Abstract
The intrinsic ruggedness of Enterococcus faecalis is responsible for its widespread distribution in nature and is often viewed as an important virulence determinant. Previously, we showed that the ClpB ATPase is negatively regulated by CtsR and is required for thermotolerance and virulence in a Galleria mellonella invertebrate model. Here, we used in silico, Northern blot and quantitative real-time PCR analyses to identify additional members of the CtsR regulon, namely the clpP peptidase and the clpC and clpE ATPases. When compared to the parent strain, virulence of the ΔctsR strain in G. mellonella was significantly attenuated.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Protein quality control, which includes the refolding or degradion of damaged proteins, plays an indispensable role in bacterial homeostasis. Under stress conditions, the synthesis of molecular chaperones and proteases is rapidly induced to minimize aggregation of denatured proteins. In addition to protein quality control, bacterial proteases also play important roles in other cellular processes by controlling the stability of central regulatory proteins (Frees et al. 2007). In low-GC Gram-positive bacteria (Firmicutes), the ClpP protease is the main system involved in protein degradation. Functional ClpP proteases have a bipartite molecular architecture, with two heptameric rings of the ClpP serine protease forming a central proteolytic chamber and one or two hexameric rings of Clp ATPases docked to either one or both apical sides of the ClpP chamber, a structure that is remarkably similar to that of the eukaryotic proteosome (Frees et al. 2007). The genomes of Gram-positive cocci typically encode up to five Clp ATPases, namely ClpB, ClpC, ClpE, ClpL and ClpX, but only ClpC, ClpE and ClpX have the recognition tripeptide that permits interaction with ClpP (Frees et al. 2007). The ClpB and ClpL proteins lack the ClpP recognition peptide and function as independent chaperones (Woo et al. 1992). Given their central role in stress reponse and cell homeostasis, it is not surprinsing that several clp genes are required for virulence and the progression of disease of many bacterial pathogens including Enterococcus faecalis, Streptococcus pneumoniae and Staphylococcus aureus (Frees et al. 2003; Ibrahim et al. 2005; Oliveira et al. 2011). In low-GC Gram positive bacteria, the levels of ClpP and selected Clp ATPases are controlled by the CtsR repressor that binds to a consensus heptad sequence located in the regulatory region of these genes (Derré et al. 1999). In Lactobacillus platarum, the CtsR regulon was characterised and CtsR is essential for stress response and growth at elevated temperatures. Moreover, the ctsR gene is co-transcribed with clpC and CtsR binds to the promoter region of the ctsR–clpC operon (Fiocco et al. 2010).
The genome of E. faecalis, a major nosocomial pathogen, encodes orthologs of the ClpB, ClpC, ClpE and ClpX ATPases and a single copy of the ClpP peptidase (Paulsen et al. 2003). In a previous study, we showed that the E. faecalis clpB gene was induced by a variety of stress conditions and negatively regulated by CtsR (Oliveira et al. 2011). In this report, we use in silico, transcriptional and mutational approaches to show that the clpC, clpE and clpP genes belong to the CtsR regulon. In addition, we showed that virulence of the ΔctsR strain is significantly attenuated in a Galleria mellonella invertebrate model.
Materials and methods
Bacterial strains and general culture conditions
The E. faecalis OG1RF and ΔctsR (Oliveira et al. 2011) strains were routinely grown in brain heart infusion (BHI) medium at 37 °C under aerobic conditions. When required for selective growth of strains erythromycin (10 µg/ml) was added to the growth medium. For Northern blot analyses, cells were grown in BHI to mid-log phase (OD600~0.5) and subjected to heat-shock at 42, 45, 48 or 50 °C for 10 min, whereas for quantitative real-time reverse-transcripatse PCR (qRT-PCR) experiments cultures were subjected to 48 °C for 30 min.
RNA methods
For each growth condition, cell pellets were collected by centrifugation and immediately treated with the RNA protect reagent (Qiagen). Total RNA was isolated by the hot acid–phenol method as described previously (Abranches et al. 2006). The crude RNA was treated with DNase I (Ambion) and then further purified by using the RNeasy mini kit (Qiagen), including an on-column DNase I digestion (Qiagen). For qRT-PCR analysis, cDNA was generated from three independent RNA samples using the Super Script first-strand synthesis system kit (Invitrogen) and gene specific primers (Table 1). The primers used for qRT-PCR were designed using the Beacon designer 2.0 software (Premier Biosoft International). qRT-PCR was carried out following the a protocol described elsewhere (Ahn et al. 2005). A Student’s t test was performed to verify significance of the qRT-PCR results. For Northern analysis, 5 µg per lane of RNA was separated on a 1.2 % agarose–formaldehyde denaturing gel and blotted to a nitrocellulose membrane as described (Oliveira et al. 2011). The membrane was probed with a fragment of the E. faecalis clpC, clpE and clpP genes labelled with [α32P] dCTP and the ready-to-go DNA labelling beads system according to the manufacturer’s recommendations (Amersham Pharmacia Biotech).
Galleria mellonella infection
For the G. mellonella infection model, 5-μl aliquots containing 1 × 106 CFU ml−1 of overnight-grown cultures of E. faecalis in sterile saline were injected into the hemocoel of each larva via the last left proleg, as described previously (Oliveira et al. 2011). Larvae injected with heat-inactivated bacteria os saline were used as controls. After injection, larvae were kept in the dark at 37 °C, and survival was recorded at selected intervals. Experiments were performed in triplicate. Kaplan–Meier killing curves were plotted and estimations of differences in survival were compared using the log rank test. P values of ≤0.05 were considered significant Experiments were performed independently three times with similar results.
Results and discussion
Promoter region and northern blot analysis of clp gene transcription
Previously, we conducted in silico, transcriptional and mutational analyses of the clpB chaperone from E. faecalis (Oliveira et al. 2011). Here, in silico analysis of the remaining clp genes present in the E. faecalis genome (clpC, clpE, clpP and clpX) identified potential binding sites for the CtsR repressor, the so-called CtsR box (Derré et al. 1999), upstream the start codon of ctsR (co-transcribed with clpC), clpE and clpP (Fig. 1). However, no CtsR box was found in the upstream region of clpX. These findings are in line with observations made with related low-GC Gram-positive bacteria whereby clpX is the only clp gene that is not under CtsR regulation in Streptococcus mutans and S. pneumoniae (Chastanet et al. 2001; Lemos and Burne 2002).
Nucleotide sequences of the promoter regions of the a clpC, b clpE, c clpP, and d clpX genes from E. faecalis. Regulatory sequences underlined, are as follows: one putative CtsR element (consensus sequence GGTCAAANANGGTCAAA) indicated in bold, the putative −35 and −10 regions, the initiation codon ATG and ribosome binding site or SD (Shine-Dalgarno)
Northern blot analysis was performed using specific probes for the clpC, clpE and clpP genes. Analysis with the specific clpC DNA probe revealed a transcript of approximately 3.0 kb, which corresponds in size to a bicistronic message containing the ctsR and clpC genes (Fig. 2a). The clpE transcript (approximately 2.6 kb) was larger than expected (Fig. 2b), suggesting that clpE is co-transcribed with a small gene (EF707) encoding for a small hypothetical protein. The detected 0.6 kb clpP mRNA was consistent with the expected size of a clpP monocistronic mRNA (Fig. 2c). Transcription of clpE and clpP was strongly induced when the growth temperature was shifted from 37 to 45 °C, with expression levels peaking between 48 and 50 °C. Transcription of clpC was induced at a lower temperature, 42 °C, but remained steady at higher temperatures (up to 50 °C). In general, qRT-PCR analysis using RNA isolated from cells grown to mid-log phase at 37 °C and shifted to 48 °C for 15 and 30 min confirmed the heat-inducibility of clpC, clpE and clpP (Fig. 3).
Analysis of genomic regions and clp genes expression with schematic representation of the genomic region of clp genes from E. faecalis V583. The arrows indicate the direction of transcription. The diagram presented is noted in the genome and shows the location and size of genes in the genome of this microrganism (www.tigr.org), molecular weight (kDa) of probable amino acid sequence and isoelectric point (pI). Northern blot analysis of clpC, clpE and clpP under conditions of thermal stress in E. faecalis growth at 37 °C (control) and subjected to heat stress at 42, 45, 48 and 50 °C for 10 min. The signal observed in autoradiograph represents hybridization with a specific probe for the indicated gene. TTS, transcription termination site
qRT-PCR analysis of the clpC, clpE, clpP and clpX genes.. Analysis expression of mRNAs from clpC (a), clpE (b), clpP (c) and clpX (d) genes in wild type cells (wt) from E. faecalis OG1RF (first two bars—black and chess), or ΔctsR strain (last two bars—white and dotted) subjected to heat shock at 48 °C for 30 min (chess and dotted bars). As a control, cells were used at 37 °C (black and white bars). (*p ≤ 0.05 student’s t test) compared to the control at 37 °C
clpC, clpE and clpP are under CtsR regulation
Previously, an E. faecalis strain bearing a complete deletion of the ctsR gene (ΔctsR) was isolated and used to demonstrate that CtsR is a repressor of clpB (Oliveira et al. 2011). We surveyed the available E. faecalis genomes for the presence of additional CtsR binding sites in non-coding regions and could only find conserved CtsR sites upstream of the clpB, ctsR (co-transcribed with clpC), clpE and clpP genes. As expected, analysis by qRT-PCR of the ΔctsR strain revealed that transcription of clpC, clpE and clpP at 37 °C was at least twofold higher when compared to the wild-type strain (Fig. 3). When transcription of the clpC, clpE and clpP genes in the ΔctsR strain was compared to the parent strain exposed to the same 48 °C heat stress, we could still observe a twofold increase in mRNA levels of clpC and clpP and fivefold increase in clpE transcription (Fig. 3). These findings suggest that there is a second, yet-to-be-identified, heat shock-responsive regulator controlling the expression of clp genes. Given that the clpX promoter region does not possess a CtsR-binding box, it was not surprising that transcription of clpX was not affected in the ΔctsR grown at 37 °C. Moreover, clpX transcription was not induced by heat shock, which indicated that clpX is not a member of the heat stress regulon. Unexpectedly, clpX transcription was strongly induced in the ΔctsR strain when compared to the parent strain subjected to the same stress, or against the ΔctsR strain grown at 37 °C (Fig. 3d). Thus, while clpX transcription was not directly regulated by CtsR, loss of CtsR indirectly affects its expression under heat stress conditions. It is tempting to speculate that this induction may be a compensatory mechanism that allows clpX to compete with the other highly expressed ClpC and ClpE proteins for binding with the ClpP peptidase. Future studies to address the underlying mechanisms controlling hyperinduction of clpC, clpE, clpP as well as the induction of clpX in the ΔctsR mutant upon heat shock will be necessary for a complete understanding of the regulatory mechanisms controlling clp gene expression.
In addition to CtsR, HrcA is another major transcriptional regulator of heat shock proteins (Narberhaus 1999). The HrcA repressor recognizes a highly conserved DNA element known as controlling invereted repeat of chaperone expression (CIRCE) and is mainly involved in the transcriptional repression of the dnaK and groE heat shock operons. In some cases, these heat shock genes can be under dual regulation by CtsR and HrcA. Specifically, the dnaK operon of S. aureus, the clpP gene from Streptococcus salivarius and the groE operon from a number of streptococci are regulated by both CtsR and HrcA (Chastanet et al. Chastanet and Msadek 2003; Lemos and Burne 2002). Sequence analysis of the promoter regions of ctsR–clpC, clpE and clpP failed to identify a CIRCE-like element, which suggests that these genes are not under HrcA regulation. Further in silico analysis using the Multiple Em for Motif Elicitation (MEME) suite also failed to identify conserved cis-acting elements within the promoter regions of these genes. Thus, the molecular factor responsible for the heat stress activation of clpC, clpE and clpP in the absence of the CtsR repressor remains to be identified.
Role of CtsR during induction of thermotolerance
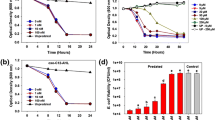
Acquired thermotolerance due to prior adaptation at sub-lethal temperatures is a common phenomenon that has been described in E. faecalis (Flahaut et al. 1997). Previously, we showed that the ClpB chaperone plays a major role in thermotolerance (Oliveira et al. 2011). To test the involvement of CtsR in thermotolerance, cultures of the wild-type and ΔctsR strains grown at 37 °C were incubated at 45 °C (sub-lethal heat stress) for 30 min and then transferred to 60 °C (lethal heat stress). Alternatively, control cultures were transferred from 37 to 60 °C. There were no significant differences in the numbers of survivors between wild-type and ΔctsR strains in cultures subjected to the 60 °C lethal stress (data not shown). Pre-incubation at 45 °C conferred nearly full protection towards a lethal treatment at 60 °C to both wild-type and ΔctsR strains (data not shown). These results indicate that CtsR repression is not required for heat stress survival or thermotolerance acquisition in E. faecalis.
Virulence of the ΔctsR is attenuated in the G. mellonella invertebrate model
By using a G. mellonella systemic infection model, we found that virulence of the ΔctsR is significantly attenuated when compared to the parent strain (p ≤ 0.001) (Fig. 4). After 30 h of infection, approximately 65 % of larvae infected with the parent strain were dead compared to 25 % death in larvae infected with the ΔctsR. Larvae used as controls were inoculated with saline solution or heat-killed cells and showed approximately 95 % survival after five days of infection. In this in vivo experiment we observed that mutant cells ΔctsR showed a significantl lower virulence when compared to wild type cells of E. faecalis OG1RF suggesting that CtsR mediates virulence of E. faecalis. This result is in agreement with, to our knowledge, the only other studies that have evaluated the virulence potential of a ctsR mutant. More specifically, a strain of Listeria monocytogenes harboring a single amino acid substitution at a conserved residue that rendered CtsR inactive showed attenuated virulence in a murine peritoneal infection model (Karatzas et al. 2003). Conversely, another group showed that their L. monocytogenes ΔctsR strain was as virulent as the parent strain but, virulence of a wild-type strain constitutively expressing CtsR was significantly attenuated, presumably because of repression of the clp genes (Nair et al. 2000). It has been shown that stability and proper folding of CtsR is mediated by the ClpC, ClpE, ClpL ATPases (Derré et al. 2000; Miethke et al. 2006; Varmanen et al. 2003; Tao and Biswas 2013). In addition to degradation of irreversibly damaged proteins, ClpP proteases perform essential roles by controlling the cellular levels of other transcriptional regulators such as the oxidative stress Spx and the DNA damage HdiR regulators (Frees et al. 2007). Thus, it appear that any imbalance in the expression levels of the CtsR and Clp proteins can have profound effects on the pathophysiology of E. faecalis and related organisms.
REFERENCES
Abranches J, Candella MM, Wen ZT, Baker HV, Burne RA (2006) Different roles of EIIABMan and EIIGlc in regulation of energy metabolism, biofilm development, and competence in Streptococcus mutans. J Bacteriol 188:3748–3758
Ahn SJ, Lemos JA, Burne RA (2005) Role of HtrA in growth and competence of Streptococcus mutans UA159. J Bacteriol 187:3028–3038
Chastanet A, Msadek T (2003) clpP of Streptococcus salivarius is a novel member of the dually regulated class of stress response genes in gram-positive bacteria. J Bacteriol 185(2):683–687
Chastanet A, Prudhomme M, Claverys JP, Msadek T (2001) Regulation of Streptococcus pneumoniae clp genes and their role in competence development and stress survival. J Bacteriol 183(24):7295–7307
Derré I, Rapoport G, Msadek T (1999) CtsR, a novel regulator of stress and heat shock response, controls clp and molecular chaperone gene expression in gram-positive bacteria. Mol Microbiol 31:117–132
Derré I, Rapoport G, Msadek T (2000) The CtsR regulator of stress response is active as a dimer and specifically degraded in vivo at 37 degrees. Mol Microbiol 38(2):335–347
Fiocco D, Capozzi V, Collins M, Gallone A, Hols P, Guzzo J, Weidmann S, Rieu A, Msadek T, Spano G (2010) Characterization of the CtsR stress response regulon in Lactobacillus plantarum. J Bacteriol 192(3):896–900
Flahaut S, Frere J, Boutibonnes P, Auffray Y (1997) Relationship between the thermotolerance and the increase of DnaK and GroEL synthesis in Enterococcus faecalis ATCC19433. J Basic Microbiol 37(4):251–258
Frees D, Qazi SN, Hill PJ, Ingmer H (2003) Alternative roles of ClpX and ClpP in Staphylococcus aureus stress tolerance and virulence. Mol Microbiol 48(6):1565–1578
Frees D, Savijoki K, Varmanen P, Ingmer H (2007) Clp ATPases and ClpP proteolytic complexes regulate vital biological processes in low GC, gram-positive bacteria. Mol Microbiol 63:1285–1295
Ibrahim YM, Kerr AR, Silva NA, Mitchell TJ (2005) Contribution of the ATP-dependent protease ClpCP to the autolysis and virulence of Streptococcus pneumoniae. Infect Immun 73(2):730–740
Karatzas KA, Wouters JA, Gahan CG, Hill C, Abee T, Bennik MH (2003) The CtsR regulator of Listeria monocytogenes contains a variant glycine repeat region that affects piezotolerance, stress resistance, motility and virulence. Mol Microbiol 49(5):1227–1238
Lemos JA, Burne RA (2002) Regulation and physiological significance of ClpC and ClpP in Streptococcus mutans. J Bacteriol 184:6357–6366
Miethke M, Hecker M, Gerth U (2006) Involvement of Bacillus subtilis ClpE in CtsR degradation and protein quality control. J Bacteriol 188(13):4610–4619
Nair S, Derré I, Msadek T, Gaillot O, Berche P (2000) CtsR controls class III heat shock gene expression in the human pathogen Listeria monocytogenes. Mol Microbiol 35(4):800–811
Narberhaus F (1999) Negative regulation of bacterial heat shock genes. Mol Microbiol 31(1):1–8
Oliveira NEM, Abranches J, Gaca AO, Laport MS, Damaso CR, Bastos MCF, Lemos JA, Marval MG (2011) clpB, a class III heat-shock gene regulated by CtsR, is involved in thermotolerance and virulence of Enterococcus faecalis. Microbiology 157:656–665
Paulsen IT, Banerjein L, Myers GSA, Nelson KE, Seshadri R, Read TD, Fouts DE, Eisen JA, Gill SR, Heidelberg JF, Tettelin H, Dodson RJ, Umayam L, Brinkac L, Beanan M, Daugherty S, DeBoy RT, Durkin S, Kolonay J, Madupu R, Nelson W, Vamathevan J, Tran B, Upton J, Hansen T, Shetty J, Khouri H, Utterback T, Radune D, Ketchum KA, Dougherthy BA, Fraser CM (2003) Role of mobile dna in the evolution of vancomycin-resistant Enterococcus faecalis. Science 299(5615):2071–2074
Tao L, Biswas I (2013) ClpL is required for folding of CtsR in Streptococcus mutans. J Bacteriol 195(3):576–584
Varmanen P, Vogensen FK, Hammer K, Palva A, Ingmer H (2003) ClpE from Lactococcus lactis promotes repression of CtsR-dependent gene expression. J Bacteriol 185(17):5117–5124
Woo KM, Kim KI, Goldberg AL, Ha DB, Chung CH (1992) The heat-shock protein ClpB in Escherichia coli is a protein-activated ATPase. J Biol Chem 267(28):20429–20434
Acknowledgments
This study was supported by Brazilian grants [Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ—Grant E-26/112.649/2012), Conselho Nacional do Desenvolvimento Científico e Tecnológico (CNPq—Grant 476119/2012-0), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES- Grant Proex 23038.001255/2011-29)] and by NIH/NIDCR award DE019783.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ana Paula Vaz Cassenego and Naira Elane Moreira de Oliveira have contributed equaly for the development of this work.
Rights and permissions
About this article
Cite this article
Cassenego, A.P.V., de Oliveira, N.E.M., Laport, M.S. et al. The CtsR regulator controls the expression of clpC, clpE and clpP and is required for the virulence of Enterococcus faecalis in an invertebrate model. Antonie van Leeuwenhoek 109, 1253–1259 (2016). https://doi.org/10.1007/s10482-016-0727-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-016-0727-0