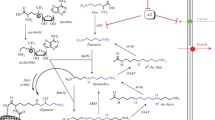
We studied the effects of some nitrogen-containing, heterocyclic, and cyclic compounds on the rate of oxidative deamination of polyamines and putrescine in tissues with a high proliferation rate. For this purpose, the specific activities of the main enzymes of polyamine oxidative degradation — spermine oxidase (SMO), polyamine oxidase (PAO), and diamine oxidase (DAO) were determined using a cell-free test system from regenerating rat liver. The compounds methyl 2-(5-formylfuran-2-yl)benzoate and 2,7-bis-[2-(diethylamino)ethoxy]-9H-fluoren-9-one (and in the form of dihydrochloride) showed mainly activating effect on oxidative degradation of putrescine, spermidine, and spermine, which indirectly indicates their antiproliferative effect. Nitrogen-free compounds inhibited this process, thus exhibiting potentially carcinogenic properties. Correlations were calculated for activity of DAO, PAO, and SMO with 5 topological indices: Wiener (W), Rouvray (R), Balaban (J) in the Trinaistich modification, detour (Ip), and electropy (Ie). The highest dependence was noted for DAO and the Balaban index (R=-0.55), for PAO and the detour index (R=0.78), and for SMO and the electropy index (R=0.53). The remaining dependencies showed insignificant correlation strength.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Polyamines Methods and Protocols. Alcázar R, Tiburcio AF, eds. Humana Press, 2018.
Miller-Fleming L, Olin-Sandoval V, Campbell K, Ralser M. Remaining mysteries of molecular biology: the role of polyamines in the cell. J. Mol. Biol. 2015;427(21):3389-3406. https://doi.org/10.1016/j.jmb.2015.06.020
Sánchez-Jiménez F, Medina MÁ, Villalobos-Rueda L, Urdiales JL. Polyamines in mammalian pathophysiology. Cell. Mol. Life Sci. 2019;76(20):3987-4008. https://doi.org/10.1007/s00018-019-03196-0
Murray-Stewart TR, Woster PM, Casero RA Jr. Targeting polyamine metabolism for cancer therapy and prevention. Biochem. J. 2016;473(19):2937-2953. https://doi.org/10.1042/BCJ20160383
Syatkin SP, Berezov TT. Metabolism of polyamines in malignant tumors.Vestn. Akad. Med. Nauk USSR. 1982;(3):10-21. Russian.
Li M, Wang Y, Ge C, Chang L, Wang C, Tian Z, Wang S, Dai F, Zhao L, Xie S. Synthesis and biological evaluation of novel alkylated polyamine analogues as potential anticancer agents. Eur. J. Med. Chem. 2018;143:1732-1743. https://doi.org/10.1016/j.ejmech.2017.10.069
Volkov SV, Kutyakov SV, Levov AN, Polyakova EI, Anh LT, Soldatova S.A.1, Soldatenkov A.T.1, Terentiev P.B. Reaction of 3-benzoyl-1-methyl-4-phenyl-γ-piperidol with arylamines and arylhydrazines. Synthesis of 3-arylamino-1-oxo-1-phenylpropanes and 1,3 diarylpyrazoles and their fragmentation under electron impact. Chem. Heterocycl. Compd. 2007;43(4):445-453. https://doi.org/10.1007/s10593-007-0064-3
Michalopoulos GK. Liver regeneration after partial hepatectomy: critical analysis of mechanistic dilemmas. Am. J. Pathol. 2010;176(1):2-13. https://doi.org/10.2353/ajpath.2010.090675
Syatkin SP. Modified method for determining protein in samples with high concentrations of lipo- and glycoproteins. Vopr. Med. Khimii. 1981;27(1):136-138. Russian.
Hu QN, Liang YZ, Fang KT. The matrix expression, topological index and atomic attribute of molecular topological structure. Journal of Data Science: JDS. 2003;1:361-389. https://doi.org/10.6339/JDS.2003.01(4).172
Lisitskaya KV, Ivanov AV, Shishkin SS, Sokueva NA, Malysheva YG3, Syatkin SP. Identification of the functional activity of synthetic polyamine analogues using a biotest system based on highly proliferating cultured human cells. Appl. Biochem. Microbiol. 2013;49(2):100-105. https://doi.org/10.1134/S0003683813020075
Syatkin SP, Neborak EV, Khlebnikov AI, Komarova MV, Shevkun NA, Kravtsov EG, Blagonravov ML, Agostinelli E. The investigation of structure-activity relationship of polyamine-targeted synthetic compounds from different chemical groups. Amino Acids. 2020;52(2):199-211. https://doi.org/10.1007/s00726-019-02778-3
Khlebnikov AI, Schepetkin IA, Kirpotina LN, Quinn MT. Computational structure-activity relationship analysis of non-peptide inducers of macrophage tumor necrosis factor-alpha production. Bioorg. Med. Chem. 2008;16(20):9302-9012. https://doi.org/10.1016/j.bmc.2008.08.078
Cervelli M, Salvi D, Polticelli F, Amendola R, Mariottini P. Structure-function relationships in the evolutionary framework of spermine oxidase. J. Mol. Evol. 2013;76(6):365-370. https://doi.org/10.1007/s00239-013-9570-3
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 177, No. 3, pp. 291-297, March, 2024
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Syatkin, S.P., Blagonravov, M.L., Hilal, A. et al. Influence of Some Heterocyclic, Cyclic, and Nitrogen-Containing Compounds on Oxidative Deamination of Polyamines in a Cell-Free Test System. Bull Exp Biol Med 177, 307–312 (2024). https://doi.org/10.1007/s10517-024-06179-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-024-06179-9