Abstract
Given the prevalence of invasive species and high rates of habitat homogenisation across the globe, understanding how these drivers interact to influence native species assemblages is crucial. In river networks, confluences create discontinuities in physical conditions, likely creating hotspots of heterogeneity that influence interactions between native and invasive fish. We examined how spatial configuration of confluences affected the outcome of interactions between native galaxiids and non-native salmonids in New Zealand alpine rivers. Electrofishing in mainstem and tributary branches of twelve replicate confluences revealed highly context-dependent distributions, contingent upon interactions between: (a) the combination of flood disturbance history in confluence branches; (b) distance to the confluence; and (c) the direction of flow, either upstream or downstream, of the confluence. Shifts in native–invasive species relative abundance were determined by the preference of large predatory salmonids for more hydrologically stable conditions, which subsequently limited the abundance of young-of-year galaxiids, and meant galaxiids were more abundant in flood-prone conditions. Distance-from-confluence effects were stronger upstream than downstream, suggesting that flow direction had an important influence on dispersal. Tributary flow regimes also predictably influenced downstream physical conditions, thereby affecting predatory salmonid distribution which likely controlled galaxiid distributions. Overall, our results reveal strong spatial context-dependency in fish assemblages in river networks, and demonstrate how flow regime influences are spatially transferred at confluences, thereby creating areas of influential riverscape heterogeneity. Understanding the influence of such heterogeneity enables ecologically significant locations to be identified, particularly for management of native species vulnerable to invaders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Invasive species and landscape-scale habitat homogenisation are arguably the current greatest threats to freshwater biodiversity around the globe (Dudgeon et al. 2006; Moorhouse and Macdonald 2014; Zeni and Casatti 2014), it is therefore pertinent to understand how environmental heterogeneity affects native-invasive species interactions in freshwater systems. Habitat heterogeneity can sometimes promote coexistence between strongly interacting native and invasive species, whereas strong native-invasive interspecific interactions in homogenous environments can often lead to extirpation of native species (Melbourne et al. 2007). Understanding how native-invasive species interactions differ depending on the spatial configuration heterogeneity will progress our understanding of invasion in freshwater ecosystems.
Conceptualizing river systems as spatially continuous habitat mosaics will be important for addressing the influence of heterogeneity on river communities (Fausch et al. 2002). Discontinuities, potentially creating hotspots of heterogeneity in river networks, are most common at tributary confluences (Kiffney et al. 2006). By joining streams with potentially different size, flow regimes, and water quality, confluences can be important sources of environmental heterogeneity in river networks (Benda et al. 2004; Kiffney et al. 2006; Rice et al. 2001). Furthermore, edge effects associated with confluence heterogeneity could be critical interfaces where biotic and abiotic processes interact to produce strong gradients in species richness and community composition (Czeglédi et al. 2015; Fernandes et al. 2004). The complexity of confluence geomorphology means determining relationships between tributary properties and aquatic communities is a major challenge (Rice 2017). A primary form of variability in river systems is the frequency and severity of flow-driven physical disturbance (Lake 2000), which structures the physical environment, including habitat heterogeneity. We focus on flood disturbances, defined as physical influences associated with high flows that alter the physical characteristics of a river channel. This is reflected in things like sediment movement, bed scouring, removal of periphyton, and can be measured by the River Disturbance Index (RDI) developed by Pfankuch (Jellyman et al. 2013; McHugh et al. 2010; Pfankuch 1975). Importantly, fish communities may respond most strongly to flow-related bed movement, compared to flow variability per se (Jellyman et al. 2013), so assessing disturbance based on physical characteristics of riverbeds is appropriate.
Recent spatial modelling indicates that landscape heterogeneity could increase coexistence between native and invading species with differing spatial niches (Maciel and Lutscher 2018). Thus, if native and invasive species vary in flow-related adaptations, spatial heterogeneity in flow disturbance could control coexistence. This also fits with the ‘environmental heterogeneity of invasions hypothesis’. This suggests that while environmental heterogeneity might enhance invader spread, it can also reduce impacts of invaders on native species through various coexistence mechanisms not possible in homogeneous environments (Melbourne et al. 2007). The juxtaposition of contrasting flow disturbance regimes between tributary, and upstream and downstream mainstem river branches at confluences could create situations where these mechanisms become important. Mobile taxa like fish can access habitats with contrasting physical characteristics (Rice 2017; Schlosser 1991), enabling individuals to move between habitat patches to complement or supplement resources (Fausch et al. 2002). However, fish also have strong habitat preferences often associated with flow-related disturbance regimes (Mims and Olden 2012; Olden and Kennard 2010). This likely means predation pressure from invasive fish will be patchy across a heterogeneous invaded riverscape. Native fish may find refuge from predation in patch types not favoured by invaders, and confluences could propagate invader influences to upstream and downstream branches depending on species mobility and the influence of directional flow. It may also be that asynchrony in the dynamics of communities in heterogeneous local branches is an additional important stabilizing mechanism (Wilcox et al. 2017). There is evidence that these types of dynamics are happening in riverine fish communities. For example, patterns of fish species composition are well known to differ between tributary and mainstem branches driven by habitat preferences, context-dependent species interactions and patterns of branch connectivity (Hitt and Angermeier 2008; Schlosser 1991; Thornbrugh and Gido 2010). This can sometimes cause source–sink dynamics across tributary and mainstem branches (Woodford and McIntosh 2010, 2011). Overall, if native–invasive species interactions are moderated by flow disturbance, spatial patterns in the relative abundance of native and non-native species in invaded confluence habitats could be determined by the configuration of disturbance, and the direction and distance from a confluence. Thus, configuration of flow disturbance around confluences could explain much context-dependence in impacts of influential invasive predators, and the heterogeneity of confluences may be important for their coexistence.
Such influences of confluence-related heterogeneity are likely to be particularly important in interactions involving non-native Salmonidae. Salmonids are one of the most widespread invasive groups in the world due to a long history of introductions to establish recreational fisheries, and their consequent effects on native fish have become a major concern (Fausch 1988; Hasegawa et al. 2016; Lowe et al. 2000; Morita 2018). For example, widely established populations of invasive trout have had severe effects on threatened native galaxiid species in the Southern Hemisphere (Habit et al. 2010; Jones and Closs 2015; McDowall 2006; McIntosh et al. 2010; Sowersby et al. 2015). Importantly, the strong interactions of non-native salmonids play out across highly spatially heterogeneous river networks, and salmonids have both strong habitat preferences and are strongly influenced by physical habitat conditions like flooding (Budy et al. 2008; Fausch et al. 2001; Jellyman et al. 2013; Jellyman and McIntosh 2020). Large trout (e.g. > 150 mm fork length), in particular, exert strong predation pressure on non-diadromous galaxiids such as Galaxias vulgaris and G. paucispondylus in New Zealand (McIntosh et al. 2010). These larger trout occur in high densities in streams with stable flow, but are often less numerous in streams disturbed by flooding (Woodford and McIntosh 2010). In comparison, non-diadromous galaxiids are often restricted to flood-prone streams when trout are present, but can reach high densities in more stable habitats when trout are absent (McIntosh 2000; Woodford and McIntosh 2010). Thus, strong trout predation in stable streams likely leads to trout-dominated assemblages, whereas native galaxiids probably persist in disturbed streams because of weaker interactions with trout in these habitats (McIntosh et al. 2010). Importantly, these interactions are size-dependent, with smaller galaxiid size classes more vulnerable, and larger trout exerting stronger predation pressure (McIntosh et al. 1994; McIntosh 2000). Like trout, these non-migratory galaxiids are capable of large riverine movements, so could easily move between river branches at confluences (Woodford and McIntosh 2011), although they typically maintain relatively small day-to-day home ranges (Cadwallader 1976). We therefore expected confluences with different configurations of flow disturbance to provide insights into the context-dependency driving patterns in native–invasive species co-occurrence in heterogeneous river networks.
To test the hypothesis that flood disturbance structures fish assemblages and allows for native and non-native species coexistence by increasing habitat heterogeneity around river confluences, we tested a series of predictions concerning how flow disturbance affected native–invasive species interactions in river confluence habitats. We first established patterns of fish assemblage structure associated with ‘stable’ and ‘disturbed’ streams, and predicted that non-native salmonids would dominate stable streams and native galaxiids would form greater proportions of assemblages in flood-disturbed streams (H1). Secondly, we expected that the relative abundance of galaxiids would vary with confluence branch (upstream mainstem, tributary or downstream mainstem), confluence configuration (spatial arrangement of stable and/or disturbed flow conditions around the confluence) and distance from the confluence (H2). Here, we expected stronger distance to confluence patterns around confluences between stable and disturbed streams, than at confluences between two similar streams. We also predicted that distance to confluence effects would be stronger upstream (compared to downstream) due to flow directionality with greater co-occurrence near the confluence in general, and co-occurrence persisting further from the confluence downstream than upstream (H3). Finally, we predicted that large predatory trout, by reducing the abundance of small galaxiids, would drive the relative abundance of trout and galaxiids (H4).
Methods
Field survey
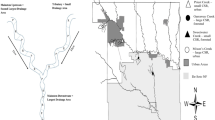
We surveyed twelve river confluences in the headwaters of the Waimakariri and Rakaia River catchments in the Canterbury high country, South Island, New Zealand (Fig. 1). This area, described in detail by Cowie et al. (1986), contains a large diversity of river types due to the presence of both large braided river floodplains creating stable, spring-fed, systems juxtaposed with highly flood-disturbed braided river channels, as well as springs associated with limestone outcrops in otherwise steep eroded catchments. All study rivers flowed through either mountain beech forests or tussock grasslands in reserves or areas subject to low-intensity grazing, so had high water quality and relatively unmodified flow regimes. We selected replicate confluences to be relatively similar within confluence configuration categories (explained below). Thus all confluences were on independent branches (i.e., not downstream of each other) on two main river catchments, had permanent flow, and were selected so that branches matched the disturbance characteristics we wanted.
Location of the twelve confluences surveyed on the eastern side of the Southern Alps, South Island, New Zealand. Coloured lines in Detail (a) represent confluence configurations which are based on the combination of stable and disturbed flow regimes. The four confluence configurations are coloured as follows: ‘disturbed-disturbed’ (red), ‘disturbed-stable’ (blue), ‘stable-disturbed’ (green) and ‘stable-stable’ (purple). For more detail of how these configurations are structured see Fig. 2
Four confluence configuration categories were selected to include different combinations of flow-driven physical disturbance. The four confluence configurations included: (a) confluences between two ‘stable’ streams, (b) confluences between a ‘stable’ upstream mainstem and a ‘disturbed’ tributary, (c) confluences with a ‘disturbed’ upstream mainstem and a ‘stable’ tributary, and finally (d) confluences between two streams with ‘disturbed’ flow regimes (Fig. 2). Confluences were therefore labelled according to their configuration of flow-driven physical disturbance (Fig. 2). Where possible, we sampled confluences between third order mainstem streams and second order tributary streams to control for the effects of stream size.
Field sampling design showing the four confluence configurations (upper panel, a–d) based on the combination of stable and disturbed flow regimes (solid and dashed lines, respectively) within the three ‘branches,’ and the arrangement of sampling reaches within a branch (lower panel). ‘Stable’ streams had low flood disturbance, and ‘disturbed’ streams had high flood disturbance. Confluence configurations were a ‘stable-stable’ (both mainstem and tributary classed as stable), b ‘stable-disturbed’ (stable mainstem and disturbed tributary), c ‘disturbed-stable’ (disturbed mainstem and stable tributary), d ‘disturbed-disturbed’ (both mainstem and tributary classed as disturbed), and each confluence involved three ‘branches:’ the tributary, upstream mainstem, downstream mainstem. Within each branch, five reaches were sampled, making a total of 15 sampling reaches per confluence replicate
To quantify flow-disturbance influences in confluence branches we used the RDI which combines visual estimates of 15 aspects of channel morphology including indicators from both the stream bed and banks to evaluate physical stability of a river channel (Pfankuch 1975; McHugh et al. 2010; Jellyman et al. 2013). While classic hydrological variables such as flood magnitude, frequency and duration are effective measures of flow variability, the RDI reflects the disturbance experienced by local stream organisms by including factors such as reach geomorphology and recent bed movement (Peckarsky et al. 2014). RDI scores were used to classify upstream mainstem and tributary branches into ‘stable’ (RDI score < 100, low flow disturbance e.g. spring-fed streams) or ‘disturbed’ (RDI score > 100, high flow disturbance e.g. braided rivers). We used RDI scores above or below 100 to broadly categorise stream habitat types (Boddy et al. 2019). We labelled confluences according to their upstream mainstem and tributary RDI scores, for example a confluence with a stable mainstem and a disturbed tributary was categorised as ‘stable-disturbed.’
Additional abiotic habitat variables were measured at every sampling reach, including: water temperature (°C, Onset HOBO pendant loggers installed in every branch of each confluence taking hourly measurements for the duration of the 2014/15 austral summer, supplemented with four spot temperature measurements per reach), substrate size (mm), water depth (cm) and velocity (three cross-sections per reach, six measurements per cross-section), wetted stream width (m), macrophyte cover (%), basic water chemistry (pH, dissolved oxygen and conductivity), and reach flow characteristics; percent of reach consisting of cascades, riffles, runs, and pools (Leathwick et al. 2008; Boddy and McIntosh 2017). Macrophyte cover and percent of reach with different flow characteristics were visually estimated and mean substrate sizes were obtained using a Wolman Walk to select 50 random substrate particles for measurement of the beta axis (Green 2003). A detailed analysis of the physical characteristics of confluences, including elevation, % riffle and run, substrate size, stream width, RDI and temperatures, and using a principal components analysis followed by permutational multivariate analysis of variance (PERMANOVA), indicated that although there was variability between replicates of a category, replicates of confluence categories grouped as expected (Boddy et al. 2019).
Each sampling reach was single-pass electrofished in an upstream direction with push-nets, but without stopnets that spanned the river width at each end of the reach. Results of this approach correlate well with more intensive quantitative techniques in these streams, both for Galaxiidae (R2 = 0.99) and Salmonidae (R2 = 0.82) abundance (Boddy et al. 2019). Fish caught were identified to species and size classes recorded based on fork length. Galaxiidae were sorted into three size classes: young of year (< 60 mm), 1+ (60–90 mm), and 2+ (> 90 mm; Woodford and McIntosh 2013). Salmonidae were also sorted into three size classes: young of year (< 50 mm; Baltz and Moyle 1984), medium (50–150 mm) and large (> 150 mm). The 150 mm size threshold was chosen to distinguish large brown and rainbow trout capable of consuming all sizes of galaxiids from medium size trout that are not (McIntosh 2000).
Each confluence survey involved sampling three ‘branches’ at the end of the austral summer in March 2015 (Fig. 2). This sampling time coincided with major growth phases of both galaxiids and trout and was well clear of both trout (winter) and galaxiid (spring) spawning times (McDowall 1990). To ensure we sampled at an appropriate scale to resolve the spatial patterns of mobile species, we sampled five ‘reaches’ in each branch, making a total of 15 sampling reaches per confluence (Fig. 2). The length of each sampled reach was five times the stream width, and the distance between reaches was five times the average reach length for that branch. The reach sampled nearest the confluence began one reach length from the confluence, so the distance sampled for each branch was 130 times the stream width from the confluence. Sampling reach length was a constant multiple of the stream width, averaged over all five reaches in a branch, to avoid incorporating variability due to sampling different proportions of the available habitat depending on stream size (Peterson and Ver Hoef 2010). Moreover, five stream widths was chosen to represent the habitat complexity in the smaller streams, and not so large that it was unachievable to sample reaches in larger rivers (> 10 m wide) in a day.
Statistical analysis
To evaluate fish-assemblage change in response to different confluence configurations, we analysed the proportion of the fish assemblage that was composed of native species. Reaches with no fish present were removed from analyses (15 reaches out of 180). Importantly, more than 98% of fish sampled were either galaxiids or salmonids, so in practice the ‘proportion native’ variable represented the relative abundance of galaxiids in reaches, and a low proportion of galaxiids meant a reach contained a high relative abundance of trout.
Generalised linear mixed-effects models with binomial error distributions were created using the lme4 package in R (Bates et al. 2015). These were used to test for a three-way interaction between the following three fixed effects which had been identified by Boddy et al. (2019) as the principal drivers of fish population patterns: confluence configuration category (i.e., stable-stable, disturbed-disturbed, stable-disturbed or disturbed-stable), branch (i.e., tributary, upstream or downstream mainstem), and distance to confluence affecting the proportion of the fish assemblage that was native. All models included confluence identity as a random effect (i.e., which of the twelve confluences the reach in question was from). There were two exceptions to this fixed effect structure: models testing the fish size class response had fish size, branch disturbance and distance to confluence as fixed effects, and the model testing the influence of large trout on small galaxias abundance had proportion of fish abundance consisting of large trout, branch and distance to confluence as fixed effects (Table 1). To understand better the mechanisms behind this three-way interaction, we also analysed branches (upstream, tributary and downstream) separately to investigate the configuration by distance interaction in more detail.
The distance to confluence variable was tested in two forms. Firstly, linear distance in meters from the confluence to the nearest end of the reach, a method that represented travel distance between reaches. Secondly, we used distance measured in reach lengths from the confluence, a method incorporating differences in stream size which assumes fish assemblage patterns shift more gradually in larger rivers than small streams. A generalised linear mixed-effects model (glmer) was created with each option and likelihood ratio tests (LRT) used to compare between models. There was little difference in variance explained, however the distance in meters model had a higher AICc score (815.3 vs. 802.2), and issues with model convergence and overdispersion. We therefore progressed modelling using distance measured in reach lengths from the confluence.
To evaluate potential spatial autocorrelation between reaches due to the nested nature of the study design, we created a spatial autocorrelation structure using Manhattan distances along each branch to include network distances between each pair of sites for each confluence. We then compared models with and without the spatial autocorrelation parameter using model outputs and AICc scores for small sample sizes to evaluate justification for inclusion in the following analysis. The inclusion of spatial autocorrelation using Manhattan distances was not supported in any of our models, with identical model outputs, no increase in predictive performance, and elevated AICc scores. This suggested our model format sufficiently accounted for the spatial structure of our data, and/or there were sufficient gaps between reaches for spatial autocorrelation not to be an issue. It was therefore excluded from our final models (more detail in Boddy et al. 2019).
Paired sets of generalised linear mixed-effect models with and without interaction terms, using the main effects identified by Boddy et al. (2019), were created to address each of our hypotheses. These model pairs were compared using likelihood ratio tests to determine the significance of interaction terms. Marginal and conditional coefficients of determination (Nakagawa et al. 2013) were used to assess model fit and were calculated using the MuMIn package (Barton 2016). Marginal R² (R²m) was used to express absolute model fit including only the fixed effects, while conditional R² (R²c) was used to express model fit including both fixed and random effects. Because confluence identity was a random effect in our model, comparing the R²m and R²c values allowed the importance of confluence-specific effects in explaining variability in the data to be evaluated. All analyses were conducted in R 3.4.3 (R Development Core Team 2016). For a description of use of RDI to characterise ‘stable’ and ‘disturbed’ reaches and how spatial autocorrelation was dealt with refer to Boddy et al. (2019).
Results
Site and habitat characteristics
Using RDI scores to split upstream mainstem and tributary branches into ‘stable’ (RDI < 100) or ‘disturbed’ (RDI > 100), we were able to characterise the combinations of physical streambed disturbance in different confluence configurations. Mainstem RDI scores in disturbed-disturbed and stable-stable configurations changed little between upstream and downstream reaches, but the configurations with both stable and disturbed conditions upstream saw substantial changes in RDI scores downstream because of tributary influence (Boddy et al. 2019). As a result of tributary influence, mainstem reaches of stable-disturbed systems became much more disturbed (i.e. higher RDI score) downstream of the tributary confluence whereas disturbed-stable mainstems became more stable (i.e. lower RDI score) downstream (Boddy et al. 2019). A similar pattern of tributary influence was seen in the average daily temperature measures (Boddy et al. 2019). While average discharge was similar between disturbed mainstems (1.38 m3/s) and stable mainstems (1.47 m3/s), disturbed mainstems were generally much wider and shallower, reflecting a major difference in channel characteristics. This geomorphology difference is one of the important aspects of the flow-driven differences between disturbed and stable branches that likely influences fish assemblage composition. Importantly, the confluence configuration categories were not confounded by location in a catchment, indicated by no consistent patterns in upstream catchment area (Boddy et al. 2019).
Fish assemblage characteristics
In total, 4,368 fish from seven different species were caught. This included five native species: alpine galaxias (Galaxias paucispondylus), Canterbury galaxias (Galaxias vulgaris), kōaro (Galaxias brevipinnis), longfin eels (Anguilla dieffenbachia) and upland bullies (Gobiomorphus breviceps); and two non-native salmonids: brown trout (Salmo trutta) and rainbow trout (Oncorhynchus mykiss). Average fish density per unit area was the lowest in disturbed-disturbed confluence configurations (0.1–0.19 fish/m2) and highest in stable-disturbed confluence configurations (0.38–1.33 fish/m2; Boddy et al. 2019). Species richness varied among sites and reaches from one to four taxa. Across sites, the most abundant taxa were G. paucispondylus (37.1% of total catch), S. trutta (28.4%), O. mykiss (18.7%) and G. vulgaris (13.1%), and the rarest taxa were G. breviceps (1.4%), G.brevipinnis (1.3%) and A. dieffenbachia (< 0.1%). Trout and galaxiids combined made up 98.6% of total fish abundance on average in a reach, so any patterns in proportion of fish that were native reflect variation in abundance of these two groups. Average proportion of the fish assemblage that was native across all sites was 44%, ranging from 89% in stable-disturbed tributary branches to 4% in stable-stable downstream mainstem branches. Below, we focus on the drivers of these changes in relative abundance of native galaxiids in these assemblages.
Relative abundance of native and introduced fish
Stable branches had significantly lower proportions of native fish in the community than disturbed branches, irrespective of confluence configuration (χ² = 37.15, df = 1, p < 0.001, Fig. 3; Table 1, “Appendix”) in our models including all sites. Overall, non-native trout dominated the fish assemblage in stable branches throughout stream networks (low % native), and galaxiids made up the majority of the fish assemblage in disturbed branches (high % native), supporting our first hypothesis. Thus, higher densities of brown and rainbow trout were associated with more stable flow conditions, whereas in disturbed branches trout were relatively less abundant and galaxiids were relatively more abundant. Our second hypothesis, that the proportion of the fish assemblage that was native would vary as a result of a three-way interaction between branch (i.e. flow direction matters), configuration (i.e. spatial context matters) and distance (i.e. proximity to confluence matters), was also supported (χ² = 50.7, df = 11, p < 0.001, R²m = 0.3, R²c = 0.68, Table 1, “Appendix”).
For tributary branches, there was no significant interaction between confluence configuration and distance from confluence affecting the proportion of the fish assemblage that was native. Therefore, the distance to confluence effects were independent of changes in fish assemblages associated with confluence configurations. Preliminary graphing indicated branch disturbance may have more influence on proportion native than confluence configuration in tributary branches, so we compared a model with ‘configuration + distance’ to a model with ‘branch disturbance + distance’. ‘Stable’ branches had RDI scores less than 100, whereas ‘disturbed’ branches had RDI scores greater than 100. There was no significant difference in the variance explained by the two models (χ² = 2.2, df = 2, p = 0.33).; however, the branch disturbance model had a slightly higher R²c score (Table 1), so branch disturbance was a marginally better predictor. The model with branch disturbance as a factor showed that a significantly higher proportion of the fish assemblage was native in disturbed tributaries than stable tributaries (χ² = 5.7, df = 1, p = 0.017, R²m = 0.28, R²c = 0.71, Fig. 4). Distance to confluence was also a significant main effect in the tributary branch disturbance model, with higher proportions of native fish close to the confluence than further upstream (χ² = 4.93, df = 1, p = 0.026).
Partial effects plot showing difference in proportion of the fish assemblage that was native in tributaries depending on whether the branch a site was located in was stable or disturbed (χ² = 5.7, df = 1, p = 0.017, R²m = 0.28, R²c = 0.71). Circles are model estimates with 95% confidence intervals, and triangles are raw data points, coloured by confluence configuration (red, disturbed-disturbed; blue, disturbed-stable; green, stable-disturbed; and purple, stable-stable)
There was also a significant interaction between confluence configuration and distance to confluence, both in upstream (χ² = 8.56, df = 3, p = 0.036, R²m = 0.36, R²c = 0.77) and downstream (χ² = 17.53, df = 3, p < 0.001, R²m = 0.39, R²c = 0.72) mainstem branches (Fig. 5; Table 1, “Appendix”). Confluence configuration influences were important to consider in these systems because effects were unique to different tributary-upstream mainstem combinations of disturbance. For example, downstream reaches had very different proportions native compared to upstream reaches in stable-disturbed configurations, but little difference was observed in disturbed-stable configurations despite both configurations having the same pairs of branch disturbance upstream. Finally, disturbed-disturbed configurations had extremely low fish abundances, both upstream and downstream, and so the proportions of native fish were highly variable. Thus our prediction that there would be stronger distance to confluence patterns in configurations with a stable and a disturbed stream than in configurations with two similarly disturbed streams was only partially supported (Fig. 5).
Partial effects plot showing interactions between confluence configuration and distance to confluence on the proportion of the fish assemblage that was native in both upstream (a) [χ² = 8.56, df = 3, p < 0.036, R²m = 0.36, R²c = 0.77], and downstream (b) reaches [χ² = 17.53, df = 11, p < 0.001, R²m = 0.39, R²c = 0.72]. Lines represent model estimates (solid, stable; and dashed, disturbed), and shapes and colours represent confluence configuration categories (red, disturbed-disturbed; blue, disturbed-stable; green, stable-disturbed; and purple, stable-stable). Lines with significantly different slopes are coded with different letters (A-C)
In upstream mainstem branches, there was some evidence of declining proportions of native fish with increasing distance from the confluence in disturbed-disturbed, disturbed-stable and stable-stable configurations, indicated by negative slope coefficients and no statistical differences between slopes for these configurations (letters in Fig. 5a). However in stable-stable upstream mainstem branches, the proportion native increased further from the confluence (Fig. 5a). Overall, regardless of distance to confluence, mainstem reaches upstream of confluences tended to have lower proportions of native fish in stable branches than disturbed branches, as we expected, but distance from confluence effects differed depending on the confluence configuration as described above.
In mainstem branches downstream of confluences, disturbed-stable and disturbed-disturbed confluence configurations both had higher proportions of native fish near the confluence than further from it indicated by positive coefficients and slopes significantly different from the other two configurations (Fig. 5b). Both configurations also had significantly different relationships between proportions of native fish and distance to confluence compared to all other configurations, with proportion native decreasing much more strongly with distance to confluence in disturbed-disturbed than disturbed-stable configurations (Fig. 5b). Stable-disturbed and stable-stable configurations had similar slope relationships, with proportions native increasing slightly with distance from the confluence in both configurations (Fig. 5b). Thus there was not clear evidence for our third hypothesis that distance to confluence effects would be stronger upstream compared to downstream due to directional flow because distance effects very much depended on the confluence configuration (Fig. 5).
Size class analysis
To examine mechanistic drivers of changes in the proportions of fish that were native, we first tested disturbance effects in tributary branches, the simplest of the branch-related patterns, and examined the relative abundance of vulnerable galaxiid size classes. Proportional abundance of each galaxiid size class differed significantly with disturbance in tributaries (χ² = 56.3, df = 2, p < 0.001, Fig. 6a; Table 1, “Appendix”). Young-of-year galaxiids made up a significantly larger proportion of total fish abundance in disturbed tributaries, increasing the relative abundance of native fish in disturbed compared to stable tributaries (Fig. 6a). Disturbed tributaries also contained significantly higher proportions of 1+ galaxiids compared to stable tributaries, but they still made up a relatively small proportion of total fish abundance (Fig. 6a). No significant difference was detected in the relative abundance of 2+ galaxiids in stable and disturbed tributary branches. Trout sizes also differed significantly with tributary disturbance (χ² = 16.7, df = 2, p < 0.001, Fig. 6b), with disturbed tributaries having significantly lower proportions of large trout (i.e. > 150 mm), likely limiting the strong predatory influence of trout in those areas (Fig. 6b).
Partial effects plot showing proportions of total fish abundance made up by each size class of galaxiid (a) [χ² = 56.3, df = 2, p < 0.001], and trout (b) [χ² = 16.7, df = 2, p < 0.001] in tributaries depending on whether the tributary was stable or flood-disturbed. Points are model estimates with 95% confidence intervals. Shapes represent different fish size classes
The relationship between the configuration of flow disturbance and changes in the proportion of fish that were native were more complex in mainstems upstream and downstream of confluence branches due to the influence of distance from confluence. For example, higher relative abundances of native fish in upstream mainstem branches near the confluence in disturbed-disturbed, disturbed-stable and stable-disturbed configurations were driven by high abundances of YOY galaxiids, which were associated with extremely low abundances of trout > 150 mm at these locations. In disturbed-disturbed and stable-disturbed reaches the abundance of YOY galaxiids was dramatically reduced moving upstream from the confluence, coinciding with higher relative abundances of trout (Fig. 5).
To further investigate the potential effects of large trout on YOY galaxiid abundance we analysed their abundance across all configurations and branches: relative abundance of YOY galaxiids significantly decreased with increasing proportions of large trout (χ² = 100.3, df = 1, p < 0.001, R²m = 0.4, R²c = 0.7, Fig. 7; Table 1, “Appendix”). This model predicted < 0.01% of the fish present at a site would be composed of YOY galaxiids if large trout (> 150 mm) made up greater than 16% of the assemblage. These results support our fourth hypothesis, that galaxiid abundance would be driven by large predatory trout. Importantly, our results reveal that this pattern was primarily driven by the vulnerability of YOY galaxiids to large trout.
Partial effects plot showing the proportion of the fish assemblage comprised of young of year (YOY) galaxiids in relation to the proportion of the fish assemblage that was made up of trout larger than 150 mm long (χ² = 100.3, df = 1, p < 0.001, R²m = 0.4, R²c = 0.7). Points represent raw data values, the line is the model estimate and the shaded area is the 95% confidence interval
Discussion
Human activities continue to fragment and homogenise habitats, so it is important to understand the role of spatial heterogeneity in invasion processes (Melbourne et al. 2007). Despite the importance of confluences as sources of heterogeneity in river systems, we need to know more about how confluences influence co-occurrence between native and non-native species (Cathcart et al. 2018). We examined how flow disturbance around confluences influenced co-occurrence of native galaxiids and introduced trout in New Zealand rivers. Our results revealed highly context-dependent distributions, which depended on confluence configuration, distance to confluence and to some extent, direction of flow. These factors created distinct patterns in relative abundance of fish driven by the configuration of disturbance around confluences.
Many of the patterns we found in species relative abundance were probably ultimately driven by the sensitivity of trout to flood disturbance. We found flood-disturbance negatively affected both brown and rainbow trout, consistent with previous New Zealand studies (Jowett 1990; Jowett and Richardson 1989; McIntosh 2000). The distribution of native fish also differed depending on flood disturbance, probably due to the well-known strong negative interactions between trout and non-migratory galaxiids (McIntosh et al. 2010). We found low proportions of galaxiids in stable branches, whereas there were high proportions of galaxiids in disturbed branches, supporting our first prediction. Importantly, these differences in galaxiid distributions were unlikely to be caused by varying galaxiid habitat preferences because the galaxiids do occupy stable habitats when barriers exclude trout (McIntosh 2000; Woodford and McIntosh 2010).
High abundances of young-of-year (YOY) galaxiids in disturbed branches, and very little co-occurrence between YOY galaxiids and large trout in stable stream branches (where trout were more numerous), drove galaxiid distributions, supporting our fourth prediction. Strong predation pressure on non-migratory galaxiids by large trout is a common feature of trout-galaxiid interactions, and is especially evident in the rarity of galaxiid fry in trout-invaded reaches (Jellyman and McIntosh 2010; Woodford and McIntosh 2010). Therefore, predation by large trout on YOY galaxiids was likely an important driver of native fish distributions across different confluence configurations. Invasive species abundance often drives their impact on native species (Kumschick et al. 2015; Latzka et al. 2016), and by limiting densities of large piscivorous trout in our study system, flood disturbance likely mediated the strength of native–invasive interactions in our riverscapes.
In a parallel study, we also found changes in fish abundance and composition evenness occurred at these confluences, and were driven by the spatial configuration of flow disturbance at the confluences (Boddy et al. 2019). Collectively, these two studies indicate that habitat preferences, movements and interspecific interactions, acting out over a complex spatial habitat mosaic, likely drive patterns in fish assemblages. Here, we found a significant interaction between flow disturbance configuration, spatial position at the confluence (tributary, upstream mainstem or downstream branch) and distance to confluence affected the relative abundance of native species, supporting our second prediction. The interaction between confluence configuration and distance to confluence suggests spatial processes such as dispersal and recolonisation are likely occurring in these systems, and influencing the relative abundance of native and invasive fish in our case. These processes have been widely anticipated (e.g., Schlosser 1991; Fausch et al. 2002), and are now being documented in riverine studies (e.g., Hitt and Angermeier 2008; Thornbrugh and Gido 2010; Woodford and McIntosh 2010). Thus, our findings are in line with a widening body of work suggesting large-scale spatial processes linked to dendritic network structures are important in structuring riverine communities.
One of the challenges in expanding knowledge of these large-scale spatial processes is identifying the underlying mechanisms involved, particularly those that might influence processes like the coexistence of native species with invaders. Stochastic processes such as recolonisation dynamics and presence of refuges may be more important in systems with high environmental variability, compared to those with low variability, where convergent processes like biological interactions might be stronger (Chase et al. 2009; Schlosser and Kallemeyn 2000). However, to develop our understanding further, we need to appreciate how these processes combine at larger scales, and specifically at network nodes such as confluences. We found that connection of stable-flowing and highly flood-disturbed streams by a confluence interacted to create important spatial patterns in species assemblages. For example, in stable-disturbed confluence configurations, stable upstream mainstems contained very low proportions of native fish (< 20% of the fish assemblage native), the disturbed tributaries had very high proportions of native fish (> 80%), and downstream there was higher co-occurrence (~ 60% native). Trout-dominated assemblages likely drove this spatial pattern in the stable upstream mainstem, whereas flood-adapted galaxiids occupied the disturbed tributary, with both of these upstream branches providing source populations to downstream reaches. Invasive trout and non-migratory galaxiids co-occur much more often than they coexist (Woodford and McIntosh 2010), and it is unknown whether these downstream reaches are sink habitats for native fish. Thus, while we can establish that heterogeneity in flood disturbance between upstream mainstem and tributary branches can increase co-occurrence between native and invasive fish species downstream of the confluence, we do not know if this heterogeneity actually facilitates coexistence such that galaxiids can successfully reproduce in these locations. Thus, these findings are consistent with our main hypothesis that flood disturbance structures fish assemblages and allows for native and non-native species coexistence by increasing habitat heterogeneity around river confluences, but cannot prove that coexistence is occurring.
The influences that tributaries have on the physical characteristics downstream of confluences, as well as the presence of source populations upstream (both native and non-native), likely play a role in determining the potential for native and non-native species coexistence. For example, stream width and associated physical conditions mediate how far downstream of ‘source’ subpopulations galaxiids located in tributaries above barriers co-occur with trout in mainstem ‘sink’ subpopulations (Woodford and McIntosh 2011). However, the stable-disturbed (i.e. disturbed tributary) confluence configuration mentioned above contrasts with the patterns found in disturbed-stable (i.e. stable tributary) confluences, which had almost entirely native fish assemblages in the mainstem, both upstream and downstream of the confluence, despite the presence of a stable tributary with very high proportions of trout. In those confluences, the tributary-resident trout appear to have relatively little effect on the mainstem galaxiid populations, either upstream or downstream. Thus, much remains to be understood about the mechanisms driving these patterns.
In our case, abiotic conditions mediating the strength of biotic interactions by limiting trout distributions is likely an important mechanism. For example, the disturbed tributary in the stable-disturbed configurations may have changed the downstream conditions to more intermediate levels of disturbance thereby moderating interspecific interactions between galaxiids and trout, and thus promote co-occurrence by limiting predation by large trout abundance downstream. There is of course evidence of important changes in physical conditions at river confluences (Benda et al. 2004; Kiffney et al. 2006; Rice et al. 2001), and that does happen at these confluences (Boddy et al. 2019), but whether it caused the more extensive galaxiid populations downstream remains to be seen. Our results, particularly from the disturbed-stable (i.e. stable tributary) configuration, do suggest however that heterogeneous disturbance conditions around confluences may extend the extent of co-occurrence between native and invasive fish species through colonist supply and/or by moderating interaction strengths.
Another important aspect of these confluence-influences is that species interactions occurring there could limit the direction and distance individuals could disperse and affect their vulnerability to invaders (Ganio et al. 2005; Jones and Schmidt 2017). We found a significant interaction between confluence configuration and distance to confluence in both mainstem upstream and downstream branches that affected the relative abundance of native fish (consistent with prediction 2). Thus, it was not simply the presence of heterogeneity, but also the spatial configuration and proximity of various habitats, that were important in determining assemblage composition. Significant distance-to-confluence effects affecting fish assemblages in branches, both upstream and downstream of confluences, show that the mobility of both groups of species involved species can propagate confluence influences in both directions. We expected that directional flow may have meant that some effects were more easily propagated downstream than upstream (prediction 3), consistent with upstream shifts in assemblages away from confluences often being abrupt (Thornbrugh and Gido 2010). This could occur for example if galaxiid fry, which are poor swimmers (Jones and Closs 2015), are more likely to colonize downstream of a source tributary than upstream. However, this pattern was not as common as expected. We were able to detect upstream shifts in the relative abundance of native fish, both in upstream mainstems and tributaries, however downstream effects were weak, so we were unable to resolve the downstream extent of confluence effects, or if interactions were affected indefinitely downstream. Nevertheless, river tributaries are capable of impacting mainstem biotic communities up to several kilometres downstream, depending on the mainstem disturbance regime (Benda et al. 2004; Rice et al. 2001; Woodford and McIntosh 2011). In our case, the behaviour of the early life history stages of galaxiids is likely to influence the extent of downstream co-occurrence depending on galaxiid swimming abilities and life stage as observed elsewhere (Jones and Schmidt 2017).
Overall, certain confluence configurations likely create opportunities for source–sink and recolonisation dynamics by enhancing riverscape spatial heterogeneity, which could explain some configuration-specific differences in distance-related patterns we observed. Nevertheless, elevated relative abundances of native fish near confluences in stable upstream branches of stable-disturbed configurations where large trout would normally eliminate galaxiids are likely indicative of sink populations of galaxiids. Galaxiids can persist in sink habitats near sources where propagule pressure is high, but because propagule pressure decreases with increasing distance to the source (Woodford and McIntosh 2011), proximity to source populations likely affects spatial patterns in co-occurrence within sink habitats in invaded riverscapes. Dramatic shifts in community composition in close proximity to confluences have also been associated with fish moving between more physiologically suitable, but food-limited habitat, and less-suitable but more resource-rich stream branches (Brewitt et al. 2017), creating so called ‘landscape complementation effects’ (Thornbrugh and Gido 2010). In the case of vulnerable native species in invaded riverscapes, ‘attractive sink’ habitats (i.e., stable branches) could represent resource-rich habitats offering higher growth rates but with a trade-off of increased mortality, and potentially reduced chances of successful reproduction (McIntosh et al. 2010; Timus et al. 2016). This could be problematic if species vulnerable to attractive sinks do not perceive any increased predation risk.
Stable-stable confluence configurations were characterised by universally low relative abundance of native galaxiids regardless of distance to confluence and were likely sink habitat resulting from strong interspecific interactions with trout (Woodford and McIntosh 2010). Thus, these homogeneous situations were universally bad for the native species we studied, consistent with the environmental heterogeneity hypothesis of invasions (Melbourne et al. 2007). In contrast, disturbed-disturbed configurations had extremely low total fish abundance, and highly variable proportions of native fish. These disturbed-disturbed situations are likely the result of disturbance-driven ‘pseudosinks’ rather than interactions between native and non-native species. For example, Woodford and McIntosh (2010) identified flood-disturbed and large streams as likely pseudosinks for non-migratory galaxiids in New Zealand, whereby very low abundances of galaxiids were isolated from source streams, and as such, susceptible to localised extinction. Therefore confluences with homogenous configurations of flow disturbance, whether stable or disturbed, tended to have lower abundance, and lower proportions of native fish, but only in the case of stable-stable confluences was this because of environmental homogeneity.
In the face of increasing homogenisation of ecosystems globally, developing and using methods to identify heterogeneity influences for management and conservation is going to be crucial. Our results, revealing the role of spatial configuration of flow disturbance around confluences in influencing the relative abundance of native and invasive fish species, in conjunction with previous studies (Brewitt et al. 2017; Peláez et al. 2017; Thornbrugh and Gido 2010), highlight the importance of spatial context in determining local assemblage characteristics. Importantly, understanding how invasive species impacts vary with physical heterogeneity, provides an opportunity to strategically prioritize management efforts at large spatial scales to minimise invader impacts (Hansen et al. 2013; Vander Zanden et al. 2017). Moreover, revealing the causes of context-dependence in invasive predator impacts, helps fill the knowledge gap around how species traits and spatial heterogeneity influence invader impacts (Kumschick et al. 2015; Latzka et al. 2016), potentially enabling targeted management of high risk locations. In addition, confluence-focussed management could promote co-occurrence, and hopefully coexistence, as an effective solution in globally common situations where invasive fish species such as trout are recreationally and economically important, but are impacting endangered native species. Management actions such as environmental flow setting or maintenance of existing heterogeneity could prioritize the protection or restoration of mutually beneficial configurations of flood disturbance in these scenarios (Chen and Olden 2017).
References
Baltz DM, Moyle PB (1984) Segregation by species and size classes of rainbow trout, Salmo gairdneri, and Sacramento sucker, Catostomus occidentalis, in three California streams. Environ Biol Fishes 10:101–110
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using Ime4. J Stat Soft 67(1)
Barton K (2016) MuMIn: multi-model inference. R package version 1.15.6. https://CRAN.R-project.org/package=MuMIn
Benda L, Poff NL, Miller D et al (2004) The network dynamics hypothesis: how channel networks structure riverine habitats. Bioscience 54:413–427
Boddy NC, McIntosh AR (2017) Temperature, invaders and patchy habitat interact to limit the distribution of a vulnerable freshwater fish. Austral Ecol 42(4):456–467
Boddy NC, Booker DJ, Mcintosh AR (2019) Confluence configuration of river networks controls spatial patterns in fish communities. Landsc Ecol 34:187–201
Brewitt KS, Danner EM, Moore JW (2017) Hot eats and cool creeks: juvenile Pacific salmonids use mainstem prey while in thermal refuges. Can J Fish Aquat Sci 74:1588–1602
Budy P, Thiede GP, McHugh P et al (2008) Exploring the relative influence of biotic interactions and environmental conditions on the abundance and distribution of exotic brown trout (Salmo trutta) in a high mountain stream. Ecol Freshw Fish 17:554–566
Cadwallader PL (1976) Home range and movements of the common river glaxias, Galaxias vulgaris Stokell (Pisces: Salmoniformes), in the Glentui River, New Zealand. Aust J Mar Freshwater Res 27:23–33
Cathcart CN, Gido KB, McKinstry MC et al (2018) Patterns of fish movement at a desert river confluence. Ecol Freshw Fish 27:492–505
Chase JM, Biro EG, Ryberg WA et al (2009) Predators temper the relative importance of stochastic processes in the assembly of prey metacommunities. Ecol Lett 12:1–9
Chen W, Olden JD (2017) Designing flows to resolve human and environmental water needs in a dam-regulated river. Nat Commun 8:2158
Cowie B, Milen PJ, Mason CR et al (1986) Waimakariri River and catchment resource survery, vol 1. North Canterbury Catchment Board and Regional Water Board, Christchurch
Czeglédi I, Sály P, Takács P et al (2015) The scales of variability of stream fish assemblages at tributary confluences. Aquat Sci 78:641–654
Dudgeon D, Arthington AH, Gessner MO et al (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev Camb Philos Soc 81:163–182
Fausch KD (1988) Tests of competition between native and introduced salmonids in streams: what have we learned? Can J Fish Aquat Sci 45:2238–2246
Fausch KD, Tanguchi Y, Nakano S et al (2001) Flood disturbance regimes influence rainbow trout invasion success among five holoarctic regions. Ecol Appl 11:1438–1455
Fausch KD, Torgersen CE, Baxter CV et al (2002) Landscapes to riverscapes: bridging the gap between research and conservation of stream fishes. Bioscience 52:483–498
Fernandes CC, Podos J, Lundberg JG (2004) Amazonian ecology: tributaries enhance the diversity of electric fishes. Science 305:1960–1962
Ganio LM, Torgersen CE, Gresswell RE (2005) A geostatistical approach for describing spatial pattern in stream networks. Front Ecol Environ 3:138–144
Green JC (2003) The precision of sampling grain-size percentiles using the Wolman method. Earth Surf Process Landf 28(9):979–991
Habit E, Piedra P, Ruzzante DE et al (2010) Changes in the distribution of native fishes in response to introduced species and other anthropogenic effects. Glob Ecol Biogeogr 19:697–710
Hansen GJ, Vander Zanden MJ, Blum MJ et al (2013) Commonly rare and rarely common: comparing population abundance of invasive and native aquatic species. PLoS One 8:e77415
Hasegawa K, Mori T, Yamazaki C (2016) Density-dependent effects of non-native brown trout Salmo trutta on the species-area relationship in stream fish assemblages. J Fish Biol 90:370–383
Hitt NP, Angermeier PL (2008) Evidence for fish dispersal from spatial analysis of stream network topology. J North Am Benthol Soc 27:304–320
Jellyman PG, McIntosh AR (2010) Recruitment variation in a stream galaxiid fish: multiple influences on fry dynamics in a heterogeneous environment. Freshw Biol 55:1930–1944
Jellyman PG, McIntosh AR (2020) Disturbance-driven consumer assemblages determine fish communities and moderate top-down effects via bottom-up constraints. J Anim Ecol 89:1175–1189
Jellyman PG, Booker DJ, McIntosh AR (2013) Quantifying the direct and indirect effects of flow-related disturbance on stream fish assemblages. Freshw Biol 58:2614–2631
Jellyman PG, McHugh PA, Simon KS et al (2017) The effects of brown trout on the trophic webs of New Zealand streams. In: Lobón-Cerviá J, Sanz N (eds) Brown trout: biology, ecology and management, 1 edn. Wiley, Hoboken, pp 570–597
Jones PE, Closs GP (2015) Life history influences the vulnerability of New Zealand galaxiids to invasive salmonids. Freshw Biol 60:2127–2141
Jones NE, Schmidt BJ (2017) Tributary effects in rivers: interactions of spatial scale, network structure, and landscape characteristics. Can J Fish Aquat Sci 74:503–510
Jowett IG (1990) Factors related to the distribution and abundance of brown and rainbow trout in New Zealand clear-water rivers. N Z J Mar Freshwater Res 24:429–440
Jowett IG, Richardson J (1989) Effects of a severe flood on instream habitat and trout populations in seven New Zealand rivers. N Z J Mar Freshwater Res 23:11–17
Kiffney PM, Greene CM, Hall JE et al (2006) Tributary streams create spatial discontinuities in habitat, biological productivity, and diversity in mainstem rivers. Can J Fish Aquat Sci 63:2518–2530
Kumschick S, Gaertner M, Vilà M et al (2015) Ecological impacts of alien species: quantification, scope, caveats, and recommendations. Bioscience 65:55–63
Lake PS (2000) Disturbance, patchiness, and diversity in streams. J North Am Benthol Soc 19:573–592
Latzka AW, Hansen GJ, Kornis MS et al (2016) Spatial heterogeneity in invasive species impacts at the landscape scale. Ecosphere 7:e01311
Leathwick JR, Elith J, Chadderton WL, Rowe D, Hastie T (2008) Dispersal, disturbance and the contrasting biogeographies of New Zealand’s diadromous and non-diadromous fish species. J Biogeogr 35(8):1481–1497
Lowe S, Browne M, Boudjelas S et al (2000) 100 of the world’s worst invasive alien species. A selection from the Global Invasive Species Database. Published by the Invasive Species Specialist Group (ISSG) a specialist group of the Species Survivial Comission (SSC) of the World Conservation Union (IUCN). Hollands Printing Ltd, New Zealand
Maciel GA, Lutscher F (2018) Movement behaviour determines competitive outcome and spread rates in strongly heterogeneous landscapes. Theor Ecol 11:351–365
McDowall RM (1990) New Zealand freshwater fishes: A natural history and guide. Heinemann Reed, Auckland
McDowall RM (2006) Crying wolf, crying foul, or crying shame: alien salmonids and a biodiversity crisis in the southern cool-temperate galaxioid fishes? Rev Fish Biol Fish 16:233–422
McHugh PA, McIntosh AR, Jellyman PG (2010) Duel influences of ecosystem size and disturbance on food chain length in streams. Ecol Lett 13:881–890
McIntosh AR (2000) Habitat- and size-related variations in exotic trout impacts on native galaxiid fishes in New Zealand streams. Can J Fish Aquat Sci 57:2140–2151
McIntosh AR, Crowl TA, Townsend CR (1994) Size-related impacts of introduced brown trout on the distribution of native common river galaxiids. N Z J Mar Freshwat Res 28:135–144
McIntosh AR, McHugh PA, Dunn N et al (2010) The impact of trout on galaxiid fishes in New Zealand. N Z J Ecol 34:195–206
Melbourne BA, Cornell HV, Davies KF et al (2007) Invasion in a heterogeneous world: resistance, coexistence or hostile takeover? Ecol Lett 10:77–94
Mims MC, Olden JD (2012) Life history theory predicts fish assemblage response to hydrologic regimes. Ecology 93:35–45
Moorhouse TP, Macdonald DW (2014) Are invasives worse in freshwater than terrestrial ecosystems? WIREs Water 2:1–8
Morita K (2018) Assessing the long-term causal effect of trout invasion on a native charr. Ecol Indic 87:189–192
Nakagawa S, Schielzeth H, O’Hara RB (2013) A general and simple method for obtaining from generalized linear mixed-effects models. Methods Ecol Evol 4(2):133–142
Olden JD, Kennard MJ (2010) Intercontinental comparison of fish life history strategies along a gradient of hydrologic variability. American Fisheries Society Symposium 73:83–107
Peckarsky BL, McIntosh AR, Horn SC, McHugh K, Booker DJ, Wilcox AC, Brown W, Alvarez M (2014) Characterizing disturbance regimes of mountain streams. Freshw Sci 33(3):716–730
Peláez OE, Azevedo FM, Pavanelli CS (2017) Environmental heterogeneity explains species turnover but not nestedness in fish assemblages of a Neotropical basin. Acta Limnol Bras 29:e117
Peterson EE, Ver Hoef JM (2010) A mixed-model moving-average approach to geostatistical modeling in stream networks. Ecology 91(3):644–651
Pfankuch DJ (1975) Stream reach inventory and channel stability evaluation. United States Department of Agriculture Forest Service, Region 1
Rice SP (2017) Tributary connectivity, confluence aggradation and network biodiversity. Geomorphology 277:6–16
Rice SP, Greenwood MT, Joyce CB (2001) Tributaries, sediment sources, and the longitudinal organisation of macroinvertebrate fauna along river systems. Can J Fish Aquat Sci 58:824–840
Schlosser IJ (1991) Stream fish ecology: a landscape perspective. Bioscience 41:704–712
Schlosser IJ, Kallemeyn LW (2000) Spatial variation in fish assemblages across a beaver-influenced successional landscape. Ecology 81:1371–1382
Sowersby W, Thompson RM, Wong BBM (2015) Invasive predator influences habitat preferences in a freshwater fish. Environ Biol Fishes 99:187–193
Thornbrugh DJ, Gido KB (2010) Influence of spatial positioning within stream networks on fish assemblage structure in the Kansas River basin, USA. Can J Fish Aquat Sci 67:143–156
Timus N, Czekes Z, Rákosy L et al (2016) Conservation implications of source-sink dynamics within populations of endangered Maculinea butterflies. J Insect Conserv 21:369–378
Vander Zanden MJ, Hansen GJA, Latzka AW (2017) A framework for evaluating heterogeneity and landscape-level impacts of non-native aquatic species. Ecosystems 20:477–491
Wilcox KR, Tredennick AT, Koerner SE et al (2017) Asynchrony among local communities stabilises ecosystem function of metacommunities. Ecol Lett 20:1534–1545
Woodford DJ, McIntosh AR (2010) Evidence of source-sink metapopulations in a vulnerable native galaxiid fish driven by introduced trout. Ecol Appl 20:967–977
Woodford DJ, McIntosh AR (2011) Location of demographic sources affects the distributions of a vulnerable native fish in invaded river networks. Freshw Biol 56:311–324
Woodford DJ, McIntosh AR (2013) Effects of introduced trout predation on non diadromous galaxiid fish populations across invaded riverscapes. Science for Conservation 320. Department of Conservation, Wellington. 23p
Zeni JO, Casatti L (2014) The influence of habitat homogenisation on the trophic structure of fish fauna in tropical streams. Hydrobiologia 726:259–270
Acknowledgements
We are grateful to Castle Hill, Brooksdale, Flock Hill and Glenthorne Stations, and to the Department of Conservation for allowing access to sampling sites. Funding was provided by a subcontract from the New Zealand National Institute for Water and Atmospheric Research under the Freshwater & Estuaries Research Programme 2, Sustainable Water Allocation (2014/15 SCI). We thank Richard White, Brandon Goeller, Sophie Hale, Simon Howard and Tom Moore for field assistance, Linda Morris for technical support, Jenny Ladley for logistical support, and the University of Canterbury for use of the Cass Field Station. Discussions with Erin Peterson at the Queensland University of Technology, Phillip Jellyman from the New Zealand National Institute for Water and Atmospheric Research, and the Freshwater Ecology Research Group (University of Canterbury), as well as comments from Jerry Closs, Winsor Lowe and several anonymous reviewers enhanced the manuscript. Procedures were conducted under Animal Ethics permit 2014/28R from the University of Canterbury.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Estimated regression parameters, standard errors, z-values and P-values for the top binomial generalised linear mixed-effect models presented in Table 1.
Estimate | Std. error | z value | P-value | |
|---|---|---|---|---|
H1) Proportion native ~ branch disturbance + branch + reach lengths from confluence + (1|confluenceID) | ||||
Intercept | 2.375 | 1.03 | 2.305 | 0.021 |
Branch disturbance Stable | − 4.576 | 1.138 | − 4.02 | <0.001 |
Branch tributary | 0.986 | 0.802 | 1.229 | 0.219 |
Branch upstream | − 0.605 | 0.595 | − 1.018 | 0.309 |
Reach lengths from confluence | − 0.072 | 0.032 | − 2.275 | 0.023 |
H2) Proportion native ~ confluence configuration x branch x reach lengths from confluence + (1|confluenceID) | ||||
Intercept | − 0.434 | 1.294 | − 0.336 | 0.737 |
Confluence configuration Disturbed-Stable | 2.754 | 1.717 | 1.604 | 0.109 |
Confluence configurationStable-Disturbed | 0.752 | 2.015 | 0.374 | 0.708 |
Confluence configuration Stable-Stable | − 2.647 | 1.893 | − 1.399 | 0.161 |
Branch tributary | 2.794 | 0.471 | 5.925 | <0.001 |
Branch upstream | 0.667 | 0.289 | 2.306 | 0.021 |
Reach lengths from confluence | − 0.046 | 0.02 | − 2.302 | 0.021 |
Confluence configuration Disturbed-Stable:BranchTributary | − 3.853 | 0.738 | − 5.22 | < 0.001 |
Confluence configuration Stable-Disturbed:BranchTributary | − 0.449 | 0.616 | − 0.729 | 0.466 |
Confluence configuration Stable-Stable:BranchTributary | − 3.978 | 1.189 | − 3.343 | <0.001 |
Confluence configuration Disturbed-Stable:BranchUpstream | − 0.578 | 0.477 | − 1.21 | 0.226 |
Confluence configuration Stable-Disturbed:BranchUpstream | − 0.55 | 0.44 | − 1.251 | 0.21 |
Confluence configuration Stable-Stable:BranchUpstream | 0.705 | 0.851 | 0.828 | 0.407 |
Confluence configuration Disturbed-Stable:Reach lengths from confluence | 0.034 | 0.027 | 1.245 | 0.213 |
Confluence configuration Stable-Disturbed:Reach lengths from confluence | 0.058 | 0.023 | 2.527 | 0.011 |
Confluence configuration Stable-Stable:Reach lengths from confluence | 0.102 | 0.037 | 2.716 | 0.006 |
BranchTributary:Reach lengths from confluence | 0.034 | 0.034 | 0.99 | 0.322 |
BranchUpstream:Reach lengths from confluence | − 0.124 | 0.055 | − 2.235 | 0.025 |
Confluence configuration Disturbed-Stable:BranchTributary:Reach lengths from confluence | − 0.177 | 0.06 | − 2.945 | 0.003 |
Confluence configuration Stable-Disturbed:BranchTributary:Reach lengths from confluence | − 0.082 | 0.042 | − 1.942 | 0.052 |
Confluence configuration Stable-Stable:BranchTributary:Reach lengths from confluence | 0.009 | 0.077 | 0.122 | 0.903 |
Confluence configuration Disturbed-Stable:BranchUpstream:Reach lengths from confluence | 0.086 | 0.06 | 1.423 | 0.154 |
Confluence configuration Stable-Disturbed:BranchUpstream:Reach lengths from confluence | 0.009 | 0.062 | 0.151 | 0.879 |
Confluence configuration Stable-Stable:BranchUpstream:Reach lengths from confluence | 0.074 | 0.074 | 0.993 | 0.32 |
H3) Upstream: proportion native ~ confluence configuration x distance to confluence + (1|confluenceID) | ||||
Intercept | 0.371 | 1.626 | 0.228 | 0.819 |
Confluence configuration Disturbed-Stable | 3.603 | 2.212 | 1.629 | 0.103 |
Confluence configuration Stable-Disturbed | − 1.867 | 2.764 | − 0.675 | 0.499 |
Confluence configuration Stable-Stable | − 2.839 | 2.401 | − 1.182 | 0.237 |
Reach lengths from confluence | − 0.147 | 0.052 | − 2.796 | 0.005 |
Confluence configuration Disturbed-Stable:Reach lengths from confluence | 0.062 | 0.056 | 1.092 | 0.274 |
Confluence configuration Stable-Disturbed:Reach lengths from confluence | 0.04 | 0.059 | 0.678 | 0.497 |
Confluence configuration Stable-Stable:Reach lengths from confluence | 0.199 | 0.075 | 2.654 | 0.007 |
H3) Tributary: proportion native ~ branch disturbance + distance to confluence + (1|confluenceID) | ||||
Intercept | 2.013 | 1.109 | 1.816 | 0.069 |
Branch disturbance stable | − 3.582 | 1.441 | − 2.486 | 0.013 |
Reach lengths from confluence | − 0.039 | 0.015 | − 2.56 | 0.011 |
H3) Downstream: proportion native ~ confluence configuration x distance to confluence + (1|confluenceID) | ||||
Intercept | 0.573 | 0.002 | 208.985 | <0.001 |
Confluence configuration Disturbed-Stable | 2.145 | 0.002 | 782.334 | <0.001 |
Confluence configuration Stable-Disturbed | − 0.255 | 1.545 | − 0.166 | 0.868 |
Confluence configuration Stable-Stable | − 4.968 | 0.002 | − 1811.26 | <0.001 |
Reach lengths from confluence | − 0.069 | 0.002 | − 25.724 | <0.001 |
Confluence configuration Disturbed-Stable:Reach lengths from confluence | 0.046 | 0.002 | 17.127 | <0.001 |
Confluence configuration Stable-Disturbed:Reach lengths from confluence | 0.082 | 0.011 | 6.907 | <0.001 |
Confluence configuration Stable-Stable:Reach lengths from confluence | 0.126 | 0.002 | 46.047 | <0.001 |
H4) proportion small galaxiids ~ proportion large trout + branch + distance to confluence + (1|confluenceID) | ||||
Intercept | − 1.624 | 0.653 | − 2.486 | 0.012 |
Proportion large trout | − 18.5 | 2.467 | − 7.498 | <0.001 |
Branch Tributary | 0.505 | 0.11 | 4.571 | <0.001 |
Branch Upstream | − 0.258 | 0.094 | − 2.73 | 0.006 |
Reach lengths from confluence | 0.004 | 0.005 | 0.878 | 0.379 |
Rights and permissions
About this article
Cite this article
Boddy, N.C., Booker, D.J. & McIntosh, A.R. Heterogeneity in flow disturbance around river confluences influences spatial patterns in native and non-native species co-occurrence. Biol Invasions 22, 3457–3475 (2020). https://doi.org/10.1007/s10530-020-02334-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02334-1