Abstract
Bioaerosols are reported to affect human health and cause chronic inflammation to the respiratory system, leading to its temporary or permanent damage. This study aims to perform the qualitative assessment of ambient air of Barrackpore, an industrial township of West Bengal, India, by analysing the airborne fungal spore diversity for two consecutive years. The spores of ambient air were trapped using Burkard 7-day volumetric sampler from June 2014 to May 2016. The association of major fungal taxa with environmental parameters was analysed by Spearman’s rank correlation coefficient and multiple regression analysis to identify the significant predictors. The daily average ambient fungal spore concentration was 3526.38 ± 2709.32 spores m−3. Ascospore, basidiospore, Periconia and Aspergillus/Penicillium spp. accounted for more than 65% of observed fungi, resulting in the major fungal taxa. A significant association of dominant fungi with meteorological parameters and air pollutants was observed. Additionally, stepwise multiple regression analysis pointed out that dewpoint, wind speed, particulate matter with an aerodynamic diameter ≤ 2.5(PM2.5) and atmospheric nitrogen dioxide concentration (NO2) are the significant predictors for dominant fungi. Analysis of daily ambient fungal spore concentration and determining their environmental determinants will give an insight into the ambient air quality of the residential area of Barrackpore, for the first time. The acquired data can be used to evaluate the health impact on the residents of an unevaluated industrial township of India.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The air quality in residences adjacent to an industrial area is often a concern point due to its adverse effect on human health. The air of an industrial area is heavily polluted with airborne particulate matters, fumes and gases which are often grouped under the umbrella term ‘aerosol’. About 25% of the ambient aerosol is of biological origin (Matthias-Maser and Jaenicke 2000). Fungal spore, an important component of ambient bioaerosols, contributes 4–11% to fine suspended particles in urban and rural air (Womiloju et al. 2003). Frequent exposure to spores at higher concentration, mainly of particle diameter 1–5 µm, may cause severe respiratory allergy (Barui and Chanda 2000; Denning et al. 2006). The prevalence of fungal allergy was estimated to be about 3–10% on the general population (Twaroch et al. 2015), and about 1 in 10 asthmatic patients of the world belong to the Indian subcontinent (Kant 2013; Koul and Patel 2015). The ambient fungal spore concentration of an area is dependent upon numerous factors like geographical position, environmental conditions, vegetation, as well as population density and human activity in that area (Adhikari et al. 2006; Haas et al. 2013). The temporal and spatial distribution of airborne fungi has been investigated widely all over the world (Aira et al. 2012; Kallawicha et al. 2017; Fang et al. 2019; Liu et al. 2019). National Allergy Bureau (NAB) of American Academy of Allergy, Asthma and Immunology, in the USA, provides online day to day pollen and mould level as well as forecasts, that can be used as a guideline to the sensitive groups in residential areas for minimizing the exposure (NAB 2019). Several studies have been performed in India regarding assessing the diversity of aeromycoflora and their incidence upon human health in different geographical regions (Garaga et al. 2019; Chakrabarti et al. 2012; Shukla and Shukla 2011). Studies performed in local areas of West Bengal indicate that local climate has an effect on ambient spore distribution (Chakraborty et al. 2003; Das and Gupta-Bhattacharya 2008; Chakrabarti et al. 2012; Roy et al. 2017; Dey et al. 2019). In this study, we aimed to investigate the relationship of airborne fungal spore concentration with environmental parameters in an unexplored biozone of Barrackpore, an industrial township of West Bengal. We monitored fungal spore concentration in ambient air of Barrackpore township for two consecutive years.
2 Material and methods
2.1 Study area
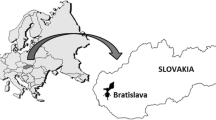
Barrackpore (22.7674° N, 88.3883° E) is a well-known industrial township of the Indian state, West Bengal. Geographically it is situated in Indo-Gangetic plain. The total area of this city is 25 km2 with a population size of 1,52,783 and male and female ratio of approximately 1.05. The sampling site is densely populated and not very far from a chemical manufacturing and an aerospace industry plant. Additionally, lots of commercial buildings, housings and a congested market area are present adjoining the site. The connecting roads have moderate to high traffic. The vegetation of the area is highly influenced by human activity. Some trees, shrubs and garden plants were present in close proximity to the spore trap. The geographical position of the sampling site is illustrated in Fig. 1.
2.2 Sampling and analysis of ambient fungal spore concentration
The daily ambient fungal spore concentration was monitored using Burkard 7-day volumetric spore trap (Burkard Manufacturing Co. Ltd, UK) following the guidelines as stated in British Aerobiology Federation (BAF 1995) from June 2014 to May 2016. The Sampler was placed on a rooftop of a residential building at a height of approximately 2 m from the ground. Fungal spores were trapped on glycerin jelly coated clean film (Melinex tape, Burkard Manufacturing Co., Rickmansworth, UK) on a rotating drum. The orifice throughput was adjusted to 10 L min−1. The drum is changed after 7 days throughout the monitoring period. The exposed tape with trapped fungal spore is cut into 48 mm length, each representing the 24 h record, mounted with distyrene, plasticizer and xylene (DPX)-mounting medium on microscope slides. Trapped spores were observedon a high-resolution microscope (Leitz, Diaplan, Germany) at 400 × magnification. Spores were identified following a standard manual (Ellis 1971; Smith et al. 1981) and counted according to British Aerobiology Federation (BAF 1995). Identification was made up to the genus level. Those which were difficult to classify, were grouped as ‘Unidentified’. The total spore count was converted to the number of spores m−3 by a conversion factor of 0.46, and daily mean spore concentration m−3 was obtained by adding all 24 h data.
2.3 Environmental parameters
The meteorological parameters were collected from the Regional Meteorological Center, Dumdum. Daily averages of temperature (T), wind speed (W), total rainfall (R), dewpoint (Td) and relative humidity (r.h.) were collected. For atmospheric pollutants, daily averages of suspended particulate matter (PM10 and PM2.5), sulphur dioxide (SO2), nitrogen dioxide (NO2), ozone (O3) and carbon monoxide (CO) were collected from the online public database of West Bengal Pollution Control Board (https://www.wbpcb.gov.in/) for Barrackpore area. For statistical analysis, we have used the average 24 h concentration. Based on atmospheric variables, four different distinguished seasons were categorized, i.e. Summer (early March to late May), Monsoon (early June to late September), Autumn (early October to late November) and Winter (early December to late February).
2.4 Statistical analysis
Data were analysed in GraphPad Prism 6 (La Jolla, California, USA) and IBM SPSS statistics version 24.0 (IBM, Armonk, New York, USA). The graph was generated using GraphPad Prism 6. The normality of all datasets was checked priorly. The consistency in spore concentrations throughout the sampling period was checked also through the unpaired t test. The temporal distribution of environmental parameters and fungal spore concentration in different seasons were checked through one-way ANOVA or Kruskal–Wallis test. The correlations between spore concentrations and individual environmental parameter were assessed by Spearman's rank correlation coefficient (r) (two-tailed). Further univariate regression analysis was performed between each fungal taxon and potential predictor variables. Variables with p value < 0.2 were included in multiple regression analysis for each fungal taxon (response variables), considering they are in linear relation (Stevens 1996). The stepwise linear regression analysis was performed (Landau and Everitt 2003) in SPSS. Changes in fungal concentration in different seasons were also assessed as a categorical variable in multiple regression analysis. Final regression model includes variables with p < 0.01. The spore concentrations were converted to logarithmic base 10 for an approximation of normality for regression analysis.
3 Results
Airborne fungal spores were monitored daily for two consecutive years by implementing Burkard 7-day volumetric spore trap. The total aerial concentration ranged from 580.6 to 12,188.40 spore m−3 with a mean ± standard deviation (SD) of 3526.37 ± 2709.32 spores m−3. Ascospores were the most prevalent taxa followed by basidiospores, Periconia, and Aspergillus/Penicillium spp. having a mean of 1471.39, 712.12, 333.19, and 216.98, respectively. More than 65% of spores belonged to these four dominant taxa. The frequency distribution of fungal spores is listed in Table 1. The unpaired t test validated consistency in spore concentration during the sampling period, as shown in Online Resource 1. The distribution of environmental parameters is shown in Table 2. The parameters varied significantly between seasons (p < 0.0001). Figure 2 describes the ambient concentrations of total spore and dominant fungal taxa in different seasons. The seasonal effect was prominent in total spores, ascospore and basidiospore during the monitoring period (p < 0.05). The data is shown in Online Resource 2.
The temporal distribution of total spore concentration and dominant taxa in each month are shown in Fig. 3. Ambient total spore concentration was higher during the post-monsoon period, especially in November with a mean concentration of 11,184 spores m−3. The lowest concentration was observed during December with a mean concentration of 667 spores m−3.
The correlation between atmospheric parameters and fungal concentrations were determined through Spearman’s rank correlation coefficient (r) and are presented in Table 3. No environmental parameters showed significant association with total fungal spore concentrations during the monitoring period. However, the major fungal taxa showed a strong correlation with both meteorological parameters and air pollutants. Temperature and dewpoint had positive effects on ascospore concentration, whereas a negative effect was observed on basidiospores and Aspergillus/Penicillium spp. concentrations. Additionally, a negative association of temperature with Periconia concentration was spotted. Rainfall and wind speed also showed a similar kind of correlation pattern with most of the major fungal taxa. The r.h. showed a negative association with both basidiospore and Aspergillus/Penicillium spp. concentrations.
Conversely to meteorological parameters, air pollutants like particulate matters (PM10 and 2.5), SO2 and NO2 showed positive effects on basidiospore and Aspergillus/Penicillium spp. and negative effects on ascospore concentrations. Periconia, by following a similar pattern, exhibited a positive association with PM2.5 and NO2 concentrations. Gaseous pollutants O3 and CO showed positive effects on Aspergillus/Penicillium spp. and negative effect on ascospore concentrations.
The stepwise multiple regression analysis is presented in Table 4. The regression models include predictor variables with p value < 0.01. The results indicate that both meteorological parameters and air pollutants are significant predictors for major fungal taxa in the study area. Wind speed had a significant positive effect on ascospores but showed a negative association with Periconia. Dewpoint showed a negative effect on Aspergillus/Penicillium spp. concentration. Air pollutant PM2.5 showed a significant positive effect on basidiospore and Aspergillus/Penicillium spp. concentrations and NO2 showed a negative association with ascospore concentration. Despite having significant predictor variables in multiple regression analysis for each dominant fungal taxon during the study period, lower goodness of fit was observed with R2 value < 0.5 (Table 4).
4 Discussion
In this study, the aerial fungal spore concentration was monitored in an industrial township of West Bengal, India, over two consecutive years. The daily average spore concentration was recorded as 3526.37 ± 2709.32 spores m−3, which is similar to other studies performed in the urban environment (Kallawicha et al. 2017; Fernández-Rodríguez et al. 2014). However, the mean concentration value varies widely and is quite lower than the observed concentration of a study done in a subtropical climate by Quintero et al. (2010). The highest concentration observed by them is > 50,000 spores m−3, as compared to > 10,000 spores m−3 observed by the present study. Although spore concentration at studied biozone is quite higher than reported maximum monthly spore concentration (approximately 7500 spores m−3) observed by Dey et al. (2019) in the nearby metropolitan city, Kolkata, which is around 30 km away from study location, it might indicate that spore concentration is dependent on local climate and activities. However, a detailed comparative study is required to explain this concentration difference. The seasonal changes influence the aerial distribution of fungal spores in the studied area. The observed spore concentration was higher towards the end of the year (October, November) and lower during December, due to winter effect. This pattern is quite similar to a 5-year study performed in Poland where higher spore concentration was observed during September–November and comparatively lower during December due to the beginning of winter (Grinn-Gofroń 2011). Daily monitoring and quantification of fungal spore load have revealed that the four observed dominant fungi ascopore, basidiospore, Periconia and Aspergillus/Penicillium spp. accounted for more than 65% of samples during the monitoring period. According to the Nomenclature Subcommittee of the International Union of Immunological Societies (IUIS) (www.allergen.org), 88 allergens are reported from the Ascomycota phylum and 23 from the Basidiomycota phylum. In the present study, the aerial concentration of the important allergenic fungi from Ascomycota phylum such as Alternaria, Aspergillus, Penicillium, Cladosporium, Curvularia and Fusarium in the studied location were quantified separately. However, we were unable to identify any reported allergenic fungi from Basidiomycota phylum in observed fungal spectrum during the monotoring period. According to Harley et al. (2009), early wheezing in infants is associated with high concentrations of ascospore and basidiospore. The mean ambient concentration of ascospores and basidiospores in studied biozone is 1471.39 and 712.12 spores m−3, respectively,which is lower than the reported threshold concentration of 1500 spores m−3 to induce allergenic inflammation, according to Chen et al. (2014). However, their clinical importance to residents of the studied area should be subjected to detailed epidemiological studies.
Spearman’s correlation coefficient (r) showed that all meteorological parameters have significant effects on most of the dominant fungi (Table 3). Various studies have confirmed that meteorological factors have an effect on the distribution of fungal spores in ambient aerosols (Filali Ben Sidel et al. 2017; Grinn-Gofroń and Bosiacka 2015; Gioulekas et al. 2004). Temperature and dewpoint of a climate zone highly affect fungal species richness and composition (Grinn-Gofroń and Bosiacka 2015; Oliveira et al. 2009; Troutt and Levetin 2001). In this study, the temperature had a significant positive association with ascospore and a negative effect on the rest major fungi (Table 3). Similarly, dewpoint was positively associated with ascospores and negatively with basidiospores and Aspergillus/Penicillium spp. The regression analysis showed that dewpoint can act as a significant predictor and had a strong negative association with Aspergillus/Penicillium spp. (Table 4). Codependency of major fungal taxa with temperature and dewpoint indicate that these two parameters have a significant contribution to the ambient fungal spore concentration in the studied location. The negative association of fungal spores with wind speed is observed by some studies (Grinn-Gofroń et al. 2018). Basidiospore, Aspergillus/Penicillium spp. and Periconia showed a significant negative correlation with wind speed (Table 3). Wind disturbance, although helps in spore dispersal, promotes mixing and dilution too, which could be a probable cause for the reverse relation. Conversely, ascospore showed a significant positive association with wind speed which could be an effect of wind direction. Some pathogenic fungi exhibited a positive relationship with wind speed, according to Rapilly (1991). Stronger wind speed can often promote higher conidial displacement in a given direction as reported by Rieux et al. (2014), thus justifying the positive correlation. The negative association of ambient dry spores with relative humidity has been published by many studies (Grinn-Gofroń et al. 2018; Ianovici 2016; Sadyś et al. 2016). Rainfall elevates atmospheric moisture content by increasing relative humidity. Magyar et al. (2009) suggested that rainfall caused wash out of the dry spores and promoted aerosolisation of wet spores, mainly ascospores.This explains the observed positive association between rainfall and ascospore concentration in the studied area (Table 3). Several studies suggested that ascospores and basidiospores are prevalant during monsoon (Singh and Dahiya 2008; Das and Gupta-Bhattacharya 2012). Similarly, Li and Kendrick (1995) observed that the relative humidity had a positive association with ascospores and basidiospores. However, it showed a negative association with Aspergillus/Penicillium. A similar observation was reported by Garaga et al. (2019), suggesting that high relative humidity lowers the abundance of Aspergillus and Penicillium spores in ambient air. Das and Gupta-Bhattacharya (2012) identified relative humidity as an important predictor for seasonal periodicity of airborne fungal spores and also observed that some basidiospore type fungi such as rust spores and smut spores showed a significant negative correlation with rainfall and relative humidity. Moreover, it was observed that in the dry environment, some basidiospore type fungi and Periconia concentration in ambient air, followed a diurnal pattern, with an increase during the evening, when the relative humidity is usually low (Sreeramulu 1959). In this study, both rainfall and relative humidity showed a strong negative association with basidiospore and Aspergillus/Penicillium spp. (Table 3).
Increasing air pollution is a major concern in India. According to a report published by GBD MAPS Working Group, PM2.5 concentration in ambient air of West Bengal was among the highest concentrations, along with Bihar and Uttarpradesh, mainly because of uncontrolled coal combustion in industries, power plants, open burning of biomass and transportation (GBD MAPS Working Group 2018). These findings are also supported by other global studies (Väkevä et al. 1999; Querol et al. 2001; Viana et al. 2006). Fine particles of urban aerosols are generally formed due to vehicular and industrial emissions. Table 2 shows moderate particulate matter concentration in the studied area, according to National Ambient Air Quality Standards, India. Atmospheric pollutants have a direct impact on ambient spore concentration (Grinn-Gofroń et al. 2011). The concentration of PM10 and PM2.5 at the study area have a significant positive correlation with basidiospores and Aspergillus/Penicillium spp., whereas negative with ascosopores. Periconia showed a positive association with PM2.5 only.
Interestingly, fungi that are positively correlated with PMs were found to be negatively correlated with temperature, probably because the temperature can accelerate the chemical reactions of hydrocarbons in PM2.5 which produces toxic components that can alter the association of microorganisms with particles in the air (Alghamdi et al. 2014). A study performed in Bejing, China, reported that concentration of Aspergillus, Penicillium and Fusarium were abundant in PM2.5 (Yan et al. 2016). Another study confirmed that fungal spores contribute with a significant percentage of organic carbon of PM2.5–10 (Zhang et al. 2010). Multiple regression analysis showed that among suspended particles, PM2.5 has a significant positive association with aerial concentrations of Aspergillus/Penicillium spp. and basidiospores and can be considered as an important source of fungal spores in studied location (Table 4).
Table 2 shows that the concentration of gaseous pollutants, i.e. SO2, NO2, O3 and CO, were within permissible range in the studied location. The contribution of gaseous pollutants in degrading human respiratory health is well known from various studies. Additionally, reactive oxygen species (ROS) and reactive nitrogen species (RNS), produced by gaseous pollutants such as O3, NO and NO2, can induce chemical alteration in allergens which may promote enhancement of allergenic potency of environmental allergens (Reinmuth-Selzle et al. 2017). Table 3 indicates that SO2 and NO2 have a positive association with basidiospores and Aspergillus/Penicillium spp. Contrarily, an adverse effect of both pollutants was observed on ascospores. Similarly to this result, Ščevková et al. (2019) reported the significant negative association between NO2 and Leptosphaeria ascospore concentration whereas the association with Agrocybe and Ganoderma basidiospores was positive. The result from multiple regression analysis further confirmed that NO2 concentration in the studied area has a significant negative effect on ascospore concentration and can act as an important predictor for the same (Table 4). Exposure to external sources of SO2, O3 and CO have a fungicidal effect upon application at a certain concentration, which has been confirmed by many studies (Korzun et al. 2008; Savi and Scussel 2014; Mansfield et al. 1991; Sommer et al. 1981). These reports lead to hypothesise that gaseous pollutant concentration has a negative impact on fungal concentration. While this pattern was only followed by ascospores in the studied area, the opposite relation was observed for Aspergillus/Penicillum spp. Yan et al. (2016) reported that both SO2, NO2 had a significant positive association with the ambient fungal community of the studied area, including dominant fungi such as Aspergillus, Penicillium, Cladosporium, Alternaria, Fusarium and Sporisorium. On the other hand, Adhikari et al. (2006) observed that the ambient O3 concentration is positively associated with Aspergillus/Penicillium spp. as a function of temperature. Atmospheric temperature controls ambient ozone concentration (Elminir 2005) and this explains the increase of O3 concentration in the summer at the studied area. Another study performed in Taipei, Taiwan, showed lower O3 level in combination with higher temperature is associated with increased airborne fungi concentration (Chao et al. 2012). The ambient O3 level was fairly low in the studied atmosphere and it varied widely in different seasons (Table 2). Little variation in CO concentration was observed in the studied atmosphere (Table 2). Aspergillus/Penicillium spp. showed a strong positive correlation with atmospheric CO concentration (Table 3). A more humid environment with the presence of CO can promote the growth of airborne bacteria (Lighthart 1973) but how it is positively correlated with Aspergillus/Penicillium spp. spores is not clearly understood. However, a detailed study is required to determine the chemical effect of these pollutant on each fungal taxon.
The R2 value of four regression models ranged from 0.075 to 0.388, which is far from the optimum value of 0.7. The lowest value was observed for Periconia (R2 = 0.075, p < 0.01) and indicates that environmental parameters have a lower influence on it. However, the values are comparable with other studies (Ponce-Caballero et al. 2013; Hummel et al. 2015; Kallawicha et al. 2017). The environmental parameters are not stagnant during the year and every parameter is co-dependent on one another. This fluctuation in parameter values could be one of the reasons for the low R2 value in the models. However, the significance of the parameters does imply that the meteorological parameters and air pollutants are important predictors in determining fungal spore concentrations in the studied area. The regression models are capable of predicting the spore concentrations for most of the major fungal taxa from observed environmental parameters, which can provide important information to improve the current approaches for managing the health effect on sensitive population by minimising the fungal spore exposure.
5 Conclusion
Biomonitoring of fungal spore diversity at Barrackpore township has revealed the temporal variation in ambient spore load for 2 years. The highest spore concentration was observed during the post-monsoon period and lowest during winter. Most of the observed fungi are from ascospore, basidiospore, Periconia and Aspergillus/Penicillium spp., hence considered as dominant fungal taxa. Multiple regression analysis showed that meteorological parameter dewpoint and wind speed along with air pollutant PM2.5 and NO2 have a significant effect on shaping the fungal spectrum at the studied biozone and were considered as important atmospheric predictor variables in determining their atmospheric concentration. This study may provide baseline information for constructing a real-time model for predicting the atmospheric concentration of fungal aerosol in different seasonal conditions. The result of this study is important for investigating the health effects of these fungal spores and can act as a preliminary piece of information for developing a forecast model for minimizing the exposure of sensitive patients.
Availability of data and material
The authors declare that all data supporting the findings of this study are available within the article and its supplementary information files.
References
Adhikari, A., Reponen, T., Grinshpun, S. A., Martuzevicius, D., & LeMasters, G. (2006). Correlation of ambient inhalable bioaerosols with particulate matter and ozone: A two-year study. Environmental Pollution, 140(1), 16–28.
Aira, M. J., Rajo, F. J., Fernández-González, M., Carmen, S. C., Elvira-Rendueles, B., Abreu, I., et al. (2012). Spatial and temporal distribution of Alternaria spores in the Iberian Peninsula atmosphere, and meteorological relationships: 1993–2009. International Journal of Biometeorology, 57(2), 265–274.
Alghamdi, M., Shamy, M., Redal, M., Khoder, M., Awad, A., & Elserougy, S. (2014). Microorganisms associated particulate matter: A preliminary study. Science of The Total Environment, 479–480, 109–116.
Barui, N. C., & Chanda, S. (2000). Aeromycoflora in the Central Milk Dairy of Calcutta, India. Aerobiologia, 16, 367–372.
British Aerobiology Federation. (1995). Airborne pollens and spores. A guide to trapping and counting. Worcester: British Aerobiology Federation.
Chakrabarti, H. S., Das, S., & Gupta-Bhattacharya, S. (2012). Outdoor airborne fungal spora load in a suburb of Kolkata, India: Its variation, meteorological determinants and health impact. International Journal of Environmental Health Research, 22(1), 37–50.
Chakraborty, P., Gupta-Bhattacharya, S., & Chanda, S. (2003). Aeromycoflora of an agricultural farm in West Bengal, India: A five-year study (1994–1999). Grana, 42(4), 248–254.
Chao, H. J., Chan, C.-C., Rao, C. Y., Lee, C.-T., Chuang, Y.-C., Chiu, Y.-H., et al. (2012). The effects of transported Asian dust on the composition and concentration of ambient fungi in Taiwan. International Journal of Biometeorology, 56(2), 211–219.
Chen, B.-Y., Jasmine Chao, H., Wu, C.-F., Kim, H., Honda, Y., & Guo, Y. L. (2014). High ambient Cladosporium spores were associated with reduced lung function in schoolchildren in a longitudinal study. Science of The Total Environment, 481, 370–376.
Das, S., & Gupta-Bhattacharya, S. (2008). Enumerating outdoor aeromycota in suburban West Bengal, India, with reference to respiratory allergy and meteorological factors. Annals of Agricultural and Environmental Medicine, 15(1), 105–112.
Das, S., & Gupta-Bhattacharya, S. (2012). Monitoring and assessment of airborne fungi in Kolkata, India, by viable and non-viable air sampling methods. Environmental Monitoring and Assessment, 184(8), 4671–4684.
Denning, D. W., O'Driscoll, B. R., Hogaboam, C. M., Bowyer, P., & Niven, R. M. (2006). The link between fungi and severe asthma: A summary of the evidence. European Respiratory Journal, 27(3), 615–626.
Dey, D., Ghosal, K., & Bhattacharya, S. G. (2019). Aerial fungal spectrum of Kolkata, India, along with their allergenic impact on the public health: A quantitative and qualitative evaluation. Aerobiologia, 35, 15–25.
Ellis, M. B. (1971). Dematiaceous hyphomycetes. Surrey: Commonwealth Mycological Institute.
Elminir, H. K. (2005). Dependence of urban air pollutants on meteorology. Science of the Total Environment, 350, 225–237.
Fang, Z., Guo, W., Zhang, J., & Lou, X. (2019). Assemblages of culturable airborne fungi in a typical urban, tourism-driven center of Southeast China. Aerosol and Air Quality Research, 19(4), 820–831.
Fernández-Rodríguez, S., Tormo-Molina, R., Maya-Manzano, J., Silva-Palacios, I., & Gonzalo-Garijo, Á. (2014). Outdoor airborne fungi captured by viable and non-viable methods. Fungal Ecology, 7, 16–26.
Filali Ben Sidel, F., Bouziane, H., Kadiri, M., El haskouri, F., & del Mar trigo, M. (2017). Fungal spores of Cladosporium in the air of tetouan meteorological parameters and forecast models. International Journal of Environmental Sciences & Natural Resources, 3(2), 43–50.
Garaga, R., Avinash, C. K. R., & Kota, S. H. (2019). Seasonal variation of airborne allergenic fungal spores in ambient PM10—A study in Guwahati, the largest city of north-east India. Air Quality, Atmosphere & Health, 12(1), 11–20.
GBD MAPS Working Group. (2018). Burden of disease attributable to major air pollution sources in India. Special Report 21. Boston, MA: Health Effects Institute. Retrieved November 26, 2019, from https://www.healtheffects.org/system/files/GBD-MAPS-SpecRep21-India-revised_0.pdf. Posted in January 2018.
Gioulekas, D., Damialis, A., Balafoutis, C., Papakosta, D., Giouleka, P., & Patakas, D. (2004). Allergenic fungal spore records (15 Years) and relationship with meteorological parameters in Thessaloniki, Greece. Allergy & Clinical Immunology International—Journal of the World Allergy Organization, 16(2), 52–59.
Grinn-Gofroń, A. (2011). Airborne Aspergillus and Penicillium in the atmosphere of Szczecin, (Poland) (2004–2009). Aerobiologia, 27(1), 67–76.
Grinn-Gofroń, A., & Bosiacka, B. (2015). Effects of meteorological factors on the composition of selected fungal spores in the air. Aerobiologia, 31(1), 63–72.
Grinn-Gofroń, A., Bosiacka, B., Bednarz, A., & Wolski, T. (2018). A comparative study of hourly and daily relationships between selected meteorological parameters and airborne fungal spore composition. Aerobiologia, 34(1), 45–54.
Grinn-Gofroń, A., Strzelczak, A., & Wolski, T. (2011). The relationships between air pollutants, meteorological parameters and concentration of airborne fungal spores. Environmental Pollution, 159(2), 602–608.
Haas, D., Galler, H., Luxner, J., Zarfel, G., Buzina, W., Friedl, H., et al. (2013). The concentrations of culturable microorganisms in relation to particulate matter in urban air. Atmospheric Environment, 65, 215–222.
Harley, K. G., Macher, J. M., Lipsett, M., Duramad, P., Holland, N. T., Prager, S. S., et al. (2009). Fungi and pollen exposure in the first months of life and risk of early childhood wheezing. Thorax, 64(4), 353–358.
Hummel, M., Hoose, C., Gallagher, M., Healy, D. A., Huffman, J. A., O'Connor, D., et al. (2015). Regional-scale simulations of fungal spore aerosols using an emission parameterization adapted to local measurements of fluorescent biological aerosol particles. Atmospheric Chemistry and Physics, 15(11), 6127–6146.
Ianovici, N. (2016). Atmospheric concentrations of selected allergenic fungal spores in relation to some meteorological factors, in Timisoara (Romania). Aerobiologia, 32(1), 139–156.
Kallawicha, K., Chen, Y.-C., Chao, H., Shen, W.-C., Chen, B.-Y., Chuan, Y.-C., et al. (2017). Ambient fungal spore concentration in a subtropical metropolis: Temporal distributions and meteorological determinants. Aerosol and Air Quality Research, 17(8), 2051–2063.
Kant, S. (2013). Socio-economic dynamics of asthma. Indian Journal of Medical Research, 138(4), 446–448.
Korzun, W., Hall, J., & Sauer, R. (2008). The effect of ozone on common environmental fungi. Clinical Laboratory Science, 21(2), 107–111.
Koul, P., & Patel, D. (2015). Indian guidelines for asthma: Adherence is the key. Lung India, 32(7), 1–2.
Landau, S., & Everitt, B. S. (2003). A handbook of statistical analyses using SPSS. London: CRC Press Company.
Li, D.-W., & Kendrick, B. (1995). A year-round study on functional relationships of airborne fungi with meteorological factors. International Journal of Biometeorology, 39(2), 74–80.
Lighthart, B. (1973). Survival of airborne bacteria in a high urban concentration of carbon monoxide. Applied Microbiology, 25(1), 86–91.
Liu, H., Hu, Z., Zhou, M., Hu, J., Yao, X., Zhang, H., et al. (2019). The distribution variance of airborne microorganisms in urban and rural environments. Environmental Pollution, 247, 898–906.
Magyar, D., Frenguelli, G., Bricchi, E., Tedeschini, E., Csontos, P., Li, D.-W., et al. (2009). The biodiversity of air spora in an Italian vineyard. Aerobiologia, 25, 99–109.
Mansfield, P. J., Bell, J. N. B., McLeod, A. R., & Wheeler, B. E. J. (1991). Effects of sulphur dioxide on the development of fungal diseases of winter barley in an open-air fumigation system. Agriculture, Ecosystems & Environment, 33(3), 215–232.
Matthias-Maser, S., & Jaenicke, R. (2000). The size distribution of primary biological aerosol particles in the multiphase atmosphere. Aerobiologia, 16(2), 207–210.
NAB. (2019). NAB Pollen Counts. Retrieved October 14, 2019, from https://www.aaaaiorg/global/nab-pollen-countsaspx.
Oliveira, M., Ribeiro, H., Delgado, J. L., & Abreu, I. (2009). The effects of meteorological factors on airborne fungal spore concentration in two areas differing in urbanisation level. International Journal of Biometeorology, 53(1), 61–73.
Ponce-Caballero, C., Gamboa-Marrufo, M., López-Pacheco, M., Cerón-Palma, I., Quintal-Franco, C., Giácoman-Vallejos, G., et al. (2013). Seasonal variation of airborne fungal propagules indoor and outdoor of domestic environments in Mérida, Mexico. Atmósfera, 26(3), 369–377.
Querol, X., Alastuey, A., Rodriguez, S., Plana, F., Mantilla, E., & Ruiz, C. R. (2001). Monitoring of PM10 and PM2.5 around primary particulate anthropogenic emission sources. Atmospheric Environment, 35(5), 845–858.
Quintero, E., Rivera-Mariani, F., & Bolaños-Rosero, B. (2010). Analysis of environmental factors and their effects on fungal spores in the atmosphere of a tropical urban area (San Juan, Puerto Rico). Aerobiologia, 26(2), 113–124.
Rapilly, F. (1991). Épidémiologieenpathologievégétale. Mycoses aériennes. Versailles: Éditions Quae.
Reinmuth-Selzle, K., Kampf, C. J., Lucas, K., Lang-Yona, N., Fröhlich-Nowoisky, J., Shiraiwa, M., et al. (2017). Air pollution and climate change effects on allergies in the anthropocene: Abundance, interaction, and modification of allergens and adjuvants. Environmental Science & Technology, 51(8), 4119–4141.
Rieux, A., Soubeyrand, S., Bonnot, F., Klein, E. K., Ngando, J. E., Mehl, A., et al. (2014). Long-distance wind-dispersal of spores in a fungal plant pathogen: Estimation of anisotropic dispersal kernels from an extensive field experiment. PLoS ONE, 9(8), e103225–e103225.
Roy, S., Chakraborty, A., Maitra, S., & Bhattacharya, K. (2017). Monitoring of airborne fungal spore load in relation to meteorological factors, air pollutants and allergic symptoms in Farakka, an unexplored bio-zone of eastern India. Environmental Monitoring and Assessment, 189(8), 370.
Sadyś, M., Adams-Groom, B., Herbert, R. J., & Kennedy, R. (2016). Comparisons of fungal spore distributions using air sampling at Worcester, England (2006–2010). Aerobiologia, 32(4), 619–634.
Savi, G. D., & Scussel, V. M. (2014). Effects of ozone gas exposure on toxigenic fungi species from Fusarium, Aspergillus, and Penicillium genera. Ozone: Science & Engineering, 36(2), 144–152.
Ščevková, J., Hrabovský, M., Kováč, J., & Rosa, S. (2019). Intradiurnal variation of predominant airborne fungal spore biopollutants in the Central European urban environment. Environmental Science and Pollution Research, 26(33), 34603–34612.
Shukla, S., & Shukla, R. V. (2011). Air borne fungal spores in the atmosphere of industrial town Korba-Chhattisgarh, India. Microbiology Journal, 1(1), 33–39.
Singh, A. B., & Dahiya, P. (2008). Aerobiological researches on pollen and fungi in India during the last fifty years: An overview. Indian Journal of Allergy and Asthma Immunology, 22(1), 27–38.
Smith, G., Allsopp, D., Onions, A. H. S., & Eggins, H. O. W. (1981). Smith's introduction to industrial mycology (7th ed.). London: Edward Arnold.
Sommer, N. F., Fortlage, R. J., Buchanan, J. R., & Kader, A. A. (1981). Effect of oxygen on carbon monoxide suppression of postharvest pathogens of fruits. Plant Disease, 65(4), 347–349.
Sreeramulu, T. (1959). The diurnal and seasonal periodicity of spores of certain pathogens in the air. Transactions of the British Mycological Society, 42(2), 177–184.
Stevens, J. (1996). Applied multivariate statistics for the social sciences (3rd ed.). Mahwah, NJ: Lawrence Erlbaum Associates, Publishers.
Troutt, C., & Levetin, E. (2001). Correlation of spring spore concentrations and meteorological conditions in Tulsa, Oklahoma. International Journal of Biometeorology, 45(2), 64–74.
Twaroch, T. E., Curin, M., Valenta, R., & Swoboda, I. (2015). Mold allergens in respiratory allergy: From structure to therapy. Allergy, Asthma & Immunology Research, 7(3), 205–220.
Väkevä, M., Hämeri, K., Kulmala, M., Lahdes, R., Ruuskanen, J., & Laitinen, T. (1999). Street level versus rooftop concentrations of submicron aerosol particles and gaseous pollutants in an urban street canyon. Atmospheric Environment, 33(9), 1385–1397.
Viana, M., Querol, X., & Alastuey, A. (2006). Chemical characterisation of PM episodes in NE Spain. Chemosphere, 62(6), 947–956.
Womiloju, T. O., Miller, J. D., Mayer, P. M., & Brook, J. R. (2003). Methods to determine the biological composition of particulate matter collected from outdoor air. Atmospheric Environment, 37(31), 4335–4344.
Yan, D., Zhang, T., Su, J., Zhao, L.-L., Wang, H., Fang, X.-M., et al. (2016). Diversity and composition of airborne fungal community associated with particulate matters in Beijing during haze and non-haze days. Frontiers in Microbiology, 7, 487.
Zhang, T., Engling, G., Chan, C.-Y., Zhang, Y.-N., Zhang, Z.-S., Lin, M., et al. (2010). Contribution of fungal spores to particulate matter in a tropical rainforest. Environmental Research Letters, 5(2), 024010.
Acknowledgements
This work is financially supported by the Department of Science and Technology, Government of India (Grant No. DST/INSPIRE Fellowship/2013/1103). The authors are thankful to the Director of the Bose Institute for providing the infrastructural facility. Sincere thanks to Chanchal Chakraborty, Soumyo Subhra Gupta, Jadab Kumar Ghosh, Kaberi Ghosh, Ashish Bera and all lab members for their technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Roy, S., Gupta Bhattacharya, S. Airborne fungal spore concentration in an industrial township: distribution and relation with meteorological parameters. Aerobiologia 36, 575–587 (2020). https://doi.org/10.1007/s10453-020-09653-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10453-020-09653-9