Abstract
The impact of non-native species, together with their pervasiveness, necessitates a means of identifying which species are most likely to pose an elevated risk of becoming invasive. This is amongst the first applications of the Aquatic Species Invasiveness Screening Kit (AS-ISK) decision support tool in Southeast Asia, and specifically for Vietnam. In total, 30 extant and horizon non-native aquatic species were screened for their potential invasiveness in Vietnam. A threshold score of 6.75 was identified for distinguishing between species likely to pose a high and low-to-medium risk of becoming invasive in Vietnam for both the basic risk assessment (BRA) and the climate change assessment (BRA + CCA) components of the screening process. However, the absence of nationally-consistent predictions on climate change impacts in Vietnam restricted the applicability of the climate change assessment component. Based on the BRA threshold, of the 30 species screened, 25 were classified as high risk, four as medium risk, and one as low risk. For the BRA, the highest-scoring species were Hypostomus plecostomus, Pomacea canaliculata and Lithobates catesbeianus. This study reliably identified and discriminated between non-invasive and potentially invasive aquatic species in Vietnam, thereby providing appropriate AS-ISK score thresholds with which to guide policy and decision-making. This suggests that the AS-ISK could be successfully employed to screen non-native aquatic species in other parts of Southeast Asia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although there is increase in knowledge and understanding of the adverse impacts exerted by invasive non-native species in some parts of the world, there remains a strong geographical skew in research effort, with Africa and Asia both seriously under-studied in terms of invasion ecology (Pyšek et al. 2008). Exceptions do exist for these continents, such as South Africa (e.g. Ellender and Weyl 2014; Weyl et al. 2016), China (e.g. Wong et al. 2009; Li et al. 2017; Wan et al. 2017) and Japan (e.g. Fausch et al. 2002; Onikura et al. 2011; Matsuzaki and Kadoya 2015). Nonetheless, the disparity in studies of non-native species can be largely explained by differing amounts of financial resources being available in different regions of the world, and in different countries within regions; this, in turn, translates into research intensity (Pyšek et al. 2006). To achieve a more geographically-balanced picture of biological invasions, researchers have called for international cooperation (Pyšek et al. 2008). However, the continuation of such regional geographical bias was recently confirmed, with only 6% of conservation science studies published in 2011–2015 being in Southeast Asia (Di Marco et al. 2017). In the present article, non-native species are defined as organisms that were introduced directly or indirectly by people outside of their natural range of distribution. Invasive non-native species are the subset of non-native species that have become established and dispersed, generating an impact on local ecosystems and species (IPBES 2019).
This relative dearth of studies is even more concerning given that the tropics contain a disproportionately high amount of the world’s biological diversity, encompassing more than three-quarters of all terrestrial, freshwater and marine species (Barlow et al. 2018). Southeast Asia, which comprises the countries of Brunei, Cambodia, Indonesia, Laos, Malaysia, Myanmar (Burma), the Philippines, Singapore, Timor-Leste, Thailand and Vietnam, is a region of exceptionally high species endemism. It is also a region of species endangerment, with the highest proportion of threatened vascular plant, reptile and mammal species when compared to other tropical regions (Sodhi et al. 2010). Indeed, Southeast Asia has been identified as the region of a looming biodiversity disaster. Drivers of biodiversity threat in the region include forest conversion and forest fires, hunting, and wildlife trade (Sodhi et al. 2004). It has previously been postulated that various abiotic and biotic factors (e.g. high species diversity) will minimise the probability of successful establishment of non-native species in undisturbed communities in the tropics (Rejmánek 1996), thus reducing the risk of impacts from invasive species. However, the scale of human disturbance and synergistic impacts that is occurring in Southeast Asia will likely undermine any such mitigation factor (Peh 2010). Indeed, tropical ecosystems are predicted to become increasingly vulnerable to biological invasion as the twenty-first century progresses (Early et al. 2016). Unfortunately, the paucity of research in Southeast Asia (Sodhi et al. 2004, 2010), including on the level and types of impacts caused by invasive non-native species (Nghiem et al. 2013), makes it difficult to establish the relative importance and long-term implications of the latter relative to other threats. As such, the situation with invasive non-native species in Southeast Asia remains very poorly documented and understood (Peh 2010; Nghiem et al. 2013).
Vietnam readily exemplifies this situation, with study of non-native species in the country nascent (Zworykin and Budaev 2013). Thus, the full extent of the establishment of invasive non-native species in Vietnam is currently unknown. For example, 956 non-native plant species (accounting for 9% of the country’s known flora) have been reported (Triet et al. 2008 in Le and Truong 2016), but the situation is likely to have changed since 2008. Similarly, for fishes, preliminary and now relatively dated reports indicate that at least nine species have naturalised in the River Mekong basin alone (Welcomme and Vidthayanom 2003; Cacot and Lazard 2009 in Zworykin and Budaev 2013), but there is no national compendium. The limited published literature that exists tends to focus on species that may affect agriculture (e.g. Dao et al. 2018; Wyckhuys et al. 2019) or human health (e.g. Shan et al. 2018). Surveys for, and/or management of, non-native species that may affect biodiversity are generally limited to protected areas (e.g. Tan et al. 2012; Le and Truong 2016).
In terms of government-led policy and management in Vietnam, a few laws (e.g. Law on Environmental Protection 2014; Law on Biodiversity 2008) make brief reference to non-native species. More specifically, regulation of non-native species that may impact on biodiversity is provided through two lists issued by the Ministry of Natural Resources and Environment. The first list covers ‘invasive’ non-native species (i.e. those that are already present in Vietnam, where they are known to have a substantial negative impact on biodiversity), while the second list is of ‘potentially invasive’ non-native species (i.e. those that are known to have impact on biodiversity in other countries, but have not yet had a significant impact in Vietnam). Separately, the Ministry of Agriculture and Rural Development has issued two lists of species (covering native and non-native species) that are considered capable of causing significant damage to agricultural plants: (1) ‘plant quarantine list I’ is of species (mainly insects, pathogens and plants) that are not yet distributed in Vietnam; and (2) ‘plant quarantine list II’ is of species that currently have a limited distribution in Vietnam. All four lists were developed based on criteria determined by the respective ministries; furthermore, various combinations of terminology, definitions and criteria are used across the four lists, as well as in other legal documents. As with other non-English speaking countries, the situation is further complicated by complexities relating to language. This is because definitions and criteria become less clear, due to increased linguistic uncertainty (Lu 2019; McGeoch et al. 2012), when legal documents, publications or research are translated into English.
The aim of the present study was to identify which extant and potential future (horizon) non-native aquatic species are likely to pose an elevated risk of becoming invasive in Vietnam. The specific objectives were to: (1) carry out screenings using a recently-developed, multilingual taxon-generic decision-support tool; (2) undertake a calibration in order to identify the threshold between non-native species that are likely to pose a high risk of becoming invasive, and those of low-to-medium risk for Vietnam; and (3) interpret the outcomes within a management context.
Materials and methods
Study area
The risk assessment area, Vietnam, spans latitudes 8°27′ to 23°23′ N and longitudes 102°08′ to 109°30′ E. The country extends 1,662 km from north to south and has a land area of ≈ 331,051 km2. There are also ≈ 1 million km2 of sea waters and ≈ 3,000 islands. If only perennial rivers and streams over 10 km are included, then Vietnam possesses ≈ 2,360 of them, with a mean density of 0.6 km/km2. Although there are nine major river systems in the country, two (the rivers Mekong and Red) are particularly important socio-economically (Ministry of Natural Resources and Environment 2010). As of 2018, the country’s population was over 95 million people, with annual population growth of 1% (World Bank Group 2019).
Vietnam’s climate is tropical, with rainy seasons that correspond to monsoon circulations. Mean annual rainfall ranges from 1400 to 2400 mm and each year the country is affected by multiple typhoons. Annual mean temperature ranges from 12.8 to 27.7 °C (Ministry of Natural Resources and Environment 2010). Projections of future climate conditions in the study region for 2050 indicate: (1) an increase in mean temperatures (ranging from 1.6 to 2.8 °C in different climate zones); (2) increased intensity of extreme weather events, including droughts, typhoons and floods; (3) increases in annual rainfall across all regions, with more extreme precipitation variability between the dry and rainy season; and (iv) a rise of 28–33 cm in sea levels (McElwee 2010; Ministry of Natural Resources and Environment 2009, 2010). However, different regions in the country are likely to have unique climate impacts, making a single national prediction difficult. Overall though, Vietnam is likely to be one of the most significantly impacted nations in the world with respect to climate change due to its long coastline, high dependence on agriculture, and relatively low levels of development in rural areas (McElwee 2010). Also, the River Mekong is recognised as one of the world’s most vulnerable mega-deltas, especially as a result of climate change-induced sea-level rise (Whitehead et al. 2019).
Toolkit description
Risk screenings were undertaken using the Aquatic Species Invasiveness Screening Kit (AS-ISK), which is available for free download at www.cefas.co.uk/nns/tools/ (Copp et al. 2016a). The AS-ISK can be used to screen 27 groups of aquatic organisms for freshwater, brackish and marine habitats and, since release of version 2.1, the AS-ISK has been available in 32 languages, including Vietnamese (Copp et al. 2021). The AS-ISK was constructed within the architecture of the Fish Invasive Screening Kit v2 (Lawson et al. 2013) using questions from the generic screening module of the European Non-native Species in Aquaculture Risk Analysis Scheme (Copp et al. 2016b). These questions were retained and/or revised, with additional questions and features, to comply with the ‘minimum standards’ (Roy et al. 2018) for the assessment of non-native species, as per the recent EC ‘Regulation No. 1143/2014 on the prevention and management of the introduction and spread of invasive alien species’. The AS-ISK consists of 55 questions: the first 49 cover the bio-geographical and biological aspects of the species under assessment and comprise the Basic Risk Assessment (BRA). The remaining six questions require the assessor to predict how future climatic conditions are likely to affect the BRA with respect to risks of introduction, establishment, dispersal and impact, and these comprise the Climate Change Assessment (CCA).
To achieve a valid AS-ISK assessment, the assessor must provide a response, justification and level of confidence to each question, with the result being a BRA score (ranging from −20 to 68) and a composite BRA + CCA score (ranging from −32 to 80). Basic AS-ISK scores < 1 suggest that the species is unlikely (i.e. poses a low risk) to become invasive in the risk assessment area, whereas scores > 1 indicate a medium-to-high risk of the species becoming invasive in the risk assessment area. The threshold BRA and BRA + CCA values that distinguish between medium and high risk levels are typically obtained through a procedure of ‘calibration’ that is specific to the risk assessment area, which is undertaken by Receiver Operating Characteristic (ROC) curve analysis (Bewick et al. 2004) (see Statistical analysis).
In the AS-ISK, confidence in responses to questions (Qs) are ranked using a 1–4 scale (1 = low; 2 = medium; 3 = high; 4 = very high), as per the Intergovernmental Panel on Climate Change (IPCC 2005; Copp et al. 2016a). Based on the confidence level (CL) allocated to each response, a confidence factor (CF) is obtained as:
where CLQi is the CL for Qi, 4 is the maximum achievable value for confidence (i.e. very high; see above) and 55 is the total number of Qs in the AS-ISK. Based on the BRA (49 Qs) and the CCA (6 Qs), the CLBRA and CLCCA are also computed. The CF ranges from a minimum of 0.25 (i.e. all 55 Qs with confidence level equal to 1) to a maximum of 1 (i.e. all 55 questions with confidence level equal to 4). Two additional CFs can be computed; namely, the CFBRA and the CFCCA (hence, similar to the CL).
Species selection and data processing
In total, 30 non-native aquatic species were screened for their potential invasiveness in the risk assessment area. Species were selected according to the following criteria: (1) extant; that is, already present in the risk assessment area (n = 26), and (2) horizon; that is, not yet reported but likely to enter the risk assessment area in the future (n = 4) (Table 1). Screenings were carried out by the first three authors (LR, KATT, TDB), who are knowledgeable in the biology and ecology of the aquatic species of the region, with GHC and LV being responsible for overseeing construction of the species list and for quality control of the generated AS-ISK database of screenings, respectively.
Following computation of the BRA and BRA + CCA scores, ROC curve analysis was used to assess the predictive ability of the AS-ISK to discriminate between species posing a high risk and those posing a medium or low risk of being invasive for the risk assessment area. For ROC curve analysis to be implemented, species need to be categorised a priori in terms of their documented invasiveness (i.e. non-invasive or invasive). In general, a priori categorisation of fishes is facilitated by the availability of online databases (i.e. FishBase). However, owing to more limited (literature-based) information being available for other groups of organisms, an ‘integrated approach’ was adopted in the present study to determine the a priori invasiveness status of species other than fishes: (1) similar to previous AS-ISK applications to fishes (i.e. Glamuzina et al. 2017; Li et al. 2017; Tarkan et al. 2017a, b; Bilge et al. 2019; Dodd et al. 2019; Clarke et al. 2020; Interesova et al. 2020; Uyan et al. 2020), a first search was made of FishBase (www.fishbase.org) and, for invertebrates, of SeaLifeBase (www.sealifebase.org), with the species categorised a priori as invasive if it was listed as a ‘potential pest’ and as non-invasive if it was listed as ‘harmless’; (2) a second search was made of the Global Invasive Species Database (GISD: www.iucngisd.org), with the species categorised a priori as invasive if it was listed therein; (3) a third search was made of the Invasive Species of Japan database (www.nies.go.jp/biodiversity/invasive/index_en.html) and the Invasive and Exotic Species of North America list (www.invasive.org), with the species categorised a priori as ‘invasive’ if listed therein; (4) if the species was absent from any of the previous databases, then a Google Scholar (literature) search was performed (using the keywords ‘invasive’, ‘invasiveness’ and ‘impact’ along with that of the species) to check whether at least one peer-reviewed reference in support was found. The latter was then taken as ‘sufficient evidence’ for categorising the species a priori as invasive; whereas, if no evidence was found, then the species was categorised a priori as non-invasive. Notably, if a species was listed as harmless in FishBase but found to be invasive in any of the other steps of the process, then the a priori categorisation of the species became that of invasive.
Statistical analysis
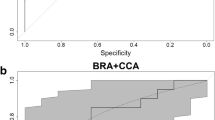
A ROC curve is a graph of sensitivity versus 1 – specificity (or alternatively, sensitivity vs. specificity) for each threshold value where, in the present context, sensitivity and specificity will be the proportion of a priori invasive and non-invasive aquatic species that are correctly identified by the AS-ISK as such. A measure of the accuracy of the calibration analysis is the Area Under the Curve (AUC), which typically ranges from 0.5 to 1; the closer to 1, the better the ability to differentiate between invasive and non-invasive species. If the AUC is equal to 1 then the test is 100% accurate because both sensitivity and specificity are 1, and there are neither ‘false positives’ (a priori non-invasive species classified as high risk, hence invasive) nor ‘false negatives’ (a priori invasive species classified as low risk, hence non-invasive). Conversely, if the AUC is equal to 0.5, then the test is 0% accurate as it cannot discriminate between ‘true positives’ (a priori invasive species classified as high risk, hence invasive) and ‘true negatives’ (a priori non-invasive species classified as low risk, hence non-invasive). AUC values are generally interpreted as follows: 0.7 ≤ AUC < 0.8 = acceptable discriminatory power, 0.8 ≤ AUC < 0.9 = excellent, 0.9 ≤ AUC = outstanding (Hosmer et al. 2013). Following ROC curve analysis, the best AS-ISK threshold value that maximises the true positives rate and minimises the false positives rate was determined using Youden’s J statistic; whereas, a ‘default’ threshold of 1 was set to distinguish between low- and medium-risk species (see Toolkit description). ROC curve analysis was carried out with the package ‘pROC’ (Robin et al. 2011) for R x64 v3.2.0 (R Development Core Team 2020) using 2000 bootstrap replicates for the confidence intervals of specificities, which were computed along the entire range of sensitivity points (i.e. 0–1, at 0.1 intervals).
Differences between mean confidence level and mean confidence factor for the BRA (CLBRA and CFBRA, respectively) and for the CCA (CLCCA and CFCCA, respectively) were tested by permutational ANOVA based on a one-factor design (i.e. Component, with two levels: BRA and CCA). Analysis was carried out in PERMANOVA+ for PRIMER v6, with normalisation of the data and using a Bray-Curtis dissimilarity measure, 9999 unrestricted permutations of the raw data (Anderson et al. 2008), and with statistical effects evaluated at α = 0.05.
Results
As ROC curve analysis yielded the same BRA and BRA + CCA thresholds when applied to all aquatic species and to fishes only, the outcomes for all aquatic species are reported here. The ROC curve for the BRA resulted in an AUC of 0.8333 (0.5067–1.0000 95% CI) and that for the BRA + CCA in an AUC of 0.8160 (0.5223–1.0000 95% CI). These AUCs were > 0.5 and had excellent discriminatory power, indicating that the AS-ISK was able to discriminate reliably between non-invasive and invasive non-native species for the risk assessment area. Youden’s J provided the same threshold of 6.75 for both the BRA and the BRA + CCA; this was used for calibration of the risk outcomes. Accordingly, the BRA threshold allowed distinction between medium-risk species, with scores within the interval [1, 6.75[, and high-risk species, with scores within ]6.75, 68]. The BRA + CCA threshold allowed distinction between medium-risk species, with scores within [1.0, 6.75[, and high-risk species, with scores within ]6.75, 80]. In contrast, species classified as low risk were those with BRA scores within [− 20, 1[ and BRA + CCA scores within [− 32, 1[. Note that thresholds reported hereafter are presented using the appropriate statistical use of interval brackets: ‘]’ and ‘[’ (www.mathwords.com/i/interval_notation.htm).
Of the 30 species screened (see Supplementary Material for AS-ISK reports), based on the BRA threshold, 25 (83.3%) were classified as high risk, four (13.3%) as medium risk, and one (3.3%) as low risk (Table 2). All 22 species categorised a priori as invasive were true positives, and amongst the eight species categorised a priori as non-invasive, one was a true negative (Cuban crocodile Crocodylus rhombifer) and three were false positives (suckermouth catfish Hypostomus plecostomus, elkhorn sea moss Kappaphycus alvarezii and whiteleg shrimp Penaeus vannamei). All four medium-risk species were categorised a priori as non-invasive. Based on the BRA + CCA threshold, 23 (76.7%) species were classified as high risk, four (13.3%) as medium risk, and three (10.0%) as low risk. Of the 22 species categorised a priori as invasive, 20 were true positives and there was one false negative (pirapitinga Piaractus brachypomus); amongst the eight species categorised a priori as non-invasive, there were two true negatives (Crocodylus rhombifer and streaked prochilod Prochilodus lineatus) and the same three false positives as for the BRA. Of the four medium-risk species, three were categorised a priori as non-invasive and one as invasive (rainbow trout Oncorhynchus mykiss).
For the BRA, the highest-scoring species (score ≥ 40, taken as an ad hoc ‘very high risk’ threshold) were suckermouth catfish Hypostomus plecostomus, channelled apple snail Pomacea canaliculata and American bullfrog Lithobates catesbeianus (from higher to lower scores). For the BRA + CCA, the highest-scoring species included all the above plus cane toad Rhinella marina. For both the BRA and the BRA + CCA, Siberian sturgeon Acipenser baerii, Mrigal carp Cirrhinus cirrhosus and Roho labeo Labeo rohita were classified as medium risk, and Crocodylus rhombifer as low risk (Table 2). The CCA resulted in an increase relative to the BRA score for five (16.7%) species, in a decrease for four (13.3%), and in no change for the remaining 21. Notably, Nile tilapia Oreochromis mossambicus and Rhinella marina achieved the largest possible positive change in score of 12, and Piaractus brachypomus and streaked prochilod Prochilodus lineatus the largest possible negative change in score of −12 (Table 2).
The mean CL (i.e. over all 55 questions) was 2.92 ± 0.06 SE, the mean CLBRA 3.00 ± 0.06 SE, and the mean CLCCA 2.26 ± 0.14 SE (hence, in all cases indicating medium to high confidence). The CLBRA was significantly higher than the CLCCA (\( F_{1,58}^{\# } \) = 25.56, P < 0.001). Similarly, mean values for CF = 0.730 ± 0.016 SE and CFBRA = 0.751 ± 0.015 SE were higher than the mean value for the CFCCA = 0.564 ± 0.034 SE, and the mean CFBRA was significantly higher than the mean CFCCA (with the same significance values as for the CLBRA vs. CLCCA comparison, due to the two indices being related).
Discussion
This study represents the first application of the AS-ISK for Vietnam and amongst the first for Southeast Asia in general. As with previous applications in other regions and countries (Glamuzina et al. 2017; Li et al. 2017; Tarkan et al. 2017a, b; Semenchenko et al. 2018; Bilge et al. 2019; Dodd et al. 2019; Clarke et al. 2020; Killi et al., 2020; Interesova et al. 2020; Uyan et al. 2020), the AS-ISK could reliably discriminate between invasive and non-invasive species in the risk assessment area. Furthermore, it is notable that only one false positive was identified, and that all species categorised a priori as invasive were ROC classified as high-risk (hence, true positives) for the BRA, and the vast majority of them also for the BRA + CCA. This result validates the adoption of the AS-ISK decision-support tool not only for Vietnam but ultimately for Southeast Asia.
The current risk screening for Vietnam established the same threshold of 6.75 for both the BRA and the BRA + CCA. Low threshold values calibrated for other risk assessment areas (i.e. Glamuzina et al. 2017; Semenchenko et al. 2018; Tarkan et al. 2017a) have been attributed to the high number of species translocated within those risk assessment areas, in particular to enclosed, artificial water bodies. The reasons for the low threshold score for Vietnam may be similar, given that Vietnam is a major hub for wildlife trade (Nguyen 2008), wildlife consumption (e.g. Venkataraman 2007), and commercial farming of wildlife (Drury 2009). It also has a culture of keeping both exotic and native animals as pets (e.g. Eaton et al. 2017) and of releasing large quantities of various species into the environment (particularly aquatic species into water bodies) during ceremonies (e.g. Tết). These activities, together with a tropical climate and broad latitudinal extent, provide substantial scope for both the introduction and establishment of non-native species in Vietnam. Overall, this suggests that non-native aquatic species, and especially fishes, pose a considerable threat to native species and ecosystems in Vietnam.
In terms of individual species, the highest-scoring species in the present study (i.e. Hypostomus plecostomus, Lithobates catesbeianus, Pomacea canaliculata and Rhinella marina) have all been categorised as invasive in the region, nearby regions, or internationally. For example, P. canaliculata has been widely introduced in Asia, including Vietnam, and has resulted in extensive damage to both agricultural land and native ecosystems, particularly wetlands (Carlsson et al. 2004; Joshi et al. 2017). Meanwhile, the situation with L. catesbeianus and Rhinella marina demonstrates the scale of knowledge gaps for non-native species in Vietnam. Both species are on the list of the ‘World’s Worst 100 Invaders’ and, in the case of L. catesbeianus, occurs in at least 41 countries on four continents, including neighbouring/nearby Asian countries e.g. Japan, Thailand, China, Taiwan (www.iucngisd.org/gisd/species.php?sc=80). There are no readily-available records for either species in Vietnam; however, given the proximity of their known non-native distribution, it is feasible that they occur in the country but have not been officially recognised.
Amongst the highest scoring of the screened species, Hypostomus plecostomus was the lone false positive. Notably, both the screening and the result for this species, as well as for Pterygoplichthys pardalis, are complicated by the fact that there is a high degree of taxonomic uncertainty for loricariids in general, and for species of the genera Hypostomus and Pterygoplichthys in particular (www.cabi.org/isc/datasheet/114927). Although Hypostomus was re-described by Armbruster (2004), additional taxonomic and systematic work is required because it is still difficult to identify most hypostomids to species level; this renders species identifications tentative if not tenuous. There are also no distinctive characteristics with which to diagnose the genus (Armbruster 2004); indeed, historically, Pterygoplichthys species have often been misidentified as hypostomids (www.fws.gov/fisheries/ans/erss/uncertainrisk/ERSS-Hypostomus-plecostomus-final.pdf; http://nas.er.usgs.gov/). Confounding issues in the present risk screening are: (1) both species have the same (multiple) common names in Vietnamese, and (2) multiple species of the genus can occur in a single location (Hoover et al. 2004). Using available literature, this makes it difficult to attribute with confidence the reported impacts to one of the two species.
The identical threshold score identified in the present study for distinguishing between medium- and high-risk species under current (BRA) and future (BRA + CCA) climate conditions contrasts other applications of the AS-ISK, where an increase in threshold value from BRA to BRA + CCA was identified for the majority of the species screened (i.e. Semenchenko et al. 2018; Bilge et al., 2019; Dodd et al. 2019; Clarke et al. 2020; Killi et al., 2020; Interesova et al. 2020; Uyan et al. 2020). The reason for the discrepancy may be attributed to the relatively low understanding of, and therefore confidence in, projected climate change impacts in Vietnam. This is further complicated by the fact that, owing to their geographical locations, various regions within Vietnam are likely to be affected differently by future changes. Overall, this makes it difficult to produce a single national projection (McElwee 2010). This climo-geographic issue is likely (if not certainly) to affect any risk screening or full assessment undertaken for any non-native species with respect to the entire country of Vietnam; it is also well demonstrated for AS-ISK screenings for Great Britain (Dodd et al. 2019) and for Italy with regard to channel catfish Ictalurus punctatus (Haubrock et al. 2021). This contrasts with other screening applications, which have tended to be for geographically-specific risk assessment areas (e.g. Glamuzina et al. 2017; Bilge et al. 2019), or for a country smaller than Vietnam and one that is therefore more climatically uniform (e.g. Belarus: Semenchenko et al. 2018).
Of note in the present study are the identical thresholds for current (BRA) and future (BRA + CCA) climate conditions when applied to all aquatic species and to fish species only. This means that this threshold can be reliably used to guide management decisions for all the taxonomic groups that were screened in the present study. However, because risk analysis is a dynamic process (Copp et al. 2016a), these risk screenings would need to be updated and revised when climate-change modelling within a climo-geographic context has been undertaken for Vietnam.
Conservation and management considerations
Research has identified that, except for unintentional hitchhiker (e.g. contaminant) species, non-native species are not randomly selected. Rather, humans tend to select species whose attributes are conducive to invasion success; furthermore, these species are introduced to areas in which the co-evolved enemies that limit the species’ abundance in their native range are absent (Buckley and Catford 2015; Rejmánek and Simberloff 2017). The main introduction vectors for aquatic non-native species to Vietnam presumably include aquaculture, captive breeding and deliberate release to the wild, as well as the pet/aquarium trade, though ranking of these is currently not possible.
The assessment of existing and potential future species (so-called horizon scanning) is crucial for identifying the most threatening of the potential invasive non-native species that do not yet occur in a risk assessment area (Roy et al. 2019). Failure to use a systematic, evidence-based approach for such assessments can lead to inconsistent and incomparable outcomes (Roy et al. 2018). To address this, the AS-ISK was enhanced to be consistent with the European Union (EU) practice of offering documents in national languages (Copp et al. 2021). Although the AS-ISK does not offer all official languages of EU member states, it does offer many of them, as well as several Asian languages (Chinese, Japanese, Korean, Filipino, Vietnamese, and Thai). Other risk screening tools exist for non-native species, such as Invasive Species Environmental Impact Assessment (Branquart 2009) and the Canadian Marine Invasive Species Tool (Drolet et al. 2016). However, few of the extant decision-support tools for non-native species are offered in more than one or a few languages. The AS-ISK (since v2.1) is the only known decision-support tool that offers the user the option of carrying out their screening in Vietnamese and, as such, serves as a means of communicating risks and uncertainties about non-native species to Vietnamese stakeholders and decision-makers (Copp et al. 2021).
As with its predecessor decision-support tool, the freshwater Fish Invasiveness Screening Kit (FISK: Copp et al. 2009; Vilizzi et al. 2019), the AS-ISK is not intended to make decisions but, rather, to inform policy and decision-makers. It is therefore an aid in the development of non-native species-related legislation, policy, and management strategies. In the context of Vietnam, the AS-ISK application and calibration presented here are intended to act as a guide for identifying which aquatic species are likely to be invasive, and thus to have adverse impacts on native biodiversity, ecosystem function and services. This information can then contribute to the establishment of invasive non-native species ‘watch’ lists and customs restrictions, as well as prioritisation of which species to control.
In Vietnam, decision-making relating to non-native (potentially) invasive species is the purview of the Ministry of Natural Resources and Environment. However, there is overlap with the Ministry of Agriculture and Rural Development, as invasive non-native species could impact both biodiversity and agriculture, and the latter ministry has remit to control species that (potentially) impact on crops, forestry, animal husbandry and fisheries. The exploration of the AS-ISK as a potential toolkit for use in Vietnam was driven by recognition by the two ministries that a more systematic approach is needed. It was facilitated though a collaboration between invasive species practitioners in Vietnam and the European developers of the AS-ISK. Initial application was challenged by the then-absence of the toolkit in Vietnamese; however, further collaboration allowed the toolkit (version 2.1 onwards) to be translated to Vietnamese. It is hoped that this functionality, together with the setting of BRA and BRA + CCA thresholds that are specific to Vietnam, will make the AS-ISK accessible to Vietnamese practitioners, researchers, and decision-makers. It is feasible that application of risk-screening toolkits such as the AS-ISK to other developing countries, particularly in Southeast Asia, could follow a similar process.
Change history
10 April 2021
A Correction to this paper has been published: https://doi.org/10.1007/s10530-021-02498-4
References
Anderson MJ, Gorley RN, Clarke KR (2008) PERMANOVA for PRIMER: guide to software and statistical methods. PRIMER-E, Plymouth
Armbruster JW (2004) Phylogenetic relationships of the suckermouth armoured catfishes (Loricariidae) with emphasis on the Hypostominae and the Ancistrinae. Zool J Linn Soc 141:1–80. https://doi.org/10.1111/j.1096-3642.2004.00109.x
Barlow J, França F, Gardner TA, Hicks CC, Lennox GD, Berenguer E, Castello L, Economo EP, Ferreira J, Guénard B, Gontijo Leal C, Isaac V, Lees AC, Parr CL, Wilson SK, Young PJ, Graham NAJ (2018) The future of hyperdiverse tropical ecosystems. Nature 559:517–526. https://doi.org/10.1038/s41586-018-0301-1
Bewick V, Cheek L, Ball J (2004) Statistics review 13: receiver operating characteristic curves. Crit Care 8:508–512. https://doi.org/10.1186/cc3000
Bilge G, Filiz H, Yapıcı S, Tarkan AS, Vilizzi L (2019) A risk screening study on the potential invasiveness of Lessepsian fishes in the south-western coasts of Anatolia. Acta Ichthyol Piscatry 49:23–31. https://doi.org/10.3750/AIEP/02422
Branquart E (2009) Guidelines for environmental impact assessment and list classification of non-native organisms in Belgium. Version 2.6. http://ias.biodiversity.be/documents/ISEIA_protocol.pdf. Accessed 19 May 2020
Buckley YM, Catford J (2015) Does the biogeographic origin of species matter? Ecological effects of native and non-native species and the use of origin to guide management. J Ecol 104:4–17. https://doi.org/10.1111/1365-2745.12501
Cacot P, Lazard J (2009) La domestication des poissons du Mékong: les enjeux et le potentiel aquacole. Cah Agric 18:125–135. https://doi.org/10.1684/agr.2009.0304
Carlsson NOL, Brönmark C, Hansson L-A (2004) Invading herbivory: the golden apple snail alters ecosystem functioning in Asian wetlands. Ecology 85:1575–1580. https://doi.org/10.1890/03-3146
Clarke SA, Vilizzi L, Lee L, Wood LE, Cowie WJ, Burt JA, Mamiit RJ, Hassina A, Davison PI, Fenwick GV, Harmer R, Skóra ME, Kozic S, Aislabie LR, Kennerley A, Le Quesne WJF, Copp GH, Stebbing PD (2020) Identifying potentially invasive non-native marine and brackish water species for the Arabian Gulf and Sea of Oman. Glob Change Biol 26:2081–2092. https://doi.org/10.1111/GCB.14964
Copp GH, Vilizzi L, Mumford J, Fenwick GV, Godard MJ, Gozlan RE (2009) Calibration of FISK, an invasiveness screening tool for non-native freshwater fishes. Risk Anal 29:457–467. https://doi.org/10.1111/j.1539-6924.2008.01159.x
Copp GH, Vilizzi L, Tidbury H, Stebbing PD, Tarkan AS, Moissec L, Goulletquer Ph (2016a) Development of a generic decision-support tool for identifying potentially invasive aquatic taxa: AS-ISK. Manag Biol Invasions 7:343–350. https://doi.org/10.3391/mbi.2016.7.4.04
Copp GH, Godard MJ, Russell IC, Peeler EJ, Gherardi F, Tricarico E, Miossec L, Goulletquer P, Almeida D, Britton JR, Vilizzi L, Mumford J, Williams C, Reading A, Rees EMA, Merino-Aguirre R (2016b) A preliminary evaluation of the European Non-native Species in Aquaculture Risk Assessment Scheme applied to species listed on Annex IV of the EU Alien Species Regulation. Fish Manag Ecol 23:12–20. https://doi.org/10.1111/fme.12076
Copp GH, Vilizzi L, Wei H, Li S, Piria M, Al-Faisal AJ, Almeida D, Atique U, Al-Wazzan Z, Bakiu R, Bašić T, Bui TD, Canning-Clode J, Castro N, Chaichana R, Çoker T, Dashinov D, Ekmekçi FG, Erős T, Ferincz Á, Ferreira T, Giannetto D, Gilles AS, Głowacki Ł, Goulletquer Ph, Interesova E, Iqbal S, Jakubčinová K, Kanongdate K, Kim J-E, Kopecký O, Kostov V, Koutsikos N, Kozic S, Kristan P, Kurita Y, Lee H-G, Leuven RSEW, Lipinskaya T, Lukas J, Marchini A, González Martínez AI, Masson L, Memedemin D, Moghaddas SD, Monteiro J, Mumladze L, Naddafi R, Năvodaru I, Olsson KH, Onikura N, Paganelli D, Pavia RT, Perdikaris C, Pickholtz R, Pietraszewski D, Povž M, Preda C, Ristovska M, Rosíková K, Santos JM, Semenchenko V, Senanan W, Simonović P, Smeti E, Števove B, Švolíková K, Ta KAT, Tarkan AS, Top N, Tricarico E, Uzunova E, Vardakas L, Verreycken H, Zięba G, Mendoza R (2021) Speaking their language – Development of amultilingual decision-support tool for communicating invasive species risks to decision makers and stakeholders. Environ Model Softw 135:104900. https://doi.org/10.1016/j.envsoft.2020.104900
Dao HT, Beattie GAC, Watson GW, Pham VL, Nguyen VL, Le DK, Nguyen TH, Nguyen DV, Holford P (2018) Citrus diaspidids in Viet Nam: new, and confirmation of previous, records based on morphological and molecular identification of taxa. J Asia-Pac Entomol 21:81–96. https://doi.org/10.1016/j.aspen.2017.09.010
Di Marco M, Chapman S, Althor G, Kearney S, Besancon C, Butt N, Maina JM, Possingham HP, Rogalla von Bieberstein K, Venter O, Watson JEM (2017) Changing trends and persisting biases in three decades of conservation science. Glob Ecol Conserv 10:32–42. https://doi.org/10.1016/j.gecco.2017.01.008
Dodd JA, Vilizzi L, Bean CW, Davison PI, Copp GH (2019) At what spatial scale should risk screenings of translocated freshwater fishes be undertaken—river basin district or climo-geographic designation? Biol Conserv 230:122–130. https://doi.org/10.1016/j.biocon.2018.12.002
Drolet D, DiBacco C, Locke A, McKenzie CH, McKindsey CW, Moore AM, Webb JL, Therriault TW (2016) Evaluation of a new screening-level risk assessment tool applied to non-indigenous marine invertebrates in Canadian coastal waters. Biol Invasions 18:279–294. https://doi.org/10.1007/s10530-015-1008-y
Drury R (2009) Reducing urban demand for wild animals in Vietnam: examining the potential of wildlife farming as a conservation tool. Conserv Lett 2:263–270. https://doi.org/10.1111/j.1755-263X.2009.00078.x
Early R, Bradley BA, Dukes JS, Lawler JJ, Olden JD, Blumenthal DM, Gonzalez P, Grosholz ED, Ibañez I, Miller LP, Sorte CJB, Tatem AJ (2016) Global threats from invasive alien species in the twenty-first century and national response capacities. Nat Commun 7:12485. https://doi.org/10.1038/ncomms12485
Eaton JA, Nguyen MDT, Willemsen M, Lee J, Chng SCL (2017) Caged in the city: an inventory of birds for sale in Ha Noi and Ho Chi Minh City, Viet Nam. TRAFFIC, Southeast Asia Regional Office, Petaling Jaya, Selangor, Malaysia. www.traffic.org/site/assets/files/1580/caged-in-the-city.pdf. Accessed 16 May 2020
Ellender BR, Weyl OL (2014) A review of current knowledge, risk and ecological impacts associated with non-native freshwater fish introductions in South Africa. Aquat Invasions 9:117–132. https://doi.org/10.3391/ai.2014.9.2.01
Fausch KD, Power ME, Murakami M (2002) Linkages between stream and forest food webs: Shigeru Nakano’s legacy for ecology in Japan. Trends Ecol Evol 17:429–434. https://doi.org/10.1016/S0169-5347(02)02572-7
Glamuzina B, Tutman P, Nikolić V, Vidović Z, Pavličević J, Vilizzi L, Copp GH, Simonović P (2017) Comparison of taxon-specific and taxon-generic risk screening tools to identify potentially invasive non-native fishes in the River Neretva catchment (Bosnia and Herzegovina and Croatia). River Res Appl 33:670–679. https://doi.org/10.1002/rra.3124
Haubrock PJ, Copp GH, Johovic I, Balzani P, Inghilesi AF, Nocita A, Tricarico E (2021) North American channel catfish, Ictalurus punctatus: a neglected but potentially invasive freshwater fish species. Biol Invas. https://doi.org/10.1007/s10530-021-02459-x
Hoover JJ, Killgore KJ, Cofranceso AF (2004) Suckermouth catfishes: threats to aquatic ecosystems of the United States? Aquat Nuisance Species Res Program Bull 4:1–13
Hosmer DW Jr, Lemeshow S, Sturdivant RX (2013) Applied logistic regression, 3rd edn. Wiley, NJ, USA
Interesova E, Vilizzi L, Copp GH (2020) Risk screening of the potential invasiveness of non-native freshwater fishes in the River Ob basin (West Siberian Plain, Russia). Reg Environ Change. https://doi.org/10.1007/s10113-020-01644-3
IPBES (2019) Global assessment report on biodiversity and ecosystem services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. In: Brondizio ES, Settele J, Díaz S, Ngo HT (eds) IPBES Secretariat, Bonn, Germany
IPCC (2005) Guidance notes for lead authors of the IPCC fourth assessment report on addressing uncertainties. Intergovernmental Panel on Climate Change, WMO & UNEP. https://www.ipcc.ch/site/assets/uploads/2018/02/ar4-uncertaintyguidancenote-1.pdf. Accessed 16 May 2020
Joshi RC, Cowie RH, Sebastian LS (eds) (2017) Biology and management of invasive apple snails. Philippine Rice Research Institute (PhilRice), Maligaya, Science City of Muñoz, Nueva Ecija
Killi N, Tarkan AS, Kozic S, Copp GH, Davison PI, Vilizzi L (2020) Risk screening of the potential invasiveness of non native jellyfishes in the Mediterranean Sea. Mar Poll Bull 150:110728. https://doi.org/10.1016/j.marpolbul.2019.110728
Law on Biodiversity (2008) https://portal.gms-eoc.org/uploads/resources/953/attachment/Vietname_Law_on_Boidiversity_28_Nov_2008_En.pdf. Accessed 16 May 2020
Law on Environmental Protection (2014) https://www.elaw.org/vietnam-environmental-impact-assessment-eia-laws. Accessed 16 May 2020
Lawson LL Jr, Hill JE, Vilizzi L, Hardin S, Copp GH (2013) Revisions of the Fish Invasiveness Screening Kit (FISK) for its application in warmer climatic zones, with particular reference to peninsular Florida. Risk Anal 33:1414–1431. https://doi.org/10.1111/j.1539-6924.2012.01896.x
Le TB, Truong QB (2016) Quick assessment of the invasiveness of non-native plant species by using eco-physiological parameters in Tram Chim National Park, Vietnam. Weed Biol Manag 16:177–185. https://doi.org/10.1111/wbm.12108
Li S, Chen J, Wang X, Copp GH (2017) Invasiveness screening of non-native fishes for the middle reach of the Yarlung Zangbo River, Tibetan Plateau, China. River Res Appl 33:1439–1444. https://doi.org/10.1002/rra.3196
Lu D (2019) Peirce’s philosophy of communication and language communication. Semiotica 230:407–423. https://doi.org/10.1515/sem-2017-0164
Matsuzaki SS, Kadoya T (2015) Trends and stability of inland fishery resources in Japanese lakes: introduction of exotic piscivores as a driver. Ecol Appl 25:1420–1432. https://doi.org/10.1890/13-2182.1
McElwee P (2010) The social dimensions of adaptation to climate change in Vietnam. Discussion Paper Number 17, The World Bank. http://documents.worldbank.org/curated/en/955101468326176513/pdf/589030NWP0EACC10Box353823B01public1.pdf. Accessed 16 May 2020
McGeoch MA, Spear D, Kleynhans EJ, Marais E (2012) Uncertainty in invasive alien species listing. Ecol Appl 22:959–971. https://doi.org/10.1890/11-1252.1
Ministry of Natural Resources and Environment (2009) Climate change, sea level rise scenarios for Vietnam. Socialist Republic of Viet Nam, Hanoi, Viet Nam. www.preventionweb.net/files/11348_ClimateChangeSeaLevelScenariosforVi.pdf. Accessed 16 May 2020
Ministry of Natural Resources and Environment (2010) Viet Nam’s second national communication to the United Nations Framework Convention on Climate Change. Socialist Republic of Viet Nam, Hanoi, Viet Nam. https://unfccc.int/resource/docs/natc/vnmnc02.pdf. Accessed 16 May 2020
Nghiem LTP, Soliman Y, Yeo DCJ, Tan HTW, Evans TA, Mumford JD et al (2013) Economic and environmental impacts of harmful non-indigenous species in Southeast Asia. PLoS ONE 8:71255. https://doi.org/10.1371%2Fjournal.pone.0071255
Nguyen SV (2008) Wildlife trading in Vietnam: situation, causes and solutions. J Environ Dev 17:145–165. https://doi.org/10.1177%2F1070496508316220
Onikura N, Nakajima J, Inui R, Mizutani H, Kobayakawa M, Fukuda S, Mukai T (2011) Evaluating the potential of invasion by non-native freshwater fishes in northern Kyushu Island, Japan, using the Fish Invasiveness Scoring Kit. Ichthyol Res 58:382–387. https://doi.org/10.1007/s10228-011-0235-1
Peh K (2010) Invasive species in Southeast Asia: the knowledge so far. Biodivers Conserv 19:1083–1099. https://doi.org/10.1007/s10531-009-9755-7
Pyšek P, Richardson DM, Jarošík V (2006) Who cites who in the invasion zoo: insights from an analysis of the most highly cited papers in invasion ecology. Preslia 78:437–468
Pyšek P, Richardson DM, Pergl J, Jarošík V, Sixtová Z, Weber E (2008) Geographical and taxonomic biases in invasion ecology. Trends Ecol Evol 23:237–244. https://doi.org/10.1016/j.tree.2008.02.002
R Development Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. www.R-project.org/
Rejmánek M (1996) Species richness and resistance to invasions. In: Orians GH, Dirzo R, Cushman JH (eds) Diversity and process in tropical forests. Springer, Berlin, pp 153–172. https://doi.org/10.1007/978-3-642-79755-2_8
Rejmánek M, Simberloff D (2017) Origin matters. Environ Conserv 44:97–99. https://doi.org/10.1017/S0376892916000333
Robin X, Turck N, Hainard A, Tiberti N, Lisacek F, Sanchez JC, Müller M (2011) PROC: an open-source package for R and S + to analyze and compare ROC curves. BMC Bioinform 12:e77. https://doi.org/10.1186/1471-2105-12-77
Roy HE, Rabitsch W, Scalera R, Stewart A, Gallardo B, Genovesi P, Essl F, Adriaens T, Booy O, Branquart E, Brunel S, Copp GH, Dean H, D'hondt B, Josefsson M, Kenis M, Kettunen M, Linnamagi M, Lucy F, Martinou A, Moore N, Nieto A, Pergl J, Peyton J, Schindler S, Solarz W, Stebbing PD, Trichkova T, Vanderhoeven S, Van Valkenburg J, Zenetos A (2018) Developing a framework of minimum standards for the risk assessment of alien species. J Appl Ecol 55:526–538. https://doi.org/10.1111/1365-2664.13025
Roy HE, Bacher S, Essl F, Adriaens T, Aldridge DC, Bishop JD, Blackburn TM, Branquart E, Brodie J, Carboneras C, Cottier-Cook EJ, Copp GH, Dean HJ, Eilenberg J, Gallardo B, Garcia M, García-Berthou E, Genovesi P, Hulme PE, Kenis M, Kerckhof F, Kettunen M, Minchin D, Nentwig W, Nieto A, Pergl J, Pescott OL, Peyton J, Preda C, Rabitsch W, Roques A, Rorke SL, Scalera R, Schindler S, Schönrogge K, Sewell J, Solarz W, Stewart AJA, Tricarico E, Vanderhoeven S, Van der Velde G, Vilà M, Wood CA, Zenetos A (2019) Developing a list of invasive alien species likely to threaten biodiversity and ecosystems in the European Union. Glob Change Biol 25:1032–1048. https://doi.org/10.1111/gcb.14527)
Semenchenko V, Lipinskaya T, Vilizzi L (2018) Risk screening of non-native macroinvertebrates in the major rivers and associated basins of Belarus using the Aquatic Species Invasiveness Screening Kit. Manag Biol Invasions 9:127–136. https://doi.org/10.3391/mbi.2018.9.2.06
Shan Lv, Guo Y-H, Nguyen HM, Sinuon M, Sayasone S, Lo NC, Zhou X-N, Andrews JR (2018) Invasive Pomacea snails as important intermediate hosts of Angiostrongylus cantonensis in Laos, Cambodia and Vietnam: implications for outbreaks of eosinophilic meningitis. Acta Trop 183:32–35. https://doi.org/10.1016/j.actatropica.2018.03.021
Sodhi NJ, Pin Koh L, Brook BW, Ng PKL (2004) Southeast Asian biodiversity: an impending disaster. Trends Ecol Evol 19:654–660. https://doi.org/10.1016/j.tree.2004.09.006
Sodhi NJ, Posa MRC, Lee TM, Bickford D, Koh LP, Brook BW (2010) The state and conservation of Southeast Asian biodiversity. Biodiv Conserv 19:317–328. https://doi.org/10.1007/s10531-009-9607-5
Tan DT, Thu PQ, Dell B (2012) Invasive plant species in the national parks of Vietnam. Forests 3:997–1016. https://doi.org/10.3390/f3040997
Tarkan AS, Sari HM, İlhan A, Kurtul I, Vilizzi L (2017a) Risk screening of non-native and translocated freshwater fish species in a Mediterranean-type shallow lake: Lake Marmara (West Anatolia). Zool Middle East 63:48–57. https://doi.org/10.1080/09397140.2017.1269398
Tarkan AS, Vilizzi L, Nildeniz T, Ekmekçi FG, Stebbing PD, Copp GH (2017b) Identification of potentially invasive freshwater fishes, including translocated species, in Turkey using the Aquatic Species Invasiveness Screening Kit (AS-ISK). Int Rev Hydrobiol 102:47–56. https://doi.org/10.1002/iroh.201601877
Triet T, Thi LTL, Nga NP (2008) Survey and assessment of the impacts of alien plant species in some national parks of Vietnam. University of Natural Science, National University of Ho Chi Minh City (in Vietnamese), Ho Chi Minh City
Uyan U, Oh C-W, Tarkan AS, Top N, Copp GH, Vilizzi L (2020) Risk screening of the potential invasiveness of non-native marine fishes in South Korea. Mar Pollut Bull 153:111018. https://doi.org/10.1016/j.marpolbul.2020.111018
Venkataraman B (2007) A matter of attitude: The consumption of wild animal products in Ha Noi, Viet Nam. TRAFFIC Southeast Asia, Greater Mekong Programme, Ha Noi, Viet Nam. https://www.traffic.org/site/assets/files/3374/matter-of-attitude-viet-nam.pdf. Accessed 16 May 2020
Vilizzi L, Copp GH, Adamovich B, Almeida D, Chan J, Davison PI, Dembski S, Ekmekçi FG, Ferincz Á, Forneck SC, Hill JE, Kim J-E, Koutsikos N, Leuven RSEW, Luna SA, Magalhães F, Marr SM, Mendoza R, Mourão CF, Neal JW (2019) A global review and meta-analysis of applications of the freshwater Fish Invasiveness Screening Kit. Rev Fish Biol Fish 29:529–568. https://doi.org/10.1007/s11160-019-09562-2
Wan F, Jiang, M, Zhan A (eds) (2017) Biological invasions and its management in China. Invading nature—Springer series in invasion ecology, vol 13. Springer, Dordrecht. https://doi.org/10.1007/978-94-024-0948-2
Welcomme RL, Vidthayanom C (2003) The impacts of introductions and stocking of exotic species in the Mekong Basin and policies for their control. MRC Technical Paper No. 9. Mekong River Commission, Phnom Penh, Cambodia. http://www.mrcmekong.org/assets/Publications/technical/tech-No9-the-impacts-of-intro-n-stocking.pdf. Accessed 16 May 2020
Weyl OLF, Daga VS, Ellender BR, Vitule JRS (2016) A review of Clarias gariepinus invasions in Brazil and South Africa. J Fish Biol 89:386–402. https://doi.org/10.1111/jfb.12958
Whitehead PG, Jin L, Bussi G, Voepel HE, Darby SE, Vasilopoulos G, Manley R, Rodda H, Hutton C, Hackney C, Tri VPD (2019) Water quality modelling of the Mekong River basin: climate change and socioeconomics drive flow and nutrient flux changes to the Mekong Delta. Sci Tot Environ 673:218–229. https://doi.org/10.1016/j.scitotenv.2019.03.315
Wong PK, Kwong KL, Qiu J-W (2009) Complex interactions among fish, snails and macrophytes: implications for biological control of an invasive snail. Biol Invasions 11:2223–2232. https://doi.org/10.1007/s10530-008-9378-z
World Bank Group (2019) World development indicators—country profile, Vietnam. https://databank.worldbank.org/views/reports/reportwidget.aspx?Report_Name=CountryProfile&Id=b450fd57&tbar=y&dd=y&inf=n&zm=n&country=VNM. Accessed 16 May 2020
Wyckhuys KAG, Hughes AC, Buamas C, Johnson AC, Vasseur L, Reymondin L, Deguine J-P, Sheil D (2019) Biological control of an agricultural pest protects tropical forests. Commun Biol 2:10. https://doi.org/10.1038/s42003-018-0257-6
Zworykin D, Budaev SV (2013) Non-indigenous armoured catfish in Vietnam: invasion and systematics. Ichthyol Res 60:327–333. https://doi.org/10.1007/s10228-013-0356-9
Acknowledgements
The participation of L. Ruykys in this study was facilitated through an assignment with the Australian Volunteers Program, which is an initiative of the Australian Government. The participation of G.H. Copp was supported by Cefas’ Science Excellence Fund.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised due to the pdf version of the article has been published with errors in punctuation, diacritics and symbols throughout the entire article and they have been corrected in the pdf version.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ruykys, L., Ta, K.A.T., Bui, T.D. et al. Risk screening of the potential invasiveness of non-native aquatic species in Vietnam. Biol Invasions 23, 2047–2060 (2021). https://doi.org/10.1007/s10530-020-02430-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02430-2