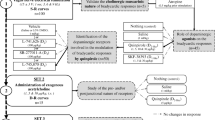
Stimulation of the serotoninergic system (5-hydroxytryptophan, 50 mg/kg; fluoxetine, 3 mg/kg) induced a significant increase in HR and a reduction in the amplitude of all waves of the heart rhythm variability. Stimulation of the dopaminergic system (L-DOPA and amantadine, 20 mg/kg each) resulted in a moderate increase in HR and amplitudes of low-frequency (LF) and very-low-frequency (VLF) waves of the heart rhythm variability. Successive blockade of nicotinic (hexamethonium, 7 mg/kg) and muscarinic cholinergic receptors (atropine, 1 mg/kg) leads to a significant decrease in the variability of cardiointervals (almost to complete levelling) both under control conditions and after stimulation of the neurotransmitter systems. Serotonin receptor blockade (promethazine, 2 mg/kg) did not affect HR, but reduced the amplitude of LF- and VLF-waves. Under conditions of serotoninergic system stimulation, the blockade of serotonin receptors was followed by a significant HR acceleration without changes in heart rhythm variability; blockade of dopamine receptors (sulpiride, 1 mg/kg) induced HR acceleration and increase in the amplitude of LF- and VLF-waves; blockade of dopamine receptors under conditions of dopamine system stimulation was followed by a significant increase in HR and a decrease in the amplitude of all waves of the heart rhythm variability. It can be hypothesized that serotonin- and dopaminergic systems affect the heart rhythm via cardiomyocyte receptors and via modulation of activity of the adrenergic and cholinergic systems. The effects of serotonin- and dopaminergic systems can be considered as synergic in the CNS, and antagonistic at the periphery.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baevsky RM, Ivanov GG, Chireykin LV, Gavrilushkin AP, Dovgalevsky PYa, Kukushkin YuA, Mironova TF, Prilutsky DA, Semenov AV, Fedorov VF, Fleishman AN, Medvedev MM. Analysis of heart rate variability using different electrocardiographic systems (part 1). Vestn. Aritmol. 2002;(24):65-86. Russian.
Belova EI. Fundamentals of Neuropharmacology. Moscow, 2006. Russian.
Bilalova GA, Kazanchikova LM, Zefirov TL, Sitdikov FG. Inotropic effect of dopamine on rat heart during postnatal ontogeny. Bull. Exp. Biol. Med. 2013;156(2):173-176.
Kuryanova EV, Zhukova YD, Tryasuchev AV, Horst NA. Influence of scopolamine, galantamine and their combination with hexametonium and atropine on the spectral characteristics of heart rhythm of nonlinear rats. Sib. Nauch. Med. Zh. 2016;36(3):5-12. Russian.
Kur’yanova EV, Tryasuchev AV, Stupin VO, Teplyi DL. Effect of Stimulation of Neurotransmitter Systems on Heart Rate Variability and β-Adrenergic Responsiveness of Erythrocytes in Outbred Rats. Bull. Exp. Biol. Med. 2017;163(1):31-36. doi: https://doi.org/10.1007/s10517-017-3731-0
Lychkova AE. Serotonergic regulation of the cardiovascular and bronchopulmonary systems. Moscow, 2012.
Nadeev AD, Zharkikh IL, Avdonin PV, Goncharov NV. Serotonin and Its Receptors in the Cardiovascular System. Eksp. Klin. Farmakol. 2014;77(5):32-37. Russian.
Nigmatullina RR, Matveeva VL, Chibireva MD. The influence of selective serotonin re-uptake inhibitor fluoxetine on inotropic function of myocardium in the ontogenesis of rats. Ross. Fiziol. Zh. 2014;100(3):348-359. Russian.
Sveshnikov DS, Kuchuk AV, Smirnov VM, Cherepanova GV. Serotonergic mechanisms of regulation of the systemic circulation vessels lumen. Kazan. Med. Zh. 2016;97(1):89-94. Russian.
Timofeeva OP, Vdovichenko ND, Kuznetsov SV. Effect of change in activity level of catecholaminergic systems on motor, respiratory, and cardiac activities in rat embryos. Zh. Evol. Biokhim. Fiziol. 2012;48(3):258-267. Russian.
Bhaskaran S, Zaluski J, Banes-Berceli A. Molecular interactions of serotonin (5-HT) and endothelin-1 in vascular smooth muscle cells: in vitro and ex vivo analyses. Am. J. Physiol. Cell Physiol. 2014;306(2):C143-C151.
Dalton DW, Feniuk W, Humphrey PP. An investigation into the mechanisms of the cardiovascular effects of 5-hydroxytryptamine in conscious normotensive and DOCA-salt hypertensive rats. J. Auton. Pharmacol. 1996;6(3):219-228.
Henze M, Tiniakov R, Samarel A, Holmes E, Scrogin K. Chronic fluoxetine reduces autonomic control of cardiac rhythms in rats with congestive heart failure. Am. J. Physiol. Heart Circ. Physiol. 2013;304(3):H444-H454.
Kubo T, Yue JL, Goshima Y, Nakamura S, Misu Y. Evidence for L-dopa systems responsible for cardiovascular control in the nucleus tractus solitarii of the rat. Neurosci. Lett. 1992;140(2):153-156.
Spasojevic N, Gavrilovic L, Kovacevic I, Dronjak S. Effects of antidepressants maprotiline and fluxilan on sympathoadrenomedullary system in stressed rats. Auton. Neurosci. 2009;145(1-2):104-107.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Byulleten’ Eksperimental’noi Biologii i Meditsiny, Vol. 168, No. 8, pp. 142-148, August, 2019
Rights and permissions
About this article
Cite this article
Kur’yanova, E.V., Stupin, V.O., Tryasuchev, A.V. et al. Effects of Blockage of Peripheral Choline, Serotonin, and Dopamine Receptors on Heart Rhythm Variability in Rats under Conditions of Stimulation of Neurotransmitter Systems. Bull Exp Biol Med 168, 193–198 (2019). https://doi.org/10.1007/s10517-019-04673-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10517-019-04673-z