Abstract
Faidherbia albida is an important tree species in the parkland agroforestry system of the Rift Valley region, central and south-eastern Ethiopia. Positive effects of F. albida on crop production are widely recognised. However, the effects of tree pruning, zone and fertiliser interactions on crop growth have not been addressed in earlier studies. A field experiment containing three levels of tree pruning (100% pruned, 50% pruned, and unpruned) as main plots, and application of recommended rates of N and P fertilisers as sub-plots, was conducted during the 2015 and 2016 growing seasons. Maize grain yield and biomass, light intensity, and soil nutrients and moisture were measured at different positions from each F. albida tree trunk (0–2, 2–4 and 4–6 m) and in crop-only plots. Biomass and yield of maize were significantly greater under tree canopies compared to crop-only plots in both the 2015 and 2016 growing seasons, regardless of pruning levels. Fertilisation significantly increased yields under tree canopies compared to crop-only plots in both years. Light intensity increased with distance from trees and with greater pruning levels. Soil carbon and nutrient concentrations and moisture content decreased with increasing distance from tree and with soil depth. These results suggest that maize production and profitability could be maintained or improved through only partial pruning of F. albida rather than pollarding, and by preferentially applying fertilisers in normal and wet years. Recommendations need to be evaluated in a total system context including other rotational crops, fuel, livestock and socio-economic factors.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Increasing demand for food has caused natural resource degradation in many regions of the world as farming practices often reduce soil quality, biodiversity and ecosystem services (Power 2010). The problem is particularly important in Sub-Saharan African countries due to the high dependency on natural resources and limited access to mechanised farming, fertiliser, and irrigation. This calls for a viable alternative to sustain or increase agricultural productivity of smallholder farms in those countries (Jose 2009; Nair 2007).
In the Central Rift Valley (CRV) of Ethiopia, farmers grow annual crops in agroforestry parkland systems where naturally regenerated and scattered trees occur along with crops (Siriri et al. 2010). Faidherbia albida is one of the main tree species in these parkland systems. Farmers have retained F. albida in order to improve soil fertility, microclimate, and crop yield, and to provide branches for fencing and firewood. Competition for light, water, and nutrients can be minimised under F. albida trees that exhibit reverse phenology, i.e. when the tree sheds its leaves during the wetter crop growing season and regrows them between seasons during drier conditions. However, this phenomenon can be disrupted by factors such as climatic conditions in areas that have transitional single and double rainfall seasons, by drought (Boffa 1999) and by pruning.
Above- and below-ground competition in agroforestry systems can be minimised by crown pruning (Semwal et al. 2002). For example, crown pruning of alnus (Alnus acuminate) and calliandra (Calliandra calothyrsus) was required to sustain bean and maize production in agroforestry systems of Uganda (Siriri et al. 2010). Either under or at a distance from trees, crop production can be improved by the application of fertilisers.
Studies that involve field experiments on F. albida trees in parklands of the CRV have demonstrated positive effects of trees on crop yields, soil fertility, and microclimate. For instance, F. albida trees scattered in crop fields apparently improved some soil properties under their canopy as compared to adjacent open plots (Kamara and Haque 1992). However, no information is available in these studies on the effects on crop productivity of farm management practices such as pruning of F. albida tree crowns and the application of fertilisers, which are two common options currently used by farmers in this parkland system. The objective of this study was to determine the impacts of crown pruning and fertiliser application on maize production under F. albida in a parkland agroforestry system in Ethiopia. Research questions addressed in the study were (a) are there interactive effects of tree shade and fertiliser applications on maize yield, and (b) how are these interactions affected by crown pruning?
Materials and methods
Study site description
The study was carried out in the parklands of the CRV of Ethiopia at Adulala watershed, which is located approximately 104 km south-east of the capital city, Addis Ababa. Adulala watershed is situated at 8°29.5′N latitude, 39°20.5′E longitude and has an average elevation of 1,688 m above sea level.
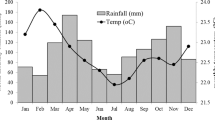
The climate of the area is characterized by a bimodal rainfall distribution with a mean annual rainfall of 820 mm. The short rainy season lasts from March to May and the long rainy season extends from June to October. During the field study, annual rainfall was 482 mm for 2015 and 1103 mm for 2016, which included the two cropping seasons (approximately June-November) used in this research. Annual mean monthly minimum and maximum temperatures were 13.9 °C and 28.5 °C.
Soil in the study area is classified as a Fluvisol (Goma 2015). The top 20 cm layer of the cultivated soil is characterised by a pH of 7.6 with total N 0.056%, available P 19 ppm (Olsen method), organic matter 4.3%, and available water holding capacity 155 mg g−1 (Goma 2015).
Natural vegetation type of the Adulala watershed is Acacia woodland, dominated by tree species such as Acacia tortilis, A. seyal and Faidherbia albida (Argaw et al. 1999; Endale et al. 2017). Crop production is mainly rainfed, and F. albida is the main agroforestry tree species in the fields of major crops such as teff (Eragrostis teff), maize (Zea mays) and wheat (Triticum aestivum L. var aestivum). Diammonium phosphate and urea fertilisers are usually applied to teff, whereas compost and manure are applied to other crops.
Experimental design and crop establishment
A total of eighteen F. albida trees were randomly selected in farmers’ fields in the Adulala watershed. The trees were scattered across several adjacent farms; within a total area of approximately 91 ha. An on-farm experiment was conducted during the growing seasons of 2015 and 2016 as a split-plot design with six replications. Main plot treatments in each replicate included three levels of tree crown pruning, i.e., unpruned, 50% pruned and 100% pruned (Fig. 1), and a crop-only plot that was located about 30 m from any tree trunk. Applications of N and P fertiliser were included as sub-plot treatments. Tree crown pruning was done by cutting tree branches at 15–20 cm from their base. The 50% pruning was done by removing branches in the bottom half of the crown length, and the 100% pruning was done by cutting all the branches (Fig. 1). Pruning of the experimental trees was done in May 2015 during the start of the experiment and newly grown branches after the first pruning were removed prior to the second season (May 2016).
A circular plot of 12-m diameter under each experimental tree was divided into four sub-plots (four quarters of the main plot that were oriented randomly). Urea (69 kg N ha−1) was added to one sub-plot, di-ammonium phosphate (23 kg P ha−1 and 9 kg N ha−1) to the second sub-plot, both urea and di-ammonium phosphate were added to the third sub-plot (78 kg N ha−1 and 23 kg P ha−1) and the fourth sub-plot was left as a control (no fertiliser applied). The four fertiliser treatments (Table 1) were allocated at random to the sub-plots under each tree and crop only plots. Fertilizers were applied by broadcasting either at sowing or after sowing, as below. Maize (Melkasa-2 variety) was sown in the third week of May during the 2015 and 2016 cropping seasons. Seeds were sown at a spacing of 0.75 m between and 0.30 m within rows in each sub-plot under each tree (a total of 18 trees, six trees per pruning level) and in the crop-only plots. DAP was applied only at sowing while urea was applied in two stages with different rates; the full rate (46 kg N ha−1) first at sowing and half the rate (23 kg N ha−1) 10 days after sowing (Table 1). Weeding was done manually in all the sub-plots every 2 weeks.
Measurements and data collection
Tree phenology
In order to estimate the relative amounts of leaves retained by tree crowns during the growing seasons, foliation (as a proportion of maximum foliation) of the unpruned trees of each treatment was observed and recorded. Maximum foliation was subjectively estimated as how much foliation there would be in a 100% foliated tree. The observation was made twice per month (May to October), in the first and third week of each month.
Light
Photosynthetically active radiation (PAR, µmol s−1 m−2) was measured under a total of nine trees (3 randomly selected trees from each of the unpruned, 50% pruned and 100% pruned trees) at different positions from the tree trunks (positions): 1, 3 and 5 m for the 0–2, 2–4, and 4–6 m positions, respectively, and in each crop-only plot. Measurements were located at the centre of each crop-only plot; and at four aspects (north, south, east and west) around each tree to provide an average value for each position. Measurements were taken after sowing at different times of the day: approximately 9:00 AM, 10:30 AM, 12:00 PM, 1:30 PM, 3:00 PM and 4:00 PM using a PAR sensor (AccuPAR model LP-80, Decagon Devices). PAR under each tree was measured for three consecutive days; the measurement under all trees was carried out between 1st June and 1st July 2016.
Soil nutrients analysis
In May 2015 prior to sowing, soil samples were taken under six randomly selected trees at two points in each tree position at depths of 0–20 cm, 20–40 cm, 40–60 cm and 60–80 cm using a core sampler. The two samples for each combination of tree position and depth were mixed to make a total of 12 composite soil samples from under each tree. At the same depths, soil samples were also taken at two randomly selected points from each of the six crop-only plots and combined to make a total of four composite samples per crop-only plot. Soil samples were air dried and analysed for organic carbon (Walkley & Black), total nitrogen (Kjeldahl) and available phosphorus (Olsen).
Soil moisture
Gravimetric water content was measured in the soil that had been sampled from three randomly selected trees from each pruning treatment (unpruned, 50% and 100% pruned trees) and crop-only plots during the growing season of 2015 at the flowering stage and in 2016 at the sowing, flowering, and physiological maturity stages. The soil samples were collected at two points in each tree position and at randomly selected two points in the crop-only plots at depths of 0–20 cm, 20–40 cm, 40–60 cm and 60–80 cm. The two soil samples for each tree position and depth as well as the samples for each crop-only plot and depth were bulked and oven dried for 24 h at 105 °C.
Crop yield and biomass
Maize total above-ground biomass and grain yield were determined by manual harvesting of all the plants from 1-m2 quadrats located randomly in all replicates of sub-plots under each tree position and crop-only plot. Harvesting was done at maturity, between the first and the third weeks of November 2015 and 2016. Grain moisture was measured using an electronic moisture tester, and grain yield was adjusted to 12% moisture content.
Statistical analysis
Differences between treatments for each parameter measured (light intensity, soil properties, crop biomass and yield) were analysed using the mixed procedure of SAS 9.4 (SAS Institute 2013). Using a split-plot design, pruning intensity, position from tree trunk and fertiliser applications were considered as fixed treatment effects while individual trees and open-plots were treated as random effects. Years were analysed separately. Significant differences (at p < 0.050) between treatment means were determined by the Tukey multiple range test.
Results
Tree phenology
Unpruned trees maintained 35–60% of maximum foliation during each growing season (Fig. 2). The pattern was similar in both seasons, increasing from May until a peak of foliage maintained in August, which then declined. Thus, foliation followed a similar pattern to rainfall in the region.
Light transmission
The amount of PAR received in the open (control) plots (52.7 ± 0.04 mol m−2 day−1) was significantly greater (p = 0.001) compared to PAR under tree crowns, which was reduced by 34%, 23% and 9% under unpruned, 50% and 100% pruned trees, respectively (Fig. 3). The amount of PAR increased with pruning and distance from the base of trees to the outer position. For example, there was an increase of PAR from 30.0 mol m−2 day−1 at the 0–2 m position of unpruned trees to 47.1 mol m−2 day−1 at the 4–6 m position of the 100% pruned trees (Fig. 3).
Soil nutrient status under tree canopies
The effect of position from F. albida trees on soil properties was significant for organic carbon, available P, and total N (p = 0.001), with greater values in at least one position under trees relative to open areas down to 60 cm depth (Table 2). The difference in soil nutrient properties in the upper 0–20 cm depth among tree positions was not significant for N or P; however organic carbon was significantly greater under the 0–2 m position (1.7 ± 0.06) relative to the 2–4 and 4–6 m positions. All soil nutrients at this soil depth were greater under tree canopies compared to open areas (Table 2).
Soil moisture
Soil moisture content ranged from 10 to 20% and generally decreased with depth (Fig. 4). Soil moisture content at the flowering stage (in 2015 and 2016) and at harvest in 2016 was significantly greater under trees compared to open areas, regardless of pruning levels for the 0–40 cm soil depth (Fig. 4, data shown for soil moisture at flowering stage in 2015 and 2016). There was no significant difference at 40–80 cm soil depth. Pruning did not significantly affect soil moisture content at any depth when measured during either growing season.
Crop yield and biomass production
Crop yield and biomass were significantly greater under trees (2–4 and 4–6 m positions, p = 0.001) compared to crop-only plots (Fig. 5, Table 3) in both growing seasons, regardless of pruning level. Yield increases in the outer position (4–6 m) were relatively larger (45–51% in 2015 and 75–76% in 2016) than the innermost positions (0–2 m) (2% in 2015 and 12% in 2016). Tree pruning significantly increased crop yield only under the 2–4 m tree position in 2015 (Fig. 5c). However, pruning significantly increased crop yield under all positions (0–2, 2–4 and 4–6 m) in 2016 (Fig. 5d), with the lowest yield (2253 kg ha−1) obtained under unpruned trees (0–2 m position).
Fertilisation with urea and DAP significantly increased yields under trees (2–4 and 4–6 m) relative to crop only-plots in both 2015 (p = 0.055) and 2016 (p = 0.001). The highest yield was obtained in 2016 from the Urea + DAP fertiliser combination (78 kg N/ha and 23 kg P/ha) under the 4–6 m tree position (Fig. 6).
Discussion
Reverse phenology of F. albida trees is promoted as a major advantage of growing this tree species with crops (Hadgu et al. 2009; Kho et al. 2001). However, close observation of their canopies during this study indicated that trees did not totally defoliate during the cropping season, maintaining 35–60% of maximum foliation. Lack of defoliation during the cropping season can be attributed to phenological disruption due to canopy pruning (Barnes and Fagg 2003). Farmers in the study area totally prune tree branches (pollarding) at intervals of 3–4 years such as recommended by Orwa et al. (2009). Reducing total canopy volume by about 35% before the onset of the rainy season, i.e. moderate pruning, might not appreciably reduce tree growth (Boffa 1999).
Results of this study demonstrate that canopy pruning results in increased PAR transmitted under trees. Lower crop biomass and yield recorded under unpruned trees (particularly under 0–2 m position) relative to the totally pruned and 50% pruned trees could partially be attributed to reduced PAR available to crops in the absence of leaf fall during the cropping seasons. The result is in agreement with Jama and Getahun (1991), who speculated that low maize yields under F. albida trees compared to crop-only plots could be the result of shading by the trees. However, the trees in that study were much younger (5 years old) than those studied in the present research (probably 35–45 years old), suggesting that the benefits associated with mature trees may take decades to develop. Faidherbia albida trees need 20–40 years to grow to a size that can significantly improve yields of understory crops (Poschen 1986). A study conducted by Suresh and Rao (1998) in a semiarid India region showed that PAR intercepted under F. albida trees was facilitated by leaf shedding (defoliation) during the crop growing period that in-turn lead to the highest sorghum (Sorghum bicolor) yield being obtained under this tree species compared to other nitrogen fixing trees that retained leaves during the cropping season such as Acacia ferruginea and Albizia lebbeck.
Significantly greater maize biomass and yield were found under F. albida tree crowns 2–6 m from the trunk compared to crop-only plots (Fig. 5). Lower or lack of yield benefit near to tree trunks (under 0–2 m positions) may be attributed to competition for resources between crops and trees in that position (Jose et al. 2000). Greater yield under trees, particularly in the 2–6 m positions could be attributed to improved soil nutrient concentrations and moisture levels associated with greater organic matter concentrations under tree canopies than in the open fields away from trees that outweighed any negative effects of shading. A similar study in the Hararghe highlands of eastern Ethiopia reported that grain yields of sorghum and maize grown under F. albida trees were increased by 76% and 36%, respectively, compared to crops grown in the open fields away from trees (Poschen 1986). The study asserted that the increase in crop yield was a result of improved soil chemical and physical conditions under tree canopies (Poschen 1986). In West Africa, a study in Niger showed that millet yield under the F. albida tree canopy was about 36% greater than the yield obtained from open fields (Kho et al. 2001). Although such cases of positive effects of F. albida tree on crop yields and biomass are well documented (Chamshama et al. 1998; Vandenbeldt and Williams 1992), some studies reported negative or no effects on some crops. In Cameroon for instance, Harmand and Njiti (1992) reported a reduction in yields of groundnut (Arachis hypogaea L.), cotton (Gossypium sp.) and sorghum by 34%, 11% and 40%, respectively. Such differences in F. albida effects on crops can be expected because of the variations and complex interactions of factors such as tree age, soil characteristics, water regime and climate (Barnes and Fagg 2003).
Fertilizer application increased crop yield on plots under tree canopies and on crop-only fields; maize yield increase in 2016 (with high rainfall) was bigger than the increase in yield in 2015 (with low rainfall). This result is consistent with nutrients being relatively less limiting than water during dry seasons (Kho et al. 2001). In the same study, Kho and colleagues reported a 36% increase in dry matter production of pearl millet (Pennisetum glaucum) under tree canopies compared to open crop-only plots. However, no increase due to trees was observed with a high rate of N fertiliser (180 N kg ha−1, Kho et al. 2001). This result suggested that the effect of F. albida on crop production is more pronounced in conditions of low soil fertility (Sileshi 2016) as nutrients are less limiting to crops at greater fertility levels. In the present study, recommended rates of fertilisers under tree canopies led to greater yield increase in 2016 (with high or average rainfall) and maize response to fertiliser was larger under the trees than in the crop-only plots. This result could be due to greater availability of water and soil nutrients other than N and P (e.g. K) under F. albida trees that were therefore less limiting to growth and thereby enabled a larger response to N and P fertiliser (Hadgu et al. 2009; Kamara and Haque 1992). This complexity is symptomatic of the response of crops to fertilisers being determined by a range of soil physical and chemical conditions (Baligar et al. 2001; Tittonell et al. 2008). Also in the present study, we note that low N and P availability limited maize growth, and it is unlikely that the N and P fertiliser rates used would have maximised growth.
Studies in Ethiopia report that concentrations of organic carbon, available P and total N decrease with increasing distance from F. albida trees and with increasing soil depth (Hadgu et al. 2009; Kamara and Haque 1992). The present study indicated that the values for those soil parameters were greater for 0–40 cm depth under trees relative to open plots and the differences for deeper 40–80 cm depth were not significant, with the exception of organic carbon, which was significantly greater under trees to a depth of 60 cm. These results are in agreement with Saka et al. (1994), who found greater levels of surface (0–15 cm) organic matter, N and Ca under tree canopies than in open areas, and no significant difference at the 30–45 cm depth. Similarly, in the Ethiopian highland Vertsols, Kamara and Haque (1992) reported greater concentrations of soil organic matter, total N, available P and exchangeable K under F. albida trees than in open fields away from trees. A study on other F. albida based systems of northern Ethiopia also revealed that soil organic matter, total N and available P were greater under tree canopies than outside canopies (Hadgu et al. 2009). Comparable results were also reported in other studies (Kho et al. 2001; Rhoades 1995; Umar et al. 2013). The mechanisms that improve soil nutrient concentrations under F. albida have been widely debated (Barnes and Fagg 2003). Some studies suggest that the factors for increased soil fertility under F. albida include deep capture and recycling of nutrients, improvement in soil biological activity, and symbiotic and asymbiotic N fixation (Rao et al. 1997; Rhoades 1995; Umar et al. 2013). Other authors have argued that improved soil fertility under tree canopies is due to lateral redistribution of nutrients, by domestic and wild animals including birds, tree roots, and wind erosion, or due to pre-existing greater soil fertility conditions favoured by establishing tree seedlings (Geiger et al. 1994).
An important effect of trees relates to soil moisture. This study show that at the flowering and maturity stages of maize, soil moisture in the top 40 cm depth was 22% and 24% greater than in the open field, respectively. Similarly, Rhoades (1995) reported increased soil moisture to a depth of 15 cm under F. albida trees compared to open areas but found no significant difference at 15–30 cm depth. Increased levels of soil moisture under tree canopies compared to open fields can be attributed several processes: (1) F. albida tree roots can take up water from deep in some soil profiles (Dupuy and Dreyfus 1992; Roupsard et al. 1999) resulting in hydraulic lift (movement of water from deeper to shallower soil layers) (Bayala et al. 2014); (2) reduced evapotranspiration compared to crop-only conditions; and (3) increases in soil organic matter that improve soil water holding capacity and moisture availability (Lal 2006; Sileshi 2016). For example, Makumba et al. (2006) reported greater (50%) soil moisture retention in gliricidia–maize than in sole maize because of a 65% increase in soil organic matter in the gliricidia–maize intercropping system in Southern Malawi.
Tree density in the CRV of Ethiopia is sparse, about 4.2 trees per hectare (Sida et al. 2018a), due to browsing by free-grazing livestock that kills naturally regenerated or planted tree seedlings (Endale et al. 2017). In addition, there is a preference by small-holder farmers to allocate their land to food crops in order to maximise food production for home consumption or sale (Abebe et al. 2013). Increasing tree population density in the study area could be a viable tree management strategy to enhance crop production in the long-term, as maize yield and biomass were greater under tree crowns compared to crop only plots. The results of the present study suggest that maize productivity under F. albida trees could be further improved by pruning and the application of fertilisers, particularly during high or average rainfall seasons. However, use of chemical fertilisers for smallholder maize production is limited by the high cost of fertilisers and the associated financial risk (Bacha et al. 2001).
Field experiments, conducted at particular points in time and space, can provide valuable information on system performance, but they are expensive, time consuming and can be of limited value in the quantitative transfer of experiences between sites. Alternatively, simulation models offer a means of quantitatively integrating complexity to evaluate system performance (Kassie et al. 2014; Smethurst et al. 2017) and to conduct virtual experiments (Luedeling et al. 2016). Therefore, we recommend that further agroforestry research in the parklands of Ethiopia includes simulation of biophysical productivity and socio-economic factors, e.g. tree-crop interactions at the plot scale, and fertilizers, manures, wood, fuel, livestock, labour, and other socio-economic factors at the farm scale. At the plot scale, the APSIM (Agricultural Production Systems sIMulator) tree-crop daily simulation model could potentially be adapted to parkland systems as it can integrate a range of biophysical factors including water, N and microclimate (Dilla et al. 2017; Sida et al. 2018b; Smethurst et al. 2017). The biophysical outputs of APSIM could then be used as inputs to farm-scale livelihood modelling options such as Simile (Muetzelfeldt and Massheder 2003), APSFarm (Rodriguez et al. 2006) and Farm-Safe (Graves et al. 2011).
References
Abebe T, Sterck F, Wiersum K, Bongers F (2013) Diversity, composition and density of trees and shrubs in agroforestry homegardens in Southern Ethiopia. Agrofor Syst 87:1283–1293
Argaw M, Teketay D, Olsson M (1999) Soil seed flora, germination and regeneration pattern of woody species in an Acacia woodland of the Rift Valley in Ethiopia. J Arid Environ 43:411–435
Bacha D, Aboma G, Gemeda A, Groote H (2001) The determinants of fertilizer and manure use in maize production in Western Oromiya, Ethiopia. In: Seventh Eastern and Southern Africa regional Maize conference proceedings
Baligar V, Fageria N, He Z (2001) Nutrient use efficiency in plants. Commun Soil Sci Plant Anal 32:921–950
Barnes R, Fagg CW (2003) Faidherbia albida monograph and annotated bibliography. Oxford Forestry Institute, Oxford
Bayala J, Sanou J, Teklehaimanot Z, Kalinganire A, Ouédraogo S (2014) Parklands for buffering climate risk and sustaining agricultural production in the Sahel of West Africa. Curr Opin Environ Sustain 6:28–34
Boffa J-M (1999) Agroforestry parklands in sub-Saharan Africa. FAO, Rome
Chamshama S, Mugasha A, Kløvstad A, Haveraaen O, Maliondo S (1998) Growth and yield of maize alley cropped with Leucaena leucocephala and Faidherbia albidain Morogoro, Tanzania. Agrofor Syst 40:215–225
Dilla A, Smethurst PJ, Barry K, Parsons D, Denboba M (2017) Potential of the APSIM model to simulate impacts of shading on maize productivity. Agroforestry Syst. https://doi.org/10.1007/s10457-017-0119-0
Dupuy NC, Dreyfus BL (1992) Bradyrhizobium populations occur in deep soil under the leguminous tree Acacia albida. Appl Environ Microbiol 58:2415–2419
Endale Y, Derero A, Argaw M, Muthuri C (2017) Farmland tree species diversity and spatial distribution pattern in semi-arid East Shewa, Ethiopia. For Trees Livelihoods 26:199–214
Geiger S, Manu A, Vandenbeldt R (1994) Variability in the growth of Faidherbia albida: the soils connection. Soil Sci Soc Am J 58:227–231
Goma M (2015) Characterization of climate variability and water harvesting system for crop production in Adulala Watershed, Central Rift Valley of Ethiopia. Haramaya University, Haramaya
Graves AR, Burgess PJ, Liagre F, Terreaux J-P, Borrel T, Dupraz C, Palma J, Herzog F (2011) Farm-SAFE: the process of developing a plot-and farm-scale model of arable, forestry, and silvoarable economics. Agrofor Syst 81:93–108
Hadgu KM, Kooistra L, Rossing WA, van Bruggen AH (2009) Assessing the effect of Faidherbia albida based land use systems on barley yield at field and regional scale in the highlands of Tigray, Northern Ethiopia. Food Security 1:337–350
Harmand J-M, Njiti CF (1992) Faidherbia albida in Northern Cameroon: Provenance trials and crop associations. Foidherbia albida in the West African Semi-Arid Tropics: Proceedings of a workshop, ICRISAT-ICRAF, pp 79–82
Jama B, Getahun A (1991) Intercropping Acacia albida with maize (Zea mays) and green gram (Phaseolus aureus) at Mtwapa, Coast Province, Kenya. Agrofor Syst 14:193–205
Jose S (2009) Agroforestry for ecosystem services and environmental benefits: an overview. Agrofor Syst 76:1–10
Jose S, Gillespie A, Seifert J, Biehle D (2000) Defining competition vectors in a temperate alley cropping system in the midwestern USA: 2. Competition for water. Agrofor Syst 48:41–59
Kamara C, Haque I (1992) Faidherbia albida and its effects on Ethiopian highland Vertisols. Agrofor Syst 18:17–29
Kassie B, Van Ittersum M, Hengsdijk H, Asseng S, Wolf J, Rötter RP (2014) Climate-induced yield variability and yield gaps of maize (Zea mays L.) in the Central Rift Valley of Ethiopia. Field Crops Res 160:41–53
Kho R, Yacouba B, Yayé M, Katkoré B, Moussa A, Iktam A, Mayaki A (2001) Separating the effects of trees on crops: the case of Faidherbia albida and millet in Niger. Agrofor Syst 52:219–238
Lal R (2006) Enhancing crop yields in the developing countries through restoration of the soil organic carbon pool in agricultural lands. Land Degrad Dev 17:197–209
Luedeling E, Smethurst PJ, Baudron F, Bayala J, Huth NI, van Noordwijk M, Ong CK, Mulia R, Lusiana B, Muthuri C (2016) Field-scale modeling of tree–crop interactions: challenges and development needs. Agric Syst 142:51–69
Makumba W, Janssen B, Oenema O, Akinnifesi FK, Mweta D, Kwesiga F (2006) The long-term effects of a gliricidia–maize intercropping system in Southern Malawi, on gliricidia and maize yields, and soil properties. Agric Ecosyst Environ 116:85–92
Muetzelfeldt R, Massheder J (2003) The Simile visual modelling environment. Eur J Agron 18:345–358
Nair P (2007) The coming of age of agroforestry. J Sci Food Agric 87:1613–1619
Orwa C, Mutua A, Kindt R, Jamnadass R, Simons A (2009) Agroforestree database: a tree species reference and selection guide version 4.0. World Agroforestry Centre ICRAF, Nairobi, KE
Poschen P (1986) An evaluation of the Acacia albida-based agroforestry practices in the Hararghe highlands of Eastern Ethiopia. Agrofor Syst 4:129–143
Power AG (2010) Ecosystem services and agriculture: tradeoffs and synergies. Philos Trans R Soc Lond B Biol Sci 365:2959–2971
Rao M, Nair P, Ong C (1997) Biophysical interactions in tropical agroforestry systems. Agrofor Syst 38:3–50
Rhoades C (1995) Seasonal pattern of nitrogen mineralization and soil moisture beneath Faidherbia albida (synAcacia albida) in central malawi. Agrofor Syst 29:133–145
Rodriguez D, DeVoil P, Cox H, Chudleigh F, Conway M, Routley R (2006) APSFARM: a whole farm systems analysis of economic and environmental indicators of contrasting farm business strategies in Central Queensland. In: Proceedings 13th of the Australian agronomy conference
Roupsard O, Ferhi A, Granier A, Pallo F, Depommier D, Mallet B, Joly H, Dreyer E (1999) Reverse phenology and dry-season water uptake by Faidherbia albida (Del.) A. Chev. in an agroforestry parkland of Sudanese west Africa. Funct Ecol 13:460–472
Saka A, Bunderson W, Itimu O, Phombeya H, Mbekeani Y (1994) The effects of Acacia albida on soils and maize grain yields under smallholder farm conditions in Malawi. For Ecol Manag 64:217–230
Semwal R, Maikhuri R, Rao K, Singh K, Saxena K (2002) Crop productivity under differently lopped canopies of multipurpose trees in Central Himalaya, India. Agrofor Syst 56:57–63
Sida TS, Baudron F, Deme DA, Tolera M, Giller KE (2018a) Excessive pruning and limited regeneration: are Faidherbia albida parklands heading for extinction in the Central Rift Valley of Ethiopia? Land Degrad Dev 29:1623–1633
Sida TS, Baudron F, Kim H, Giller KE (2018b) Climate-smart agroforestry: Faidherbia albida trees buffer wheat against climatic extremes in the Central Rift Valley of Ethiopia. Agric For Meteorol 248:339–347
Sileshi GW (2016) The magnitude and spatial extent of influence of Faidherbia albida trees on soil properties and primary productivity in drylands. J Arid Environ 132:1–14
Siriri D, Ong C, Wilson J, Boffa J, Black C (2010) Tree species and pruning regime affect crop yield on bench terraces in SW Uganda. Agrofor Syst 78:65
Smethurst PJ, Huth NI, Masikati P, Sileshi GW, Akinnifesi FK, Wilson J, Sinclair F (2017) Accurate crop yield predictions from modelling tree-crop interactions in gliricidia-maize agroforestry. Agric Syst 155:70–77
Suresh G, Rao J (1998) Intercropping sorghum with nitrogen fixing trees in semiarid India. Agrofor Syst 42:181–194
Tittonell P, Vanlauwe B, Corbeels M, Giller KE (2008) Yield gaps, nutrient use efficiencies and response to fertilisers by maize across heterogeneous smallholder farms of western Kenya. Plant Soil 313:19–37
Umar BB, Aune JB, Lungu OI (2013) Effects of Faidherbia albida on the fertility of soil in smallholder conservation agriculture systems in eastern and southern Zambia. AJAR 8:173–183
Vandenbeldt R, Williams J (1992) The effect of soil surface temperature on the growth of millet in relation to the effect of Faidherbia albida trees. Agric For Meteorol 60:93–100
Acknowledgements
This research was conducted as part of the Trees-for-Food-Security project (http://aciar.gov.au/aifsc/projects/trees-food-security-improving-sustainable-productivity-farming-systems-and-evergreen), under the auspices of the CGIAR research programme on Forests, Trees and Agroforestry. The project was managed by ICRAF with financial support from ACIAR and project partners (including CSIRO). We thank staff at Melkassa Agricultural Research Centre and ICRAF Ethiopia for field and laboratory support. We thank Professor Catherine Muthuri for her professional advice at the inception of the project proposal. We thank development agents and farmers at Adulala village for their assistance and cooperation during field work. We also thank Chris Harwood, Daniel Mendham and anonymous reviewers for comments on earlier drafts of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dilla, A.M., Smethurst, P.J., Barry, K. et al. Tree pruning, zone and fertiliser interactions determine maize productivity in the Faidherbia albida (Delile) A. Chev parkland agroforestry system of Ethiopia. Agroforest Syst 93, 1897–1907 (2019). https://doi.org/10.1007/s10457-018-0304-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10457-018-0304-9