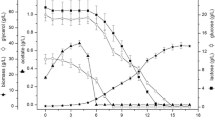
A recombinant form of pneumolysin from Streptococcus pneumoniae was obtained. By using Vector NTI Advance 11.0 bioinformatic analysis software, specific primers were designed in order to amplify the genome fragment of strain No. 3358 S. pneumoniae serotype 19F containing the nucleotide sequence encoding the full-length pneumolysin protein. A PCR product with a molecular weight corresponding to the nucleotide sequence of the S. pneumoniae genome fragment encoding the full-length pneumolysin was obtained. An expression system for recombinant pneumolysin in E. coli was constructed. Sequencing confirmed the identity of the inserted nucleotide sequence encoding the full-length recombinant pneumolysin synthesized in E. coli M15 strain. Purification of the recombinant protein was performed by affinity chromatography using Ni-Sepharose in 8 M urea buffer solution. Confirmation of the recombinant protein was performed by immunoblotting with monoclonal antibodies to pneumolysin.
Article PDF
Similar content being viewed by others

Avoid common mistakes on your manuscript.
References
Brooks LRK, Mias GI. Streptococcus pneumoniae’s virulence and host immunity: aging, diagnostics, and prevention. Front. Immunol. 2018;9:1366. https://doi.org/10.3389/fimmu.2018.01366
Weiser JN, Ferreira DM, Paton JC. Streptococcus pneumoniae: transmission, colonization and invasion. Nat. Rev. Microbiol. 2018;16(6):355-367. https://doi.org/10.1038/s41579-018-0001-8
Briles DE, Paton JC, Mukerji R, Swiatlo E, Crain MJ. Pneumococcal vaccines. Microbiol. Spectr. 2019;7(6). https://doi.org/10.1128/microbiolspec.GPP3-0028-2018
Wagner A, Weinberger B. Vaccines to prevent infectious diseases in the older population: immunological challenges and future perspectives. Front. Immunol. 2020;11:717. https://doi.org/10.3389/fimmu.2020.00717
Masomian M, Ahmad Z, Gew LT, Poh CL. Development of next generation streptococcus pneumoniae vaccines conferring broad protection. Vaccines (Basel). 2020;8(1):132. https://doi.org/10.3390/vaccines8010132
Nishimoto AT, Rosch JW, Tuomanen EI. Pneumolysin: pathogenesis and therapeutic target. Front. Microbiol. 2020;11:1543. https://doi.org/10.3389/fmicb.2020.01543
Vorobyev DS, Semenova IB, Volokh YuV, Romanenko EE, Baturo AP, Mikhailova NA. Properties of native protein-containing antigens of Streptococcus pneumoniae. Zh. Mikrobiol. Epidemiol. Immunol. 2019;(1):22-28. Russian. https://doi.org/10.36233/0372-9311-2019-1-22-28
Eutsey RA, Powell E, Dordel J, Salter SJ, Clark TA, Korlach J, Ehrlich GD, Hiller NL. Genetic stabilization of the drug-resistant PMEN1 Pneumococcus lineage by its distinctive DpnIII restriction-modification system. mBio. 2015;6(3):e00173. https://doi.org/10.1128/mBio.00173-15
Kaloshin AA, Gatypova EV, Mikhailova NA. Obtaining recombinant forms of the outer membrane protein F of pseudomonas aeruginosa and assessment of their immunogenic properties. Appl. Biochem. Microbiol. 2011;47(8):780-788. https://doi.org/10.1134/S0003683811080060
Petukhova ES, Vorobyev DS, Sidorov AV, Semenova IB, Volokh YV, Leonova AY, Sidorova AV, Mikhailova NA. Immunization with recombinant pneumolysin induces the production of antibodies and protects mice in a model of systemic infection caused by Streptococcus pneumoniae. Bull. Exp. Biol. Med. 2020;168(4):485-487. https://doi.org/10.1007/s10517-020-04736-6
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 174, No. 12, pp. 723-727, December, 2022
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Vorobyev, D.S., Sidorov, A.V., Kaloshin, A.A. et al. Preparation a Recombinant Form of Pneumolysin Protein from Streptococcus pneumoniae. Bull Exp Biol Med 174, 749–753 (2023). https://doi.org/10.1007/s10517-023-05785-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-023-05785-3