearing (Decocq et al. 2016; Valdés et al. 2020). Forest fragments function as habitats for forest species, as scarce shelters and migration stepping-stones, and provide several ecosystem services (Decocq et al. 2016; Kowalska et al. 2021). The biodiversity of forest fragments is highly influenced by landscape structure, including landscape composition (proportion of land-use types) and configuration (spatial pattern of fragments) (Fahrig 2013). Biodiversity usually increases with fragment size (Šipek et al. 2022) and age (Valdés et al. 2020).
Another threat to the biodiversity of forest fragments is biological invasion (Petrášová et al. 2013; Šibíková et al. 2019; Becerra and Simonetti 2020). European temperate lowland forest communities are among the most invaded forest habitats (Walter et al. 2005; Jansen et al. 2011; Medvecká et al. 2014). Phytosociological analysis has revealed that at least one alien plant species was found in 90% of lowland forest associations (Jansen et al. 2011). Vulnerability of a native forest community is determined by a number of environmental factors such as light (Marinšek and Kutnar 2017), nutrient availability (Kowalska et al. 2021), disturbance regime and propagule pressure (Jansen et al. 2011; Petrášová et al. 2013). Invasions in lowland forests are largely promoted by anthropogenic activities such as agriculture, forestry, transportation, ornamental planting in nearby settlements and urban areas, or even the deposition of garden waste in forests and at their edges (Petrášová et al. 2013; Rusterholz et al. 2012; Sladek and Strgulc Krajšek 2019; Šipek and Šajna 2020).
In agriculture and forestry, most cultivated plant species are of alien origin (Reichard and White 2001; Petrášová et al. 2013). Besides intentionally introducing economically important crops and trees, unintentional alien plant introduction is especially high in agriculture ecosystems, contributing mainly to numerous alien weed species. Even though weeds are primarily restricted to arable or disturbed land, they can become established on forest edges, or even penetrate into the forest interior if forest fragments mingle with arable land and if abiotic conditions at the micro-level are favorable (Honnay et al. 2002). However, the impact of agricultural intensification on plant invasion from arable land into forest fragments has rarely been studied (but see Honnay et al. 2002; Ohlemüller et al. 2006; Weber and Gibson 2007).
The increasing urbanization rate and proximity to urban areas increase both the diversity and cover of alien plant species (Borgmann and Rodewald 2005; Bartuszevige et al. 2006; Maheu-Giroux and de Blois 2007; Okimura and Mori 2018); however, the pattern of increase is not always clear (Trentanovi et al. 2013). Forest fragments near human settlements are at particularly high invasion risk from ornamental alien plant species (Pergl et al. 2016; Gaggini et al. 2017; Ward and Amatangelo 2018; Guo et al. 2019; Fornal-Pieniak et al. 2019). Horticulture is recognized as the main source of escaped and naturalized alien plant species and represents a continuously increasing species pool (Reichard and White 2001; Pergl et al. 2016; Gaggini et al. 2017; van Kleunen et al. 2018; Beaury et al. 2021). Moreover, most of the worst invasive alien plants were introduced for ornamental purposes (Reichard and White 2001). Garden waste deposits consisting of ornamental alien plant propagules are often found in (peri-)urban forest fragments thus promoting new escapes by and spread of ornamental alien plant species into (semi-)natural forest communities (Sullivan et al. 2005; Rusterholz et al. 2012; Gaggini et al. 2017; Šipek and Šajna 2020).
Studying alien plant species richness and composition in lowland forest fragments of various sizes in relation to the surrounding land-cover is crucial to understanding invasion patterns in a landscape context. However, the focus in studies is often on the perpendicular distance from a forest edge to particular type of land-cover (Honnay et al. 2002; Ohlemüller et al. 2006; Weber and Gibson 2007), whereby the fragment size and forest edge length are neglected. To consider the importance of fragment size, we focused on the length of the shared edge between a particular land-cover and a forest fragment and on the proportion of particular land-cover in a buffer zone bordering the fragments. Because different groups of alien plants are characteristic for specific land-cover (e.g., weeds prevail in agricultural land and ornamental plants in urban areas), we expected that the specific land-cover surrounding a forest fragment would influence the composition of alien plants in that fragment. Our motivation was to assess whether the effect of surrounding land-cover type decreases with increasing fragment size. In general, forest fragmentation increases the amount of forest edge relative to the amount of forest interior for each fragment; a fragment’s increased perimeter/area ratio is consequently followed by greater edge effects. In urban and agricultural landscapes, this can promote alien plant invasion, since an increased forest edge can result in a greater probability of alien plants penetration and establishment in a forest (Pauchard and Alaback 2004; McDonald and Urban 2006; Maheu-Giroux and de Blois 2007; Vilà and Ibáñez 2011; LaPaix et al. 2012; González-Moreno et al. 2013; Marinšek and Kutnar 2017). Therefore, the concern exists that in degraded and largely fragmented landscapes, dominated by agricultural land (or urban areas), where the forest is represented mainly by small remnants, they can lose their forest-dwelling sanctuary role for forest specialist species. What is more, these forest remnants might even become nurseries for alien plant species instead.
An example of extensive hardwood lowland forests largely converted into agricultural land with a few remnants left is the sub-Pannonian Quercus-Carpinus forest on the former floodplains of the Mura and Drava rivers in NE Slovenia (Čarni et al. 1998). We hypothesized that the richness and composition of vascular plant species and especially of alien plant species established in the forest fragments can be explained by three sets of environmental drivers. To test our hypothesis, we examined the effect of (1) landscape structure (fragment size and shape, proportion of land-cover types in a buffer zone surrounding a forest fragment), (2) abiotic conditions in the forest fragment, and (3) anthropogenic disturbance (urbanization level and human activities inside a fragment) on plant species richness and composition.
Methods
Study area
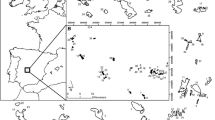
The study was conducted on the alluvial plains of the Mura and Drava rivers in NE Slovenia, which represents the westernmost part of the Pannonian Basin (Fig. 1). Both rivers are tributaries to the Danube. The lowland Apaško polje (220 m a.s.l.) along the Mura river covers an area of 36 km2. The lowland Dravsko polje (250 m a.s.l.) represents a larger area of approximately 260 km2 and begins downstream of Maribor. Both plains belong to the sub-Pannonian region exhibiting a temperate continental climate with mean annual precipitation of 874 mm (Apaško polje) and 972 mm (Dravsko polje) and an equal mean annual temperature of 10.9 °C in 2019. Although the precipitation regime is favorable, droughts often occur during the summer (GGN 2016).
Zonal vegetation in the study area belongs to the Illyrian Quercus-Carpinus betulus forest (EUNIS habitat classification) lying between the central European, Balkan and Pannonian Quercus forests (Davies et al. 2004). Prevailing trees are Q. robur L., Q. petraea (Mattuschka) Liebl., Carpinus betulus L., Prunus padus L., Fraxinus sp. Tourn. Ex L., and Ulmus sp. L., with Populus sp. L. and Salix sp. L. on the riverbanks. On both lowlands, the rivers have been regulated, and the once extensive Quercus-Carpinus forests have been transformed into arable land (Repe 2004). Today intensive agriculture is the prevailing land-use, followed by forests, urban areas and meadows. Forest fragments of different sizes and shapes occur in a gradient from the agricultural toward the urban matrix. Characteristic landscape elements of both plains are long roadside villages of family houses with backyard gardens, vineyards and arable fields behind (Ščap 2018; Vodeb et al. 2016). The population of Apaško polje is more rural than the inhabitants of Dravsko polje, where the majority live in compact urban settlements.
Fragment measurements
Using Google Earth Pro (2020), we drew polygons of 48 sampled forest fragments and determined the perimeter and the area of the forest fragments (Table S1). Further, we measured the lengths of their edges neighboring the existing land-cover types (urban areas, arable land, roads, meadows and rivers or watercourses).
We computed three forest fragment shape indices P/A, FRAC and SHPI, to obtain the shape complexity of forest fragments. P/A is perimeter (p)—area (a) ratio (\(P/A=\frac{p}{a}\), \(P/A>0\)). Larger P/A values suggest a more complex and irregular shape. FRAC indicates fractal dimension (Mandlebrot 1977) and was calculated as \(FRAC=2\frac{{\text{log}}(p)}{{\text{log}}(a)}\); values are between 1 and 2. FRAC approaching value 1 indicates less complex shapes like squares and circles, while FRAC approaching value 2 suggests shapes that are more complex. SHPI (Patton 1975) is calculated as \(SHPI=\frac{p}{2\sqrt{\pi \bullet a}}\), \(SHPI>1\).With increasing SHPI, the complexity of the fragment increases.
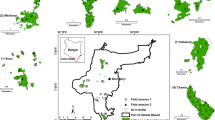
In ArcGIS (ESRI 2014), three buffers of different widths were defined: 500 m, 200 m and 20 m from the edge of the fragment (Fig. 2). In the buffer, we extracted the land-cover type proportions from the Corine Land Cover 2018 data layer (EEA 2018). We combined the Corine Land Cover categories to get six main land-cover types: urban, roads, arable, rivers (watercourses), meadows (combining traditional mosaic land-use) and forests.
Schematic representation of sampling design. The green square represents the sampled forest fragment. Surrounding land-cover is shown by a maize symbol (arable land-cover), or by houses (urban land-cover), for which the length of edge shared with the forest fragment is shown by yellow and red lines, respectively; and the proportion of various land-covers in 3 buffers of different width: 20 m, 200 m and 500 m
The building index (number of buildings) was determined for each buffer zone from the building data layer accessible on the geodetic data site of the Surveying and Mapping Authority of the Republic of Slovenia, Ministry of the Environment and Spatial Planning (Building Cadastre 1999). The building index was used in the analysis evaluating alien plant species richness. However, it was excluded from the generalized linear models (GLMs) because of the strong correlation with urban areas.
Forest fragments were classified into three classes according to urbanization level estimated according to the Corine Land Cover proportion of urban areas around forest fragments in a 500 m buffer zone: low (26 forest fragments)—up to 0.02%, medium (10 forest fragments)—from 0.02 to 10% and high (12 forest fragments)—above 10% of urban areas. The classification of the fragments according to the urbanization level into three classes was based on our preliminary analyses. We searched for proportions of urbanization levels where differences in alien plant species richness and composition were observed while simultaneously creating classes with a sufficient number of fragments that can be used in the analysis. The urbanization level was included in ordination analysis as a grouping variable.
Field sampling: Vegetation survey, abiotic conditions, and anthropogenic disturbance
The vascular flora was sampled in 25 and 23 forest fragments in Apaško polje and Dravsko polje, respectively. Sampling was done twice in 2019, first in spring (April and May) and second in autumn (August and September) to cover the entire vegetation season. We sampled plants in the forest fragments by walking around each fragment near the edge and crossing it diagonally. We increased the sampling effort with increasing fragment size until new vascular plant species were hard to detect. We listed and determined all vascular plant species to the species level according to Martinčič et al. (2007), with the exception of grasses and sedges, which were instead assigned to a higher taxonomic level in the absence of flowers (fruits).
The recorded plant species were classified into several categories: native plants (Martinčič et al. 2007), alien plants (Jogan et al. 2012), invasive alien plants which are a subgroup of alien plants (Jogan et al. 2012) and archaeophytes (Jogan et al. 2012). The archeophytes were treated as a separate group because of their uncertain origin and long presence, which might show different distribution patterns than recently established alien plant species not yet able to spread widely. Plants belonging to the alien plant category were further classified into 4 subgroups: ornamental plants—further divided according to life forms: ornamental herbaceous, ornamental bush and ornamental trees; arable weeds; trees planted for silviculture; and fruit trees. Some of the alien plant species did not match any of these categories and were classified as “Other”. Trees planted for silviculture, fruit trees and “Other” were excluded from modelling, because of their very low presence in these forest fragments.
Based on the presence of native plant species, we assigned Ellenberg indicator values (EIV; Ellenberg et al. 1992) of light, temperature, soil moisture, soil reaction and nutrients calculated in JUICE software (Tichý 2002) to each fragment. It must be emphasized that this is a very rough assessment of ecological conditions, and one, which deteriorates as the fragment size and its heterogeneity increase. However, an average EIV can be an adequate rough estimate for the ecological separation of individual forest fragments.
In each fragment, we recorded the presence of waste and its type (garden waste or other) and the presence of footpaths as an indicator of anthropogenic disturbance.
Statistical analysis
Differences in landscape characteristics and plant species richness between Apaško polje and Dravsko polje were tested with Student’s t-test and ANOVA. To assess whether the data met assumptions of normal distribution and homogeneity of variance Shapiro–Wilk and Levene tests were used, respectively. If the data violated those assumptions, we log-transformed the data or used appropriate nonparametric tests (Welch t-test, the Wilcoxon rank sum test and Kruskal–Wallis test). Since the landscape structure in the Apaško polje and Dravsko polje areas was similar, in further analysis of predicting plant species richness and plant species composition in the lowland forest fragments data from both plains were combined.
Model fitting
To predict plant species richness in lowland forest fragments according to the 3 sets of explanatory variables (Table 1), we fitted several generalized linear models (GLMs). We fitted GLMs using the function “glm”, which allows the input of both numerical and categorical explanatory variables. GLMs were fitted with Poisson error distribution and a log link as proposed for count data.
Model fitting steps:
-
(i)
Calculation of correlation matrix to assess correlation among variables, highly correlated variables were omitted from model building.
We defined the buffer zone width with the greatest impact on the plant species richness according to the strongest Pearson correlation coefficients (r) among land-cover proportions in different buffer widths. Land-cover proportions occurring in the buffer with the greatest impact (200 m buffer) were further used in GLMs to assess the significance of individual explanatory variables. A similar process was done with shape indices. SHPI explained the most variance of plant species richness and was therefore included in GLMs.
-
(ii)
Assessment of spatial autocorrelation of response variables data.
Spatial autocorrelation of response variables data (richness of plant species) was assessed using Moran’s I test based on a matrix of inverse distance weights (function “Moran.I”, package ape; Paradis and Schliep 2019). According to Moran’s I, we found evidence of spatial autocorrelation in our data (p < 0.05, Table S2), which was expected as closer forest fragments share a similar matrix that affects plant species richness in a similar way.
-
(iii)
Model building.
To explain the overall plant species richness in fragments and the richness of alien plant species and other above-defined plant categories we used three sets of explanatory variables included in GLMs (Table 1). The effects of (1) landscape structure (fragment size, shape, and the proportion of adjacent land-cover), (2) abiotic conditions assessed by EIV for light, temperature, humidity and nutrients and (3) anthropogenic disturbance (human activity inside a fragment detected as the presence of garden waste, other types of waste and footpaths) were tested using GLMs.
For all full models (all explanatory variables were included), the variance inflation factor was calculated using the “vif” function (package car; Fox and Weisberg 2019) to avoid multicollinearity among variables. In all models, variance inflation factor was below 2.5, which is less than the limit value of 10 as proposed by Lindborg (2007).
-
(iv)
Simplification of the full models by stepwise variable selection.
We selected the best models by using the “step” function. This is a stepwise algorithm returning the final model according to the lowest Akaike information criterion.
-
(xxii)
Summary of the final model and assessment of the spatial autocorrelation of the model residuals.
Lastly, we visually estimated whether the models matched assumptions using the »plot« function for model diagnostics in R. The spatial autocorrelation of final model residuals was assessed visually by plotting the residuals on a map. We concluded that spatial autocorrelation was generally low as there was no visible interpretable pattern of plotted residuals. This indicates that all important covariates accounting for the spatial structure of the data were included in the models. Therefore, we consider the final models valid. We calculated D-squared (D2) for all final models, which is the amount of deviance accounted for by the GLMs and is equivalent to the better known R2 in linear modelling (Guisan and Zimmermann 2000).
Plant community composition analysis
Ordination methods were used to analyze the effect of landscape structure, abiotic conditions and anthropogenic disturbance on plant species composition in the forest fragments.
The main ecological gradient in the distribution of alien plants was examined by detrended correspondence analysis (DCA). Because of the short environmental gradient revealed by DCA analysis, linear methods were subsequently used. Principal component analysis (PCA) was used to display each forest fragment in a multidimensional space where each dimension is defined by the richness of one plant species group according to the plant species classifications defined above. We used the matrix of forest fragments × plant species groups to obtain comprehensive plant species composition and the composition of alien plant groups in forest fragments.
Redundancy analysis (RDA) was used to explore the interaction among plant species composition and environmental explanatory variables (forest fragment configuration and Ellenberg indicator values). Because there were zeroes in the data set, we applied redundancy analysis with Hellinger transformation of species composition data (Legendre and Gallagher 2001). Significant explanatory variables were chosen using the forward selection method in the adespatial package with the»forward.sel« function. To assess the significance of the selected model, a Monte Carlo permutation test with 999 permutations was used. Adjusted p values were calculated using Holm’s correction. Ordination analyses were performed using a vegan package (Oksanen et al. 2022).
All statistical analyses were done in R programming language version 3.6.3 (R Development Core Team 2023).
Results
Forest fragment landscape characteristics
The forest fragments in Apaško polje and Dravsko polje had similar perimeters (123–12,076 m; t-test, t = 0.46, p = 0.65), areas (0.1–260 ha; t-test, t = 0.30, p = 0.76) and shape complexity (SHPI: 1.08–2.25; Wilcoxon test, W = 349, p = 0.21).
The prevailing adjacent land-cover in both lowlands in the buffer zones 20, 200 and 500 m around the forest fragment and along the forest edge was arable land (Fig. 3) therefore further analysis was performed on merged data. The following land-covers in smaller proportions are forests, meadows, urban areas, roads and rivers (Fig. 2, Table S3). Roads and urban land-cover were typical of the more densely populated Dravsko polje, while in Apaško polje a rural matrix prevailed.
Plant species richness in forest fragments
In these 48 lowland forest fragments, we identified 482 vascular plant species, ranging from 42 to 184 species per fragment. Based on the t-test statistic (Student or Welch t-test) overall plant species richness and native plant richness were similar between forest fragments in Apaško polje and Dravsko polje (mean of 88.32 vs. 89.22 for overall plant species richness and 73.56 vs. 69.61 for native plant richness, respectively). The proportion of native plants was significantly lower in Dravsko polje than in Apaško polje (mean of 78% vs. 84%; Table S4).
In total, 85 alien plants species were identified, ranging from 2 to 38 species per fragment. Among the listed alien plants, 17 were classified as invasive (Table 2). The numbers and proportion of alien plants were significantly higher for Dravsko polje (mean of 11.57 vs. 8.08 and 12% vs. 9%, respectively), while there was no difference in invasive alien plant species between both lowlands (mean of 4.87 vs. 5.20 and 5% vs. 6% for Dravsko polje and Apaško polje, respectively). Archaeophytes were recorded from 1 to 15 species per fragment (30 different species in total) and their species richness did not differ between Apaško polje and Dravsko polje (mean of 6.68 vs. 8.04), while their proportion was significantly higher for Dravsko polje (mean of 9% vs. 7%; Table S4). The species richness of alien plant subgroups differed significantly between Apaško polje and Dravsko polje in the number of weed species (mean of 2.40 vs. 3.91) and fruit trees (mean of 0.08 vs. 0.35), while there were no differences for the other groups. Apaško polje had a higher proportion of herbaceous ornamental plants (mean of 42% vs. 31%), while for Dravsko polje the proportion of ornamental bushes was higher (mean of 11% vs. 4%; Table S4).
The most common invasive alien plants in forest fragments were Solidago gigantea (81% presence) followed by Robinia pseudoacacia (73% presence), Erigeron annuus and Phytolacca americana (67% presence) (Table 2). The most comprehensive list of invasive alien plant species for Slovenia is from 2012 (Jogan et al. 2012). According to this list, an additional invasive species is recognized: Ph. americana, which was previously classified as naturalized. However, over a period of 10 years, the species has significantly expanded its range across Slovenian forests and was present in our study site in 67% of sampled forest fragments.
Environmental drivers of plant species richness in forest fragments
The overall plant species richness in lowland forest fragments was determined by landscape structure (Tables S5 and S6), abiotic conditions and anthropogenic disturbance. The shape complexity of forest fragments was a significant predictor of all plant species group richness, while the inclusion of forest fragment area in the models did not improve them. The richness of all plant species groups increased with increasing fragment shape complexity (Tables 3, 4).
The forest fragment shape index SHPI, the proportion of urban areas, roads and the presence of footpaths have a positive effect on overall plant species richness, while the effect of meadows and nutrients was negative. The deviance explained in the final model accounted for 68%. A similar set of variables excluding urban areas accounted for 63% of deviance explained for native plant species richness (Fig. 4A, B; Table 3).
Prediction accuracy of species richness models: A overall plant species, B native plant species, C alien plant species (APS), D invasive alien plant species (IAPS) and E archaeophytes. The green line represents fitted values: points below and above the green line are overestimated low species richness and underestimated high species richness, respectively
SHPI, the proportion of urban areas and light positively affected alien plant species richness while the proportion of arable land and nutrients exhibited a negative effect. The final model accounted for 79% of deviance explained. In contrast, the proportion of urban areas in the buffers and abiotic conditions were not important predictors of invasive alien plant species richness (62% of deviance explained; Fig. 4C, D; Table 3).
Archaeophytes were positively associated by the shape index SHPI, the proportion of arable land in a 200 m buffer, light and garden waste in forest fragments. However, the final model accounted for only 45% of deviance explained (Fig. 4E, Table 3).
Ornamental alien plant species richness increased with increasing shape index SHPI and the proportion of urban areas in a 200 m buffer, while increasing the proportion of arable land and nutrients had a negative effect. The deviance explained in the final model accounted for 78% (Fig. 5A; Table 4).
Prediction accuracy of alien plant species richness models: A ornamental plants, B ornamental herbaceous plants, C ornamental bushes, D ornamental trees and E weeds. The green line represents fitted values: points below and above the green line are overestimated low species richness and underestimated high species richness, respectively
Species richness of individual groups of ornamental plants according to their life strategy (ornamental herbaceous, ornamental bushes and ornamental trees) was best explained with a similar set of variables (SHPI, the proportion of urban areas and arable land in the buffer surrounding a forest fragment and nutrients in the soil), with only slight differences among specific ornamental groups. However, the deviance explained in the final models was lower for ornamental herbaceous, ornamental bushes and ornamental trees when compared to all ornamental plants combined (55%, 76% and 57% vs. 78%, respectively; Fig. 5B–D; Table 4).
The building index explained 56% of alien ornamental plant species richness variance and 94% of ornamental bush richness variance in forest fragments. Regardless of its high predictive value, the building index was omitted from the models because of a strong correlation with the proportion of urban areas in buffers, which exceeded R2 > 0.70 (Fig. 6).
Weed species richness increased with increasing shape complexity and light, while the effect of moisture was negative. Variables indicating the presence of anthropogenic disturbance in the forest fragments did not affect weed richness. The final model accounted for 44% of deviance explained (Fig. 5E; Table 4).
Environmental drivers of plant species composition in forest fragments
According to the PCA and redundancy analysis, the plant species composition between Apaško polje and Dravsko polje is similar (results not shown). The results of the PCA show a separation of forest fragments according to their surrounding urbanization level, which coincides well with the invasion of various plant groups (Fig. 7). The ordination of plant species groups (Fig. 7A) explained 86.5% of the total variation. The first principal component, covering 70.7% of the variance, was strongly negatively influenced by the richness of overall (− 0.96), native (− 0.89), alien (− 0.87), invasive alien plants (− 0.79) and archaeophytes (− 0.64). The second PCA axis explained 15.8% of the variance and was negatively correlated to the archaeophytes (− 0.65) and positively to the IAPS (0.49). Moreover, the ordination of only alien plant groups (Fig. 7B) explained 58.9% of the total variation. The first principal axis (40.1%) was strongly positively influenced by all ornamental species groups (0.75–0.90) and fruit trees (0.62), while weeds (− 0.62) and trees planted for silviculture (− 0.76) contributed negatively to the second PCA axis, explaining 18.8% of the variance. In contrast to the above PCA, forest fragments were separated according to the urbanization level. The urbanization level in the buffer surrounding a forest fragment determined invasions by alien plant species (Fig. 7B).
PCA ordination of A overall plant species composition and B alien plant species in relation to low, medium or high urbanization level surrounding forest fragment. (SR overall plant species richness, NAT native plant species richness, AS alien plant species, IAS invasive alien plant species, Arch Archaeophytes)
Redundancy analysis of alien plant groups confirmed that differences in alien plant species composition in forest fragments can be explained by the urbanization level in the surrounding matrix. However, the separation is unclear, instead showing a partial distinction of forest fragments in low and medium vs. high urbanized matrix. The composition of all alien plant species groups was best explained with the following environmental variables: the share of urban area in 200 m buffer and Ellenberg indicator values for light and nutrients (Fig. 8, Table 5). The final model accounted for 35.46% of the total explained variance.
Biplot of redundancy analysis (RDA) of alien plant species groups and environmental variables in the surrounding urbanization gradient. RDA1 and RDA2 accounted for 19.53 and 14.08% of explained variance, respectively. The color of the circles indicates the urbanization gradient: red—low, green—medium, black—high
Discussion
We assess the impact of landscape structure with an emphasis on various land-cover types on overall richness and particularly on alien plant species richness and composition in lowland forest fragments located in agricultural landscapes with detailed examinations along forest fragment perimeters and in their wider surrounding area.
Small fragments, even tiny ones are of relevance; however, their existence and functionality are threatened by human disturbance. In our study sites, even though at two different lowlands, persistent agricultural use resulted in very similar forest fragments according to their size and shape variability. The landscape composition surrounding the fragments was similar as well and is summarized below. Buffer zones in the distance of 20–500 m around forest fragments consisted mainly of arable land (over 70%), followed by other agricultural uses (e.g., meadows and traditional mosaic land-cover). On average, 6% of the buffer zones accounted for urbanized areas and roads, both types more frequent in Dravsko polje. The vicinity of urbanized areas to forest fragments reflects increased human pressure assessed by the presence of footpaths and waste disposal in forest fragments as well as in the increased urbanization level in the matrix.
Our study indicates the significance of preserved fragments of the sub-Pannonian Quercus-Carpinus forest. We recognize these as biodiversity refugia, and even the smallest fragments no bigger than 0.1 ha can support as many as 45 native plant species among which 13% were even characteristic ancient forest species. On the other hand, the same type of forest fragment was not resistant to invasion, and 9% of the fragment’s flora comprised alien plant species (5% invasive alien plant species), while the most invaded fragment harbored as many as 20% alien species. The total number of listed native plants in the sampled forest fragments exceeded 10% of national flora (Martinčič et al. 2007), clearly indicating the importance of forest fragment conservation in a rural–urban matrix regardless of its area (Decocq et al. 2016).
Fragment shape complexity is a major driver of plant species richness in forest fragments
The most important variables affecting species richness in forest fragments are fragment area, isolation, habitat heterogeneity and the age of the stand (Decocq et al. 2016; Šipek et al. 2022). However, their effects can vary from region to region. Generally, native and alien plant species richness were associated with similar fragment characteristic variables such as shape complexity SHPI. Matrix variables were also important, but their effect was specific to each plant species group. However, not all studies confirm the effect of the matrix on alien plant species richness. Similar to our results, Dauber et al. (2003) have shown the importance of grassland fragment characteristics on plant species richness, but contrary to our findings, land-cover adjacent to the fragments did not affect plant richness. Pauchard and Alaback (2006) concluded that in North American Pinus forests, the matrix affected native plant species richness, while surprisingly, there was no effect on alien plant species richness.
Fragment shape affects the invasion susceptibility of the habitat because it determines how strongly the fragment is embedded into the matrix, which is the source of alien plant propagules. Contrary to our expectations, increasing the proportion of edge toward the forest area itself (P/A ratio) in our study did not result in higher native plant or alien plant species richness; instead, this relationship was negative. On the other hand, larger shape complexity SHPI resulted in increased native as well as alien plant species richness, which is in accordance with the study of Slovakian Pannonian floodplain forests (Petrášová-Šibíková et al. 2017).
By definition, decreasing a fragment’s size increases the proportion of its edge toward its surroundings, thereby changing the fluxes of energy, nutrients and organisms among neighboring habitats (Forman 1995; Cadenasso et al. 2003). Consistent with the fundamental biogeography theory, we found a general trend that with increasing fragment area, the richness of overall plant species as well of the plant species subgroups increased (Hobbs 1988), but the area predictor did not improve any of our models and was therefore excluded from the final models. However, many studies have succeeded in finding a positive relationship between forest area and alien plants (Petrášová-Šibíková et al. 2017). Even more, the richness of alien plants is often found to be greater in smaller fragments because of the edge effect (Guirado et al. 2006; Ohlemüller et al. 2006). The reason for the inconsistency in the effect of fragment area on alien plant species richness might also lie in the sampling methodology, while usually a few smaller plots are sampled in the forest regardless of their size while the majority of the larger area is overlooked. Consequently, the relative sampled area is different in smaller and larger fragments and can result in a number of unnoticed alien plant species in larger fragments, especially those with only a small number of individuals. The latter is true for the majority of not (yet) invasive naturalized plants and for those with subspontaneous occurrence.
Adjacent land-cover and urbanization drive richness and determine alien plant species composition in forest fragments
When comparing forest fragments, changes in alien plant species richness may not be accompanied by changes in species composition. Besides unrevealing factors driving plant species richness, we also explore factors influencing alien plant species composition, which is determined by multiple factors. Primarily, alien plant species composition is determined by the surrounding landscape (González-Moreno et al. 2013) which is the source of alien plant propagules as well as local environmental conditions that enable alien plants to establish and persist. Urbanization affects alien plant species composition and has a positive effect on alien plant abundance, richness, and diversity (Vilà and Ibáñez 2011), whereas the effect on native plant species is typically small, or there is no discernible effect (Wang et al. 2020). The higher level of urbanization in Dravsko polje than in Apaško polje is exhibited in the significantly higher richness and proportion of alien plants in Dravsko polje, even though similar native plant richness was recorded in both lowlands. In our study, forest fragments in a low urbanized matrix had a lower richness of alien plant species than similar-sized forest fragments embedded in a high urbanized matrix. Contrary to our expectations the effect of urbanization level was less pronounced in the smallest forest fragments. However, proximity to urban areas alone does not always result in increased invasion; instead, invasion depends on proximity to settlements because alien plant propagule pressure here is higher (Vidra and Shear 2008). Settlements with gardens in their backyards are major propagule sources of ornamental plants, which are mainly of foreign origin (Pergl et al. 2016).
We found a very strong correlation between overall ornamental alien plant and ornamental bush richness in forest fragments and the building index, which could serve as a surrogate for propagule pressure. Forests near settlements are under high human pressure, resulting in numerous disturbances. Forest fragments are crisscrossed with footpaths—corridors of highly disturbed, continuously trampled forest habitat. Walkers can introduce plant propagules from the surrounding landscape, which become caught in their clothing and footwear and increase the number of non-forest plants in forest habitats. Illegal garden waste disposal in forest fragments additionally increased alien plant species richness, mainly ornamentals (Rusterholz et al. 2012). In Slovenia, approximately 10% of garden owners dispose of garden waste in nearby forests, and doing this increases the risk of establishment and spread of ornamental plants into natural habitats (Šipek and Šajna 2020). Ornamental alien plants established in forests in the vicinity of garden waste are usually few and subspontaneous, but they may become established, naturalized and even invasive in the future (Haeuser et al. 2018; Klonner et al. 2019).
On the other hand, arable land-cover decreased alien plant as well as invasive alien plant richness; however, it had no effect on native plant richness, which is consistent with other studies showing that the matrix had a higher impact on alien than native plants (González-Moreno et al. 2013). Surprisingly, even alien weeds were in a negative relationship with an increasing proportion of arable land in the matrix; nevertheless, weeds are confined to arable land and are able to persist in forest edges (Honnay et al. 2002). Ohlemüller et al. (2006) previously found the negative effect of agricultural land-cover around forest fragments on alien plant richness, but the effect on native plant richness is unknown, while Daniels et al. (2019) even found a positive relationship between the percentage of arable land and invasive plant cover in forests.
Riparian zones such as riverbanks and floodplain forests are among the most highly invaded habitats in Europe (Castro-Díez and Alonso 2017; Marinšek and Kutnar 2017), which we can confirm from our field observations. Forest edges near rivers were covered by several invasive alien plants (Fallopia sp., Impatiens glandulifera, Solidago gigantea and others). Therefore, we would expect to find a positive relationship between invasive alien plant species richness and the presence of rivers in the forest fragments buffer zone. This was true only when the river was the sole explanatory variable included in the GLM (results not shown; p < 0.0001). However, when the river variable was included in the models beside other explanatory variables, it was not a significant predictor of alien or invasive alien plant species richness. Instead, other landscape variables were more important (e.g., shape of the fragment and the proportion of arable land). Another reason could be the relatively small number of fragments along the rivers. Only 8 fragments (17%) had a river (including streams and melioration canals) in a 200 m buffer zone, and 9 fragments (18%) had a river in at least a part of the forest edge. Besides that, even if the river was present in a forest buffer, environmental conditions of the forest fragment may prevent the establishment of alien or invasive alien plant species otherwise associated with river banks and floodplains. Similar to our results, Daniels et al. (2019) could not improve models predicting invasive plant cover in forests by including corridors such as roads and streams as explanatory variables in the models. Instead, other landscape variables were better predictors.
High-resource habitats are often more susceptible to invasion than those with limited resources (Funk 2013). Variables indicating abiotic conditions of the forest fragments in this study explained a considerably smaller proportion of plant species richness variance than landscape characteristics; however, they did influence alien plant species composition. As expected, weeds penetrating the forest fragments mainly from open agricultural landscapes were positively associated with greater light availability (Honnay et al. 2002). On the other hand, ornamental herbaceous plant species were positively associated with greater nutrient availability. We expected a positive effect of Ellenberg indicator values for nutrients on the richness of alien or invasive alien plant species. However, the analyses revealed a negative effect. Two main reasons could account for these findings. Firstly, the lack of indicator values of alien plant species may contribute to this result. The group of alien plant species is very heterogeneous, including a wide variety of ornamental species as well as weed species that are less dependent on nutrient rich sites. The second reason for this unexpected negative effect may be the relatively high values for nutrients in all forest fragments ranging from 5.48 to 6.96 (mean nutrient value was 6.20).
Conclusion
When we overlook small forest fragments, especially if they are embedded in an urban–rural matrix, the landscape can lose its spatial coherence established in an otherwise unforested matrix. Our results suggest larger forest fragments are not per se more resistant to penetration and invasion of alien plant species; instead, invasion depends on the urbanization level of the matrix. Forest fragments preserved in an intensive agricultural matrix can, on the one hand, support native plant species richness—–functioning as a biodiversity sanctuary in otherwise degraded landscapes, while at the same time the agricultural matrix act as a barrier against alien plant penetration from urbanized areas into the forest fragments. Therefore, conserving larger fragments of natural habitats is not by itself sufficient but should also take into account the matrix which is a source of anthropogenic disturbances and alien plant propagules. Finally, the high native plant species diversity of the smallest fragments, even with several forest specialist species indicates their high conservation value.
References
Bartuszevige AM, Gorchov DL, Raab L (2006) The relative importance of landscape and community features in the invasion of an exotic shrub in a fragmented landscape. Ecography 29:213–222. https://doi.org/10.1111/j.2006.0906-7590.04359.x
Beaury EM, Patrick M, Bradley BA (2021) Invaders for sale: the ongoing spread of invasive species by the plant trade industry. Front Ecol Environ 19:1540–9295. https://doi.org/10.1002/fee.2392
Becerra P, Simonetti JA (2020) Native and exotic plant species diversity in forest fragments and forestry plantations of a coastal landscape of central Chile. Bosque 41(2):125–136. https://doi.org/10.4067/S0717-92002020000200125
Borgmann KL, Rodewald AD (2005) Forest restoration in urbanizing landscapes: interactions between land-uses and exotic shrubs. Restor Ecol 13:334–340. https://doi.org/10.1111/j.1526-100X.2005.00042.x
Botkin D, Saxe H, Araujo MB, Betts R, Bradshaw RHW, Cedhagen T, Chesson P, Dawson TP, Etterson JR, Faith DP, Ferrier S, Guisan A, Skjoldborg HA, Hilbert DW, Loehle C, Margules C, New M, Sobel MJ, Stockwell DRB (2007) Forecasting effects of global warming on biodiversity. Bioscience 57:227–236. https://doi.org/10.1641/B570306
Building Cadastre (1999) Surveying and Mapping Authority of the Republic of Slovenia. https://eprostor.gov.si/imps/srv/eng/catalog.search#/metadata/5aa2b97b-2ea3-427f-a9fb-d7827c61c115. Accessed 10 Apr 2020
Cadenasso ML, Pickett STA, Weathers KC, Jones CG (2003) A framework for a theory of ecological boundaries. Bioscience 53:750–758. https://doi.org/10.1641/0006-3568(2003)053[0750:AFFATO]2.0.CO;2
Čarni A, Jarnjak M, Oštir-Sedej K (1998) Past and present forest vegetation in NE Slovenia derived from old maps. Appl Veg Sci 1:253–258. https://doi.org/10.2307/1478955
Castro-Díez P, Alonso A (2017) Effects of non-native riparian plants in riparian and fluvial ecosystems: a review for the Iberian Peninsula Pilar. Limnetica 36:525–541. https://doi.org/10.23818/limn.36.19
Chapman JI, Myers AL, Burky AJ, McEwan RW (2015) Edge effects, invasion, and the spatial pattern of herb-layer biodiversity in an old-growth deciduous forest fragment. Nat Areas J 35(3):439–451. https://doi.org/10.3375/043.035.0307
Daniels MK, Iacona GD, Armsworth PR, Larson ER (2019) Do roads or streams explain plant invasions in forested protected areas? Biol Invasions 21:3121–3134. https://doi.org/10.1007/s10530-019-02036-3
Dauber J, Hirsch M, Simmering D, Waldhardt R, Otte A, Wolters V (2003) Landscape structure as an indicator of biodiversity: matrix effects on species richness. Agric Ecosyst Environ 98:321–329. https://doi.org/10.1016/S0167-8809(03)00092-6
Davies CE, Moss D, Hill MO (2004) EUNIS habitat classification revised 2004. European Topic Centre on Nature Protection and Biodiversity, Paris
Decocq G, Andrieu E, Brunet J, Chabrerie O, De Frenne P, De Smedt P, Deconchat M, Diekmann M, Ehrmann S, Giffard B et al (2016) Ecosystem services from small forest patches in agricultural landscapes. Curr For Rep 2:30–44. https://doi.org/10.1007/s40725-016-0028-x
Ellenberg H, Weber HE, Dull R, Wirth V, Werner W, Paulissen D (1992) Zeigerwerte von Pflanzen in Mitteleuropa. Scr Geobot 18:1–248
ESRI (2014) ArcGIS 10.2.2 for Desktop. CA: Environmental Systems Research Institute, Redlands
European Environment Agency (EEA) (2018) Data and Maps: Copernicus Land Monitoring Service - Corine Land Cover. https://www.eea.europa.eu/data-and-maps/data/copernicus-land-monitoring-service-corine. Accessed 10 Apr 2020
Fahrig L (2013) Rethinking patch size and isolation effects: the habitat amount hypothesis. J Biogeogr 40:1649–1663. https://doi.org/10.1111/jbi.12130
Forman RTT (1995) Land mosaics: the ecology of landscapes and regions. Cambridge University Press, Cambridge
Fornal-Pieniak F, Ollik M, Schwerk A (2019) Impact of different levels of anthropogenic pressure on the plant species composition in woodland sites. Urban For Urban Green 38:295–304. https://doi.org/10.1016/j.ufug.2019.01.013
Fox J, Weisberg S (2019) An R companion to applied regression, 3rd edn. Sage, Thousand Oaks CA
Funk JL (2013) The physiology of invasive plants in low-resource environments. Conserv Physiol 1:cot026. https://doi.org/10.1093/conphys/cot026
Gaggini L, Rusterholz H-P, Baur B (2017) Settlements as a source for the spread of non-native plants into Central European suburban forests. Acta Oecol 79:18–25. https://doi.org/10.1016/j.actao.2016.12.008
González-Moreno P, Pino J, Gassó N, Vilà M (2013) Landscape context modulates alien plant invasion in Mediterranean forest edges. Biol Invasions 15:547–557. https://doi.org/10.1007/s10530-012-0306-x
Google Earth Pro (2020) https://www.google.com/intl/sl/earth/versions/#earth-pro. Accessed 10 Sept 2019
Gozdnogospodarski načrt (GGN) za mariborsko gozdnogospodarsko območje 2016–2025 (2016) Zavod za gozdove Slovenije, OE Maribor, Maribor
Guirado M, Pino J, Rodà F (2006) Understory plant species richness and composition in metropolitan forest archipelagos: effects of forests size, adjacent land-use and distance to the edge. Glob Ecol Biogeogr 15:50–62. https://doi.org/10.1111/j.1466-822X.2006.00197.x
Guisan A, Zimmermann NE (2000) Predictive habitat distribution models in ecology. Ecol Modell 135:147–186. https://doi.org/10.1016/S0304-3800(00)00354-9
Guo W-Y, van Kleunen M, Pierce S, Dawson W, Essl F, Kreft H, Maurel N, Pergl J, Seebens H, Weigelt P, Pyšek P (2019) Domestic gardens play a dominant role in selecting alien species with adaptive strategies that facilitate naturalization. Global Ecol Biogeogr 28:628–639. https://doi.org/10.1111/geb.12882
Haddad NM, Brudvig LA, Clobert J, Davies KF, Gonzalez A, Holt RD, Lovejoy TE, Sexton JO, Austin MP, Collins CD, Cook WM, Damschen EI, Ewers RM, Foster BL, Jenkins CN, King AJ, Laurance WF, Levey DJ, Margules CR, Melbourne BA, Nicholls AO, Orrock JL, Song D-X, Townshend JR (2015) Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci Adv. https://doi.org/10.1126/sciadv.1500052
Haeuser E, Dawson W, Thuiller W, Dullinger S, Block S, Bossdorf O, Carboni M, Conti L, Dullinger I, Essl F, Klonner G, Moser D, Münkemüller T, Parepa M, Talluto MV, Kreft H, Pergl J, Pyšek P, Weigelt P, Winter M, Hermy M, Van der Veken S, Roquet C, van Kleunen M (2018) European ornamental garden flora as an invasion debt under climate change. J Appl Ecol 55:2386–2395. https://doi.org/10.1111/1365-2664.13197
Harsch MA, Zhou Y, Leung R, Phillips A, Rinnan S, Kot M (2017) Moving forward: insights and applications of moving habitat models for climate change ecology. J Ecol 105:1169–1181. https://doi.org/10.1111/1365-2745.12724
Hobbs ER (1988) Species richness of urban forest patches and implications for urban landscape diversity. Landscape Ecol 1:141–152. https://doi.org/10.1007/BF00162740
Honnay O, Verheyen K, Hermy M (2002) Permeability of ancient forest edges for weedy plant species invasion. For Ecol Manag 161:109–122. https://doi.org/10.1016/S0378-1127(01)00490-X
Jansen F, Ewald J, Zerbe S (2011) Ecological preferences of alien plant species in North-Eastern Germany. Biol Invasions 13:2691–2701. https://doi.org/10.1007/s10530-011-9939-4
Jogan N, Bačič M, Strgulc P, Krajšek S (2012) Neobiota Slovenije: Invazivne tujerodne vrste v Sloveniji ter vpliv na ohranjanje biotske raznovrstnosti in trajnostno rabo virov. Univerza v Ljubljani (Biotehniška fakulteta). Končno poročilo. CRP »Konkurenčnost Slovenije 2006–2013«, Ljubljana
Klonner G, Wessely J, Gattringer A, Moser D, Dullinger I, Hülber K, Rumpf SB, Block S, Bossdorf O, Carboni M, Conti L, Dawson W, Haeuser E, Hermy M, Münkemüller T, Parepa M, Thuiller W, Van der Veken S, Verheyen K, van Kleunen M, Essl F, Dullinger S (2019) Effects of climate change and horticultural use on the spread of naturalized alien garden plants in Europe. Ecography 42:1548–1557. https://doi.org/10.1111/ecog.04389
Kowalska A, Affek A, Wolski J, Regulska E, Kruczkowska B, Zawiska I, Kołaczkowska E, Baranowski J (2021) Assessment of regulating ES potential of lowland riparian hardwood forests in Poland. Ecol Indic 120:106834. https://doi.org/10.1016/j.ecolind.2020.106834
LaPaix R, Harper K, Freedman B (2012) Patterns of exotic plants in relation to anthropogenic edges within urban forest remnants. Appl Veg Sci 15:525–535. https://doi.org/10.1111/j.1654-109X.2012.01195.x
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280. https://doi.org/10.1007/s004420100716
Lindborg R (2007) Evaluating the distribution of plant life-history traits in relation to current and historical landscape configurations. J Ecol 95:555–564. https://doi.org/10.1111/j.1365-2745.2007.01232.x
Maheu-Giroux M, de Blois S (2007) Landscape ecology of Phragmites australis invasion in networks of linear wetlands. Landsc Ecol 22:285–301. https://doi.org/10.1007/s10980-006-9024-z
Mandlebrot BB (1977) Fractals, Form, Chance and Dimension. W.H Freeman and Co., New York
Marinšek A, Kutnar L (2017) Occurrence of invasive alien plant species in the floodplain forests along the Mura River in Slovenia. Period Biol 119(4):251–260. https://doi.org/10.18054/pb.v119i4.4933
Martinčič A, Wraber T, Jogan N, Podobnik A, Turk B, Vreš B, Ravnik V, Frajman B, Strgulc Krajšek S, Trčak B, Bačič T, Fischer MA, Eler K, Surina B (2007) Mala flora Slovenije. Ključ za določanje praprotnic in semenk. Četrta, dopolnjena in spremenjena izdaja. Tehniška založba Slovenije, Ljubljana
McDonald RI, Urban DL (2006) Edge effects on species composition and exotic species abundance in the North Carolina Piedmont. Biol Invasions 8:1049–1060. https://doi.org/10.1007/s10530-005-5227-5
Medvecká J, Jarolímek I, Senko D, Svitok M (2014) Fifty years of plant invasion dynamics in Slovakia along a 2.500 m altitudinal gradient. Biol Invasions 16:1627–1638. https://doi.org/10.1007/s10530-013-0596-7
Ohlemüller R, Walker S, Wilson JB (2006) Local vs. Regional factors as determinants of the invasibility of indigenous forest fragments by alien plant species. Oikos 112:493–501. https://doi.org/10.1111/j.0030-1299.2006.1388
Okimura T, Mori AS (2018) Functional and taxonomic perspectives for understanding the underlying mechanisms of native and alien plant distributions. Biodivers Conserv 27:1453–1469. https://doi.org/10.1007/s10531-018-1503-4
Oksanen J, Simpson G, Blanchet F, Kindt R, Legendre P, Minchin P, O'Hara R, Solymos P, Stevens M, Szoecs E, Wagner H, Barbour M, Bedward M, Bolker B, Borcard D, Carvalho G, Chirico M, De Caceres M, Durand S, Evangelista H, FitzJohn R, Friendly M, Furneaux B, Hannigan G, Hill M, Lahti L, McGlinn D, Ouellette M, Ribeiro Cunha E, Smith T, Stier A, Ter Braak C, Weedon J (2022) vegan: Community Ecology Package. R package version 2.6-4. https://CRAN.R-project.org/package=vegan
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528. https://doi.org/10.1093/bioinformatics/bty633
Patton DR (1975) A diversity index for quantifying habitat “edge.” Wildl Soc Bull 3:171–173
Pauchard A, Alaback PB (2004) Influence of elevation, land use and landscape context on patterns of alien plant invasions along roadsides in protected areas of south-central Chile. Conserv Biol 18:238–248. https://doi.org/10.1111/j.1523-1739.2004.00300.x
Pauchard A, Alaback PB (2006) Edge type defines alien plant species invasions along Pinus contorta burned, highway and clearcut forest edges. For Ecol Manag 223:327–335. https://doi.org/10.1016/j.foreco.2005.11.020
Pergl J, Sádlo J, Petřík P, Danihelka J, Chrtek J, Hejda M, Moravcová L, Perglová I, Štajerová K, Pyšek P (2016) Dark side of the fence: ornamental plants as a source of wildgrowing flora in the Czech Republic. Preslia 88:163–184
Petrášová M, Jarolímek I, Medvecká J (2013) Neophytes in Pannonian hardwood floodplain forests—history, present situation and trends. For Ecol Manag 308:31–39. https://doi.org/10.1016/j.foreco.2013.07.041
Petrášová-Šibíková M, Bacigál T, Jarolímek I (2017) Fragmentation of hardwood floodplain forests—How does it affect species composition? Community Ecol 18:97–108. https://doi.org/10.1556/168.2017.18.1.11
R Core Team (2023) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reichard SH, White P (2001) Horticulture as a pathway of invasive plant introductions in the United States. Bioscience 51:103–113. https://doi.org/10.1641/0006-3568(2001)051[0103:HAAPOI]2.0.CO;2
Repe B (2004) Vegetation of Slovenia, in: Orožen Adamič, M. (Ed.), Slovenia: a geographical overview. Association of the Geographical Societies of Slovenia: Založba ZRC, Ljubljana, p. 5762
Rusterholz HP, Wirz D, Baur B (2012) Garden waste deposits as a source for non-native plants in mixed deciduous forests. Appl Veg Sci 15:329–337. https://doi.org/10.1111/j.1654-109X.2011.01175.x
Ščap D (2018) Apaško polje po poti strpnosti skozi zgodovino. Občina, Apače
Šibíková M, Jarolímek I, Hegedüšová K, Májeková J, Mikulová K, Slabejová D, Škodová I, Zaliberová M, Medvecká J (2019) Effect of planting alien Robinia pseudoacacia trees on homogenization of Central European forest vegetation. Sci Total Environ 687:1164–1175. https://doi.org/10.1016/j.scitotenv.2019.06.043
Šipek M, Šajna N (2020) Public opinions and perceptions of peri-urban plant invasion: the role of garden waste disposal in forest fragments. Manag Biol Invasions 11(4):733–746. https://doi.org/10.3391/mbi.2020.11.4.08
Šipek M, Kutnar L, Marinšek A, Šajna N (2022) Contrasting responses of alien and ancient forest indicator plant species to fragmentation process in the temperate lowland forests. Plants 11(23):3392. https://doi.org/10.3390/plants11233392
Sladek P, Strgulc Krajšek S (2019) Forsitije (Forsythia Vahl) v Sloveniji. [In Slovenian: Forsythias (Forsythia Vahl) in Slovenia]. Hladnikia 44:10–29
Sullivan JJ, Timmins SM, Williams PA (2005) Movement of exotic plants into coastal native forests from gardens in northern New Zealand. N Z J Ecol 29:1–10
Tichý L (2002) JUICE, software for vegetation classification. J Veg Sci 13:451–453. https://doi.org/10.1111/j.1654-1103.2002.tb02069.x
Trentanovi G, von der Lippe M, Sitzia T, Ziechmann U, Kowarik I, Cierjacks A (2013) Biotic homogenization at the community scale: disentangling the roles of urbanization and plant invasion. Divers Distrib 19:738–748. https://doi.org/10.1111/ddi.12028
Valdés A, Lenoir J, De Frenne P, Andrieu E, Brunet J, Chabrerie O, Cousins SAO et al (2020) High ecosystem service delivery potential of small woodlands in agricultural landscapes. J Appl Ecol 57:4–16. https://doi.org/10.1111/1365-2664.13537
van Kleunen M, Essl F, Pergl J, Brundu G, Carboni M, Dullinger S, Early R, González-Moreno P, Groom QJ, Hulme PE, Kueffer C, Kühn I, Máguas C, Maurel N, Novoa A, Parepa M, Pyšek P, Seebens H, Tanner R, Touza J, Verbrugge L, Weber E, Dawson W, Kreft H, Weigelt P, Winter M, Klonner G, Talluto MV, Dehnen-Schmutz K (2018) The changing role of ornamental horticulture in alien plant invasions. Biol Rev 93:1421–1437. https://doi.org/10.1111/brv.12402
Vidra RL, Shear TH (2008) Thinking locally for urban forest restoration: a simple method links exotic species invasion to local landscape structure. Restor Ecol 16:217–220. https://doi.org/10.1111/j.1526-100X.2008.00387.x
Vilà M, Ibáñez I (2011) Plant invasions in the landscape. Landsc Ecol 26:461–472. https://doi.org/10.1007/s10980-011-9585-3
Vodeb V, Mladenovič L, Nikšič M, Cotič B, Šuklje Erjavec I, Praper S, Gulič A, Sendi R, The Urban planning Institute of the Republic of Slovenia, Miklavčič T, Robič G, Uršič N, Ministry of the Environment and Spatial Planning, Spatial Planning, Construction and Housing Directorate (2016) National report on urban development—Habitat III. Ministry of the Environment and Spatial Planning, Spatial Planning, Construction and Housing Directorate, Ljubljana. https://www.gov.si/assets/ministrstva/MOP/Publikacije/9d3d8bd710/porocilo_urbani_razvoj_HabitatIII_en.pdf. Accessed 17 Mar 2020
Walter J, Essl F, Englisch T, Kiehn M (2005) Neophytes in Austria: Habitat preferences and ecological effects. In: Nentwig W, et al (eds.) Biological invasions—from ecology to control. NEOBIOTA, vol 6, pp 13–25
Wang M, Li J, Kuang S, He Y, Chen G, Huang Y, Song C, Anderson P, Łowicki D (2020) Plant diversity along the urban–rural gradient and its relationship with urbanization degree in Shanghai, China. Forests 11:171. https://doi.org/10.3390/f11020171
Ward SG, Amatangelo KL (2018) Suburban gardening in Rochester, New York: exotic plants preference and risk of invasion. Landsc Urban Plan 180:161–165. https://doi.org/10.1016/j.landurbplan.2018.09.004
Weber JS, Gibson KD (2007) Exotic plant species in old-growth forest in Indiana. Weed Sci 55:299–304. https://doi.org/10.1614/WS-06-164.1
Acknowledgements
We acknowledge Andrej Kosednar for his valuable help with the fieldwork. We are grateful to the two anonymous reviewers for their constructive comments and suggestions, which have enhanced this article.
Funding
The study was partly supported by the Slovene Ministry of Higher Education, Science and Technology within research programs P1-0403 and J1-2457.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by MŠ. The first draft of the manuscript was written by MŠ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Šipek, M., Šajna, N. Lowland forest fragment characteristics and anthropogenic disturbances determine alien plant species richness and composition. Biol Invasions 26, 1595–1614 (2024). https://doi.org/10.1007/s10530-024-03269-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-024-03269-7