Abstract
We tested apoptosis levels in in vitro irradiated T-lymphocytes from breast cancer (BC) patients with radiotherapy-induced late effects. Previous results reported in the literature were revised. We also examined the effect of TP53 Arg72Pro polymorphism on irradiation-induced apoptosis (IA). Twenty BC patients, ten with fibrosis and/or telangiectasias and ten matched controls with no late reactions, were selected from those receiving radiotherapy between 1993 and 2007. All patients were followed-up at least 6 years after radiotherapy. Using the combination of both CD3 and CD8 antibodies the in vitro IA was measured in CD3, CD8 and CD4 T-lymphocytes, and CD8 natural killer lymphocytes (CD8 NK) by flow cytometry. The TP53 Arg72Pro genotype was determined by sequencing. Patients with late radiotherapy toxicity showed less IA for all T-lymphocytes except for the CD8 NK. CD8 NK showed the highest spontaneous apoptosis and the lowest IA. IA in patients with toxicity appears to be lower than the control patients only in TP53 Arg/Arg patients (P = 0.077). This difference was not present in patients carrying at least one Pro allele (P = 0.8266). Our data indicate that late side effects induced by radiotherapy of BC are associated to low levels of IA. CD8 NK cells have a different response to in vitro irradiation compared to CD8 T-lymphocytes. It would be advisable to distinguish the CD8 NK lymphocytes from the pool of CD8+ lymphocytes in IA assays using CD8+ cells. Our data suggest that the 72Pro TP53 allele may influence the IA of patients with radiotherapy toxicity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The success of radiotherapy in eradicating a tumour depends on the total radiation dose given, but the tolerance of the normal tissues surrounding the tumour limits this dose [1]. The majority of patients tolerate well standard radiation therapy, but up to 10 % of patients suffer from acute to life threatening adverse effects arising from the intrinsic sensitivity of normal tissue [2]. As there is a direct relation between radiation dose and tumour control, the development of side effects in a minority limits the dose that can be safely prescribed to the majority of patients [1, 3]. Furthermore, radiotherapy side-effects impact negatively on quality-of-life [4].
Development of a test to predict side effects should enable individualized radiation dose prescription. It would increase cancer cure, while reducing the number of survivors suffering from the consequences of treatment [5]. Since the early 1990s, various cell-based in vitro assays have been developed to quantify cellular radiosensitivity and predict the outcome after radiotherapy [5]. These studies showed a relationship between the biomarkers measured and normal tissue toxicity, although, in general, cell-based assays lack the sensitivity, specificity and reproducibility required for a routine clinical test [1, 5, 6].
Crompton et al. [7] reported lower levels of irradiation-induced apoptosis (IA), measured by flow cytometry on CD4 and CD8 cells, in patients with severe reactions to radiotherapy when compared to healthy and cancer patients who displayed normal toxicity. In a prospective study of 399 patients treated by radiotherapy for different cancer types, Ozahin et al. [8] demonstrated that a lower CD4 or CD8 apoptosis correlated to higher risk of late adverse effects. Recently, other authors have also shown that patients with localized cervical carcinoma or advanced head and neck cancer and late toxicity induced by radiotherapy displayed a low apoptotic response compared with patients with normal tissue response [9, 10].
One of the most important apoptotic regulatory proteins is the tumour suppressor protein p53 (encoded by TP53 gene). Acting as a transcription factor, p53 responds to diverse cellular stresses to regulate cell cycle arrest, apoptosis, senescence, and DNA repair (reviewed by Riley et al. [11]). TP53 has a common single-nucleotide polymorphism (SNP), that results in either proline or arginine at amino-acid position 72 (Arg72Pro) (dbSNP: rs1042522) in the p53 protein. This polymorphism is located in a p53 proline-rich domain, which is necessary for the protein to fully induce apoptosis (reviewed by Whibley et al. [12]). Some evidences support that these two p53 variants exhibit different biochemical properties, one of which is that p53-72Arg is a more efficient inducer of apoptosis than p53-72Pro [12, 13].
In our study we tested in ten breast cancer (BC) patients with fibrosis or/and telangiectasias and 10 patients without late lesions induced by radiotherapy: (1) the levels of lymphocyte apoptosis induced by in vitro irradiation using the combination of both CD3 and CD8 antibodies to specifically identify T-lymphocytes; and (2) the effect on the IA of the TP53 Arg72Pro polymorphism. We also reviewed the previous studies reported in the literature.
Patients and methods
Patients
The late toxicity after radiotherapy was reviewed in the patient records treated with radiation for BC between 1993 and 2007. About one hundred patients per year received radiotherapy during this period. After breast-conserving surgery, the patients received adjuvant systemic treatment (chemotherapy, hormone therapy or combined). The patients received a standard breast irradiation treatment with two tangential fields, each one with a wedge: a lateral and a medial. Planning was based on 2D simulation, including verification and quality assurance. The patients were given whole breast radiotherapy with conventional fractionation, 50 Gy, 2 Gy/fraction with 60Co unit. Treatment added a tumorectomy electron boost (9–16 MeV) from a lineal accelerator, with doses ranging from 14 to 20 Gy. The dose was 20 Gy at 85 % isodose. All patients have been followed-up at least 6 years after radiotherapy (6–19 years). The occurrence of acute and late effects of radiotherapy were monitored and documented by the physician during the standard patient follow-up along and after radiotherapy using Radiation Therapy Oncology Group toxicity grading system.
For the case–control study it was possible to recruit ten BC patients with fibrosis and/or telangiectasias (Table 1). Each case was matched with one control that showed no late change in breast appearance. There is no statistically significant difference among the two groups of patients for treatment, demographic and tumour characteristics (Table 2). Supplementary Table S1 shows the tumour staging for the ten BC patients with late toxicity and for the ten control patients. Three BC patients with late toxicity had criteria for BRCA1/BRCA2 analysis, and no mutation was identified (Supplementary Table S1). The ethical committee of the hospital approved the study and all women participating signed an informed consent.
Radiation-induced apoptosis assay
The protocol of IA assay was performed as previously reported [7, 14] with some modifications (Supplementary Materials). Briefly, two fresh blood cultures, in RPMI 1640 supplemented with 20 % foetal bovine serum, were set up for each patient. Immediately after, one was treated with 8 Gy at room temperature at a dose rate of 3 Gy/min with a 6 MV photon beam. Both cultures were then placed into a 37 °C incubator with 5 % CO2. After 48 h of incubation, the cell pellet was labelled with FITC-conjugated anti-CD8 and APC-conjugated anti-CD3 monoclonal antibodies. The erythrocytes were lysed and the cell pellet was stained with propidium iodide and treated with RNase. The cells were subsequently examined using a FACScan flow cytometer. Data from at least 10,000 lymphocytes per culture were acquired.
Apoptosis was measured in the following cell types: (a) total T-lymphocytes (CD3+); (b) CD8 T-lymphocytes (CD3+ CD8+); (c) CD4 T-lymphocytes (CD3+ CD8−) and; (d) CD8 subset of natural killer (NK) lymphocytes (CD3− CD8+).
DNA genotyping
The TP53 Arg72Pro SNP: c.215G > C was genotyped by Sanger sequencing using BigDye Terminator v3.1 Cycle Sequencing Kit, on a 3130xl Genetic Analyzer. PCR amplification was performed using primers designed to include the SNP: 5′-CCTGGTCCTCTGACTGCTCT-3′ (forward) and 5′-CAGGCATTGAAGTCTCATGG-3′ (reverse).
Statistical analysis
The outcome variables analyzed were the percentage of spontaneous apoptotic cells and the IA ratio, calculated as the percentage of apoptotic cells induced by 8 Gy divided by the basal percentage of apoptotic cells. As each cell type was studied in the same patients, the between-group differences in basal apoptosis and IA ratio were analyzed using generalized linear models (GzLMs) for repeated measures. Also, a lognormal distribution was specified as it fitted well both outcome variables. Sequential Bonferroni corrections for multiple comparisons were used for post hoc comparisons if appropriate. The analysis included three factors: (1) cell type; (2) clinical toxicity due to radiotherapy; and (3) TP53 Arg72Pro polymorphism genotype. The outcome variables are presented as geometric means with 95 % confidence intervals (95 % CIs), since this is the best way when data follow a lognormal distribution.
The demographic and clinical data were compared between the patients without late toxicity and patients presenting late toxicity by using a t test for continued variables or Yates corrected Chi square test for categorial variables (Table 2).
In order to estimate the classification properties of the ratios of IA, area under the receiver-operating characteristic curves (AUC ROC) and the respective 95 % CIs were calculated, as well as a cut-off value according to Youden’s index (maximum of sensitivity + specificity −1).
Statistical computations were performed using the packages SAS® 9.2 (GzLMs), Stata 11 (AUC), and STATISTICA 8.0 (Graphic charts).
Results
Spontaneous apoptosis
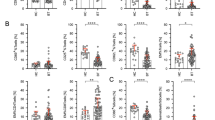
The results of the GzLMs analysis for repeated measures indicated that the clinical toxicity factor does not affect the percentage of spontaneous apoptosis (P = 0.806), since there were no differences between patients with and without late effects induced by radiotherapy (Fig. 1). In contrast, the factor of cell type showed a significant effect on the basal percentage of apoptosis (P < 0.0001), as the CD8+ subset of natural killer lymphocytes (CD8 NK) showed higher basal percentage of apoptosis than the other cell types, irrespective of clinical toxicity status (P = 0.0001 for CD8NK vs. CD3, CD8NK vs. CD4 and CD8NK vs. CD8, Bonferroni test) (Fig. 1). Supplementary Table S2 shows the age, the number of cells evaluated and the percentage of spontaneous apoptosis for the four untreated cell types.
Spontaneous apoptosis levels (geometric means ± 95 % CI) in non irradiated cultures compared between patients with and without late effects induced by radiotherapy, and among the different cell types. No differences in the spontaneous apoptosis between patients with and without late effects induced by radiotherapy. The CD8 subset of natural killer lymphocytes (CD8 NK) showed the highest values of spontaneous apoptosis compared with the rest of analyzed cell types
Ratio of the radiation-induced apoptosis (IA)
The ratio of apoptosis induced by 8 Gy was affected by both the clinical toxicity of the patients and the cell type (P = 0.049 and P = 0.0003, respectively). The patients with fibrosis and telangiectasias demonstrated a lower rate of IA than the patients without side effects (Fig. 2) for the CD3, CD4 and CD8 T-lymphocytes although this difference was only significant for the CD3 cells (P = 0.004 for CD3; P = 0.060 for CD4; P = 0.097 for CD8, Bonferroni test). In contrast, the IA ratio of the CD8 NK cells was similar between patients with or without late lesions (P = 0.389, Bonferroni test) (Fig. 2).
Irradiation-induced apoptotic (IA) ratios (geometric mean ± 95 % CI) compared between patients with and without late effects induced by radiotherapy, and among the different cell types. Patients with late radiotherapy toxicity showed less IA for all T-lymphocytes except for the CD8 NK. CD8 NK showed the lowest IA
Considering the cell type effect, the CD8 T-lymphocytes showed IA ratios higher than other cells irrespectively of the patient toxicity status (CD3 vs. CD8 P = 0.0026; CD4 vs. CD8 P = 0.0015; CD8 vs. CD8NK P = 0.0006, Bonferroni test), while the ratio values of CD8 NK lymphocytes were lower (CD3 vs. CD8 NK P = 0.0181; CD4 vs. CD8 NK P = 0.0431; CD8 vs. CD8 NK P = 0.0006, Bonferroni test) (Fig. 2). Supplementary Table S3 shows the total number of evaluated cells, the % of apoptotic cells induced by 8 Gy, and the IA ratio per each cell type and patient.
The GzLMs analysis for repeated measures including the Arg72Pro TP53 genotype factor showed, although marginally non-significant and irrespectively of cell type, that the IA ratio in patients with late toxicity was lower than that of patients without late toxicity only in patients with Arg72Arg genotype (six control patients vs. seven patients suffering late toxicity, P = 0.0776; Bonferroni test). No such difference was present in carriers of at least one Pro allele (four control patients vs. three patients with late toxicity, P = 0.8266; Bonferroni test) (Fig. 3). Permutation analysis confirmed that the all above obtained P-values were robust (Supplementary Table S4).
Irradiation-induced apoptotic (IA) ratios of the all analysed cell types (geometric mean ± 95 % CI) compared between 72Arg and 72Pro TP53 allele carriers with or without clinical toxicity. IA in patients with toxicity is lower than the control patients only in TP53 Arg/Arg patients (P = 0.077). This difference was not present in patients carrying at least one Pro allele (P = 0.8266)
The ROC curve analyses of IA ratios of CD3, CD4, CD8, and CD8 NK showed that the area under the curve was greater for CD3 (0.91; 95 % CI, 0.683–0.988) than for CD4 (0.80; 95 % CI, 0.563–0.943), CD8 (0.77; 95 % CI, 0.509–0.913) and CD8 NK (0.47; 95 % CI, 0.231–0.685). The ratio ≥6.46 of CD3 cells had the highest sensitivity (100 %) and specificity (80 %) as a cut-off value. Therefore, the 100 % of patients without late radiotherapy toxicity exhibited CD3 IA ratio values ≥6.46, while the 80 % of patients with late lesions had values lower than 6.46 (Fig. 4).
Discussion
In our study, the BC patients suffering from severe late complications due to radiotherapy exhibited lower levels of IA in total T-lymphocyte (CD3) compared to patients without complications. A similar trend was obtained from CD4 and CD8 T-lymphocytes, although statistically not significant. Regarding the prediction of patients at risk for late complications after radiotherapy, ROC analysis showed that a cut-off value of 6.46 of CD3 IA ratio correctly classifies 90 % of the patients. These results together with others previously reported [7–10, 15–17, 24] seem to support that IA determined in blood lymphocytes can be a reliable method to predict late tissue toxicity due to radiotherapy. However, in our study two patients (SP-3 and SP-15) with late skin lesions displayed IA higher than 6.46 (Fig. 4). Tumour staging of both patients are similar to those of the other patients (Supplementary Table S1). The SP-15 patient, that presented bilateral BC and telangiectasia and fibrosis grade 3 in both events (Table 1), exhibited a high level of IA (9.76) (Fig. 4). Moreover, no pathogenic BRCA1/BRCA2 variant was identified in the SP-15 patient (Supplementary Table S1). The severe sensitivity to radiotherapy in this patient could be due to other mechanisms than those leading to a low IA. Larger groups of patients, including other tumour sites, are needed to study these IA differences.
Some other studies in cancer patients reported no correlation between individual response to radiotherapy and IA. Table 3 shows the studies published to date and the differences in experimental conditions that could in part, explain these contradictory results. Thus, there was no relation between low apoptosis and clinical toxicity when lymphoblastoid cell lines (LCLs) or frozen lymphocytes rather than fresh ones were used [18–21], when the cells were irradiated with doses lower than 8 Gy [18–24] and, when the TUNEL assay was employed to analyze apoptosis [21, 22] (Table 3).
The mechanism responsible for low apoptotic response in lymphocytes of cancer patients with late side effects remains unclear. Lymphocytes from individuals with radiation hypersensitivity diseases caused by germ-line mutations in genes implied in DNA repair as ataxia telangiectasia and Nijmegen breakage syndrome also display a reduced apoptotic induction after in vitro irradiation [7, 15, 22, 25]. An inverse association between initial number of DNA double-strand breaks (DSBs) induced by in vitro irradiation and the IA has also been described in patients with locally advanced BC [26, 27]. Therefore, there is a potential link between cellular response to DNA DSBs and decreased IA [27]. Likewise, Henríquez-Hernández et al. [28] compared in a group of 12 locally advanced BC patients the significantly regulated pathways and biological process of differentially expressed genes and found common routes between clinical toxicity and DNA DSBs, clinical toxicity and apoptosis, and DNA DSBs and apoptosis. Thus, initial DNA damage, IA and clinical toxicity seem to share a common genetic background [28].
On the other hand, Crompton et al. [15], showed a decreased IA of CD4 and CD8 T-lymphocytes in two individuals with germ-line heterozygous mutation in the NF1 gene that causes the Neurobrimatosis I Syndrome (NF1). The loss of the NF1 gene product, neurofibromin, results in the presence of activated RAS, which in turn inhibits apoptosis and stimulates cell proliferation [29]. Further, it has been described that individuals with NF1 are at increased risk of developing secondary malignant neoplasms after radiotherapy [29]. In addition, a recent study demonstrates that Nf1 ± mutant mice develop more malignancies after irradiation than matched wild type mice [30]. All these findings support that the decreased apoptosis after in vitro irradiation found in patients with late complications induced by radiotherapy could be attributed to impaired response mechanisms to radiation cell exposure such as DNA DSBs repair and RAS signal transduction pathway.
Given that a subset of NK cells express CD8 on their surface, the analysis with only anti-CD8 mixes up two distantly related lymphoid lineages [31, 32]. We thus chose double surface immunostaining using the combination of anti-CD3 and anti-CD8 for distinguishing T and NK cells expressing CD8 on their surface. Using this approach we found two clear different responses to in vitro irradiation: the CD8 T-lymphocytes showed the greatest sensitivity to radiation while the CD8 NK cells subset were the most radio-resistant cells (Fig. 2). These data concur with previous reports showing that the total NK cells (without differentiating the subset expressing CD8) were the most radio-resistant cells [33–35] and that CD8+ lymphocytes were more sensitive to irradiation than CD4+cells [8, 15, 24, 33]. In contrast, Vokurkova et al. [32] analyzing the proportion of live intact cells, described that the CD3− CD8+ NK subset was more radiosensitive than CD4 and CD8 T-lymphocytes.
In addition, in our study the subset of CD8 NK cells showed the highest level of spontaneous apoptosis compared to the remaining cell types (Fig. 1), while the IA ratios of this type of NK cells in toxicity and non-toxicity patients were similar (Fig. 2). According to our results, and in order to avoid a source of variation, it would be advisable to perform the IA assay in CD8 cells with the combination of anti-CD3 and anti-CD8 to distinguish the CD8 NK subset from the pool of CD8+ lymphocytes.
The TP53 Arg72Pro polymorphism encodes two p53 variants that significantly affect the biological activity of p53 [12, 13]. The apoptotic potential of the different forms of the protein depends on the cell type and cellular environment [36]. The current consensus is that p53-Arg72 is more effective at inducing apoptosis and protecting stressed cells from neoplastic development than p53-Pro72 [12]. However, it is not yet understood how universal these functional differences between p53-Pro72 and p53-Arg72 might be in different cell types or whether they are relevant in vivo [12]. In particular, the apoptotic activity of the codon 72 variants in human lymphocytes irradiated in vitro is not known.
Although slightly non-significant, the IA ratio of the total analysed cells in patients with late toxicity was lower than that of patients without late toxicity only in patients with Arg72Arg genotype. This difference was not present in patients carrying at least one Pro allele (Fig. 3). Our finding, even though based on a low number of individuals, suggests that the Pro allele may influence the lymphocyte apoptotic response to in vitro irradiation in patients suffering from late toxicity after radiotherapy.
In summary, our results show that radiotherapy-induced late toxicity is associated to low levels of IA. Given that the response to irradiation of CD8 NK cells is different from that of the T-lymphocytes, it would be advisable to distinguish the CD8 NK lymphocytes from the pool of CD8+ lymphocytes in IA assays using CD8+ cells. Because of the potential functional role of the TP53 Arg72Pro polymorphism on apoptosis, larger and prospective studies are needed to validate the influence of 72Pro TP53 allele on IA.
References
Barnett GC, West CM, Dunning AM, Elliott RM, Coles CE, Pharoah PD, Burnet NG (2009) Normal tissue reactions to radiotherapy: towards tailoring treatment dose by genotype. Nat Rev Cancer 9:134–142
Popanda O, Marquardt JU, Chang-Claude J, Schmezer P (2009) Genetic variation in normal tissue toxicity induced by ionizing radiation. Mutat Res 667:58–69
Bentzen SM (2006) Preventing or reducing late side effects of radiation therapy: radiobiology meets molecular pathology. Nat Rev Cancer 6:702–713
Barker CL, Routledge JA, Farnell DJ, Swindell R, Davidson SE (2009) The impact of radiotherapy late effects on quality of life in gynaecological cancer patients. Br J Cancer 100:1558–1565
West CM, Barnett GC (2011) Genetics and genomics of radiotherapy toxicity: towards prediction. Genome Med 3:52
Fernet M, Hall J (2008) Predictive markers for normal tissue reactions: fantasy or reality? Cancer Radiother 12:614–618
Crompton NE, Miralbell R, Rutz HP, Ersoy F, Sanal O, Wellmann D, Bieri S, Coucke PA, Emery GC, Shi YQ, Blattmann H, Ozsahin M (1999) Altered apoptotic profiles in irradiated patients with increased toxicity. Int J Radiat Oncol Biol Phys 45:707–714
Ozsahin M, Crompton NE, Gourgou S, Kramar A, Li L, Shi Y, Sozzi WJ, Zouhair A, Mirimanoff RO, Azria D (2005) CD4 and CD8 T-lymphocyte apoptosis can predict radiation-induced late toxicity: a prospective study in 399 patients. Clin Cancer Res 11:7426–7433
Bordon E, Henriquez Hernandez LA, Lara PC, Pinar B, Fontes F, Rodriguez Gallego C, Lloret M (2009) Prediction of clinical toxicity in localized cervical carcinoma by radio-induced apoptosis study in peripheral blood lymphocytes (PBLs). Radiat Oncol 4:58
Bordon E, Henriquez-Hernandez LA, Lara PC, Ruiz A, Pinar B, Rodriguez-Gallego C, Lloret M (2010) Prediction of clinical toxicity in locally advanced head and neck cancer patients by radio-induced apoptosis in peripheral blood lymphocytes (PBLs). Radiat Oncol 5:4
Riley T, Sontag E, Chen P, Levine A (2008) Transcriptional control of human p53-regulated genes. Nat Rev Mol Cell Biol 9:402–412
Whibley C, Pharoah PD, Hollstein M (2009) p53 polymorphisms: cancer implications. Nat Rev Cancer 9:95–107
Dumont P, Leu JI, Della Pietra AC, George DL, Murphy M (2003) The codon 72 polymorphic variants of p53 have markedly different apoptotic potential. Nat Genet 33:357–365
Ozsahin M, Ozsahin H, Shi Y, Larsson B, Wurgler FE, Crompton NE (1997) Rapid assay of intrinsic radiosensitivity based on apoptosis in human CD4 and CD8 T-lymphocytes. Int J Radiat Oncol Biol Phys 38:429–440
Crompton NE, Shi YQ, Emery GC, Wisser L, Blattmann H, Maier A, Li L, Schindler D, Ozsahin H, Ozsahin M (2001) Sources of variation in patient response to radiation treatment. Int J Radiat Oncol Biol Phys 49:547–554
Azria D, Ozsahin M, Kramar A, Peters S, Atencio DP, Crompton NE, Mornex F, Pelegrin A, Dubois JB, Mirimanoff RO, Rosenstein BS (2008) Single nucleotide polymorphisms, apoptosis, and the development of severe late adverse effects after radiotherapy. Clin Cancer Res 14:6284–6288
Foro P, Algara M, Lozano J, Rodriguez N, Sanz X, Torres E, Carles J, Reig A, Membrive I, Quera J, Fernandez-Velilla E, Pera O, Lacruz M, Bellosillo B (2014) Relationship between radiation-induced apoptosis of T-lymphocytes and chronic toxicity in patients with prostate cancer treated by radiation therapy: a prospective study. Int J Radiat Oncol Biol Phys 88:1057–1063
Finnon P, Kabacik S, Mackay A, Raffy C, A’hern R, Owen R, Badie C, Yarnold J, Bouffler S (2012) Correlation of in vitro lymphocyte radiosensitivity and gene expression with late normal tissue reactions following curative radiotherapy for breast cancer. Radiother Oncol 105:329–336
Greve B, Dreffke K, Rickinger A, Konemann S, Fritz E, Eckardt-Schupp F, Amler S, Sauerland C, Braselmann H, Sauter W, Illig T, Schmezer P, Gomolka M, Willich N, Bolling T (2009) Multicentric investigation of ionising radiation-induced cell death as a predictive parameter of individual radiosensitivity. Apoptosis 14:226–235
Greve B, Bolling T, Amler S, Rossler U, Gomolka M, Mayer C, Popanda O, Dreffke K, Rickinger A, Fritz E, Eckardt-Schupp F, Sauerland C, Braselmann H, Sauter W, Illig T, Riesenbeck D, Konemann S, Willich N, Mortl S, Eich HT, Schmezer P (2012) Evaluation of different biomarkers to predict individual radiosensitivity in an inter-laboratory comparison-essons for future studies. PLoS One 7:e47185
Wistop A, Keller U, Sprung CN, Grabenbauer GG, Sauer R, Distel LV (2005) Individual radiosensitivity does not correlate with radiation-induced apoptosis in lymphoblastoid cell lines or CD3+ lymphocytes. Strahlenther Onkol 181:326–335
Barber JB, West CM, Kiltie AE, Roberts SA, Scott D (2000) Detection of individual differences in radiation-induced apoptosis of peripheral blood lymphocytes in normal individuals, ataxia telangiectasia homozygotes and heterozygotes, and breast cancer patients after radiotherapy. Radiat Res 153:570–578
Brzozowska K, Pinkawa M, Eble MJ, Muller WU, Wojcik A, Kriehuber R, Schmitz S (2012) In vivo versus in vitro individual radiosensitivity analysed in healthy donors and in prostate cancer patients with and without severe side effects after radiotherapy. Int J Radiat Biol 88:405–413
Schnarr K, Boreham D, Sathya J, Julian J, Dayes IS (2009) Radiation-induced lymphocyte apoptosis to predict radiation therapy late toxicity in prostate cancer patients. Int J Radiat Oncol Biol Phys 74:1424–1430
Shi YQ, Li L, Sanal O, Tezcan I, Emery GC, Blattmann H, Crompton NE (2001) High levels of delayed radiation-induced apoptosis observed in lymphoblastoid cell lines from ataxia-telangiectasia patients. Int J Radiat Oncol Biol Phys 49:555–559
Henriquez-Hernandez LA, Carmona-Vigo R, Pinar B, Bordon E, Lloret M, Nunez MI, Rodriguez-Gallego C, Lara PC (2011) Combined low initial DNA damage and high radiation-induced apoptosis confers clinical resistance to long-term toxicity in breast cancer patients treated with high-dose radiotherapy. Radiat Oncol 6:60
Pinar B, Henriquez-Hernandez LA, Lara PC, Bordon E, Rodriguez-Gallego C, Lloret M, Nunez MI, De Almodovar MR (2010) Radiation induced apoptosis and initial DNA damage are inversely related in locally advanced breast cancer patients. Radiat Oncol 5:85
Henriquez-Hernandez LA, Pinar B, Carmona-Vigo R, Bordon E, Rodriguez-Gallego C, Flores-Morales A, Lara PC (2013) Common genomic signaling among initial DNA damage and radiation-induced apoptosis in peripheral blood lymphocytes from locally advanced breast cancer patients. Breast 22:28–33
Sharif S, Ferner R, Birch JM, Gillespie JE, Gattamaneni HR, Baser ME, Evans DG (2006) Second primary tumors in neurofibromatosis 1 patients treated for optic glioma: substantial risks after radiotherapy. J Clin Oncol 24:2570–2575
Choi G, Huang B, Pinarbasi E, Braunstein SE, Horvai AE, Kogan S, Bhatia S, Faddegon B, Nakamura JL (2012) Genetically mediated nf1 loss in mice promotes diverse radiation-induced tumors modeling second malignant neoplasms. Cancer Res 72:6425–6434
Cooper MA, Fehniger TA, Caligiuri MA (2001) The biology of human natural killer-cell subsets. Trends Immunol 22:633–640
Vokurkova D, Sinkora J, Vavrova J, Rezacova M, Knizek J, Ostereicher J (2006) CD8+ natural killer cells have a potential of a sensitive and reliable biodosimetric marker in vitro. Physiol Res 55:689–698
Bordon E, Henriquez-Hernandez LA, Lara PC, Pinar B, Rodriguez-Gallego C, Lloret M (2011) Role of CD4 and CD8 T-lymphocytes, B-lymphocytes and Natural Killer cells in the prediction of radiation-induced late toxicity in cervical cancer patients. Int J Radiat Biol 87:424–431
Louagie H, Van EM, Philippe J, Thierens H, De RL (1999) Changes in peripheral blood lymphocyte subsets in patients undergoing radiotherapy. Int J Radiat Biol 75:767–771
Philippe J, Louagie H, Thierens H, Vral A, Cornelissen M, De RL (1997) Quantification of apoptosis in lymphocyte subsets and effect of apoptosis on apparent expression of membrane antigens. Cytometry 29:242–249
Wiggs JL, Hewitt AW, Fan BJ, Wang DY, Figueiredo Sena DR, O’Brien C, Realini A, Craig JE, Dimasi DP, Mackey DA, Haines JL, Pasquale LR (2012) The p53 codon 72 PRO/PRO genotype may be associated with initial central visual field defects in caucasians with primary open angle glaucoma. PLoS One 7:e45613
Acknowledgments
This study was supported by the ‘‘Fondo de Investigación Sanitaria’’ [FIS 05/2181] from the Spanish Health Ministry and the ‘‘Fundación de Investigación Médica Mutua Madrileña’’ [FMMA 2008–2012] grants. Dr Sara Gutiérrez-Enríquez is currently funded by a Miguel Servet contract awarded by the Instituto de Salud Carlos III of the “Ministerio Español de Economía y Competitividad”. We are grateful to the BC donor patients involved in this study.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Fuentes-Raspall, M.J., Caragol, I., Alonso, C. et al. Apoptosis for prediction of radiotherapy late toxicity: lymphocyte subset sensitivity and potential effect of TP53 Arg72Pro polymorphism. Apoptosis 20, 371–382 (2015). https://doi.org/10.1007/s10495-014-1056-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-014-1056-2