Abstract
Agroforestry can improve predator recruitment by increasing the diversity of microhabitats and modifying the microclimate. Four treatment plots were defined with three tree-shading levels in a 20 years old agroforestry system combining organic vegetable crops with hybrid walnut trees. Temperature and canopy openness were recorded in each treatment in July and September 2015 and arthropods were sampled using pitfall traps at two dates (2 weeks in July and 2 weeks in September). The daily activity pattern of the main taxa was then estimated using dry pitfall traps for 7 days in July and 8 days in September. Agroforestry significantly limited the daily temperature extremes in the day and night (± 1.5 °C). We observed a significant effect of treatment on species distribution. In July, the main xerophilic species, Pseudoophonus rufipes (Coleoptera, Carabidae), was less abundant in the two most shaded plots (− 25%). Pardosa hortensis (Arachnida, Lycosidae) showed significant differences in activity-density and diurnal activity between treatments. This spider was more active between 10:00 and 14:00 in the two most shaded treatments especially in tomatoes (more than 20% of the daily activity) compared to the control (13%). The activity-density of this species was also higher in the two shaded treatments than in the control (> 20%). Our results highlight that agroforestry, by buffering climate extremes, is likely to modify predatory arthropod activity and possibly the associated services such as biocontrol.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Agroforestry systems are defined as agroecosystems associating trees with agricultural crops and/or pasture (Batish et al. 2007). In the last 20 years, they have been spreading throughout Europe and have real potential in the production of sustainable food (Eichhorn et al. 2006; Quinkenstein et al. 2009). These systems have economic and environmental benefits for farmers and society because they take advantage of complementary ecosystem and plant services (Moreno et al. 2017).
The most common agroforestry set-up is a combination of tree rows and crop alleys with adequate spacing to allow easy access for agricultural machinery (Eichhorn et al. 2006). In these systems, the tree rows are usually not cultivated and left as grassy and/or wildflower strips (Batish et al. 2007). This extra vegetation stratum increases the diversity of available niches (Collins et al. 2002; Ranjha and Irmler 2014; Saska et al. 2007), and potentially increases the associated taxonomic biodiversity (Malézieux et al. 2009; Tscharntke et al. 2011). The specificity of agroforestry systems makes them of particular interest for conservation biocontrol (CBC; Pumariño et al. 2015). CBC is defined as a strategy aimed at maintaining or increasing the local abundance of beneficials, especially using the conservation of shelter habitats (natural or not) around crops and suitable practices (Bianchi et al. 2006; Griffiths et al. 2008; Simon et al. 2010). However, a study on a temperate agroforestry apple orchard found that the increased vegetal diversity had only marginal effects on arthropod communities and CBC (Kranz et al. 2018), possibly due to a lack of specific tri trophic relationships. CBC strategies are considered key to sustainable production (Zehnder et al. 2007). This is mostly the case in organic production systems where pests cannot be regulated using synthesized chemical products. Instead these systems rely on natural pesticides and the development of new plant protection strategies based on mechanical control and local biodiversity to maximize natural CBC, as reviewed in Zehnder et al. (2007).
Several studies highlighted the impacts of recent climate change on ecosystems and agro-ecosystems (Fuhrer et al. 2014; Greenwood et al. 2016; Walther et al. 2002). In the Mediterranean area, the climate tends to be drier and hotter during summer and has higher rainfall during autumn and winter (Diffenbaugh et al. 2007; Giorgi and Lionello 2008). Arthropods are ectothermic and will thus be directly affected by these changes (Guo et al. 2009; Musolin 2007). In response, the diversity, structure and ecological traits of communities could be affected (Lindberg et al. 2002; Rainio and Niemelä 2003; Walther et al. 2002). Berthe et al. (2015) also recently showed impacts of soil warming experiments on ground beetle community in corn fields. They found reduced diversity in carabids exposed to a 2 °C heat increase.
In addition to creating new habitats, trees modify microclimate conditions as they have an impact on air circulation and provide shade, which results in differences in ground and air temperature as well as hygrometry, depending on the seasons (Chen et al. 1999; Lasco et al. 2014; Lin 2007; Pezzopane et al. 2011). Shady microclimates can modify vegetation composition (De Frenne et al. 2013) and associated wildlife (Greenwood et al. 2016). In agroforestry systems, management of tree crowns is a way to modulate microclimate to promote plant growth (Jones et al. 1998) and is likely to affect pest and prey arthropods (Guenat et al. 2017; Karungi et al. 2015; Mariño et al. 2016). Temperature and humidity are important factors driving the local density and/or activity (“activity-density”) of ground predators (Buchholz et al. 2013; Honek 2013; Saska et al. 2013).
Several factors play a role in establishing the diel activity patterns of different species and the changes in these rhythms, which are probably an expression of both physiological and ecological demands (Park 1941). Edgar (1969) focused on hunting behavior and suggested that spiders are more active in the morning because it is easier to capture prey (flies) during this period. Competition avoidance mechanisms could be involved in structuring spider diel activity (Krumpalova and Tuf 2013). Frampton et al. (2001) showed that collembola are sensitive to daily changes in temperature and habitat moisture, which could lead to changes in prey availability for upper trophic levels on a short time scale (within hours). Results of the multiple possible interactions between biotic and abiotic factors can thus affect arthropod diel activity (Dondale et al. 1972) and determine the potential chances of prey and predator individuals encountering one another (Pfannenstiel and Yeargan 2002; Vucic-Pestic et al. 2011; Weber et al. 2008).
It is already known that the daily and seasonal patterns of ground predator activity vary between open and closed habitat types, such as forest and grasslands (Krumpalova and Tuf 2013; Thiele 1977; Tuf et al. 2012). Tuf et al. (2012) characterized changes in circadian rhythm for several carabids in open versus forest habitats and concluded that epigeic carabid activity in the forest is higher mostly between 21:00 and midnight while peak activity in clear-cut areas was in the afternoon. Atienza et al. (1996) showed that in central Spain a diurnal Carabidae (Angoleum nitidus) drastically reduced its activity at midday when the soil surface temperature reached 47.5 °C on sunny days, but not on cloudy days with lower temperatures (37.5 °C). Edgar (1969) made a similar observation of the spider Pardosa amentata, which ceased diurnal activity during the hottest period of the warm days in the Netherlands.
Currently, there is no information on the possible effects of agroforestry systems on epigeal fauna diel activity. Since mature trees in an agroforestry system can modify the microclimate and buffer climatic extremes, it was hypothesized that agroforestry could thus have effects on their daily and seasonal activity. The aim of this study was to assess the impact of microclimate conditions created by trees on the main generalist predators (spiders and Carabidae) in vegetable crops. Our first hypothesis was that agroforestry pruning management strategies would affect air temperature. Secondly, we examined whether ground predator circadian rhythms are modified in these different microclimatic conditions. Thirdly, the intensity of these modifications throughout the seasons was investigated.
Materials and methods
Site description
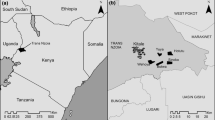
The experimental site was an agroforestry system located in an 11 ha commercial organic vegetable farm in southern France at Vézénobres (44° 03′ 11″North, 4° 08′ 10″East, elevation 151 m). This agroforestry site was originally set-up by the French Institute of Agronomic Research (in 1996) and cropped with cereals associated with poplars. Since 2010, the site is owned by farmers practicing organic production and is a participative research site involving the farmers, AGROOF society and the Avignon INRA center.
The climate is Mediterranean and locally influenced by the Cevennes Mountains, resulting in a generally warm climate, drought in the summer and heavy rainfall episodes in September and October. There are 2616 h of total sun per year and an average yearly rainfall of 740 mm (Météo France). 2015 was characterized by a warm and dry summer without precipitation before heavy rainfalls in October. The soil is a sandy (56%) loam (36%) with a very uniform profile throughout the treatments and no rocks. The pH is 8 at 0–20 cm and 8.5 from 40 to 80 cm. Total organic matter between 0 and 20 cm is around 2.5% in both treatments, and between 2 and 1.5% from 20 to 60 cm.
Within the site, hybrid walnut trees (Juglans nigra L. × Juglans regia L.) were grown with vegetable crops. The trees were planted in 1996. They are spaced 10 m from each other and were on average 17 m high. Conventional diversified non-organic crops (cereals, vegetables in the north and south, riparian forest in the west and forest in the east (pines, green oak) surrounded the site. In February 2015, four treatment plots were defined with three tree-shading agroforestry treatments corresponding to three levels of pruning: pollarding (AF−), strong-pruning (AF+) and light-pruning (AF++). A control plot without trees was also present for comparison (Fig. 1). The branch volume (m3) removed from the trees in each treatment was: 0.015 (AF++), 0.084 (AF+) and 0.3 (AF−), and the chipped wood volumes (branches under 7 cm in diameter) per tree per treatment were and 0.48, 0.275 and 0.055, for AF−, AF+ and AF++ respectively. The aim of the pruning was to provide three shading levels, creating three different microclimatic conditions. An illustration of the result is shown in the “Appendix”. The other agronomical practices were the same (crops, pesticides, irrigation and ground cover management). Light pruning is traditionally applied in most agroforestry systems every year to shape the trees, only lower branches are removed to preserve the trunk for timber, and the higher branches are left to follow their natural development. In the strong pruning treatment the lower branches and branches covering alley crops were removed, at about 2 m from the trunk. This treatment results in a hedgerow shape and was carried out with cutter bar equipment. Pollarding was carried out to cut off all the branches of the crown leaving only the bases of the major branches. The resulting wood was used as rameal wood chips to cover crops, in equal proportions.
Each of the four treatments corresponded to a square of 30*40 m, or 1200 m2 and included crop alleys and grass-strips in the tree rows. In each treatment, lettuce, carrots and tomatoes were organically grown separately, between two tree rows. Each crop resulted in five shallow beds measuring 25 m long and 1 m wide, separated by six walking spaces of 0.8 m. Each crop in the same treatment (9 m wide including shallow beds and walking spaces) was separated from the others by a grass strip, at the base of tree rows, measuring 25 m long by 1 m wide.
Tomatoes were planted in May at a density of 3 plants m−2. Lettuce was planted in June at a density of 16 plants m−2. Weeds were manually removed throughout all experiments and the soil was covered with a 3 cm layer of wood chips to limit weed growth. Water was supplied using drip irrigation, adjusted for each treatment. The crops were fertilized with castor cake (200 g m−2). A wheat strip separated each treatment plot and the surrounding area was composed of similar crops.
Due to the large size of each plot needed to obtain realist conditions regarding microclimate, the treatments could not be replicated. Interpretation of results thus relied on the gradient of shading associated with the pruning treatments, to detect relevant effects on Carabidae and spiders (we did not only consider the significant differences between treatments but also ranked the means according to the gradient).
Characterization of the microclimate
The average air temperature was monitored using four data logger sensors per treatment (Ibutton DS1923) at a timescale of 15 min. Sensors were placed in April 2015 and left until the end of September. In each treatment, four sensors were placed above the soil in the crops (at 2 m and 1 m) to measure the overall air temperature. Sensors were placed under a wooden shelter to avoid direct sun.
The percentage of canopy openness was calculated using hemispherical photographs in three seasons: before leaves appeared in April, after leaves developed in July and in the late vegetative season in September. The images were analyzed with Gap Light Analyzer software (Cary Institute of Ecosystem Studies 1999). For each treatment, the mean canopy openness was calculated based on 12 images taken in 12 different locations prior to each sampling period in each AF treatment. Leaves were present on the trees at the two arthropod sampling dates. The photos were taken at a fixed height (1.75 m) to avoid crop shade. Leaves appeared in mid-April, after the first canopy openness assessment, so the calculated percentage of canopy openness at this period represented the shading due to branches only.
Sampling of arachnid and ground beetle species at the seasonal scale
Two sampling sessions were carried out in July and August, and two in September and October. Pitfall traps (8 cm diameter, 11 cm height) were filled with conservation liquid (100 g NaCl per liter of water) and left for one complete week on the third of July, 8th of August, 10th of September and the 1st October 2015. In each treatment, five pitfalls traps separated by 5 m were placed in crops (tomato and lettuce) (Leather 2005). In total 40 traps were monitored at each sampling session.
The spiders and carabid beetles caught were conserved in alcohol (70%) and then identified to the species level using a binocular microscope and identification keys for carabids (Jeannel 1941, 1942; Coulon et al. 2011, 2012), and arachnids. The reference genitalias for French species were found at http://arachno.piwigo.com/index.php (Oger 2015). Juveniles were identified using the site https://araneae.unibe.ch/ (Nentwig et al. 2015). The species identified for each season were considered as reference species for daily activity experiments during which the main taxa were then identified with a low magnification lens or the naked eye without preservation.
Sampling design for daily spider and carabid activity patterns
Daily activity was studied in July and September with the expectation of more pronounced microclimatic effects during the warmest period, due to tree shading. In each treatment, two lines of five pitfall traps were placed in lettuce and tomato crops. All pitfall traps were spaced 5 m apart from each other in a row. Sampling design (wet and dry sampling methods) is shown in Fig. 1. In total, 80 traps (20 per treatment, 10 in lettuce and 10 in tomato) were monitored every day during the different sampling periods:
For summer community activity, sampling started on the 13th of July for 7 days and the hours of assessment were from sunrise to sunset: 6, 10, 12, 18 and 22 h. The individuals monitored at 6 h were assigned to the night activity period.
For early autumnal community activity, sampling started on the 17th of September for 8 days and the hours of assessment were from sunrise to sunset: 7, 10, 12, 18 and 21 h. The individuals monitored at 7 h were assigned to the night period of activity.
This sampling pattern separated day and night activity and also days into different periods. Individuals caught during an interval were assigned to the corresponding periods: from 6 to 10 h was AM, 10–14 h was midday, 14–18 h was PM, 18–22 h was dusk and 22–6 h was night.
Immediately after visual identification, individuals were released 1 m away from the trap in the same habitat. Immediate identification to the species level was made when specific criteria could be observed with the naked eye or low magnification lens, on the basis of the species list established during the previous wet sampling sessions. When determinant criteria could not be observed, individuals were assigned to an upper taxonomic class.
For carabids, individuals from the genera Poecilus, Pterostichus, Pseudoophonus, Dolichus, and Cylindera were identified at the species levels.
For arachnids, distinction was made between Lycosidae, Gnaphosidae and other spider families. Distinction between Pardosa and Trochosa genera was made using general habitus, size and leg stripes.
Statistical analysis
Statistical analyses were performed using R software (v 3.1.2). Computed standard errors are provided by the symbol “±” in the text.
Microclimate data
Differences in canopy openness at each date were assessed with one-way ANOVA. Mean values were computed using each photograph as an individual, after validation of homoscedasticity and normality of the residuals.
At each date, temperature differences between treatment periods were assessed using mixed models with repeated-measures, considering sensors as random effects, and followed by HSD Tukey tests. Differences in global (overall treatment daily means) temperatures between July and September were assessed using a Kruskal–Wallis rank sum test.
Activity-density patterns
Only the most abundant and easily recognizable species were assessed.
Activity-density for each trap was first averaged for each sampling period (July and September), resulting in a single value per trap in July and September.
The difference in average activity-density between treatments within crops measured with pitfall traps was then computed using an ANOVA, with activity-density for each pitfall trap as the explanation variable and treatments and habitats as fixed effects.
Daily activity patterns
To determine the daily pattern, counts for each period (AM, midday, PM, dusk and night) and pitfall trap, were divided by the total catches for this species that day in that trap. The aim of this transformation was to compare the percentage of activity by period to minimize the influence of differences in density between traps. Values were thus comprised between 0 and 1, and the sum of values for each pitfall trap in a day was 0 or 1. Pitfall traps that remained devoid of one species during the entire day were removed from the activity analysis for this species.
To assess the effect of canopy treatments on daily activity patterns, we used mixed models (“lmerTest” R library, Kuznetsova et al. 2016) with the proportion of activity per period set as the dependent variable, the treatment in interaction with crops as the fixed variable and the pitfall traps as a random effect. Models were made for each period and day was included as a fixed effect. The significance was assessed using the L ratio (Zuur 2009). Normality of the residuals was visually checked and data square root or log transformed if necessary. Seasonal intra treatment differences were assessed using mixed models in the same conditions but adding a new explanatory variable “season”. An HSD Tukey test was then used to compute intra treatment comparisons. Global activity (average activity over all treatments) was also computed to compare seasonal global activity. The homogeneity of residuals was visually assessed at each time point (Zuur 2009).
Results
Microclimate
The pruning treatments had a significant positive effect on the percentage of canopy openness for each date and reflected the intensity of tree pruning or pollarding (P < 0.001; F = 147.8; Df = 6; Fig. 2). Smaller differences occurred in April before leaves appeared than in July and September. In July, differences in canopy openness between treatments were correlated with the pruning intensity gradient (AF++ 21.9%, AF+ 37.9%, AF− 60.1%, control 92%). From July to September, canopy openness increased by 2% in AF++ and AF+ and decreased by 7% in AF−. Canopy openness was above 90% in the control plot in all three seasons.
Percent canopy openness for each pruning treatment and control plot at three dates in 2015: April, July and September (AF++ = light pruning, AF+ = strong pruning, AF− = pollarding. Different letters indicate significant differences after Tukey test comparisons within a date. Error bars represent standard error of the mean
Pruning treatments affected temperature at the hourly scale in both seasons (Fig. 3). The biggest diurnal difference between treatments occurred between 10 AM and 14 PM in July, and between 14 and 18 PM in September (with mean values of 27.4 °C and 15.89 °C in July and September respectively). Significantly warmer temperatures were observed under trees (AF++, AF+ , AF−) at night in both months (Table 1). Although the differences in temperatures in AF+ and AF− were not significant, the trend in average values correlated with increasing canopy openness and an average difference of approximately 1 °C between the control and AF++ in both seasons was observed. The overall temperatures in July and September were significantly different (Kruskal–Wallis test, df = 1, P < 0.01) with an average difference of 10.5 °C.
Carabid and arachnid species: assessment of the dominant species in July
In total, 477 carabids and 2342 arachnids were caught over 1 week in July using wet pitfall traps. Pseudoophonus rufipes (De Geer 1774) was the dominant ground beetle species, with 72.7% of the total catch of 347 individuals. Poecilus cupreus (Linne 1758), Asaphidion curtum (Heyden 1870), Cylindera germanica (Linne 1758), and Poecilus kugelanni (Panzer 1797) individuals represented 18.4% of the total catches. Other species represented less than 10% of the total catch (Table 2). Lycosid species Pardosa hortensis (Thorell 1872) dominated the arachnid community, with 58% of total arachnid catches. Pardosa sp., Phalangium sp., Trochosa sp., Pardosa proxima (C. L. Koch 1847) and Erigone dentipalpis (Wider 1834) represented 33.1% of the catches. Pardosa sp. individuals were juvenile stages.
Carabids and arachnids: assessment of the dominant species in September
Three hundred and seventeen carabids and 1140 arachnids were caught during the sampling period. P. rufipes was the dominant carabid species, accounting for 63.5% of the total catch with 141 individuals caught (Table 3). Pterostichus madidus (Fabricius 1775), Calathus fuscipes (Goeze 1777), Pterostichus melanarius (Illiger 1798) and Carabus violaceus (Fabricius 1787) represented 24.7% of the total catches. Other species represented only 10% of the total catch.
Pardosa juveniles (44.3% of total catches), Phalangium sp., Zelotes sp. and Trochosa juveniles dominated the arachnid community.
Weekly activity-density of the main taxa in July using dry pitfall traps
Two thousand and fourteen arachnids (1575 Pardosa sp.) and 598 carabids (497 P. rufipes) were caught during the summer daily activity sampling period using dry pitfall traps.
The interaction between crops and pruning treatment had a marginally significant effect on Pardosa sp. activity-density (ind. day−1 trap−1) (F = 2.57, Df = 3, P = 0.06). For lettuce, activity-density was significantly lower in the control and AF− compared to AF+ (respectively 0.52 ± 0.04, 0.65 ± 0.05 vs 0.96 ± 0.07) and intermediate in AF++ (0.72 ± 0.06). For tomato, activity-density was significantly lower in the control compared to AF++ (0.35 ± 0.34 vs 0.58 ± 0.05), and intermediate in AF− and AF+ (0.49 ± 0.05 and 0.47 ± 0.04 respectively) (Fig. 4a). The main species of ground beetle P. rufipes (497 catches) was also significantly affected by an interaction between crops and treatments (F = 3.21, Df = 3, P < 0.03). Significantly higher activity-density was found in the two most open treatments in lettuce compared to AF+ and AF++ (0.27 ± 0.04 in control and 0.32 ± 0.05 in AF− vs 0.010 ± 0.02 and 0.13 ± 0.03 in AF+ and AF++ respectively), while in tomato, activity in AF++ (0.08 ± 0.02) was significantly lower compared to the control (0.22 ± 0.03) with intermediary activity in AF+ (0.19 ± 0.04) and AF− (0.19 ± 0.03) (Fig. 4c).
Mean (+SE) abundance-activity of the two main ground predators per day and per trap in the four treatments (AF++ = light pruning, AF+ = strong pruning, AF− = pollarding) in July (a, b) and September (c, d). Letters indicate a significant difference (P < 0.05, Tukey HSD contrasts method). Error bars represent standard error of the mean
Weekly activity-density of the dominant taxa in September using dry pitfall traps
No difference in activity between treatments was found for juvenile Pardosa sp. (F = 0.38, Df = 3, P = 0.76) in this season (366 individuals), (AF++: 0.11 ± 0.01, AF+: 0.12 ± 0.01, Control: 0.10 ± 0.01, AF−: 0.10 ± 0.01) (Fig. 4b). We found significantly more individuals in lettuce than in tomato plots (F = 9.88, Df = 3, P = 0.002).
P. rufipes (335 individuals) had a significantly (F = 10.18, Df = 3, P < 0.001) higher daily activity-density (ind. day−1 trap−1) in AF+ (0.20 ± 0.02) than in the control (0.10 ± 0.01) and AF− (0.11 ± 0.01), and there was no difference between AF− and the control (Fig. 4d). Activity was lower in AF++ than in all other treatments. No interaction was found between crops and treatment (F = 1.65, df = 3, P = 0.18).
Daily activity patterns
Pardosa sp.
In July, Pardosa sp. had a mainly diurnal activity pattern in all the treatments, with more than 62% of its activity from AM to late PM (6–18 h) (Fig. 5a). The interaction between crops and treatments had a significant effect on Pardosa sp. daily activity (F = 2.09, Df = 12, P < 0.0001). In lettuce, in the early morning, activity was higher in the control compared to AF++ (26% ± 4 vs 11% ± 2). At midday, activity was higher in AF++ and AF+ compared to AF− (25% ± 4, 18% ± 3 vs 6% ± 2) and intermediary in the control (17% ± 4), while during late PM, activity was significantly higher in AF+ compared to AF++ and control (39% ± 4 vs 26% ± 4 and 18% ± 4). In tomato plots, significant differences occurred at midday, with significantly more activity in AF++ and AF+ compared to AF− and control (31% ± 5 and 25% ± 5 vs 6% ± 2 and 10% ± 4 in this order). At dusk, activity was significantly higher in AF− compared to AF++ (21% ± 4 vs 7% ± 3).
Percentage activity throughout the day for the spider Pardosa sp. (a, b) and the Carabidae Pseudoophonus rufipes (c, d) during the daily sampling experiment in July and September. Error bars show the standard error, different letters indicate significant differences between values (P < 0.05, Tukey HSD contrasts method). Values were compared between the two seasons. Error bars represent standard error of the mean
In September, significant differences were no longer observed between treatments at any period except in the morning (F = 3.14, df = 12, P < 0.0001), when there was higher activity in the control (19.6%) than in AF− (2.8%) and AF+ (4.7%). No interaction was found between crop and treatment (F = 1.01, df = 12, P = 0.43) (Fig. 5b). Compared to July, spiders showed significantly (F = 3.22, df = 12, P < 0.001) increased activity at midday in all treatments (AF+: + 28%; AF−: + 37.3%; control: + 19.7%, P < 0.05), except in AF++ (+ 6%). Overall activity between 6 AM and 6 PM was approximately 83.2%.
Pseudoophonus rufipes
In July, P. rufipes was mainly active (averaged activity for overall treatments) at dusk (25.1% ± 2.5) and during the night (73.5% ± 2.5), with a significantly (F = 5.99, df = 12, P < 0.001) higher activity at night in the control (83.1% ± 4.7) than in AF++ (63% ± 5.8) and AF− (64.4% ± 5.0) (P < 0.05) (Fig. 5c). The interaction between crop and treatment was not significant (F = 1.01, Df = 12, P = 0.44).
In September, P. rufipes was more active during the day (AM: 2.54%, Midday: 15.0%, PM: 20.6%), with less activity at dusk (18.54%) and high activity during the night (43.29%). However, no difference between treatments was found for any period and no interaction between crops and treatment was found (F = 1.22, Df = 12, P = 0.26) (Fig. 5d). Overall daily activity increased by 36.4% from July to September (AM, mid and PM periods). Night activity significantly decreased (F = 3.1, df = 12, P < 0.001) in all treatments (− 46% in control, − 24% in AF−, − 27% in AF+ and − 23% in AF++; P < 0.05).
Discussion
Microclimate treatment effect
In this study, we clearly showed that agroforestry, under a warm and dry Mediterranean climate, modifies air temperature during summer and at the beginning of autumn. The magnitude of these changes was clearly associated with pruning practices, and thus canopy openness.
The effect of tree pruning on the dominant ground beetle species Pseudoophonus rufipes
The two main predatory arthropods had very distinct reactions when faced with climate extremes. In July, the ground beetle P. rufipes behaved differently between treatments, with a significantly lower overall activity-density in the AF++ and AF+ plots than in the AF− and control plot. These differences changed in September but did not follow the canopy openness gradient, perhaps due to the less important effect of temperature.
Pruning management had a slight effect on the circadian rhythm of this mostly nocturnal species. A recent report from Berthe et al. (2015) showed that P. rufipes activity-density increased in warmer conditions (+ 2 °C). In our study, P. rufipes showed preference for conditions with the coldest temperatures during its activity period (night) and warmest temperatures during its resting period (day). Higher activity-density at night in the more open treatments could suggest a potential preference for the optimal temperature when faced with climate extremes in July, as these differences disappeared in September.
The absence of a clear pattern between treatments suggests that the activity-density of this species was affected by non-monitored parameters, which could explain differences between treatments in the two seasons. Numerous factors other than temperature are also affected by the presence of trees, such as weed composition (Batish et al. 2007) and then food resources. This omnivorous species was previously shown to prefer particular seeds (Petit et al. 2014).
This species can be found in most countries in Europe, southern Russia, northern China, Afghanistan, Iran and Morocco (Ghahari et al. 2010) as well as in several different European agricultural landscapes (Allegro and Sciaky 2003; Kromp 1999). This suggests that it has a great ability to adapt to varied environmental conditions and especially to contend with soil perturbation such as tillage (Miñarro and Dapena 2003).
Effect of pruning treatment on the dominant arachnid Pardosa sp.
In contrast to the ground beetle P. rufipes, in July the diurnal wolf spider (mostly P. hortensis) was more abundant in the agroforestry plots compared to the control. Moreover, strong differences in circadian rhythms appeared between the treatments, with reduced activity around midday in the control and strongly pruned treatment (especially in tomatoes) and more activity in the control than in the three agroforestry plots in the AM period in July. Closely related species, such as P. amentata, were reported to be more active during the morning than afternoon on warm sunny days to avoid “very warm” temperatures, associated with reduced activity (Edgar 1969).
Krumpalova and Tuf (2013) showed that for three Pardosa species (P. lugubris, P. amentata and P. prativaga), peak activity happened earlier in an open field compared to a forest (3 h earlier). The higher midday activity in AF++ and AF+ could be explained by tree morphology: in these treatments, tree crowns overlapped, shading the soil homogeneously, while the stronger pruning in AF− allowed direct heating of some parts of the ground at midday. Ground temperatures in this period might have been too warm and prevented arachnid activity. In our study, activity was high between 14:00 and 18:00 in all treatments despite warm air temperatures (above 33.9 °C). Additional sampling points during this period could have helped to see if activity is homogeneous during this sampling time, or condensed in late PM, especially in the most open treatment and control. It is likely that crop type also influenced conditions under the trees, such as microclimate, and could explain the variability observed for this species in July. The measured effect of the interaction between crops and trees on daily activity shows that the impact of agroforestry on epigeal arthropods can also depend on crop type and management. Indirect effects of prey availability could also be involved in circadian rhythm differences.
Perspectives
It is often claimed that agroforestry is a way to buffer climate extremes (Lasco et al. 2014; Lin 2007). An interesting aspect of the “buffer” effect of agroforestry is that during warm climatic episodes, higher temperatures can be found during the night under trees compared to open fields. Depending on species life traits and strategies, this microclimate effects are likely to enhance or reduce species activity-density and activity. This experiment shows how agroforestry is likely to modulate activity-density of ground dwelling arthropods which could possibly affect CBC such as demonstrated in a tropical context (Guenat et al. 2017; Poeydebat et al. 2017; Pumariño et al. 2015).
Limitations
It is technically difficult to set up multiple similar agroforestry sites especially because of the low number of commercial agroforestry systems with aged trees in Europe, and this limits the possibility of carrying out a more general analysis. In particular, to study microclimate large enough blocks are required, as a single tree could not constitute a large enough sample unit.
In contrast, replicated block experiments can lead to more accurate control over explanatory variables but would not have allowed similar and good commercial conditions (the farmer could not care for larger areas). Generating a gradient of canopy openness within a split plot design was the best compromise between farmer practices and research needs.
Throughout the sampling, individuals may have been recaptured. However, it was assumed that this effect was minimal as the recapture rate is generally low even with longer sampling experiments. For example, in an agricultural context sampling a medium sized ground beetle species, the recapture rate was between 7 and 16% for 3385 individuals caught in 46 days (Joyce et al. 1999).
Conclusions
This study clearly demonstrated the effect of trees on air temperature in temperate agroforestry crops. Our findings also showed how canopy management minimizes the intensity of these effects. Temperatures under the canopies were cooler during the day and warmer at night. This was highlighted by the daily activity assessment, which found contrasting trends in July for the two more abundant carabids and the arachnids P. rufipes and P. hortensis. P. rufipes was more abundant in the control and AF− treatments whereas P. hortensis showed the opposite distribution. Secondly, the activity of these species within a day could differ depending on the percentage of canopy openness, especially in July. A combination of different abundance levels with optimum microclimates for maximum activity, could result in differences in terms of pest regulation. Consumption of pests by ground predators is not the only factor acting in CBC, but our results showed continuous activity during the day for the wolf spider P. hortensis, whereas it decreased in adverse climatic conditions. The changes to the microclimate due to mature trees in agroforestry systems appear to influence beneficial arthropods through various drivers, and further studies should focus on potential predation and pest CBC in agroforestry systems.
References
Allegro G, Sciaky R (2003) Assessing the potential role of ground beetles (Coleoptera, Carabidae) as bioindicators in poplar stands, with a newly proposed ecological index (FAI). For Ecol Manag 175:275–284
Atienza JC, Farinos GP, Zaballos JP (1996) Role of temperature in habitat selection and activity patterns in the ground beetle Angoleus nitidus. Pedobiologia 40:240–250
Batish D, Kohli R, Jose S, Singh H (eds) (2007) Ecological basis of agroforestry. CRC Press, Boca Raton
Berthe SCF, Derocles SAP, Lunt DH, Kimball BA, Evans DM (2015) Simulated climate-warming increases Coleoptera activity-densities and reduces community diversity in a cereal crop. Agric Ecosyst Environ 210:11–14. https://doi.org/10.1016/j.agee.2015.05.001
Bianchi FJJ, Booij CJ, Tscharntke T (2006) Sustainable pest regulation in agricultural landscapes: a review on landscape composition, biodiversity and natural pest control. Proc R Soc B Biol Sci 273:1715–1727. https://doi.org/10.1098/rspb.2006.3530
Buchholz S, Rolfsmeyer D, Schirmel J (2013) Simulating small-scale climate change effects-lessons from a short-term field manipulation experiment on grassland arthropods: simulating small-scale climate change effects. Insect Sci 20:662–670. https://doi.org/10.1111/j.1744-7917.2012.01556.x
Chen J, Saunders SC, Crow TR, Naiman RJ, Brosofske KD, Mroz GD, Brookshire BL, Franklin JF (1999) Microclimate in forest ecosystem and landscape ecology variations in local climate can be used to monitor and compare the effects of different management regimes. Bioscience 49:288–297
Collins KL, Boatman ND, Wilcox A, Holland JM, Chaney K (2002) Influence of beetle banks on cereal aphid predation in winter wheat. Agric Ecosyst Environ 93:337–350
Coulon J, Pupier R, Quénnec E, Ollivier E, Richoux P (2011) Faune de France n°94 Coléoptères carabiques: complément et mise à jour volume 1. Fédération Française des Sociétés de Sciences Naturelles, 352 p
Coulon J, Pupier R, Quénnec E, Ollivier E, Richoux P (2012) Faune de France n°95 Coléoptères carabiques: complément et mise à jour. Fédération Française des Sociétés de Sciences Naturelles, 337 p
De Frenne P, Rodriguez-Sanchez F, Coomes DA, Baeten L, Verstraeten G, Vellend M, Bernhardt-Romermann M, Brown CD, Brunet J, Cornelis J, Decocq GM, Dierschke H, Eriksson O, Gilliam FS, Hedl R, Heinken T, Hermy M, Hommel P, Jenkins MA, Kelly DL, Kirby KJ, Mitchell FJG, Naaf T, Newman M, Peterken G, Petrik P, Schultz J, Sonnier G, Van Calster H, Waller DM, Walther G-R, White PS, Woods KD, Wulf M, Graae BJ, Verheyen K (2013) Microclimate moderates plant responses to macroclimate warming. Proc Natl Acad Sci 110:18561–18565. https://doi.org/10.1073/pnas.1311190110
Diffenbaugh NS, Pal JS, Giorgi F, Gao X (2007) Heat stress intensification in the Mediterranean climate change hotspot. Geophys Res Lett. https://doi.org/10.1029/2007GL030000
Dondale CD, Redner JH, Semple RB (1972) Diel activity periodicities in meadow arthropods. Can J Zool 50:1155–1163. https://doi.org/10.1139/z72-154
Edgar WD (1969) Prey and feeding behaviour of adult females of the wolf spider Pardosa amentata (Clerck). Neth J Zool 20:487–491
Eichhorn MP, Paris P, Herzog F, Incoll LD, Liagre F, Mantzanas K, Mayus M, Moreno G, Papanastasis VP, Pilbeam DJ, Pisanelli A, Dupraz C (2006) Silvoarable Systems in Europe—Past, Present and Future Prospects. Agrofor Syst 67:29–50. https://doi.org/10.1007/s10457-005-1111-7
Frampton GK, Van den Brink PJ, Wratten SD (2001) Diel activity patterns in an arable collembolan community. Appl Soil Ecol 17:63–80. https://doi.org/10.1016/S0929-1393(00)00128-1
Fuhrer J, Gregory PJ, International CAB (eds) (2014) Climate change impact and adaptation in agricultural systems. CABI climate change series. CABI, Boston
Ghahari H, Avgin SS, Ostovan H (2010) Carabid beetles (Coleoptera: Carabidae) collected from different ecosystems in Iran with new records. Turk. J, Entomol, p 34
Giorgi F, Lionello P (2008) Climate change projections for the Mediterranean region. Glob Planet Change 63:90–104. https://doi.org/10.1016/j.gloplacha.2007.09.005
Greenwood O, Mossman HL, Suggitt AJ, Curtis RJ, Maclean IMD (2016) Using in situ management to conserve biodiversity under climate change. J Appl Ecol 53:885–894. https://doi.org/10.1111/1365-2664.12602
Griffiths GJK, Holland JM, Bailey A, Thomas MB (2008) Efficacy and economics of shelter habitats for conservation biological control. Biol Control 45:200–209. https://doi.org/10.1016/j.biocontrol.2007.09.002
Guenat S, Kaartinen R, Jonsson M (2017) Shade trees decrease pest abundances on brassica crops in Kenya. Syst, Agrofor. https://doi.org/10.1007/s10457-017-0159-5
Guo K, Hao S-G, Sun OJ, Kang L (2009) Differential responses to warming and increased precipitation among three contrasting grasshopper species. Glob Change Biol 15:2539–2548. https://doi.org/10.1111/j.1365-2486.2009.01861.x
Honek A (2013) The effect of temperature on the activity of Carabidae (Coleoptera) in a fallow field. EJE 94:97–104
Jeannel R (1941) Faune de France n°39, Coléoptères carabiques. Fédération Française des Sociétés de Sciences Naturelles, 575 p
Jeannel R (1942) Faune de France n°40, Coléoptères carabiques. Fédération Française des Sociétés de Sciences Naturelles, 606 p
Jones M, Sinclair FL, Grime VL (1998) Effect of tree species and crown pruning on root length and soil water content in semi-arid agroforestry. Plant Soil 201:197–207
Joyce KA, Holland J, Doncaster C (1999) Influences of hedgerow intersections and gaps on the movement of carabid beetles. Bull Entomol Res. https://doi.org/10.1017/S000748539900067X
Karungi J, Nambi N, Ijala AR, Jonsson M, Kyamanywa S, Ekbom B (2015) Relating shading levels and distance from natural vegetation with hemipteran pests and predators occurrence on coffee. J Appl Entomol 139:669–678. https://doi.org/10.1111/jen.12203
Kranz AJ, Wolz KJ, Miller JR (2018) Effects of shrub crop interplanting on apple pest ecology in a temperate agroforestry system. Syst, Agrofor. https://doi.org/10.1007/s10457-018-0224-8
Kromp B (1999) Carabid beetles in sustainable agriculture: a review on pest control efficacy, cultivation impacts and enhancement. Agric Ecosyst Environ 74:187–228
Krumpalova Z, Tuf IH (2013) Circadian rhythms of ground living spiders: mechanisms of coexistence strategy based on the body size. Pol J Ecol 61:575–586
Kuznetsova A, Brockhoff PB, Christensen RHB (2016) lmerTest: tests in linear mixed effects models. R package version 2.0-33. https://CRAN.R-project.org/package=lmerTest
Lasco RD, Delfino RJP, Espaldon MLO (2014) Agroforestry systems: helping smallholders adapt to climate risks while mitigating climate change. Wiley Interdiscip Rev Clim Change 5:825–833. https://doi.org/10.1002/wcc.301
Leather SR (ed) (2005) Insect sampling in forest ecosystems. Methods in ecology. Blackwell Pub, Malden
Lin BB (2007) Agroforestry management as an adaptive strategy against potential microclimate extremes in coffee agriculture. Agric For Meteorol 144:85–94. https://doi.org/10.1016/j.agrformet.2006.12.009
Lindberg N, Engtsson JB, Persson T (2002) Effects of experimental irrigation and drought on the composition and diversity of soil fauna in a coniferous stand. J Appl Ecol 39:924–936
Malézieux E, Crozat Y, Dupraz C, Laurans M, Makowski D, Ozier-Lafontaine H, Rapidel B, Tourdonnet S, Valantin-Morison M (2009) Mixing plant species in cropping systems: concepts, tools and models. A review. Agron Sustain Dev 29:43–62. https://doi.org/10.1051/agro:2007057
Mariño YA, Pérez M-E, Gallardo F, Trifilio M, Cruz M, Bayman P (2016) Sun vs. shade affects infestation, total population and sex ratio of the coffee berry borer (Hypothenemus hampei) in Puerto Rico. Agric Ecosyst Environ 222:258–266. https://doi.org/10.1016/j.agee.2015.12.031
Miñarro M, Dapena E (2003) Effects of groundcover management on ground beetles (Coleoptera: Carabidae) in an apple orchard. Appl Soil Ecol 23:111–117. https://doi.org/10.1016/S0929-1393(03)00025-8
Moreno G, Aviron S, Berg S, Crous-Duran J, Franca A, de Jalón SG, Hartel T, Mirck J, Pantera A, Palma JHN, Paulo JA, Re GA, Sanna F, Thenail C, Varga A, Viaud V, Burgess PJ (2017) Agroforestry systems of high nature and cultural value in Europe: provision of commercial goods and other ecosystem services. Syst, Agrofor. https://doi.org/10.1007/s10457-017-0126-1
Musolin DL (2007) Insects in a warmer world: ecological, physiological and life-history responses of true bugs (Heteroptera) to climate change. Glob Change Biol 13:1565–1585. https://doi.org/10.1111/j.1365-2486.2007.01395.x
Nentwig W, Blick T, Gloor D, Hänggi A, Kropf C (2015) Spiders of Europe. Version 10.2015. www.araneae.unibe.ch. Accessed Oct 2015
Oger P (2015) Les araignées de Belgique et de France. http://arachno.piwigo.com/index.php?/categories. Accessed Oct 2015
Park O (1941) Concerning Community Symmetry. Ecology 22:164. https://doi.org/10.2307/1932211
Petit S, Boursault A, Bohan DA (2014) Weed seed choice by carabid beetles (Coleoptera: Carabidae): Linking field measurements with laboratory diet assessments. Eur J Entomol 111:615
Pezzopane JRM, de Souza PS, Rolim GDS, Gallo PB (2011) Microclimate in coffee plantation grown under grevillea trees shading. Acta Sci Agron. https://doi.org/10.4025/actasciagron.v33i2.7065
Pfannenstiel RS, Yeargan KV (2002) Identification and diel activity patterns of predators attacking Helicoverpa zea (Lepidoptera: Noctuidae) eggs in soybean and sweet corn. Environ Entomol 31:232–241. https://doi.org/10.1603/0046-225X-31.2.232
Poeydebat C, Tixier P, De Lapeyre De Bellaire L, Carval D (2017) Plant richness enhances banana weevil regulation in a tropical agroecosystem by affecting a multitrophic food web. Biol Control 114:125–132. https://doi.org/10.1016/j.biocontrol.2017.08.009
Pumariño L, Sileshi GW, Gripenberg S, Kaartinen R, Barrios E, Muchane MN, Midega C, Jonsson M (2015) Effects of agroforestry on pest, disease and weed control: a meta-analysis. Basic Appl Ecol 16:573–582. https://doi.org/10.1016/j.baae.2015.08.006
Quinkenstein A, Wöllecke J, Böhm C, Grünewald H, Freese D, Schneider BU, Hüttl RF (2009) Ecological benefits of the alley cropping agroforestry system in sensitive regions of Europe. Environ Sci Policy 12:1112–1121. https://doi.org/10.1016/j.envsci.2009.08.008
Rainio J, Niemelä J (2003) Ground beetles (Coleoptera: Carabidae) as bioindicators. Biodivers Conserv 12:487–506
Ranjha MH, Irmler U (2014) Movement of carabids from grassy strips to crop land in organic agriculture. J Insect Conserv 18:457–467. https://doi.org/10.1007/s10841-014-9657-1
Saska P, Vodde M, Heijerman T, Westerman P, van der Werf W (2007) The significance of a grassy field boundary for the spatial distribution of carabids within two cereal fields. Agric Ecosyst Environ 122:427–434. https://doi.org/10.1016/j.agee.2007.02.013
Saska P, van der Werf W, Hemerik L, Luff ML, Hatten TD, Honek A (2013) Temperature effects on pitfall catches of epigeal arthropods: a model and method for bias correction. J Appl Ecol 50:181–189. https://doi.org/10.1111/1365-2664.12023
Simon S, Bouvier J-C, Debras J-F, Sauphanor B (2010) Biodiversity and pest management in orchard systems. A review. Agron Sustain Dev 30:139–152
Thiele H-U (1977) Carabid beetles in their environments. Springer, Berlin
Tscharntke T, Clough Y, Bhagwat SA, Buchori D, Faust H, Hertel D, Hölscher D, Juhrbandt J, Kessler M, Perfecto I, Scherber C, Schroth G, Veldkamp E, Wanger TC (2011) Multifunctional shade-tree management in tropical agroforestry landscapes—a review. J Appl Ecol 48:619–629. https://doi.org/10.1111/j.1365-2664.2010.01939.x
Tuf IH, Dedek P, Vesely M (2012) Does the diurnal activity pattern of carabid beetles depend on season, ground temperature and habitat? Arch Biol Sci. https://doi.org/10.2298/ABS1202721T
Vucic-Pestic O, Ehnes RB, Rall BC, Brose U (2011) Warming up the system: higher predator feeding rates but lower energetic efficiencies. Glob Change Biol 17:1301–1310. https://doi.org/10.1111/j.1365-2486.2010.02329.x
Walther G-R, Post E, Convey P, Menzel A, Parmesan C, Beebee TJ, Fromentin J-M, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
Weber DC, Pfannenstiel RS, Lundgren JG (2008) Diel predation pattern assessment and exploitation of sentinel prey: new interpretations of community & individual behaviors. In: Proceedings of the third international symposium on biological control of arthropods, Christchurch, New Zealand, pp 485–494
Zehnder G, Gurr GM, Kühne S, Wade MR, Wratten SD, Wyss E (2007) Arthropod pest management in organic crops. Annu Rev Entomol 52:57–80. https://doi.org/10.1146/annurev.ento.52.110405.091337
Zuur AF (ed) (2009) Mixed effects models and extensions in ecology with R. Statistics for biology and health. Springer, New York
Acknowledgements
We thank the foundations Fondation de France, Fondation Humus, Terra Symbiosis and Fondation Picard who financially supported the ARBRATATOUILLE project. We wish to express considerable thanks to Virginie and Denis Florès who actively participated in the project and gave us permission to work on their farm.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
See Fig. 6.
Rights and permissions
About this article
Cite this article
Martin-Chave, A., Béral, C., Mazzia, C. et al. Agroforestry impacts the seasonal and diurnal activity of dominant predatory arthropods in organic vegetable crops. Agroforest Syst 93, 2067–2083 (2019). https://doi.org/10.1007/s10457-018-0309-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-018-0309-4