Abstract
Tick-borne rickettsioses belong to the important emerging infectious diseases worldwide. We investigated the potential human exposure to rickettsiae by determining their presence in questing ticks collected in an urban park of Budapest and a popular hunting and recreational forest area in southern Hungary. Differences were found in the infectious risk between the two habitats. Rickettsia monacensis and Rickettsia helvetica were identified with sequencing in questing Ixodes ricinus, the only ticks species collected in the city park. Female I. ricinus had a particularly high prevalence of R. helvetica (45 %). Tick community was more diverse in the rural habitat with Dermacentor reticulatus ticks having especially high percentage (58 %) of Rickettsia raoultii infection. We conclude that despite the distinct eco-epidemiological traits, the risk (hazard and exposure) of acquiring human pathogenic rickettsial infections in both the urban and the rural study sites exists.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Tick-borne rickettsioses are produced by spotted fever group rickettsiae and cause an expanding spectrum of clinical signs. Until recently, Mediterranean spotted fever caused by Rickettsia conorii was considered the only tick-borne rickettsiosis in Europe (Oteo and Portillo 2012). In the last decade many other species and subspecies of Rickettsia have been discovered and implicated as human pathogens, and new rickettsial syndromes have been described. Lack of (local) awareness among medical health professionals and availability of diagnostic tools (case definitions, serology, PCR etc.) hamper the identification of clinical cases.
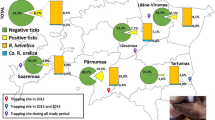
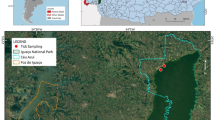
The aim of this study was to investigate potential human exposure to rickettsiae by determining their presence in questing ticks collected in an urban park of Budapest (Margaret Island), and a popular hunting and recreational forest area in Hungary (Gemenc). Ticks collected with flagging in 2011–2012 were identified and stored in 70 % ethanol until DNA extraction by alkaline hydrolysis then analyzed with a multiplex qPCR assay as previously described (de Bruin et al. 2015). This included a primer pair specific for R. helvetica gltA gene and another amplifying a different region of gltA of rickettsiae in general (Stenos et al. 2005). Positive samples were analyzed with conventional PCR and sequencing (de Bruin et al. 2015).
We collected 534 Ixodes ricinus from Margaret Island and 162 ticks (34 I. ricinus, 64 Dermacentor reticulatus, 2 D. marginatus and 62 Haemaphysalis concinna) from Gemenc. Rickettsia helvetica was detected in 139 (26 %) out of 534 and 9 (26.5 %) out of 34 questing I. ricinus from the urban park and the rural site, respectively (Table 1). The qPCR specific for Rickettsia spp. was positive in 88 (16.5 %) out of 534 I. ricinus ticks from the urban and 41 (25.3 %) out of 162 ticks from the rural habitat. Prevalence of rickettsiae in I. ricinus did not differ significantly in the two study sites (Table 1). However, both components of infection risk, the hazard (density of infected I. ricinus) and exposure (human presence) is higher in the urban park (Földvári et al. 2014) compared to the rural site. Female I. ricinus ticks in the urban parks were found to have a particularly high prevalence of R. helvetica (44.6 %) suggesting a higher infection risk when humans are bitten by this tick stage. The significantly higher prevalence of R. helvetica and Rickettsia spp. in the adult stages of I. ricinus compared to nymphs from the urban habitat (Fisher’s exact test: p < 0.05) suggests the important role of transstadial infection in the eco-epidemiology of these pathogens (Table 1).
From the urban habitat 22 R. monacensis and 9 R. helvetica out of 534 questing I. ricinus were identified with the less sensitive conventional PCR and sequencing. Compared to the average ratio of these two rickettsiae in other European studies (Rizzoli et al. 2014; Špitalská et al. 2014), the relatively high prevalence of R. monacensis (originally described from a city park in Germany (Rizzoli et al. 2014)) appears unique probably as a consequence of the eco-epidemiology of the closed island park habitat (Földvári et al. 2014).
Rickettsiae were detected in 57.8 % of D. reticulatus which is much higher compared to the prevalence of 15.5 % reported previously in questing ticks of the same species collected throughout the country (Hornok et al. 2010). We identified R. raoultii infection with sequencing in 31 qPCR-positive D. reticulatus samples from the rural habitat. Tick-borne lymphadenopathy (TIBOLA) patients (Lakos 1997) and R. slovaca and R. raoultii in ticks removed from them have been reported in Hungary (Földvári et al. 2013). Both female (53.7 %) and male (65.2 %) D. reticulatus ticks in the present study had high prevalence of Rickettsia spp. This corroborates with previous findings about the equal role of both tick sexes and both Dermacentor spp. in TIBOLA epidemiology (Földvári et al. 2013).
Our results showed considerable difference between the dominant rickettsial agents in the city park (R. helvetica and R. monacensis) and natural forest habitat (R. raoultii). This is due to the differences of these habitats in their vector diversity. In urban settings, usually I. ricinus dominates (Rizzoli et al. 2014), whereas in natural habitats there is a more diverse tick community even visible in the small rural sample size of the present study (Szekeres et al. 2015). This more diverse tick community extends the range of possible human pathogenic rickettsiae, including newly emerging ones. Both our study sites have frequent human visitors: Margaret Island is a popular recreational and jogging park in the center of Budapest and Gemenc is a popular hunting and hiking area with over 50,000 tourists per year. Since all rickettsiae (R. helvetica, R. monacensis and R. raoultii) detected in this study are proven human pathogens (Fournier et al. 2000; Jado et al. 2007; Jia et al. 2014), we can conclude that despite the distinct eco-epidemiological traits, the risk (hazard and exposure) of acquiring rickettsial infections in both the urban and the rural study sites exists.
References
de Bruin A, van Leeuwen AD, Jahfari S et al (2015) Vertical transmission of Bartonella schoenbuchensis in Lipoptena cervi. Parasit Vectors 8:176. doi:10.1186/s13071-015-0764-y
Földvári G, Rigó K, Lakos A (2013) Transmission of Rickettsia slovaca and Rickettsia raoultii by male Dermacentor marginatus and Dermacentor reticulatus ticks to humans. Diagn Microbiol Infect Dis 76:387–389. doi:10.1016/j.diagmicrobio.2013.03.005
Földvári G, Jahfari S, Rigó K et al (2014) Candidatus Neoehrlichia mikurensis and Anaplasma phagocytophilum in Urban Hedgehogs. Emerg Infect Dis 20:496–498
Fournier P-E, Grunnenberger F, Jaulhac B et al (2000) Evidence of Rickettsia helvetica Infection in Humans, Eastern France. Emerg Infect Dis 6:389–392
Hornok S, Meli ML, Perreten A et al (2010) Molecular investigation of hard ticks (Acari: Ixodidae) and fleas (Siphonaptera: Pulicidae) as potential vectors of rickettsial and mycoplasmal agents. Vet Microbiol 140:98–104. doi:10.1016/j.vetmic.2009.07.013
Jado I, Oteo JA, Aldámiz M et al (2007) Rickettsia monacensis and Human Disease, Spain. Emerg Infect Dis 13:1405–1407
Jia N, Zheng Y, Huo Q et al (2014) Human Infections with Rickettsia rauoltii, China. Emerg Infect Dis 20:8–10
Lakos A (1997) Tick-borne lymphadenopathy-a new rickettsial disease? Lancet 350:1006. doi:10.1016/S0140-6736(05)64072-X
Oteo JA, Portillo A (2012) Tick-borne rickettsioses in Europe. Ticks Tick Borne Dis 3:271–278. doi:10.1016/j.ttbdis.2012.10.035
Rizzoli A, Silaghi C, Obiegala A et al (2014) Ixodes ricinus and its transmitted pathogens in urban and peri-urban areas in Europe: new hazards and relevance for public health. Front Public Heal 2:251. doi:10.3389/fpubh.2014.00251
Špitalská E, Boldiš V, Derdáková M et al (2014) Rickettsial infection in Ixodes ricinus ticks in urban and natural habitats of Slovakia. Ticks Tick Borne Dis 5:161–165. doi:10.1016/j.ttbdis.2013.10.002
Stenos J, Graves SR, Unsworth NB (2005) A highly sensitive and specific real-time PCR assay for the detection of spotted fever and typhus group rickettsiae. Am J Trop Med Hyg 73:1083–1085
Szekeres S, Coipan EC, Rigó K et al (2015) Candidatus Neoehrlichia mikurensis and Anaplasma phagocytophilum in natural rodent and tick communities in Southern Hungary. Ticks Tick Borne Dis 6:111–116. doi:10.1016/j.ttbdis.2014.10.004
Acknowledgments
We are grateful to the Gemenc Forest and Game Co. Ltd. who supported our work in the area. This study was partially supported by European Union grant FP7-261504 EDENext and was catalogued by the EDENext Steering Committee (www.edenext.eu) as EDENext437. G. F. was supported by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences and an NKB and Research Faculty grants from the Faculty of Veterinary Science, Szent István University. S. Sz. and H. S. were supported by EurNegVec Cost Action TD1303. S. Sz. was supported by the city Council of Hajdúböszörmény, the Campus Hungary Scholarship and TÁMOP-4.2.2.B-10/1-2010-0011.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Szekeres, S., Docters van Leeuwen, A., Rigó, K. et al. Prevalence and diversity of human pathogenic rickettsiae in urban versus rural habitats, Hungary. Exp Appl Acarol 68, 223–226 (2016). https://doi.org/10.1007/s10493-015-9989-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10493-015-9989-x