Abstract
Egeria densa and Elodea canadensis are two common invasive submerged macrophytes in streams and rivers worldwide. We conducted a six-week growth experiment with monocultures of E. densa and E. canadensis and mixed communities of the two species under five different shade levels (35%, 63%, 79%, 90% and 95%). Our aim was to test the effect of shade and competition on colonization of vegetative propagules of these species in flowing waters. We found that biomass accrual in both species was greatly reduced at high shade (≥ 79%) and that E. densa individuals accrued more biomass than E. canadensis individuals at all shade levels. At the least shade level (35%), interspecific competition with E. densa stimulated biomass accrual of E. canadensis individuals, compared to those within an E. canadensis monoculture. At higher shade (≥ 63%), this facilitation response disappeared, although E. canadensis plants continued to have longer stem length and a greater degree of branching than E. densa plants at ≤ 79% shade. At the highest shade level, competition with E. canadensis reduced biomass accrual of E. densa. Hence, shade influenced the competitive interactions between these two species. At low shade, the competitiveness of E. canadensis was enhanced, but in the field E. densa appears to be the dominant species in streams, especially those with higher levels of shade and less frequent disturbance by flushing flow events.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Macrophyte beds affect stream ecosystem structure by modifying flow velocity, substrate conditions, stream depth and cross-sectional area (Sand-Jensen 2002; Champion and Tanner 2000). They also play a critical role in the functioning of stream ecosystems as they influence primary production (Alnoe et al. 2015), nutrient cycling (Levi et al. 2015) and biodiversity (e.g., Bell et al. 2013). Since streams are naturally unstable ecosystems with frequent disturbances due to hydrological events, vegetation dynamics in these systems are usually intense (Riis 2008; Riis and Sand-Jensen 2006). Many stream macrophyte species propagate via stem fragments, most often generated during hydrological events (Riis 2008; Riis and Sand-Jensen 2006). Retention of fragmentsis facilitated by the presence of obstacles in the stream bed, including established macrophyte beds, woody debris and incursions of riparian vegetation into the channel (Riis and Sand-Jensen 2006). Trapped stem fragments are a primary source of colonizing propagules which can create new populations, or re-establish existing ones, following disturbance events.
The first interaction that macrophyte propagules have with the recipient environment is likely to be competition for limiting resources (Vila and Weiner 2004). Competition occurs between species (interspecific) and within species (intraspecific) due to the need to acquire resources for growth, and this influences the relative abundance and dominance of species (Thiébaut et al. 2019; Maestre et al. 2009; Milne et al. 2007). Interspecific competition can be strong amongst species, especially those which are biologically and ecologically similar. For example, Hofstra et al. (1999) found that the relative competitive potential of the Hydrocharitaceae Hydrilla verticillata was low when it was competing with morphologically similar species in the same family (i.e., Egeria densa, Elodea canadensis and Lagarosiphon major), but it was high when competing with Ceratophyllum demersum (Ceratophyllaceae), which is morphologically distinct. For this study, Egeria densa Planch. and Elodea canadensis Michx. were selected as being two common non-native, and invasive, submerged aquatic macrophytes in New Zealand streams and rivers (Champion et al. 2013; Riis et al. 2012). They are morphologically similar so we anticipated that there would be strong interspecific competition between the two species. However, E. densa is regarded as the more invasive of the two species in New Zealand according to an aquatic weed risk assessment model (Champion et al. 2014).
A non-native species is one that has arrived to a new region other than the region where it evolved (Davidson et al. 2011). High competitive ability is considered to be the primary factor dictating the invasion potential of an exotic species (Vila and Weiner 2004). Recent studies have examined alterations in dominance of invasive species when competing for resources such as nutrient, light and carbon (Connolly et al. 2001; Martin and Coetzee 2014; Eller et al. 2015). Several studies have investigated the controlling effects of these biophysical factors for colonization and growth of exotic macrophyte species (Martin and Coetzee 2014; Riis et al. 2012), whereas less is known about the effect of interspecific competition on the colonization success of these species in streams.
Light is an essential resource for photosynthesis; hence, it is a prominent limiting factor affecting the distribution of submerged aquatic macrophytes (Lacoul and Freedman 2006). In streams, shade from riparian vegetation affects light availability, especially along stream margins, and this may limit the invading ability of exotic stream macrophytes (Evangelista et al. 2017). Shade tolerance and light-related plastic adaptations are advantageous in competitive environments (Barko and Smart 1981; Lacoul and Freedman 2006). In general, increasing shade negatively affects growth rate, biomass accrual and canopy formation, while it positively affects stem elongation in monoculture stands (Riis et al. 2012; Zefferman 2014). This study aimed to investigate the influence of shade, and thus limitation of light, on competition between E. densa and E. canadensis, from the initial colonization stage. More specifically, we tested the influence of shade on competition in colonizing monocultures of each species and in mixed communities of the two species in an outdoor recirculating mesocosm which mimicked a slow-flowing large stream system. We examined the influence of shade on the competitive traits of individual plants growing within the different community compositions. In streams, macrophyte competitiveness is conferred by morphological traits that maximize plant photosynthesis. For example, high stem length and high branching degree are considered to be beneficial traits for competition because they increase light harvesting (Riis et al. 2012). We also measured overall species biomass accrual to evaluate the overall effect of shade and competition on the community.
Methodology
Egeria densa and E. canadensis are well known invaders in lakes, rivers and streams in many parts of the world including New Zealand (Zefferman 2014; Champion et al. 2013; Riis et al. 2012). Both species mainly propagate by stem fragments (Champion et al. 2013; Riis et al. 2012). For this study, plant specimens were collected from the Waitoa River (37.7517° S, 175.7171° E), North Island, New Zealand. Specimens were stored in a chiller at 4 °C for 48 h. Then apical shoots were harvested and planted into pots and placed in the experimental mesocosm.
Shoots were planted into 800 mL (9 cm diameter, 12.5 cm deep) plastic pots filled with 1/3 garden soil at the bottom and 2/3 washed sand on top. Commercial grade garden soil was used to ensure sufficient nutrient availability for plant growth in the substrate (similar to Riis et al. 2012). Total carbon (C) and nitrogen (N) in sediment samples (three replicates, sand and garden soil separately) were analyzed using a C/N analyzer (Procedure MAM, 01-1090, Elementar, Germany). Sediment total recoverable phosphorus (TRP) was determined using an ICP-MS (after a nitric/hydrochloric acid digestion, Method—EPA 200.2). Sediment C, N and TRP contents were 0.030 ± 0.006%, 0.020 ± 0.001% and 0.010 ± 0.001% in sand and 5.15 ± 0.92%, 0.27 ± 0.03% and 0.050 ± 0.002% in garden soil, respectively.
Replicate apical shoots (10 cm length) of one or both species were planted into each pot. Similar biomass was introduced to each pot. Egeria densa was approximately twice as heavy by weight (wet, dry) as E. canadensis. Six apical shoots from each species were set aside for initial quantification of biomass (grams dry weight, gdw). This confirmed that the weight ratio of E. densa: E. canadensis was 1.95:1. The mean dry weight of E. densa and E. canadensis 10-cm-long apical tips used in the study was 0.059 ± 0.019 g and 0.030 ± 0.007 g, respectively. Hence, two apical shoots of E. densa were planted in each pot for pots only with E. densa (hereafter Ed pots and individual plants named Ed), while four apical shoots were introduced for pots only with E. canadensis (hereafter Ec pots and individual plants named Ec). Mixed community pots were planted with one apical tip of E. densa and two apical tips of E. canadensis, hence introducing similar biomass to each pot (hereafter named mixed pots and each species grown in mixed community was named as Ed-Mix and Ec-Mix). This study was planned as a simple replacement design, in which all the experimental pots received similar initial biomass, with the mixed communities receiving approximately half the biomass of each species relative to the monocultures (Harper 1977). A similar approach has been used previously to study interactions between E. densa and Stuckenia pectinata in monoculture and mixed culture along environmental gradients (Borgnis and Boyer 2016). A total of 15 plant pots were assigned to each shade treatment (3 competition treatments × 5 replicates), yielding a total of 75 plant pots (15 pots × 5 shade treatments). The plants were grown for 6 weeks from March 8 to April 19, 2018 (early to mid-austral autumn). We selected this time of year and experiment durations broadly representative of the period of peak macrophyte growth and biomass accrual, and a typical duration between scouring flood flow events in North Island, New Zealand, lowland agricultural streams. Six weeks was considered an adequate time period for colonization in a new habitat in the latter part of the growing season.
The experiment was set up in a large recirculating outdoor mesocosm (20 m long, 1.5 m wide, water depth—0.75 m) at the Ruakura experimental facility (see Redekop et al. 2016 for image). The tank was filled with clear potable water [Hamilton City Water Supply, typically containing 470 µg NO3–N L−1, < 10 µg NH4–N L−1 and 5 µg PO4−3–P L−1 with a total alkalinity of 30.4 mg L−1 (0.6 mEq L−1) and pH 7.5]. Water with relatively low nutrient concentrations was used to minimize the growth of phytoplankton and epiphyton, which would likely further reduce light availability. We considered that the garden soil added to pots at depth would provide sufficient nutrients in porewater to enable macrophyte growth, without generating a significant flux of nutrients into the overlying water of the mesocosm. Flow velocity in the mesocosm was adjusted to be in the range of 0.05–0.08 ms−1 by adjusting the rotating velocity of a paddle wheel. Flow velocities of this magnitude are typical of macrophyte-dominated lowland streams in North Island, New Zealand, during summer (see Champion and Tanner 2000). Light availability was manipulated by using shade cloth covers. Nominal percentages of shading relative to ambient light provided by the cloth covers were 30%, 50%, 70%, 80% and 90%. Earlier experiments at this facility showed that this range of shading supported the growth of both species (Kankanamge et al. 2019). Ambient light in daylight hours varied from 75 to 2915 µmol m−2 s−1 during the experimental period (Radiation data from Ruakura climate station). Daylight hours were between approximately 6 AM and 6 PM, while the peak values were usually observed in between 12 and 3 PM. Actual shading provided by the shade cloth covers was calculated by measuring the available light at the base of the tank (filled with water) in the absence of plants and ambient light above the shade cloth cover by using a Licor UWQ 9346 underwater quantum sensor connected to a LI-1500 light sensor logger. Measurements were made on March 29, April 4 and April 16 at mid-day, and percentage shade at the base of the tank was calculated and averaged, with the values being 35%, 63%, 79%, 90% and 95%, respectively. The average photosynthetically available radiation (PAR) experienced by plants in each shade treatment during the six-week experiment was 28, 16, 9, 4 and 2 mol m−2 d−1, respectively.
Competitive morphology traits were measured at the end of the experiment. Individual plants were carefully separated. For each plant, we measured the main stem length and the length of each branch separately. Thereafter, shoots and roots were separated and plant material was dried in an oven at 80 °C for 48 h. Biomass accrual was measured as total shoot dry weight, root dry weight, root/shoot ratio, dry weight/unit length, length of main stem and length of each branch. Relative growth rate (RGR), branching degree (number of branches/main stem length), lateral spread (total plant length/main stem length) and weight of a unit length (dry biomass/ total stem length) were calculated using the above measurements. Relative growth rate was calculated based on the mean of the natural logarithm of the dry weights according to Hoffman and Poorter (2002).
Data processing
All analyses were performed in SPSS 21.0 software. Two-way ANOVA was used to evaluate the influences of shade and plant community composition on plant growth and morphology. One-way ANOVA was used to isolate the effect of shade on each species grown in different community compositions. Tukey multiple comparison tests were used to identify significantly different groups.
Measured percentage shade at the base of the mesocosm was used as the shade level to which plants were exposed. The influence of shading on intraspecific and interspecific competition was studied on individual plants and pots. Growth and morphology results were compared at the individual plant level, while biomass accrual results were compared at the pot level. Pot-level biomass production was considered to reflect species behavior in an individual patch or in a mixed community.
Results
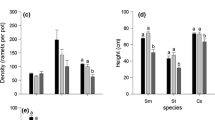
Shoots of both species were able to grow under all shade conditions. However, shade and community composition had effects on the growth and morphology of both species. Overall, total biomass accrual in pots was significantly reduced when shade increased (one-way ANOVA, F = 77.06, P < 0.001). At 35% shade and 95% shade, total biomass accrual in pots differed among plant community compositions but at shade levels 63%, 79% and 90% biomass accrual was similar for E. densa monocultures, E. canadensis monocultures and mixed communities (Fig. 1). At 35% shade, monoculture E. canadensis communities accumulated significantly lower biomass than mixed communities and monoculture E. densa communities, which had similar biomass. At 95% shade, monoculture E. densa communities accrued more biomass than monoculture E. canadensis communities and mixed communities.
Increasing shade from 35 to 95% generally reduced biomass accrual for individual plants of both species, especially E. densa (Table 1). At higher shade levels, especially ≥ 79%, biomass accrual of both E. canadensis and E. densa was reduced. Egeria densa plants were primarily affected by shade. The presence of E. canadensis generally had little effect on biomass accrual, growth and morphological traits of E. densa except at high shade (95%) where biomass accrual and RGR were reduced (Table 1, Fig. 2). In contrast, both shade and community composition affected the biomass accrual, growth and morphology of E. canadensis (Table 1). Interspecific competition had a positive influence on biomass accrual of E. canadensis plants at 35% shade. E. canadensis individuals accrued a similar biomass to E. densa individuals when the two species were grown together at this shade level (Fig. 2). Furthermore, E. canadensis plants had a longer main stem length, and greater lateral spread and branching degree than E. densa plants when grown together at 35% shade (Fig. 2), although the RGR of E. canadensis plants was lower. However, at higher shading levels the facilitation received by E. canadensis due to the presence of E. densa disappeared, although main stem length of E. canadensis plants continued to be longer than E. densa plants at 63% and 79% shade. At the highest shade level (95%), the presence of E. canadensis was found to reduce biomass accrual and RGR of E. densa individuals. Egeria densa plants accrued a higher biomass compared to E. canadensis plants at all shade levels, except 95% shade level (Fig. 2). Biomass allocation to roots reduced as shade increased for both species with no apparent effect of intra- or inter-specific competition.
Effect of shade on morphological and physiological traits after six weeks based on measurements of individual plants. Mean ± SD (n = 5). Letters indicate significant differences (P < 0.05) between the growth conditions at a given shade level according to one-way ANOVA analysis and post-hoc Tukey tests. Ed, E. densa plants in monoculture; Ed-Mix, E. densa plants in mixed community, Ec, E. canadensis plants in monoculture; Ec-Mix, E. canadensis plants in mixed community; DW, dry weight; RGR, relative growth rate, A Dry weight per plant, B Relative growth rate, C Root/shoot ratio, D Main stem length, E Lateral spread, F Branching degree
Discussion
Shading and interspecific competition interacted to affect growth responses of the two stream macrophytes, E. canadensis and E. densa. Shade had a strong influence on biomass accrual, growth rates and morphology of both E. canadensis and E. densa as observed in several previous studies of these, and other stream macrophyte species (Kankanamge et al. 2019; Zefferman 2014; Riis et al. 2012; Barko and Smart 1981). The effects of interspecific competition were more subtle than those of shade especially for E. densa. Furthermore, interactive effects between shade and community composition were significant for E. canadensis but not for E. densa. Our results imply that the competitiveness of E. canadensis with E. densa is increased at low shade (35%) as E. canadensis showed longer main stems, higher lateral spread and branching degree than E. densa. E. canadensis also had demonstrably longer stems compared to E. densa with shading of 63% and 79% supporting its potential competitive nature under these conditions. However, its propensity for lower biomass accrual across all shade levels tested likely means that E. densa will still dominate in mixed communities. Field observations from New Zealand streams and rivers tend to support this expectation. E. canadensis usually occurs at low abundance when E. densa is present (e.g., Champion and Tanner 2000), with E. canadensis only tending to proliferate in the absence of E. densa, or in streams with high rates of flushing flows (F. Matheson, P. Champion Pers. Observs.).
This study supports the concept that competition and facilitation interactions between species may vary along environmental stress gradients (Maestre et al. 2009). Along the shade gradient provided in this study, facilitation received by E. canadensis from interactions with E. densa at low shade (high light availability) shifted towards a neutral interaction between these two species at high shade (low light availability). Meanwhile, E. densa experienced a neutral interaction with E. canadensis at low shade (high light availability), which shifted towards a competitive interaction at high shade (low light availability). Similar variations in species interactions along environmental gradients have been observed in other studies (Li et al. 2018; Hao et al. 2013; Milne et al. 2007), and there are numerous examples which provide evidence that such interactions play a determinant role in dominance of species in mixed communities (Thiébaut et al. 2019; Hao et al. 2013, Maestre et al. 2009 and references there in).
The results of this study suggest that the presence of E. densa facilitates E. canadensis colonization when light availability is high and a similar biomass of propagules is available. Such interactions may have potential to alter the community composition in streams. Most previous studies have examined interactions between a native species and an invasive species, while this study has documented interactions between two exotic, invasive species with similar morphology. This may explain the relatively subtle interactive effects that we have observed especially for E. densa. Further, the results might differ when the propagule density varies (Thiébaut et al. 2019; You et al. 2016). Here, the design enables us only to evaluate the influence by the presence of the other, not to study the influence of propagule density, since it is a simple replacement experiment as used in other studies (Borgnis and Boyer 2016 as an example) for evaluating the difference between monoculture and mixed culture. Nevertheless, in this study our results do indicate that even at twice the propagule number (density) E. canadensis cannot compete with E. densa in terms of biomass accrual at shade levels ranging from 35 to 95%. Potentially, E. canadensis might be more competitive than E. densa in some stream systems, particularly those that have regular flushing flow events, which frequently reset the plant community, and if the relative supply of E. canadensis to E. densa propagules is higher, i.e., ratio > 2.
In conclusion, our study has shown that shade has a strong influence on the growth and competitive traits of two common stream macrophyte species with similar morphology, E. densa and E. canadensis. When grown in mixed culture with high light availability, growth of E. canadensis plants was facilitated by the presence of E. densa. At higher shade levels, the interaction shifted to neutral then competitive, with E. densa affected by E. canadensis presence at high shade yet still clearly dominating under these conditions. Biomass accrual was greater for E. densa plants at all shade levels, suggesting that this species is likely to be the superior competitor in stream and river systems.
References
Alnoe AB, Riis T, Andersen M, Baattrup-Pedersen A, Sand Jensen K (2015) Whole stream metabolism in nutrient poor calcareous streams on Oland. Sweden Aquat Sci 77(2):207–219
Barko JW, Smart RM (1981) Comparative influences of light and temperature on the growth and metabolism of selected submersed freshwater macrophytes. Ecol Monogr 51(2):219–235
Bell N, Riis T, Suren AM, Baattrup-Pedersen A (2013) Distribution of invertebrates within beds of two morphological contrasting stream macrophyte species. Fund Appl Limnol 183:309–321
Borgnis E, Boyer KE (2016) Salinity tolerance and competition drive distributions of native and invasive submerged aquatic vegetation in the upper San Francisco estuary. Estuar Coast 39:707–717
Champion PD, Tanner CC (2000) Seasonality of macrophytes and interaction with flow in a New Zealand lowland stream. Hydrobiologia 441(1):1–12
Champion PD, de Winton MD, Clayton J (2014) A risk assessment based proactive management strategy for aquatic weeds in New Zealand. Manag Biol Invasions 5(3):233–240
Champion P, Rowe D, Smith B, Wells R, Kilroy C, de Winton M (2013) Freshwater pests of New Zealand. https://www.niwa.co.nz/sites/niwa.co.nz/files/sites/default/files/pest_guide_lndscp_feb_2013_pdf. Accessed 4 December 2018
Connolly J, Wayne P, Bazzaz FA (2001) Interspecific competition in plants: how well do current methods answer fundamental questions? Am Nat 157:107–125
Davidson AM, Jennions M, Nicotra AB (2011) Do invasive species show higher phenotypic plasticity than native species and if so, is it adaptive? A meta-analysis. Ecol Lett 14:419–431
Eller F, Alnoe AB, Boderskov T, Guo W, Kamp AT, Sorrell BK, Brix H (2015) Invasive submerged freshwater macrophytes are more plastic in their response to light intensity than to the availability of free CO2 in air-equilibrated water. Freshw Biol 60:929–943
Evangelista HB, Michelan TS, Gomes LC, Thomaz SM (2017) Shade provided by riparian plants and biotic resistance by macrophytes reduce the establishment of an invasive Poaceae. J Appl Ecol 54:648–656
Hao B, Wu H, Shi Q, Liu G, Xing W (2013) Facilitation and competition among foundation species of submerged macrophytes threatened by severe eutrophication and implications for restoration. Ecol Eng 60:76–80
Harper JL (1977) Population biology of plants. Academic Press, New York
Hoffmann WA, Poorter H (2002) Avoiding bias in calculation of relative growth rate. Ann Bot (Lond) 80:32–47
Hofstra DE, Clayton J, Green JD, Auger M (1999) Competitive performance of Hydrilla verticillata in New Zealand. Aquat Bot 63:305–324
Kankanamge CE, Matheson FE, Riis T (2019) Shading constrains the growth of invasive submerged macrophytes in streams. Aquat Bot 158:103125
Lacoul P, Freedman B (2006) Environmental influences on aquatic plants in freshwater ecosystems. Environ Rev 14:89–136
Levi PS, Riis T, Alnoe AB, Peipoch P, Maetzke K, Bruus C, Baattrup- Pedersen A (2015) Macrophyte complexity controls nutrient uptake in lowland streams. Ecosystems 18(5):914–931
Li F, Yang N, Zhu L, Xie Y, Yang G, Hu C, Chen X, Deng Z (2018) Competition and facilitation of two wetland macrophytes under different water levels and nutrient-heterogeneous conditions. Freshw Sci 37(2):296–306
Maestre FT, Callaway RM, Valladares F, Lortie CJ (2009) Refining the stress-gradient hypothesis for competition and facilitation in plant communities. J Ecol 97:199–205
Martin GD, Coetzee JA (2014) Competition between two aquatic macrophytes, Lagarosiphon major (Ridley) Moss (Hydrocharitaceae) and Myriophyllum spicatum Linnaeus (Haloragaceae) as influenced by substrate sediment and nutrients. Aquat Bot 114:1–11
Mony C, Koschnick TJ, Haller WT, Muller S (2007) Competition between two invasive Hydrocharitaceae (Hydrilla verticillata (L.f.) (Royle) and Egeria densa (Planch)) as influenced by sediment fertility and season. Aquat Bot 86:236–242
Milne JM, Lang P, Murphy KJ (2007) Competitive interactions between Salvinia auriculata Aubl., Limnobium laevigatum (Humb. and Bonpl. ex Willd.) Heine, and other free-floating aquatic macrophytes under varying nutrient availability. Fund Appl Limnol 169:169–176
Redekop P, Hofstra D, Hussner A (2016) Elodea canadensis shows a higher dispersal capacity via fragmentation than Egeria densa and Lagarosiphon major. Aquat Bot 130:45–49
Riis T (2008) Dispersal and colonisation of plants in lowland streams: success rates and bottlenecks. Hydrobiologia 596:341–351
Riis T, Sand-Jensen K (2006) Dispersal of plant fragments in small streams. Freshw Biol 51:274–286
Riis T, Olesen B, Clayton JS, Lambertini C, Brix H, Sorrell BK (2012) Growth and morphology in relation to temperature and light availability during the establishment of three invasive aquatic plant species. Aquat Bot 102:56–64
Sand-Jensen K (2002) Drag and reconfiguration of freshwater macrophytes. Freshw Biol 48:271–283
Thiébaut G, Rodriguez-Perez H, Jambon O (2019) Reciprocal interactions between the native Mentha aquatica and the invasive Ludwigia hexapetala in an outdoor experiment. Aquat Bot 157:17–23
Vila M, Weiner J (2004) Are invasive plant species better competitors than native plant species? Evidence from pair-wise experiments. Oikos 105:229–238
You WH, Han CM, Fang LX, Du DL (2016) Propagule pressure, habitat conditions and clonal integration influence the establishment and growth of an invasive clonal plant Alternanthera philoxeroides. Front Plant Sci 7:568
Zefferman E (2014) Increasing canopy shading reduces growth but not establishment of Elodea nuttallii and Myriophyllum spicatum in stream channels. Hydrobiologia 734:159–170
Acknowledgements
Funding for this study was provided by the New Zealand Ministry for Business, Innovation and Employment to the National Institute of Water and Atmospheric Research (Strategic Science Investment Project: SMARTer Riparian & Wetland Strategies). The authors thank M. Crump and D. Rendle for technical support. We also thank Champion P for his valuable comments on anearlier version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Télesphore Sime-Ngando
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ellawala Kankanamge, C., Matheson, F.E. & Riis, T. Shading may alter the colonization pattern and dominance between two invasive submerged aquatic plant species. Aquat Ecol 54, 721–728 (2020). https://doi.org/10.1007/s10452-020-09770-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-020-09770-0