Abstract
HIV remains a threat to global public health, disproportionately affecting countries across Sub-Saharan Africa. Although treatment and access to care have improved, prevention remains critical to ending new HIV infections by 2030. A variety of prevention strategies exist, yet their effectiveness is difficult to measure and variable due to the nature of the interventions and vulnerability of the intervention during implementation. This systematic review of 51 studies synthesizes data on the implementation and evaluation of evidence-based HIV prevention interventions across Sub-Saharan Africa. Studies were included if they occurred between January 1, 2008, and December 31, 2019, inclusive in Sub-Saharan Africa, were written in English, implemented an HIV prevention intervention in the field, and had reportable results. Using a modified social-ecological model for HIV prevention, we divided studies into three categories: interventions targeting health systems (n = 16), communities (n = 8), and individuals (n = 27). Across all categories, the data emphasized preventing mother-to-child transmission (PMTCT), medical interventions, and psychosocial interventions. The most successful programs bundled several interventions that were integrated into the health system. There is a notable lack of interventions targeting key populations and there are significant rates of loss to follow up (LTFU) across many studies. This review provides insight into the prioritization of evidence-based HIV prevention interventions across Sub-Saharan Africa.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Human Immunodeficiency Virus (HIV) is a major public health challenge worldwide, with more than 38 million estimated to be living with HIV in 2019 [1]. Successful implementation of prevention strategies, access to testing, and linkage to treatment have resulted in a 23% decline in new infections worldwide since 2010 [1]. While this is largely attributed to HIV treatment and improvements in care quality and accessibility in recent years, prevention remains critical to ending new HIV infections [2]. HIV primary prevention strategies include condom use, harm reduction programs such as needle exchanges for individuals who inject drugs, pre-exposure prophylaxis (PrEP), behavioral interventions to reduce risk, and programs to diminish exposure in healthcare settings [3]. Secondary prevention strategies, which block integration of the virus once exposed, include voluntary male medical circumcision (VMMC), post exposure prophylaxis (PEP), and prevention of vertical transmission in pregnant and/or breastfeeding women [3, 4]. An additional effective prevention strategy is universal coverage of treatment given data supporting undetectable = untransmittable (U = U) to sexual partners [5].
HIV prevention interventions can represent large scale, multi sector, public health initiatives that are best employed utilizing a local population approach, which centers programs around collaborations between governments, local civil society organizations, and local communities [4]. Multidisciplinary teams achieve better and more expansive results, reinforcing the importance of strengthening collaborations between governments, civil society organizations, and community actors [4, 6]. Yet, real-world evaluations are challenged by the difficulty in quantifying the efficacy of any single intervention within a health program and large gaps exist in measuring the sustainability and accessibility of programs [4]. The monitoring and evaluation strategies for health programs that do exist often focus predominantly on process outputs, while effectiveness and impact has traditionally been more challenging to measure. Additionally, the counterfactual of disease (i.e., the prevention of disease) is challenging to measure without well-established longitudinal cohorts. The heterogeneity of affected populations requires locally tailored solutions accessing key populations such as sex workers, men who have sex with men (MSM), and IV drug users, yet how these interventions apply across populations with differing health infrastructure is difficult to quantify [4, 7].
Countries across Sub-Saharan Africa are disproportionally affected by HIV, recording more than 70% of the global burden [8]. Among the 20.7 million people living with HIV (PLWH) in Sub-Saharan Africa, 73% of adults and 58% of children are on antiretroviral therapy. Despite improving access to this therapy in addition to improved testing, HIV incidence in Sub-Saharan Africa continues to rise, necessitating improved prevention efforts [9]. Improved prevention of HIV in Sub-Saharan Africa could have a large impact on reducing the global HIV burden. While appreciating the diversity of Sub-Saharan Africa and the differing challenges neighboring regions face, reviewing this literature in whole can provide valuable lessons for impactful future work. This paper aims to collate HIV prevention interventions in countries across Sub-Saharan Africa to inform HIV policy makers, governments, civil society organizations, and local community members of effective strategies and lessons learned. Moreover, this review can support the development and implementation of data driven prevention programs.
Methods
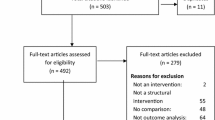
We performed a systematic review of studies examining interventions along the HIV care cascade. A query of search terms was completed in PubMed, Embase, Scopus, Wed of Science, and the Cochrane Library using a four-concept search focused on HIV/AIDS, HIV Care Cascade, Service/Program delivery, and Sub-Saharan Africa. Search strategy concepts were developed in collaboration with a professional librarian (KL) and the Johns Hopkins University Ending AIDS in West Africa (EAWA) study team (CL, GT, BH, EH, SR). An overview of the search strategies is available in Appendix 1. Studies published between January 1, 2008 and December 31, 2019 were screened for inclusion. Figure 1 shows the PRISMA diagram for systematic review stages, including the number of studies excluded and reasons for exclusion. Studies were categorized into one of the stages of the HIV care cascade (prevention, testing/diagnosis, linkage to care/antiretroviral initiation, or adherence) and each stage was treated as an independent, focused systematic review (CL, GT, BH, EH, SR). Only studies focused on HIV prevention were included in this review.
Study screening
Our objective was to evaluate studies that implemented an intervention to improve prevention strategies and examine novel prevention techniques. Articles initially underwent a title review by two authors (CL, GT, BH, EH, SR). Titles were included if they mentioned an HIV prevention intervention such as HIV counseling and testing, prevention of vertical transmission (also referred to as mother-to-child transmission), pre-/post-exposure prophylaxis, medical circumcision, and other interventions. Titles were excluded if they were not written in English or did not mention the implementation of an HIV prevention intervention. After passing title review, studies underwent abstract and full text review by a single author (CL, GT, BH, EH, SR). If a study met any of the following criteria, it was excluded: an intervention was not implemented, the study did not report results (i.e. study protocols, feasibility studies), PLWH were not the focus of the study, the study did not occur in Sub-Saharan Africa, the intervention was not on the care cascade, the study was a simulation (i.e., not a real-world evaluation but rather a modeling activity), the study was not implemented in the field, or if the study was descriptive in nature and did not report implementation outcomes.
Data extraction and conceptual framework
Primary data was used for data extraction, which included: country of study, type of intervention implemented, and key findings and conclusions (SFR, ADP, BH). The conceptual framework is based upon a modified social ecological model that underscores the multiple levels of HIV vulnerability requiring targeted multi-level prevention program implementation [10]. In our model, we stratified prevention interventions into three levels: those focused on health system factors (n = 16), those targeting strengthening communities (n = 8), and those targeting individual vulnerabilities (n = 27) (Fig. 2) (SFR, ADP, BH). Within each category, interventions were further divided based upon their topic area: prevention of mother to child transmission (PMTCT), medical interventions, and behavioral interventions (Fig. 3).
Results
Context
Fifty-one HIV prevention studies were included in the review. Nearly one third (n = 16/51) of studies examined structural or systems level interventions. Among these, ten found positive results suggestive of HIV prevention efficacy, two found mixed results, one found negative results in which the intervention was associated with worse outcomes, and three studies were not assessed against a reference group. Community based HIV prevention studies represented only 15% of interventions (n = 8/51). Among these, six studies found positive results, one found mixed outcomes, and one found no measurable intervention effect. Overall, more than 50% of interventions (n = 27/51) were implemented at the individual level. Among these interventions, twelve found encouraging results in which the intervention was associated with an increase in HIV prevention behaviors or a decrease in HIV risk behaviors, five had mixed results or no intervention efficacy, and ten had outcomes that were not assessed against a reference group (i.e. pilot or feasibility studies).
Interventions targeted prevention of HIV transmission utilizing several intervention strategies. Twelve utilized biomedical interventions such as PrEP, PEP, contraceptive use, and VMMC [11,12,13,14,15,16,17,18,19,20,21,22]; nine utilized integration of care services [23,24,25,26,27,28,29,30,31]; seven studies utilized HIV and/or family planning counseling [21, 32,33,34,35,36,37]; five studies utilized risk reduction counseling [38,39,40,41,42]; six interventions targeted improvement of follow up [43,44,45,46,47,48]; four utilized peer support [49,50,51,52]; four utilized quality improvement [53,54,55,56]; two examined partner involvement [57, 58]; two utilized psychosocial support/motivational interviewing [59, 60]; and one examined youth-based services [61].
Thirteen studies were conducted in South Africa, seven in Malawi, five each in Democratic Republic of Congo (DRC) and Kenya, four each in Uganda and Zambia, two each in Ethiopia and Tanzania, and one each in Burkina Faso, Cameroon, Mozambique, and Zimbabwe. Five studies were conducted across multiple sites (Table 1).
Health system level interventions
Sixteen studies addressed HIV prevention through systems level interventions, defined as improving the function of an organization and the delivery of its services to the community. Thirteen studies [23,24,25,26,27,28,29,30,31, 53,54,55,56] targeted vertical transmission or PMTCT, one study [11] utilized the medical intervention of PrEP while two studies [33, 61] employed the psychosocial interventions of HIV counseling and family planning and youth friendly healthcare services (Fig. 2).
Integration into PMTCT service
Among the PMTCT interventions, nine studies [23,24,25,26,27,28,29,30,31] focused on integrating specialized HIV care into routine antenatal and/or pediatric care while four studies [53,54,55,56] targeted organizational level quality improvement. Among the studies focused on integrating specialized HIV care into routine antenatal and/or pediatric care, four found improvement in key prevention indicators of antenatal follow up rate, CD4 + test rate, baby PCR testing, initiation of HAART, and initiation of AZT [24, 26, 28, 30]; two found mixed results of improved service coverage but poor follow up [23, 27]; and one found a decrease in the efficacy of HIV care when services were decentralized to antenatal clinics [25].
Four strategies stood out as improving the uptake of HIV services to prevent mother-to-child transmission of HIV. The first was implementation of national guidelines to support PMTCT interventions in antenatal care, the second focused on the delivery of high-quality services within health facilities, the third included community engagement to promote male partner engagement in both antenatal care and PMTCT services, while the last used community health care workers (CHW) to identify women who would benefit from antenatal services, which was followed by directing them to care.
The four studies that addressed HIV care quality improvement [53,54,55,56] all yielded positive results in outcome indicators of facility delivery, linkage to care, antenatal care visit completion, antiviral (ART) initiation, percentage of HIV exposed infants (HEI) issued antiretroviral prophylaxis, infant and young child feeding (IYCF) uptake, survival, and/or male partner involvement. Interventions were aimed at strengthening systems that collected PMTCT data, strengthening facility/community linkages through community engagement, improving outcomes of male partner involvement in antenatal care, strengthening HIV commodities and increasing community health worker engagement, and reorganizing service delivery to integrate nutrition and IYCF.
Integration into other routine care service
Integration of HIV prevention interventions into other routine services was identified as a highly effective strategy. For example, integrating PrEP into routine family planning services found that adherence and incidence improved in the intervention group [11], while another study randomized CHW to a one-week training intervention and found that participants in the intervention arm were significantly more likely to receive partner HIV testing [33]. Rosenberg et al. assessed the integration of youth friendly services, defined as spaces tailored to the needs of youth and providers with specific adolescent care training, to address the unique challenges adolescent girls and young women (AGYW) face in accessing HIV related health services. They found that those receiving AGYW services were more likely to receive testing, contraception, and condoms when compared to the standard of care [61].
Community-based interventions
Eight studies utilized community-based HIV interventions, defined as interventions in which members of a given community serve a direct role in the intervention. Four studies [43, 49,50,51] examined interventions aimed at PMTCT, one study utilized a medical intervention of ART combined with counseling-based interventions [21], and three studies utilized psychosocial interventions [32, 38, 39].
Community PMTCT
Several studies engaged community health workers (CHW) to improve antenatal care appointment adherence, increase ART adherence, and encourage partner testing. Two studies utilized peer mentors instead of CHW [50, 51]. Two interventions found encouraging results [43, 50], one found mixed results [51], and one found no impact on behaviors associated with prevention [49].
Among the CHW interventions, Herlihy et al. found that community-based follow up increased CD4 testing, ART initiation among pregnant women, and HIV testing among infants [43]. Peltzer et al. found that “Protect Your Family” risk reduction sessions in addition to standard of care were not associated with differences in infant HIV status at 6 weeks and at 12 months, maternal adherence at 32 weeks, reported infant adherence at 6 weeks, or knowledge of PMTCT or HIV [49].
In the peer-support studies, Phiri et al. found that peer-support, education, and tracing were associated with increased uptake and retention in option B+ (a maternal ART program) in both facility-based and community-based settings when compared to standard of care, with a slightly greater effect when implemented within the community [50]. Richter et al. found that peer-mentorship from other woman living with HIV (WLH) was associated with an increased likelihood of encouraging partner testing and a reduced likelihood of depressed mood and infant stunting. However, the intervention was associated with significantly reduced adherence to ART during pregnancy when compared to standard of care [51].
Community medical intervention
Utilizing a counseling-based intervention with ART, Hayes et al. randomized groups to receive a combination prevention intervention with ART, a combination prevention intervention with ART provided according to local guidelines, or standard of care [21]. Both intervention groups had positive results, although implementation with local guidelines had a greater reduction in adjusted incidence rate and improved viral suppression at 24 months when compared to the prevention intervention with ART alone.
Community psychosocial interventions
The three psychosocial interventions included the use of a mobile job aid to assist CHW facilitation and two risk reduction counseling interventions, with two studies finding positive and one study finding mixed results. Among those finding positive results, Agarwal et al. utilized a mobile job aid to assist CHW facilitation of family planning and HIV counseling services, which demonstrated a 15% increase in HIV testing among past intervention participants compared to new users [32]. Karamagi et al. implemented and tested a quality improvement (QI) for behavioral change model utilizing a combination of community health workers, local leaders, and local peers, which resulted in a decreased proportion of AGYW reporting multiple sex partners and transactional sex [39]. Jones et al. found mixed results when implementing an intervention utilizing CHWs to examine the efficacy of a pre-existing, evidence-based intervention with the QI teams targeting high-risk AGYW. They found consistent condom use increased, intimate partner violence decreased, but the intervention failed to reduce alcohol consumption [38].
Individual interventions
Twenty-seven studies addressed HIV prevention at the individual level. Of these, eleven studies [12, 34,35,36, 44,45,46,47,48, 57, 58] targeted PMTCT; nine focused on medical interventions such as PrEP uptake [13,14,15,16,17,18], PEP [20], VMMC [19], and contraceptive use [22] and seven used psychosocial interventions, such as risk reduction counseling [40,41,42], peer support [52], motivational interviewing [59], counseling [60] and family planning [37].
Individual PMTCT
Among the studies targeting PMTCT, there were interventions aimed at improving antenatal care appointment attendance, adherence to ART, and partner engagement in antenatal care.
Among the five studies targeting improving antenatal visit follow up, three [46,47,48] implemented interventions, while two [44, 45] described reasons for attrition along the PMTCT cascade. Kassaye et al. used SMS messaging to improve retention in care through direct messaging between HIV positive mothers and CHWs and educational reinforcement messaging. There was no difference in ART use between intervention and control arms, but there was a decrease in the number of HIV positive infants at 6 weeks [48]. Yotebieng et al. (2016, 2016) compensated their intervention group with increasing payments for attendance at scheduled clinic visits and accepted PMTCT services, which resulted in a significant improvement in retention in care. In a follow up study, the same intervention was implemented to assess its efficacy for ART adherence, which found a decreased proportion of participants with undetectable viral loads at 6 weeks post-partum, revealing compensation was not effective for this outcome measure [46, 47]. In assessing the perceptual factors associated with loss to follow up (LTFU), Yotebieng et al. (2017) found that LTFU was less likely if women believed HIV was a “very serious” problem for her baby, if women believed it would be “very likely” to pass HIV to their baby if she did not take medication, and if she anticipated cost would be a barrier to attending clinic visits [45]. These factors were antagonistic to the cash incentives Yotebieng utilized in their previous interventions. Finally, Fayorsey et al. evaluated the effectiveness of active patient follow up compared to standard of care in order to identify challenges associated with treatment attrition and found that attrition along the PMTCT cascade is largely due to pregnancy complications, infant deaths, and transferring out of care [44].
Four studies assessed ART adherence in PMTCT. Using a combination of a peer mentoring program and “Mamekhava”, a culturally adapted cognitive behavioral intervention, Futterman et al. compared adherence to PMTCT practices among HIV positive pregnant women at a control site and an intervention site [35]. Self-reports of adherence to PMTCT practices were over 90% at both study sites, although Mamekhaya participants had greater adherence at follow-up visits and increased HIV knowledge scores. Fatti et al. employed community health workers, who provided individual HIV prevention counseling through three monthly home or clinic-based HIV tests (couples and individual)[34]. Hamela et al. utilized traditional birth attendants (TBAs) trained to administer Nevirapine (NVP) at home visits to increase the number of women receiving NVP after delivery [36]. Bisio et al. assessed the factors influencing ART compliance, and found that older age, telephone availability, and occupation were associated with improved adherence [12].
Two studies utilized partner involvement interventions [57, 58] to improve PMTCT cascade care. Sifunda et al. employed a two phase, two condition longitudinal study, finding that a lack of partner involvement correlated with increased depressive symptoms and significantly more HIV-infected infants and infant deaths [58]. Nyondo et al. randomized study participants to either send an invitation card to their male partners to accompany them to PMTCT services, or to standard of care [57]. They found that women in the invitation card group were 50% more likely to be accompanied by their male partners than those in the standard of care group.
Individual medical interventions
Biomedical interventions included PrEP [13,14,15,16,17,18], PEP [20], VMMC [19], and contraception use [22]. Amico et al. assessed adherence approaches in four PrEP clinical trials, which all had variable adherence rates despite adherence-specific education [13]. Psaros et al. also measured adherence as an ancillary study within an efficacy trial of Tenofovir PrEP and employed a counseling intervention for those with < 80% adherence measured by unannounced pill counts [17]. The mean adherence increased significantly in the month after the first intervention. Eakle et al. assessed adherence as an outcome measure of a treatment and prevention program for female sex workers, where women were given PrEP if HIV negative and ART if HIV positive [15]. LTFU was significantly more likely among those taking PrEP than those taking ART.
Assessing challenges with attrition in PrEP trials, Wynne et al. found that only 3% of visits were missed, with reasons including participants migrating for better employment, time constraints resulting from commercial sex work, and perceptions of excessive visit frequency given negative HIV status [18]. Assessing the efficacy of PrEP itself, Baeten et al. implemented a prospective study focused on PrEP and ART initiation for heterosexual, serodiscordant couples and found a 96% reduction in HIV incidence when compared to the simulated incidence [14]. Haberer et al. employed an ART monitoring and adherence counseling intervention to improve compliance [16].
Abrahams et al. utilized PEP telephone support for sexual assault survivors, measuring adherence as the primary outcome [20]. The intervention group received both a leaflet and phone calls, while the control arm only received a leaflet. Though not statistically significant, adherence was higher in the intervention arm. Assessing medical circumcision as prevention, Odeny et al. utilized an SMS intervention to improve the proportion of men returning for follow up after medical male circumcision [19]. Those that received the SMS intervention had significantly higher follow up rates than those in the control arm. Stephenson et al. assessed contraceptive use uptake among serodiscordant and concordant couples and found that after the intervention, contraceptive use was almost universal [22].
Individual psychosocial interventions
Three studies utilized risk reduction counseling [40,41,42]. Kalichman et al. compared the efficacy of a 60-minute risk reduction session to a 20-minute HIV- sexually transmitted infection (STI) educational session, finding that patients in the risk reduction group were less likely to obtain an STI [40]. Miller et al. randomized participants to standard counseling, five behavioral intervention sessions, or behavioral intervention plus 12 weeks of antiretrovirals and found that those in all arms had a decrease in the number of condomless sex acts over a week span, with an increased percentage of those in the behavioral intervention plus antiretroviral group suppressed than those in the behavioral group or standard counseling group [42]. Maman et al. used counseling to change pregnant women’s sexual behaviors to reduce risk of HIV and MTCT with those randomized to the intervention arm receiving enhanced HIV pre- and post-test counseling, legal support and access to support groups [41]. There were no intervention effects on incident STIs for either HIV negative or HIV positive participants, although the intervention was associated with a decrease in risk of past 30-day inconsistent condom use at nine-months among HIV negative women. The intervention did not affect inconsistent condom use among HIV positive women.
Konate et al. utilized peer support, which consisted of peer education on HIV prevention, communication sessions, condom distribution, regular HIV counseling and testing, and free ART if needed, among high- risk women [52]. While condom use at last sexual intercourse increased, HIV incidence tended to be higher during the first eight months of follow-up than thereafter. Corneli et al. utilized motivational interviewing to reduce HIV transmission risk behaviors, particularly in acute HIV infection when individuals are most infectious [59]. Patients randomized to receive the intervention (education sessions plus motivational interviewing) felt the counseling provided them with necessary information and emotional support at the time of diagnosis, but there were few differences regarding self-reported sexual behaviors between the intervention and control arms. Jani et al. utilized a psychosocial intervention to reduce HIV risk among migrant adolescents, which included 3-months of client centered, individual, group, and creative arts counseling [60]. HIV testing rates increased and knowledge of a place to test for HIV increased for both females and males, with a greater effect for males for both outcomes. Napierala utilized HIV counseling and family planning, which assessed the effect of a two month male and female condom intervention on female condom acceptability and use among sexually active women, which found increases in female condom use [37].
Discussion
This systematic review identified 51 studies that implemented and evaluated HIV prevention strategies. The most impactful interventions implemented system level interventions that combined several HIV prevention foci (e.g. both biomedical and behavioral). Among systems level interventions that found results associated with an increase in HIV prevention behaviors or knowledge, several themes emerged. First, successful interventions often targeted multiple junctions along the HIV care cascade. For example, South Africa’s National Department of Health leveraged data-driven quality improvement process indicators to identify facility bottlenecks along the PMTCT care continuum, for example the proportion of HIV positive women receiving CD4 + testing to proportion initiated on AZT [24]. These indicators were reported quarterly, using “stoplight” visual aids and resulted in significant improvement across all seven indicators. The second theme found was bundled interventions: for example, Gamell et al. found success with a bundled intervention that had four separate strategies -- implementation of a dedicated unit for PMTCT, an electronic medical record system, hospital-based HIV testing, and early infant diagnosis [26]. The third key lesson was shifting the focus of service initiation from the individual patient to the health system. Moses et al. found that implementation of opt-out testing within antenatal care visits led to a 99% acceptance of HIV testing among pregnant women when compared to an opt-in model [30]. Bachanas et al. found that provider-initiated counseling on HIV prevention strategies greatly increased prevention knowledge among HIV positive patients [33]. Malaju et al. found that provider-initiated counseling on HIV testing was associated with increased likelihood of attending all antenatal care visits [28]. Finally, integration of relevant care services improved the likelihood of uptake of HIV prevention interventions. For example, Mansoor et al. demonstrated significant uptake of topical PrEP when offered within family planning clinics, and Rosenberg et al. found improved uptake of HIV testing, condoms, and contraception among youth when HIV prevention was integrated within a youth-friendly behavioral health center when compared to standard of care clinics [11, 61].
LTFU was a significant challenge among both successful and unsuccessful interventions for community-based and individual level interventions. Rates of LTFU were as high as 71% in some studies [15]. A study of 44,377 women living with HIV revealed that more than 1 in every 3 women (35%) were lost to follow up [23]. Unexpectedly, subsequent analysis found that Option B+, an intervention to reduce MTCT, was associated with increased rates of attrition in the intervention group compared to the standard of care. LTFU is thus not only challenging from an evaluation perspective, but may also signal challenges with continued engagement strategies. Innovative solutions to decrease attrition and thus maintain engagement in care must be sought. Yotebieng et al. examined the efficacy of conditional crash transfer as a means to overcome this attrition [45,46,47]. While small cash transfers to cover cost of transport to the clinic increased the likelihood of retention, nearly one in five participants in the intervention group were still lost to follow up.
The majority of studies focused on interventions targeting the prevention of HIV transmission via mother to child transmission or heterosexual sexual transmission. No studies examined other modes of transmission such as sex between men, injection drug use (IDU), or nosocomial infections. While these behaviors may not be homogeneously prevalent among unique geographic and demographic groups in Sub-Saharan Africa, these high-risk groups may represent a hidden or missed opportunity [62]. One study in Bangkok identified that acute HIV detection and immediate treatment reduced onward transmission by 89% among MSM [63]. Furthermore, strategies such as providing a safe blood supply to prevent nosocomial transmission are found to be more cost effective compared to media campaigns and condom distribution [64]. Additionally, IDU interventions such as needle exchange and drug treatment programs demonstrate the benefit of changing baseline drug-related and sex-related risk behaviors following individual participation in an outreach-based HIV risk reduction intervention.
Most studies evaluated intervention efficacy through assessment of outcomes indirectly associated with HIV prevention. These outcomes included knowledge of HIV transmission and prevention, HIV testing, initiation of ART, self-report of HIV prevention behaviors, and adherence to ART via self-report or pill count, among many others. Few studies analyzed measures directly associated with HIV prevention such as incidence. While measurement of variables directly associated with HIV transmission is costly and may be logistically unfeasible in some settings, the efficacy of many of the interventions for prevention of new infections requires assumptions that may not always hold. For example, Yotebieng et al. found that conditional cash transfer increased the likelihood of retention and engagement in care among pregnant women with HIV [47]. However, in a subsequent study, though compensation was indeed associated with reduced attrition, the intervention had no impact on adherence to ART or HIV viral load, which are outcome variables arguably more directly associated with prevention of HIV transmission. Additionally, the heterogeneity of outcome variables across studies limits the comparison of efficacy between different interventions, limiting the practical utility of study results for policymakers and funders. Future studies should prioritize measures directly or closely associated with HIV prevention. Adoption of standardized outcome indicators may unveil a lack of efficacy for interventions previously presumed effective and better identify transmission-reducing interventions in the future.
Limitations
This review of studies is not without limitations. Effective HIV prevention programs are informed by the local context, making generalization of programs across settings of limited value. Although all studies took place in Sub-Saharan Africa, the region includes a diverse collection of countries and settings, which limits the generalizability of results even within the region. Factors associated with specific contexts should be acknowledged and accounted for when comparing results of studies within the review and when using the review to inform future programs and policies in various settings. The nature of the studies in the review itself is another important limitation and consideration. Although observational and other non-intervention studies were excluded, there is heterogeneity in the rigor of study methods ranging from randomized controlled trials to non-randomized and pilot studies to correlational data. This produces a disparity in the strength of study results, thus making conclusions challenging to compare and with varying levels of utility. Additionally, there is a lack of data on the cost effectiveness of interventions.
Conclusions
Our systematic review provides meaningful insight into successful HIV prevention programs in Sub-Saharan Africa. Our findings highlight the demonstrable challenges of implementing and evaluating the effectiveness of HIV prevention interventions. The most effective interventions were multifaceted, integrated into the health system, and included the following components: national guidelines to standardize quality care, specialized services bundled into routine care, and community engagement often utilizing CHWs. There remain two significant areas to be addressed: the first is identifying solutions to overcome significant rates of LTFU and the second is targeting HIV prevention interventions to key-populations that would otherwise missed.
Data Availability
Data are fully available without restriction. No primary data were collected as part of this manuscript. The reviewed data are available in all references cited.
Code Availability
Not applicable.
References
November 25 CSH govDate last updated:. 2020. Global Statistics [Internet]. HIV.gov. 2020 [cited 2021 Feb 22]. Available from: https://www.hiv.gov/hiv-basics/overview/data-and-trends/global-statistics.
Africa NRC (US) P on D and RP for AA in S-S, Cohen B, Trussell J. Primary HIV-Prevention Strategies [Internet]. Prev. Mitigating AIDS Sub-Sahar. Afr. Res. Data Priorities Soc. Behav. Sci. National Academies Press (US); 1996 [cited 2021 Feb 25]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK232826/.
Beyrer C. Consolidated Guidelines on Hiv Prevention, Diagnosis, Treatment and Care for Key Populations [Internet]. Place of publication not identified: World Health Organization; 2016 [cited 2021 Feb 22]. Available from: http://proxy.library.carleton.ca/loginurl=https://www.deslibris.ca/ID/10063272.
UNAIDS. Prevention gap report [Internet]. 2016 [cited 2021 Feb 25]. Available from: https://www.unaids.org/en/resources/documents/2016/prevention-gap.
February 24 CSH govDate last updated:. 2020. HIV Treatment as Prevention [Internet]. HIV.gov. 2020 [cited 2021 Feb 25]. Available from: https://www.hiv.gov/tasp.
Magnus M, Castel A. Breaking Down the Siloes: Developing Effective Multidisciplinary HIV Research Teams. AIDS Behav. 2016;20:273–80.
Torrone EA, Levandowski BA, Thomas JC, Isler MR, Leone PA. Identifying gaps in HIV prevention services. Soc Work Public Health. 2010;25:327–40.
Kharsany ABM, Karim QA. HIV Infection and AIDS in Sub-Saharan Africa: Current Status, Challenges and Opportunities. Open AIDS J. 2016;10:34–48.
HIV and AIDS in East and Southern Africa regional overview [Internet]. Avert. 2019 [cited 2021 Oct 18]. Available from: https://www.avert.org/professionals/hiv-around-world/sub-saharan-africa/overview.
Baral S, Logie CH, Grosso A, Wirtz AL, Beyrer C. Modified social ecological model: a tool to guide the assessment of the risks and risk contexts of HIV epidemics. BMC Public Health. 2013;13:482.
Mansoor LE, Yende-Zuma N, Baxter C, Mngadi KT, Dawood H, Gengiah TN, et al. Integrated provision of topical pre-exposure prophylaxis in routine family planning services in South Africa: a non-inferiority randomized controlled trial. J Int AIDS Soc. 2019;22:e25381.
Bisio F, Masini G, Blasi Vacca E, Calzi A, Cardinale F, Bruzzone B, et al. Effectiveness of a project to prevent HIV vertical transmission in the Republic of Congo. J Antimicrob Chemother. 2013;68:1862–71.
Amico KR, Mansoor LE, Corneli A, Torjesen K, van der Straten A. Adherence Support Approaches in Biomedical HIV Prevention Trials: Experiences, Insights and Future Directions from Four Multisite Prevention Trials. AIDS Behav. 2013;17:2143–55.
Baeten JM, Heffron R, Kidoguchi L, Mugo NR, Katabira E, Bukusi EA, et al. Integrated Delivery of Antiretroviral Treatment and Pre-exposure Prophylaxis to HIV-1-Serodiscordant Couples: A Prospective Implementation Study in Kenya and Uganda. PLoS Med. 2016;13:e1002099.
Eakle R, Gomez GB, Naicker N, Bothma R, Mbogua J, Escobar MAC, et al. HIV pre-exposure prophylaxis and early antiretroviral treatment among female sex workers in South Africa: Results from a prospective observational demonstration project. PLOS Med Public Library of Science. 2017;14:e1002444.
Haberer JE, Baeten JM, Campbell J, Wangisi J, Katabira E, Ronald A, et al. Adherence to Antiretroviral Prophylaxis for HIV Prevention: A Substudy Cohort within a Clinical Trial of Serodiscordant Couples in East Africa. PLoS Med [Internet]. 2013 [cited 2020 May 24];10. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3769210/.
Psaros C, Haberer JE, Katabira E, Ronald A, Tumwesigye E, Campbell JD, et al. An intervention to support HIV pre-exposure prophylaxis (PrEP) adherence in HIV serodiscordant couples in Uganda. J Acquir Immune Defic Syndr 1999. 2014;66:522–9.
Wynne J, Muwawu R, Mubiru MC, Kamira B, Kemigisha D, Nakyanzi T, et al. Maximizing participant retention in a phase 2B HIV prevention trial in Kampala, Uganda: The MTN-003 (VOICE) Study. HIV Clin Trials. 2018;19:165–71.
Odeny TA, Bailey RC, Bukusi EA, Simoni JM, Tapia KA, Yuhas K, et al. Text messaging to improve attendance at post-operative clinic visits after adult male circumcision for HIV prevention: a randomized controlled trial. PLoS ONE. 2012;7:e43832.
Abrahams N, Jewkes R, Lombard C, Mathews S, Campbell J, Meel B. Impact of telephonic psycho-social support on adherence to post-exposure prophylaxis (PEP) after rape. AIDS Care Taylor & Francis. 2010;22:1173–81.
Hayes RJ, Donnell D, Floyd S, Mandla N, Bwalya J, Sabapathy K, et al. Impact of a universal testing and treatment intervention on HIV incidence in Zambia and South Africa: results of the HPTN 071 (PopART) community-randomized trial. N Engl J Med. 2019;381:207–18.
Stephenson R, Vwalika B, Greenberg L, Ahmed Y, Vwalika C, Chomba E, et al. A randomized controlled trial to promote long-term contraceptive use among HIV-serodiscordant and concordant positive couples in Zambia. J Womens Health 2002. 2011;20:567–74.
Ahoua L, Tiendrebeogo T, Arikawa S, Lahuerta M, Aly D, Journot V, et al. PMTCT care cascade and factors associated with attrition in the first four years after Option B + implementation in Mozambique. Trop Med Int Health TM IH. 2020;25:222–35.
Bhardwaj S, Barron P, Pillay Y, Treger-Slavin L, Robinson P, Goga A, et al. Elimination of mother-to-child transmission of HIV in South Africa: rapid scale-up using quality improvement. South Afr Med J Suid-Afr Tydskr Vir Geneeskd. 2014;104:239–43.
Edmonds A, Feinstein L, Okitolonda V, Thompson D, Kawende B, Behets F. Decentralization Does Not Assure Optimal Delivery of PMTCT and HIV-Exposed Infant Services in a Low Prevalence Setting. J Acquir Immune Defic Syndr 1999. 2015;70:e130–9.
Gamell A, Glass TR, Luwanda LB, Mapesi H, Samson L, Mtoi T, et al. Implementation and Operational Research: An Integrated and Comprehensive Service Delivery Model to Improve Pediatric and Maternal HIV Care in Rural Africa. J Acquir Immune Defic Syndr 1999. 2016;73:e67–75.
Horwood C, Haskins L, Vermaak K, Phakathi S, Subbaye R, Doherty T. Prevention of mother to child transmission of HIV (PMTCT) programme in KwaZulu-Natal, South Africa: an evaluation of PMTCT implementation and integration into routine maternal, child and women’s health services. Trop Med Int Health TM IH. 2010;15:992–9.
Malaju MT, Alene GD. Assessment of utilization of provider-initiated HIV testing and counseling as an intervention for prevention of mother to child transmission of HIV and associated factors among pregnant women in Gondar town, North West Ethiopia. BMC Public Health. 2012;12:226.
Mandala J, Kasonde P, Badru T, Dirks R, Torpey K. HIV Retesting of HIV-Negative Pregnant Women in the Context of Prevention of Mother-to-Child Transmission of HIV in Primary Health Centers in Rural Zambia: What Did We Learn? J Int Assoc Provid AIDS Care. 2019;18:2325958218823530.
Moses A, Zimba C, Kamanga E, Nkhoma J, Maida A, Martinson F, et al. Prevention of mother-to-child transmission: program changes and the effect on uptake of the HIVNET 012 regimen in Malawi. AIDS Lond Engl. 2008;22:83–7.
Penda CI, Tejiokem MC, Sofeu CL, Ndiang ST, Ateba Ndongo F, Kfutwah A, et al. Low rate of early vertical transmission of HIV supports the feasibility of effective implementation of the national PMTCT guidelines in routine practice of referral hospitals in Cameroon. Paediatr Int Child Health. 2019;39:208–15.
Agarwal S, Lasway C, L’Engle K, Homan R, Layer E, Ollis S, et al. Family Planning Counseling in Your Pocket: A Mobile Job Aid for Community Health Workers in Tanzania. Glob Health Sci Pract. 2016;4:300–10.
Bachanas P, Kidder D, Medley A, Pals SL, Carpenter D, Howard A, et al. Delivering Prevention Interventions to People Living with HIV in Clinical Care Settings: Results of a Cluster Randomized Trial in Kenya, Namibia, and Tanzania. AIDS Behav. 2016;20:2110–8.
Fatti G, Shaikh N, Jackson D, Goga A, Nachega JB, Eley B, et al. Low HIV incidence in pregnant and postpartum women receiving a community-based combination HIV prevention intervention in a high HIV incidence setting in South Africa. PLoS ONE. 2017;12:e0181691.
Futterman D, Shea J, Besser M, Stafford S, Desmond K, Comulada WS, et al. Mamekhaya: a pilot study combining a cognitive-behavioral intervention and mentor mothers with PMTCT services in South Africa. AIDS Care. 2010;22:1093–100.
Hamela G, Kabondo C, Tembo T, Zimba C, Kamanga E, Mofolo I, et al. Evaluating the benefits of incorporating traditional birth attendants in HIV prevention of mother to child transmission service delivery in Lilongwe, Malawi. Afr J Reprod Health. 2014;18:27–34.
Napierala S, Kang M-S, Chipato T, Padian N, van der Straten A. Female Condom Uptake and Acceptability in Zimbabwe. AIDS Educ Prev. 2008;20:121–34.
Jones D, Weiss SM, Arheart K, Cook R, Chitalu N. Implementation of HIV Prevention Interventions in Resource Limited Settings: The Partner Project. J Community Health. 2014;39:151–8.
Karamagi E, Sensalire S, Nabwire J, Byabagambi J, Awio AO, Aluma G, et al. Quality improvement as a framework for behavior change interventions in HIV-predisposed communities: a case of adolescent girls and young women in northern Uganda. AIDS Res Ther. 2018;15:4.
Kalichman SC, Cain D, Eaton L, Jooste S, Simbayi LC. Randomized Clinical Trial of Brief Risk Reduction Counseling for Sexually Transmitted Infection Clinic Patients in Cape Town, South Africa. Am J Public Health American Public Health Association. 2011;101:e9–17.
Maman S, Moodley D, McNaughton-Reyes HL, Groves AK, Kagee A, Moodley P. Efficacy of Enhanced HIV Counseling for Risk Reduction during Pregnancy and in the Postpartum Period: A Randomized Controlled Trial. PLOS ONE Public Library of Science. 2014;9:e97092.
Miller WC, Rutstein SE, Phiri S, Kamanga G, Nsona D, Pasquale DK, et al. Randomized Controlled Pilot Study of Antiretrovirals and a Behavioral Intervention for Persons With Acute HIV Infection: Opportunity for Interrupting Transmission. Open Forum Infect Dis. 2019;6:ofy341.
Herlihy JM, Hamomba L, Bonawitz R, Goggin CE, Sambambi K, Mwale J, et al. Integration of PMTCT and Antenatal Services Improves Combination Antiretroviral Therapy Uptake for HIV-Positive Pregnant Women in Southern Zambia: A Prototype for Option B+? J Acquir Immune Defic Syndr 1999. 2015;70:e123–9.
Fayorsey RN, Chege D, Wang C, Reidy W, Peters Z, Syengo M, et al. Mother Infant Retention for Health (MIR4Health): Study Design, Adaptations, and Challenges With PMTCT Implementation Science Research. J Acquir Immune Defic Syndr 1999. 2016;72 Suppl 2:S137-144.
Yotebieng M, Moracco KE, Thirumurthy H, Edmonds A, Tabala M, Kawende B, et al. Conditional cash transfers improve retention in PMTCT services by mitigating the negative effect of not having money to come to the clinic. J Acquir Immune Defic Syndr 1999. 2017;74:150–7.
Yotebieng M, Thirumurthy H, Moracco KE, Edmonds A, Tabala M, Kawende B, et al. Conditional Cash Transfers to Increase Retention in PMTCT Care, Antiretroviral Adherence, and Postpartum Virological Suppression: A Randomized Controlled Trial. J Acquir Immune Defic Syndr 1999. 2016;72:S124–9.
Yotebieng M, Thirumurthy H, Moracco KE, Kawende B, Chalachala JL, Wenzi LK, et al. Conditional cash transfers increase uptake of and retention in PMTCT care: A randomized controlled trial. Lancet HIV. 2016;3:e85–93.
Kassaye SG, Ong’ech J, Sirengo M, Kose J, Matu L, McOdida P, et al. Cluster-Randomized Controlled Study of SMS Text Messages for Prevention of Mother-to-Child Transmission of HIV in Rural Kenya. AIDS Res Treat. 2016;2016:1289328.
Peltzer K, Weiss SM, Soni M, Lee TK, Rodriguez VJ, Cook R, et al. A cluster randomized controlled trial of lay health worker support for prevention of mother to child transmission of HIV (PMTCT) in South Africa. AIDS Res Ther [Internet]. 2017 [cited 2021 Feb 28];14. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5732507/.
Phiri S, Tweya H, van Lettow M, Rosenberg NE, Trapence C, Kapito-Tembo A, et al. Impact of Facility- and Community-Based Peer Support Models on Maternal Uptake and Retention in Malawi’s Option B + HIV Prevention of Mother-to-Child Transmission Program: A 3-Arm Cluster Randomized Controlled Trial (PURE Malawi). J Acquir Immune Defic Syndr 1999. 2017;75 Suppl 2:S140–8.
Richter L, Rotheram-Borus MJ, Van Heerden A, Stein A, Tomlinson M, Harwood JM, et al. Pregnant women living with HIV (WLH) supported at clinics by peer WLH: a cluster randomized controlled trial. AIDS Behav. 2014;18:706–15.
Konate I, Traore L, Ouedraogo A, Sanon A, Diallo R, Ouedraogo J-L, et al. Linking HIV Prevention and Care for Community Interventions Among High-Risk Women in Burkina Faso—The ARNS 1222 “Yerelon” Cohort. JAIDS J Acquir Immune Defic Syndr. 2011;57:50.
Akama E, Mburu M, Mutegi E, Nyanaro G, Otieno JP, Ndolo S, et al. Impact of a Rapid Results Initiative Approach on Improving Male Partner Involvement in Prevention of Mother to Child Transmission of HIV in Western Kenya. AIDS Behav. 2018;22:2956–65.
Kanyuuru L, Kabue M, Ashengo TA, Ruparelia C, Mokaya E, Malonza I. RED for PMTCT: an adaptation of immunization’s Reaching Every District approach increases coverage, access, and utilization of PMTCT care in Bondo District, Kenya. Int J Gynaecol Obstet Off Organ Int Fed Gynaecol Obstet. 2015;130(Suppl 2):68–73.
Nsubuga-Nyombi T, Karamagi E, Nabitaka L, Namukose S, Calnan J, Nyakwezi S, et al. Increasing HIV-Free Survival of Infants: Reorganizing Care Using Quality Improvement for the Optimal Health and Nutrition of HIV-Positive Women and Their Exposed Infants in Uganda. J Int Assoc Provid AIDS Care. 2019;18:2325958219857724.
Phaswana-Mafuya N, Peltzer K, Ladzani R, Mlambo G, Davids A, Phaweni K, et al. Pre- and post-intervention assessment of a PMTCT-programme-strengthening initiative in a rural area of the Eastern Cape, South Africa. Afr J AIDS Res AJAR. 2011;10:83–93.
Nyondo AL, Choko AT, Chimwaza AF, Muula AS. Invitation Cards during Pregnancy Enhance Male Partner Involvement in Prevention of Mother to Child Transmission (PMTCT) of Human Immunodeficiency Virus (HIV) in Blantyre, Malawi: A Randomized Controlled Open Label Trial. 10: PLOS ONE. Public Library of Science; 2015. p. e0119273.
Sifunda S, Peltzer K, Rodriguez VJ, Mandell LN, Lee TK, Ramlagan S, et al. Impact of male partner involvement on mother-to-child transmission of HIV and HIV-free survival among HIV-exposed infants in rural South Africa: Results from a two phase randomised controlled trial. PLoS ONE. 2019;14:e0217467.
Corneli A, Pettifor A, Kamanga G, Golin C, McKenna K, Ou S-S, et al. HPTN 062: A feasibility and acceptability pilot intervention to reduce HIV transmission risk behaviors among individuals with acute and early HIV infection in Lilongwe, Malawi. AIDS Behav. 2014;18:1785–800.
Jani N, Vu L, Kay L, Habtamu K, Kalibala S. Reducing HIV-related risk and mental health problems through a client-centred psychosocial intervention for vulnerable adolescents in Addis Ababa, Ethiopia. J Int AIDS Soc. 2016;19:20832.
Rosenberg NE, Bhushan NL, Vansia D, Phanga T, Maseko B, Nthani T, et al. Comparing Youth-Friendly Health Services to the Standard of Care Through “Girl Power-Malawi”: A Quasi-Experimental Cohort Study. J Acquir Immune Defic Syndr 1999. 2018;79:458–66.
ELOPRE L, KUDROFF K, WESTFALL AO, OVERTON ET, MUGAVERO MJ. The Right People, Right Places, and Right Practices: Disparities in PrEP access among African American Men, Women and MSM in the Deep South. J Acquir Immune Defic Syndr 1999. 2017;74:56–9.
Kroon EDMB, Phanuphak N, Shattock AJ, Fletcher JLK, Pinyakorn S, Chomchey N, et al. Acute HIV infection detection and immediate treatment estimated to reduce transmission by 89% among men who have sex with men in Bangkok. J Int AIDS Soc. 2017;20:21708.
Söderlund N, Lavis J, Broomberg J, Mills A. The costs of HIV prevention strategies in developing countries. Bull World Health Organ. 1993;71:595–604.
Funding
The authors are grateful for the support of the National Institute of Mental Health (Award Number R01 MH110358) and the National Institute of Nursing Research of the National Institutes of Health (NIH) (Award Number R01 NR016650).
Author information
Authors and Affiliations
Contributions
BH, SB and GT conceived the study; CL, GT, BH, EH, and SR developed the initial search strategy and completed the initial search, SFR, ADP, NV, and BH completed data abstraction for the selected articles and updated the search; SFR, ADP, and BH contributed to the synthesis of the results and the writing of the manuscript; all authors reviewed, edited and approved of the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethics approval
Not applicable.
Informed consent
This study is a systematic review of the peer-reviewed published literature so informed consent was not needed as no patient level were included in this study.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
Appendix 1: Overview of search strategies. (Supplementary material 2).
Appendix 2: Appendix of all HIV prevention articles included in this review. (Supplementary material 3).
Rights and permissions
About this article
Cite this article
Rapaport, S.F., Peer, A.D., Viswasam, N. et al. Implementing HIV Prevention in Sub-Saharan Africa: A Systematic Review of Interventions Targeting Systems, Communities, and Individuals. AIDS Behav 27, 150–160 (2023). https://doi.org/10.1007/s10461-022-03751-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10461-022-03751-0