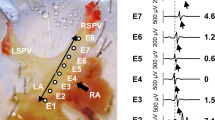
A number of pharmacological drugs have side effects that contribute to the occurrence of atrial fibrillation, the most common type of cardiac rhythm disorders. The clinical use of antihistamines is widespread; however, information regarding their anti- and/or proarrhythmic effects is contradictory. In this work, we studied the effects and mechanisms of the potential proarrhythmic action of the first-generation antihistamine chloropyramine (Suprastin) in the atrial myocardium and pulmonary vein (PV) myocardial tissue. In PV, chloropyramine caused depolarization of the resting potential and led to reduction of excitation wave conduction. These effects are likely due to suppression of the inward rectifier potassium current (IK1). In presence of epinephrine, chloropyramine induced spontaneous automaticity in the PV and could not be suppressed by atrial pacing. Chloropyramine change functional characteristics of PV and contribute to occurrence of atrial fibrillation. It should be noted that chloropyramine does not provoke atrial tachyarrhythmias, but create conditions for their occurrence during physical exercise and sympathetic stimulation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ball J, Carrington MJ, McMurray JJ, Stewart S. Atrial fibrillation: profile and burden of an evolving epidemic in the 21st century. Int. J. Cardiol. 2013;167(5):1807-1824. https://doi.org/10.1016/j.ijcard.2012.12.093
Kornej J, Börschel CS, Benjamin EJ, Schnabel RB. Epidemiology of Atrial Fibrillation in the 21st Century: Novel Methods and New Insights. Circ. Res. 2020;127(1):4-20. https://doi.org/10.1161/CIRCRESAHA.120.316340
Kaakeh Y, Overholser BR, Lopshire JC, Tisdale JE. Drug-induced atrial fibrillation. Drugs. 2012;72(12):1617-1630. https://doi.org/10.2165/11633140-000000000-00000
van der Hooft CS, Heeringa J, van G, Kors JA, Kingma JH, Stricker BH. Drug-induced atrial fibrillation. J. Am. Coll. Cardiol. 2004;44(11):2117-2124. https://doi.org/10.1016/j.jacc.2004.08.053
Tamargo J, Caballero R, Delpón E. Drug-induced atrial fibrillation. Expert Opin. Drug Saf. 2012;11(4):615-634. https://doi.org/10.1517/14740338.2012.698609
Maciag A, Farkowski MM, Chwyczko T, Beckowski M, Syska P, Kowalik I, Pytkowski M, Wozniak J, Dabrowski R, Szwed H. Efficacy and safety of antazoline in the rapid cardioversion of paroxysmal atrial fibrillation (the AnPAF Study). Europace. 2017;19(10):1637-1642. https://doi.org/10.1093/europace/euw384
Farkowski MM, Maciąg A, Żurawska M, Kołakowski K, Gardziejczyk P, Kowalik I, Szwed H, Pytkowski M. Rapid pharmacological cardioversion of recent-onset atrial fibrillation using antazoline in elderly patients. Pol. Arch. Intern. Med. 2022;132(1):16120. https://doi.org/10.20452/pamw
Ösken A, Zehir R, Ösken S, Yaylacı S, Aydın E, Şahinkuş S, Can Y. Cetirizine-Induced Atrial Fibrillation. Arch. Med. Health Sci. 2016;4(2):258-260. https://doi.org/10.4103/2321-4848.196192
Ali Z, Ismail M, Khan F, Sajid H. Association of H1-antihistamines with torsade de pointes: a pharmacovigilance study of the food and drug administration adverse event reporting system. Expert Opin. Drug Saf. 2021;20(1):101-107. https://doi.org/10.1080/14740338.2021.1846717
Uvelin A, Hajduković D, Vrsajkov V, Kolak R, Lazukić A, Vicković S, Gojković Z. A case of recurrent arrhythmia in an acute pancreatitis patient — pathophysiological explanation using shortage of “repolarization reserve”. Acta Clin. Croat. 2013;52(4):515-522.
Budihna M, Drevensek G, Burjak M, Kocijancic S. Effects of chloropyramine and famotidine on postischaemic and posthypoxic myocardial damage in isolated rat hearts. Pflugers Arch. 1996;431(6, Suppl 2):R217-R218. https://doi.org/10.1007/BF02346346
Vedder VL, Reinberger T, Haider SMI, Eichelmann L, Odenthal N, Abdelilah-Seyfried S, Aherrahrou Z, Breuer M, Erdmann J. pyHeart4Fish: Chamber-specific heart phenotype quantification of zebrafish in high-content screens. Front. Cell Dev. Biol. 2023;11:1143852. https://doi.org/10.3389/fcell.2023.1143852
Masani F. Node-like cells in the myocardial layer of the pulmonary vein of rats: an ultrastructural study. J. Anat. 1986;145:133-142.
Doisne N, Maupoil V, Cosnay P, Findlay I. Catecholaminergic automatic activity in the rat pulmonary vein: electrophysiological differences between cardiac muscle in the left atrium and pulmonary vein. Am. J. Physiol. Heart Circ. Physiol. 2009;297(1):H102-H108. https://doi.org/10.1152/ajpheart.00256.2009
Egorov YV, Rosenshtraukh LV, Glukhov AV. Arrhythmogenic Interaction Between Sympathetic Tone and Mechanical Stretch in Rat Pulmonary Vein Myocardium. Front. Physiol. 2020;11:237. https://doi.org/10.3389/fphys.2020.00237
Haïssaguerre M, Jaïs P, Shah DC, Takahashi A, Hocini M, Quiniou G, Garrigue S, Le Mouroux A, Le Métayer P, Clémenty J. Spontaneous initiation of atrial fibrillation by ectopic beats originating in the pulmonary veins. N. Engl. J. Med. 1998;339(10):659-666. https://doi.org/10.1056/NEJM199809033391003
Kuzmin VS, Ivanova AD, Filatova TS, Pustovit KB, Kobylina AA, Atkinson AJ, Petkova M, Voronkov YI, Abramochkin DV, Dobrzynski H. Micro-RNA 133a-3p induces repolarization abnormalities in atrial myocardium and modulates ventricular electrophysiology affecting ICa,L and Ito currents. Eur. J. Pharmacol. 2021;908:174369. https://doi.org/10.1016/j.ejphar.2021.174369
Isenberg G, Klockner U. Calcium tolerant ventricular myocytes prepared by preincubation in a “KB medium”. Pflugers Arch. 1982;395(1):6-18. https://doi.org/10.1007/BF00584963
Ivanova AD, Filatova TS, Abramochkin DV, Atkinson A, Dobrzynski H, Kokaeva ZG, Merzlyak EM, Pustovit KB, Kuzmin VS. Attenuation of inward rectifier potassium current contributes to the α1-adrenergic receptor-induced proarrhythmicity in the caval vein myocardium. Acta Physiol. (Oxf). 2021;231(4):e13597. https://doi.org/10.1111/apha.13597
Tsuneoka Y, Irie M, Tanaka Y, Sugimoto T, Kobayashi Y, Kusakabe T, Kato K, Hamaguchi S, Namekata I, Tanaka H. Permissive role of reduced inwardly-rectifying potassium current density in the automaticity of the guinea pig pulmonary vein myocardium. J. Pharmacol. Sci. 2017;133(4):195-202. https://doi.org/10.1016/j.jphs.2016.12.006
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 176, No. 12, pp. 734-740, December, 2023
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Egorov, Y.V., Filatova, T.S., Abramov, A.A. et al. Suprastin (Chloropyramine) Causes Proarrhythmic Deterioration of Excitation Conduction, Depolarization and Potentiates Adrenergic Automaticity in the Pulmonary Veins Myocardium. Bull Exp Biol Med 176, 761–766 (2024). https://doi.org/10.1007/s10517-024-06104-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-024-06104-0