Abstract
The invasive Australian swamp stonecrop, Crassula helmsii, is a perennial amphibious herb originating from Australia and New Zealand. In freshwater wetlands of North-western Europe, this alien plant species is invasive due to its efficient colonization of empty niches. The establishment of dense C. helmsii growth is threatening native biodiversity and functioning of freshwater ecosystems, especially oligotrophic wetlands with high disturbance and nutrient enrichments. As the effects of these potential drivers of ecosystem degradation are generally difficult to determine in the field, we tested the competitive strength of C. helmsii in a greenhouse experiment with two native competitor species of the same habitat type, Pilularia globulifera and Littorella uniflora. Sods dominated by either of the native species, as well as bare soils, were collected from the field and manually infested with propagules of C. helmsii. Settlement and growth of C. helmsii was assessed after five weeks. In addition, the effect of nutrient enrichment by water bird feces on competition was studied by adding waterfowl droppings. C. helmsii was able to settle successfully in all treatments, but P. globulifera and L. uniflora dominance reduced settlement success and growth of C. helmsii. On vegetated sods, the addition of waterfowl droppings had a low effect on the performance of C. helmsii, however, this treatment significantly increased biomass production on bare soils with low nutrient availability. We conclude that both absence of native competitors and eutrophication, including guanotrophication by waterfowl, explain the establishment success and invasiveness of C. helmsii. Given the fact that eradication of C. helmsii is very challenging, our results imply that management should focus on a combination of increasing local species densities and abating eutrophication. This will strongly limit the window of opportunity for invasion of C. helmsii and enhance resistance by native plant communities.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Australian swamp stonecrop Crassula helmsii (T.Kirk) Cockayne is a perennial amphibious plant. Native to Australia and New Zealand, this species is presumed to have been introduced to Europe for ornamental use in ponds (Dawson and Warman 1987; Robert et al. 2013). It is highly invasive in Northwestern Europe and is increasingly dominating moorlands, dune pools and other freshwater to slightly brackish wetlands, thereby threatening endangered native species and protected ecosystems (Dawson and Warman 1987; Leach and Dawson 1999; OEPP/EPPO 2007; Newman 2013; Brouwer et al. 2017; Prinz et al. 2019; Smith and Buckley 2020). Eradication of C. helmsii has been found to be both difficult and costly (Van der Loop et al. 2018), and effective eradication is only possible under certain conditions (Dawson and Warman 1987; Robert et al. 2013; Van Kleef et al. 2017). Only when infestations are small and isolated, and thorough measures are taken, can eradication be successful (Brouwer et al. 2017; Hussner et al. 2017; Van Kleef et al. 2017; Van der Loop et al. 2018). Since these conditions are rarely met, it is necessary to explore alternative options to control this species.
The biotic resistance of a community may reduce the success of biological invasions (e.g., Moulton and Pimm 1983; Case 1990; Kennedy et al. 2002; Levine et al. 2003). Although the mere presence of native vegetation does not enable ecosystems to resist invasions given the ongoing propagule pressure. The dominance of the invasive species can be repressed by competition (Levine et al. 2003). Furthermore, when the native vegetation has similar traits or if most available niches are occupied, new species are less likely to become established (Funk et al. 2008). Disturbances often create new available niches, which can be rapidly occupied by alien invasive species (e.g., Hobbs 1989, 1991; Hobbs and Huenneke 1992; Rejmánek 1999).
In this paper, we test the role of native species in the invasion success of C. helmsii in both oligo—and eutrophic ecosystems, to explore whether active management focusing on harnessing native species can reduce the settlement and dominance of C. helmsii. Two native species were selected, Pilularia globulifera L. and Littorella uniflora (L.) Ascherson to provide competition with C. helmsii. Both species occur in habitats susceptible to invasions of C. helmsii and are regularly found in high abundance, indicating that they may be strong competitors (e.g., Arts and Leuven 1988; Bloemendaal and Roelofs 1988). P. globulifera grows on soils with a relatively high organic content, whereas L. uniflora thrives on more oligotrophic, mineral soils. Both species form dense swards limiting space and available nutrients for C. helmsii, potentially affecting its settlement and growth. We therefore hypothesize that these species are able to suppress the settlement and growth of C. helmsii and consequently increase the biotic resistance to invasions of the alien plant species.
Additionally we study the effects of water birds on invasions of C. helmsii. Endo—and epizoochorous transport by water birds is believed to be an important vector for C. helmsii. Geese droppings contain many poorly digested plant fragments, often still capable of vegetative regeneration (Denys et al. 2014). Not only can these two processes greatly enhance propagule pressure over large areas, but nutrient release from feces also works as a natural fertilizer (Dawson and Warman 1987; Robert et al. 2013; Dean et al. 2015; Brouwer et al. 2017; Hussner et al. 2017). The enrichment of nutrients from feces is therefore hypothesized to facilitate the invasiveness of C. helmsii in oligotrophic ecosystems (Brouwer et al. 2017; Van Kleef et al. 2017), but only in the absence of competitors.
A greenhouse experiment was carried out to test the strength of biotic resistance exerted by native plant communities on invasive C. helmsii in oligotrophic and nutrient rich environments. An additional treatment with waterfowl feces was included to assess the potential facilitation of invasion through guanotrophication.
Materials and methods
Experimental setup
Experiments were carried out between August and November 2017 in the greenhouse facilities of Radboud University (Nijmegen, The Netherlands). Temperature was set on 20.0 and 17.1 °C during day and night, respectively. Assimilation lighting (16 h light, 8 h dark) was fixed at 174 µmol m− 2 s− 1. Vegetated and bare sods (26.5 × 36.5 cm) were collected from two soft water lakes in the Netherlands (i.e. Moerven and Korenburgerven). Sods containing a closed vegetation of L. uniflora and bare soils (n = 16 for each) were collected in the Moerven (nature reserve Huis ter Heide, 51°36′31.11″ N, 5°1′45.05″ E). Sods containing a closed vegetation of P. globulifera and bare soils (n = 16 for each) were collected in the Korenburgerven (nature reserve Korenburgerveen, 51°59’29.87"N, 6°38’43.91"E). In the latter area, C. helmsii was present. Vegetated and bare ground soil composition were assumed to be equal is origin, as extractions were just a few meters apart. The sods were placed in plastic containers without draining and manually cleared from all C. helmsii biomass. Following two weeks without any new shoot formation, the sod was considered free from C. helmsii. Germination from possible seed banks in sods under experimental conditions are never recorded and features of germinating plants from seeds and sprouting from fragments, as used in this experiment, remarkably differ.
After two weeks of acclimatization to the environmental conditions of the greenhouse complex, sods were infested with C. helmsii collected from the Akkerenven (51°25’32.73"N, 4°21’31.66"E, The Netherlands). To simulate the settlement of C. helmsii in the field, the plant was fragmented with each plant piece containing one node and two leaves, but no roots. Hussner (2009) reported this as the minimum fragment size for regeneration. Experiments with fragments of this size mimic the dispersal of propagules by waterfowl or human assisted spread via the treat of footwear. In total, 60 C. helmsii fragments were placed within three rows with 20 fragments per sod. We used sods with bare soils (n = 30) and sods that were completely covered (100%) with L. uniflora (n = 16) or P. globulifera (n = 16).
For each treatment, half the sods received waterfowl feces which were collected in an urban and agricultural area in Arnhem (51°59′15.86″ N, 5°54′7.58″ E) and Nijmegen (51°52′44.56″ N, 5°51′49.28″ E), the Netherlands, respectively. Feces from two different species of waterfowl, Anser anser L. (Anatidae), and Alopochen aegyptiacus L. (Tadorninae), were collected in equal amounts. In order to minimize the risk of infestation by waterfowl droppings, we collected these from sites confirmed to have no C. helmsii infestation, and where waterfowl populations were predominantly sedentary birds. Before application, feces were mixed and molded in homogenous droppings of 6 gram FW (i.e. the average weight of a dropping). Nutrient content was not determined. Literature data show that geese dropping release between 371 and 500 µmol inorganic nitrogen and 40–46 mg phosphate per gram of feces (Liu et al. 2014; Frazão 2008). An amount of 6 feces was added to the duplicated sods. During the experiment approximately 13.37–18.00 mmol inorganic nitrogen, and approximately 1.4–1.7 g phosphate per sod was added. We replicated 8 sods per treatment combination, except treatment bare soils of P. globulifera with feces n = 06, for a total of 62 replicates. Table 1 shows an overview of all treatment combinations. For 8 weeks the sods were watered with rainwater every 48 h until the water level was at soil surface. Water loss only occurred by evapotranspiration. At the start of the experiment, and every two weeks thereafter, soil porewater samples were taken using rhizons. Seedlings of plant species other than L. uniflora, P. globulifera and C. helmsii, were manually removed from all sods. After 2 weeks, aphids were observed in P. globulifera vegetation, and therefore all sods were treated once with the biological control Capsanem and Entonem, nematodes Steinernema carpocapsae and S. feltiae respectively (both 0.5 million nematodes per m2 soil surface) which successfully eradicated the aphids with no visible harm of any plant species. Settlements of C. helmsii propagules were counted in week 2, 5 and 8. Propagules were considered ‘settled’ when they produced new biomass such as new nodes, leaves or roots. Nine weeks after inoculation of the sods, C. helmsii biomass as well as soil and vegetation samples from P. globulifera and L. uniflora were harvested. One quarter of the vegetation area was harvested for analysis, and dry weights were multiplied by 4. After harvest and thorough rinsing to remove any soil particles, all plant samples were put in a drying stove for 48 h at 70 °C to determine dry weight.
Chemical analyses
Porewater pH was measured with a standard combined glass Ag/AgCl pH electrode (Orion Research, Beverly, CA, USA) connected to a pH meter (Tim800; Radiometer analytical, Lyon, France). Alkalinity was estimated by titration to pH 4.2 with 0.1 mmol L− 1 HCl using an auto burette (ABU901, Radiometer, Lyon, France). An auto-analyser 3 system (Bran and Lubbe, Norderstedt, Germany) was used to measure concentrations of nitrate and ammonium colourimetrically using hydrazine sulphate (Kamphake et al. 1967) and salicylate (Grasshof and Johannsen 1972) respectively. An inductively coupled plasma spectrometry (ICP-OES icap 6000; ThermoFischer scientific, Waltham, MA, USA) was used to measure the concentrations of calcium, iron, magnesium, potassium, phosphorus and sulphate. Plant nitrogen content was measured by grinding dried plant material and measuring the concentration on a CN analyser (model EA NA 1500, Thermo Fisher Scientific). Soil (0.5 L) of every sod was collected and similarly analysed for its chemical content of plant available nitrogen and phosphorus content.
Statistical analysis
Since assumptions for normality a homogeneity of variance were not met for either soil and plant chemistry, nor for C. helmsii response parameters, treatment effects were tested for significance using General Linear Models. Effects on settlement was tested using a quasipoisson error distribution on a log-linked GLM model, since significant overdispersion was present when using a standard Poisson error distribution. All other effect parameters were tested using a Gamma GLM with identity link. Data was tested and visualized using the statistical programme R version 3.5.2 (Team 2018). Treatment and/or treatment combination effects were tested for significance by a priori testing approach using a full factorial predictor structure (single treatment and all treatment interaction effects) in all response models. For all tested response parameter, box plots have been drawn to provide visual representation of treatment effects (Figs. 1, 2, 3, 4, 5).
Results
Soil nutrient status
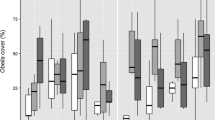
The Huis Ter Heide soil was characterized as nutrient poor mineral soil with low plant available inorganic nitrogen (N) and phosphorus (P) concentrations (Table 2; Fig. 1). The Korenburgerveen soil was characterized as a mineral soil substantially richer in plant available N as well as P concentration, which was emphasized by a clearly visible organic layer. Average water levels of both experimental systems were 0–1 cm below soil level. Plant carbon limitation by aquatic growth was absent. Feces treatment did not significantly increase plant available P or inorganic N in in the soils below the feces. Two sods (replicates of treatment combinations C− F+ P. globulifera) represented significant outliers for multiple parameters, these were excluded from all statistical analysis.
Nutrient concentrations in pore water (a, c, start of experiment) and plant-available inorganic N and PO43− in the soils (b, d, end of experiment, extractable from soil) for nutrient poor and rich soils (P. globulifera treatment C− F+: n = 06; all other treatments: n = 08). C+ and C− indicate treatments with and without competition by native vegetation, respectively; treatments with and without feces addition are indicated by F+ and F−, respectively
Tissue chemistry of C. helmsii
Plant tissue N:P ratios (g/g) of C. helmsii strongly differed between locations after experiment. For the nutrient poor site they were 19.28 (which may be indicative of P limitation (Koerselman and Meuleman 1996; Güsewell 2004) whereas at the nutrient rich site tissue N:P ratio was 4.54 (Fig. 2). Limitations of N and P occur when ratios are ~ 10 and ~ 20, respectively. At N:P ratios between ~ 10 and ~ 20, either N or P can be limiting or plant growth is co-limited by both nutrients (Koerselman and Meuleman 1996). The addition of feces lowered N:P ratios for plants from Huis Ter Heide to 9.83, suggesting that plants became less P-limited by this treatment.
Settlement of C. helmsiisettlement of C. helmsii
Settlement of C. helmsii on bare soils was high (83.94% of propagules), and much higher than in treatments with native plant species present (26.83%, Fig. 3, Competition: t = −4.99, p < 0.001). Nutrient status of the system had no significant effect on settlement on bare soils (Table 2; Nutrient rich: t = −0.29, p = 0.77). However, the competition effect of the native species vegetation was much stronger in the nutrient rich treatments dominated by P. globulifera (33.06% lower settlement) than in the nutrient poor treatments dominated by L. uniflora (Table 2; Competition x Nutrient rich: t = −5.40, p < 0.001).
Unexpectedly, the addition of waterfowl feces did not affect C. helmsii settlement, neither in the bare treatments nor in the treatments with local vegetation present (Fig. 3; Table 2). This was the case for both the nutrient rich and nutrient poor treatments (Fig. 3; Table 2).
The number of settled propagules (max = 60) of Crassula helmsii on the nutrient low mineral soils (a) and the nutrient rich organic soil (b) in all treatment combinations (n = 08). Presence or absence of competitions is indicated by C+ and C−. Treatments with and without by feces addition are indicated by F+ and F−, respectively
Biomass
The realized biomass of C. helmsii was much higher on bare soil than in the presence of native plant species competition (Fig. 4, Competition: t = −3.30, p < 0.01). For the nutrient-poor and -rich site, realized biomass at the end of the experiment on bare soils was 29.9 and 281.5 times higher, respectively (Fig. 4; Table 2; Nutrient rich: t = −2.57, p < 0.05). Competition with native vegetation resulted in a strong reduction in C. helmsii realized biomass, indicating growth rates close to zero for both the nutrient poor and rich system (Fig. 4). As a consequence of higher realized biomass on bare soil, the competition effect was significantly stronger in nutrient rich sites (Table 2; Competition x Nutrient rich: t = −2.60, p < 0.05).
The addition of waterfowl feces only affected C. helmsii growth for nutrient poor, bare soil sites (Table 2: Feces: t = 2.08, p < 0.05), where realized biomass at the end of the experiment was doubled. Growth was also stimulated for these sites when native vegetation was present (Table 2: Competition x feces: t = −2.04, p < 0.05). However, the absolute increase under competition was minimal compared to no competition (Fig. 2). The stimulating effect was absent in nutrient rich bare soil (Table 2: nutrient rich x feces: t = −0.39, p = 0.7) sods, as well as under competition in nutrient rich sods (Table 2: Competition x Nutrient rich x feces: t = 0.38, p = 0.71).
Mean realized biomass at the end of the experiment (g dry weight) of Crassula helmsii after adding 60 propagules on the nutrient low mineral soils (a) and the nutrient rich organic soil (b) over all treatments (n = 08). With and without competition by native vegetation indicated by C+ and C−. Treatments with and without feces addition are indicated by F+ and F−, respectively
The total biomass of the native species was not increased by the nutrient enrichment from feces for both L. uniflora (p = 0.6) and P. globulifera (p = 0.3) (Fig. 5).
Discussion
Settlement of C. helmsii was very high on bare soils in both nutrient-rich and nutrient-poor treatments, but strongly regulated by the presence of native vegetation. In addition, competition with native species almost nullified growth of this invasive species in both systems. Although guanotrophication did not affect settlement, growth of C. helmsii was stimulated by bird droppings but only on bare, nutrient poor soils (Fig. 6).
Treatments including waterfowl feces and Crassula helmsii growth four weeks after start of the experiment. Upper panels: nutrient-poor, mineral sod with closed Littorella uniflora vegetation (a1) and bare sod (a2) from Huis ter Heide. Lower panel: nutrient-rich, organic sod with closed Pilularia globulifera vegetation (b1) and bare sod (b2) from Korenburgerveen
Biotic resistance to invasion by the presence of native vegetation: settlement constraints
Under absence of competitors on bare soil, C. helmsii showed high settlement and growth rates under both nutrient poor and nutrient rich conditions. This underlines the high risk of invasiveness of C. helmsii in situations when native vegetation is absent or has declined, and bare soil is present. These bare conditions can be a result of natural disturbance (e.g., drought, overgrazing), but are also created by nature managers. For example, in ecological restoration projects with large-scale dredging or topsoil removal practices to re-create nutrient-poor conditions and restore biodiversity. Ecosystem disturbance leading to bare spots, both unintended and intended, therefore results in a highly increased risk of major colonization by invasive alien species in these wetlands, as reported in previous studies (Hobbs 1989, 1991; Hobbs and Huenneke 1992; Rejmánek 1999). Brouwer et al. (2017) described that the development of already settled C. helmsii plants on the shores of moorland pools is strongly reduced by competitors (such as L. uniflora and Hypericum elodes L.). Here, we show that a closed vegetation of L. uniflora in nutrient-poor, mineral systems and P. globulifera in more nutrient-rich, organic soil conditions strongly reduces the settlement of C. helmsii, supporting our hypothesis that native species strongly increase the biotic resistance to invasions of alien plant species. Settlement of new introductions is more complicated than the growth of already settled plants because of the absence of roots and thus the lagging of nutrient uptake. The settlement of C. helmsii was clearly suppressed under dominance of native vegetation, which resulted in the absence of open niches. Our results show that P. globulifera in the nutrient-richer system is better in suppressing settlement than L. uniflora in the more nutrient poor system. L. uniflora has a rosette with an open leaf canopy, resulting in a relatively open sward structure, whereas in P. globulifera the sward is much denser. This dense vegetation probably functions as a physical barrier for settlement, effectively withholding C. helmsii propagules to reach the soil with newly formed roots. Yet, since P. globulifera lacks winter greenness, there is still a high risk that C. helmsii will be able settle in winter, partially overgrowing the native vegetation, thereby cancelling out the competition gradually each year.
Biotic resistance to invasion by the presence of native vegetation: growth constraints
In addition to affecting settlement, native species have an even stronger effect on growth of C. helmsii. The growth of this species is reduced in the presence of competitors, with both competitors showing the capability to reduce growth to near zero growth during the experiment. As L. uniflora swards are markedly less dense than P. globulifera swards, it may be expected that this should enable C. helmsii to reach higher growth rates compared to the competition with P. globulifera. However, the root system of L. uniflora, occurring under nutrient-poor conditions, is very extensive and dense, and adapted to an efficient nutrient and carbon uptake (Kuntz et al. 2014; Brouwer et al. 2017), which might still have suppressed the growth of C. helmsii by nutrient competition due to its extensive root system. This suggests that competition for nutrients is more important than competition for space and light in explaining growth suppression under these conditions.
P. globulifera forms a less deep and dense root system, and is less well adapted to grow under highly oligotrophic soil conditions (Bloemendaal and Roelofs 1988). In addition, competition for nutrients is not likely under the nutrient rich soil conditions in which P. globulifera was grown in this experiment. Here, competition for space and light might be the causal mechanism in suppressing growth of C. helmsii. The nature of competition may also result from allelopathic interaction with the competitive species besides strong competition for nutrients, space and light (Rice 2012). Denton (2003) suggest that L. uniflora might possibly suppress C. helmsii by some chemical effects. However, Brouwer et al. (2007) show improved growth of C. helmsii when mixed with L. uniflora. Our results clearly indicate competition by the latter species as the most important factor. There are no indications growth restrains from allelopathic factors of C. helmsii. Future investigations into the role of allelopathic interactions in determining competitive strength can provide more insight in the relative importance of this factor on competition between native and invasive plant species.
Role of nutrient availability; effects of guanotrophication
For bare soil conditions, growth of C. helmsii was higher under nutrient rich site conditions compared to nutrient poor conditions, similar to the studies of Hussner (2009) and Brouwer et al. (2017). Plant tissue N:P ratio suggested more P-limited, or NP co-limited biomass production of C. helmsii at the nutrient poor treatment (Koerselman and Meuleman 1996), whereas at the nutrient rich treatment tissue N:P ratio suggested more N-limited biomass production, similar to Brouwer et al. (2017) (Fig. 2). However, the availabilities of both N and P were much higher in the nutrient rich treatment than at the nutrient poor treatment.
Nutrient enrichment by waterfowl feces increased C. helmsii growth, but only on the bare, nutrient-poor soil. For the more nutrient-rich site, C. helmsii growth did not appear to be limited by nutrient availability, and growth suppression by P. globulifera must have been functioning via different mechanisms than by L. uniflora. The addition of feces on the bare, nutrient-poor soil sods appears to have removed P limitation or NP co-limitation. This stimulating effect by guanotrophication under nutrient poor conditions indicates that anthropogenic ecosystem disturbance at the landscape-scale has carry-over effects to nature reserves, by increasing the vulnerability of these ecosystems to invasive alien species. In the last decades, geese populations (A. anser, Anser albifrons Scorpoli, Branta canadensis L., and the shelduck-sheldgoose species A. aegyptiacus, among others) have increased exponentially in the Netherlands and other parts of Europe (Larsson et al. 1988; Madsen et al. 1999; Berndt et al. 2002; Rehfisch et al. 2002; Vermeersch et al. 2004; Van der Jeugd et al. 2006). Intensive land use and increased use of fertilizer increased grassland productivity, increasing overall food availability for geese (Van Eerden 1996; Fox et al. 2005; Van Eerden et al. 2005; Van der Jeugd et al. 2006). In the Netherlands, wintering geese increased by a factor of 10 between 1975 and 2014 (Van der Jeugd et al. 2006; Sovon 2015). In addition to causing eutrophication, the presence of waterfowl also increases the propagule pressure and settlement probability of C. helmsii via avian transport. Hence, increased waterfowl pressure on oligotrophic ecosystems enhances the invasiveness of C. helmsii via several mechanisms that positively influence each other. The existence of such positive feedback mechanism has also been described for other invasive alien species (e.g., Blossey and Notzold 1995; Davis 2009; Engelhardt 2011; Matzek 2011; Pasari et al. 2011) and stresses the need for policy makers to account for these landscape level interactions in nature conservancy legislation.
Implications for invasive species management
Our study shows that the presence of bare soils, functioning as empty niches, strongly creates a window of opportunity for aggressive C. helmsii colonization. In the Netherlands, these open niches often arise when former agricultural land is reclaimed for rehabilitation of nature areas. Restoration of these high production ecosystems generally takes place by removing the nutrient rich topsoil to enable the development of more biodiverse vegetation. However, when residual nutrients are retained in the system, these can facilitate the growth of C. helmsii (Dawson and Warman 1987; Dean et al. 2015; Brouwer et al. 2017; Van Kleef et al. 2017), stressing the need to maximize nutrient removal in such restoration practices. Given our findings that C. helmsii was also well able to colonize bare oligotrophic ecosystems, this can be expected to be insufficient to prevent its invasion.
We show that competition by native species vegetation highly suppresses the invasiveness of C. helmsii, suggesting that measures aimed at increasing biotic resistance of an ecosystem are likely to be effective at preventing C. helmsii dominance. Therefore, it is advisable to minimize the duration of bare soil presence after natural disturbances or as a result of management activities (Dean et al. 2015; Brouwer et al. 2017; Van Kleef et al. 2017). Given the low success levels of eradication efforts (Van der Loop et al. 2018), ecosystem-based measures focusing on low nutrient status and fast native plant recolonization, may provide a viable alternative to traditional eradication management (Van Kleef et al. 2016; Van der Loop et al. 2018). Such an approach aims at limiting settlement chance and growth conditions for C. helmsii. These measures consist of reducing nutrients on site, e.g., by removing nutrient rich top soils or limiting nutrient influx via surface water, and by the prevention of guanotrophication by water birds. A promising novel approach to boost biotic resistance is the stimulation of succession by native vegetation in newly created nature reserves or after high-disturbance nature management practices. The presence of bare soil will be limited to a shorter time frame by introducing highly competitive native plant species adapted to the specific habitat conditions (e.g., via spreading vegetative propagules or seeds). This will effectively reduce the vulnerability with respect to C. helmsii infestation. Although this prevention strategy does not guarantee that C. helmsii will not colonize the system, it may strongly hamper its invasiveness after settlement and thus reduce its ecological impact. In the Netherlands, several field experiments on cost-effectivity of system oriented management measures to increase resistance of habitats to plant invasion are currently in progress and assessments of the results will become soon available.
Most of the prevention efforts of reducing the introduction, and effects of invasive species are aimed at composing risk assessments, reducing introduction pathways and maintain an early detection and removal management.
Although these prevention strategies are important to reduce the introductions and spread of alien species, prevention can be achieved by reducing disturbances in nature areas and correct nature management as well.
References
Arts GHP, Leuven RSEW (1988) Floristic changes in shallow soft waters in relation to underlying environmental factors. Freshw Biol 20:97–111
Berndt RK, Koop B, Struwe-Juhl B (2002) Vogelwelt Schleswig-Holsteins: Band 5: Brutvogelatlas. Wachholtz, Hamburg, p 464
Bloemendaal F, Roelofs J (1988) Waterplanten en waterkwaliteit. Koninklijke Nederlandse Natuurhistorische Vereniging, Utrecht, p 189
Blossey B, Notzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887–889
Brouwer E, Denys L, Lucassen ECHET, Buiks M, Onkelinx T (2017) Competitive strength of Australian swamp stonecrop (Crassula helmsii) invading moorland pools. Aquat Invasion 12:321–331
Case TJ (1990) Invasion resistance arises in strongly interacting species-rich model competition communities. Proc Natl Acad Sci 87:9610–9614
Davis MA (2009) Invasion biology. Oxford University Press, Oxford, p 264
Dawson F, Warman E (1987) Crassula helmsii (T. Kirk) Cockayne: Is it an aggressive alien aquatic plant in Britain? Biol Conserv 42:247–272
Dean CE, Day J, Gozlan RE, Diaz A (2015) Grazing vertebrates promote invasive Swamp stonecrop (Crassula helmsii) abundance. Invasion Plant Sci Manag 8:131–138
Denton J (2013) Could shoreweed be useful for Crassula control. Conserv Land Manag 11:18–19
Denys L, Packet H, Jambon W, Scheers K (2014) Dispersal of the non-native invasive species Crassula helmsii (Crassulaceae) may involve seeds and endozoochorous transport by birds. New J Bot 4:104–106
Engelhardt K (2011) Eutrophication, aquatic. In: Simberloff D, Rejmánek M (eds) Encyclopedia of biological invasions. University of California Press, Berkeley, pp 209–213
Fox A, Madsen J, Boyd H, Kuijken E, Norriss D, Tombre I, Stroud D (2005) Effects of agricultural change on abundance, fitness components and distribution of two arctic-nesting goose populations. Glob Change Biol 11:881–893
Frazão JFT (2008) Eutrophication of Dutch softwater lakes due to the presence of waterfowl, in particular geese. Rapport 138 afdeling Aquatische Oecologie & Milieubiologie, Radboud University Nijmegen, pp 26–44
Funk JL, Cleland EE, Suding KN, Zavaleta ES (2008) Restoration through reassembly: plant traits and invasion resistance. Trends Ecol Evol 23:695–703
Grasshof K, Johannsen H (1972) New sensitive and direct method for automatic determination of ammonia in seawater. J Du Conseil 34:516–521. https://doi.org/10.1093/icesjms/34.3.516
Güsewell S (2004) N:P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–266
Hobbs RJ (1989) The nature and effects of disturbance relative to invasions. In: Drake J, Mooney H, Di Castri F, Groves R, Kruger F, Rejmanek M, Williamson M (eds) Biological invasions: a global perspective. Wiley, Chichester, pp 389–405
Hobbs RJ (1991) Disturbance of a precursor to weed invasion in native vegetation. In: Plant protection quarterly, Australia pp 99–104
Hobbs RJ, Huenneke LF (1992) Disturbance, diversity, and invasion: implications for conservation. Conserv Biol 6:324–337
Hussner A (2009) Growth and photosynthesis of four invasive aquatic plant species in Europe. Weed Res 49(5):506–515
Hussner A, Stiers I, Verhofstad M, Bakker E, Grutters B, Haury J, van Valkenburg J, Brundu G, Newman J, Clayton J (2017) Management and control methods of invasive alien freshwater aquatic plants: a review. Aquat Bot 136:112–137
Kamphake LJ, Hannah SA, Cohen JM (1967) Automated analysis for nitrate by hydrazine reduction. Water Res 1:205–216. https://doi.org/10.1016/0043-1354(67)90011-5
Kennedy TA, Naeem S, Howe KM, Knops JM, Tilman D, Reich P (2002) Biodiversity as a barrier to ecological invasion. Nature 417:636–638
Koerselman W, Meuleman AF (1996) The vegetation N:P ratio: a new tool to detect the nature of nutrient limitation. J Appl Ecol 33:1441–1450
Kuntz K, Heidbüchel P, Hussner A (2014) Effects of water nutrients on regeneration capacity of submerged aquatic plant fragments. Annales de Limnologie Int J Limnol 50(2):155–162
Larsson K, Forslund P, Gustafsson L, Ebbinge BS (1988) From the high Arctic to the Baltic: the successful establishment of a Barnacle Goose Branta leucopsis population on Gotland, Sweden. Ornis Scand 3:182–189
Leach J, Dawson H (1999) Crassula helmsii in the British Isles—an unwelcome invader. Brit Wildl 10:234–239
Levine JM, Vila M, Antonio CM, Dukes JS, Grigulis K, Lavorel S (2003) Mechanisms underlying the impacts of exotic plant invasions. Proc R Soc Lond B Biol Sci 270:775–781
Liu Y, Hefting MM, Verhoeven JT, Klaassen M (2014) Nutrient release characteristics from droppings of grass-foraging waterfowl (Anser brachyrhynchus) roosting in aquatic habitats. Ecohydrology 7(4):1216–1222
Madsen J, Cracknell G, Fox A (1999) Goose populations of the western Palearctic. Wetlands International Publication 48. Wetlands International, Wageningen
Matzek V (2011) Superior performance and nutrient-use efficiency of invasive plants over non-invasive congeners in a resource-limited environment. Biol Invasion 13:3005–3014
Moulton MP, Pimm SL (1983) The introduced Hawaiian avifauna: biogeographic evidence for competition. Am Nat 121:669–690
Newman JR (2013) CEH Information Sheet 12: Crassula helmsii, Australian Swamp Stonecrop. Centre for Ecology & Hydrology, CAPM, CEH Wallingford, Crowmarsh Gifford, Wallingford, Oxon, p 1
OEPP/EPPO (2007) Data sheets on quarantine pests. Crassula helmsii. EPPO Eur Mediterr Plant Prot Organ 37:225–229
Pasari J, Selmants P, Young H, O’Leary J, Zavaleta E (2011) Nitrogen enrichment. In: Simberloff D, Rejmánek M (eds) Encyclopedia of biological invasions. University of California Press, Berkeley, pp 488–492
Prinz M, Peppler-Lisbach C, Weidhüner A, Freund H (2019) Crassula helmsii (T. Kirk) Cockayne: Standortansprüche, Verbreitung und Vergesellschaftung eines invasiven Neophyten auf Norderney. Tuexenia 39:267–286
Rehfisch MM, Austin GE, Holloway SJ, Allan JR, O’Connell M (2002) An approach to the assessment of change in the numbers of Canada Geese Branta canadensis and Greylag Geese Anser anser in southern Britain. Bird Study 49:50–59
Rejmánek M (1999) Invasive plant species and invasible ecosystems. In: Sandlund OT, Schei PJ, Viken A (eds) Invasive species and biodiversity management. Kluwer, Dordrecht, pp 79–102
Rice EL (2012) Allelopathy. Academic Press, New York, p 421
Robert H, Lafontaine RM, Beudels-Jamar RC, Delsinne T (2013) Risk analysis of the Australian swamp stonecrop, Crassula helmsii (Kirk) Cockayne—risk analysis report of non-native organisms in Belgium. Royal Belgian Institute of Natural Sciences for the Federal Public Service Health, Food chain safety and Environment, Brussels, pp 13–26
Smith T, Buckley P (2020) Biological Flora of the British Isles: Crassula helmsii. J Ecol 108(2):797–813
Sovon (2015) Aantallen ganzen in Nederland Sovon Vogelonderzoek Nederland Nijmegen. https://www.sovon.nl/nl/content/aantallen-ganzen-nederland. Accessed 13 Feb 2020
Team RC (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. 2012. http://www.R-project.org. Accessed 13 Feb 2020
Van der Jeugd HP, Voslamber B, van Turnhout C, Sierdsema H, Feige N, Nienhuis J, Koffijberg K (2006) Overzomerende ganzen in Nederland: grenzen aan de groei? Sovon-onderzoeksrapport 2006/02. SOVON Vogelonderzoek Nederland, Beek-Ubbergen
Van der Loop JMM, de Hoop L, van Kleef HH, Leuven RSEW (2018) Effectiveness of eradication measures for the invasive Australian swamp stonecrop Crassula helmsii. Manag Biol Invasion 3(9):343–355
Van Eerden M (1996) The response of Anatidae to changes in agricultural practice: long-term shifts in the carrying capacity of wintering waterfowl. Gibier Faune Sauvage. Game Wildl 13:681–706
Van Eerden MR, Drent RH, Stahl J, Bakker JP (2005) Connecting seas: western Palaearctic continental flyway for water birds in the perspective of changing land use and climate. Glob Change Biol 11:894–908
Van Kleef HH, Brouwer E, van der Loop JMM, Buiks M, Lucassen ECHET (2017) Systeemgerichte bestrijding van watercrassula. Stichting Bargerveen Nijmegen, 89 p
Van Kleef HH, van der Loop JMM, Nyssen BJM, Brouwer E (2016) Systeemgericht beheer als duurzame oplossing tegen invasieve exoten. De Levende Natuur 117(6):251–255
Vermeersch G, Anselin A, Devos K, Herremans M, Stevens J, Gabriëls J, van der Krieken B (2004) Atlas van de Vlaamse broedvogels 2000–2002. Mededelingen van het Instituut voor Natuurbehoud 23:1–496
Acknowledgements
This study was financially supported by the Programme Kennisnetwerk Ontwikkeling en Beheer Natuurkwaliteit (O + BN) of the Dutch Ministry of Agriculture, Nature and Food quality (LNV) and the Province of Noord-Brabant. Furthermore, we are grateful to nature managers of our research sites and volunteers from Natuurmonumenten for their support. The authors would like to thank the staff of Stichting Bargerveen and Radboud University for their advice and technical assistance during the experiment and harvest. We thank Conor Strong MSc, the associate editor Dr. Deah Lieurance and two anonymous reviewers for their constructive comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van der Loop, J.M.M., Tjampens, J., Vogels, J.J. et al. Reducing nutrient availability and enhancing biotic resistance limits settlement and growth of the invasive Australian swamp stonecrop ( Crassula helmsii ) . Biol Invasions 22, 3391–3402 (2020). https://doi.org/10.1007/s10530-020-02327-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02327-0