Abstract
This study determined whether the acute cartilage response, assessed by cartilage thickness and echo intensity, differs between patients with early-mild knee osteoarthritis (OA) and healthy controls. We recruited 56 women aged ≥ 50 years with Kellgren–Lawrence (KL) grade ≤ 2 (age, 70.6 ± 7.4 years; height, 153.7 ± 5.2 cm; weight, 51.9 ± 8.2 kg). Based on KL grades and knee symptoms, the participants were classified into control (KL ≤ 1, asymptomatic, n = 27) and early-mild knee OA groups (KL 1 and symptomatic, KL 2, n = 29). Medial femoral cartilage thickness and echo intensity were assessed using ultrasonographic B-mode images before and after treadmill walking (15 min, 3.3 km/h). To investigate the acute cartilage response, repeated-measures analysis of covariance (groups × time) with adjusted age, external knee moment impulse, steps during treadmill walking, and cartilage thickness at pre-walking was performed. A significant interaction was found at the tibiofemoral joint; after walking, the cartilage thickness was significantly decreased in the early-mild knee OA group compared to the control group (p = 0.002). At the patellofemoral joint, a significant main effect of time was observed, but no interaction was detected (p = 0.802). No changes in cartilage echo intensity at either the tibiofemoral or patellofemoral joints, and no interactions were noted (p = 0.295 and p = 0.063). As acute cartilage response after walking, the thickness of the medial tibiofemoral joint in the early-mild knee OA was significantly reduced than that in the control group. Thus, greater acute deformation after walking might be a feature found in patients with early-mild knee OA.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Articular cartilage undergoes immediate deformation in response to mechanical loading as a physiological response [16]. This deformation, attributed to fluid outflow from the cartilage [16], can be detected in vitro using the compression test for the cartilage sample [29]. It can be detected in vivo using magnetic resonance imaging (MRI) or ultrasound imaging devices as a decline in cartilage thickness. Previous studies identified cartilage deformation following weight-bearing exercises such as walking [8, 9], running [8], squatting [5], and jumping [10]. In addition, the extent of cartilage deformation is influenced by its composition. Increased acute deformation may result from proteoglycan loss and altered collagen orientation by cartilage degeneration, which reduces the joint’s ability to retain water content within the cartilage [2, 4]. A previous study reported greater cartilage deformation after running in patients with anterior cruciate ligament injury and cartilage degeneration versus healthy controls [7]. Therefore, acute cartilage deformation after mechanical loading has attracted considerable attention as an important indicator of cartilage quality reduction, which precedes cartilage thickness loss.
However, it is unknown whether acute cartilage deformation is associated with early cartilage degeneration in patients with knee osteoarthritis (OA), as observed in young adults, because the mechanical properties of the cartilage are probably altered by age-related change [1, 13]. Ho et al. [12] reported no significant differences in cartilage deformation after treadmill walking between patients with knee OA and healthy older controls [12]. However, they did not adequately clarify the features of cartilage deformation in patients with knee OA because of the limited sample size (n = 9) and failure to distinguish between medial and lateral OA. Acute cartilage deformation is influenced not only by the degree of cartilage degeneration, but also by the amount of mechanical loading. Thus, in a sufficiently large number of patients with knee OA, it is necessary to investigate acute cartilage deformation caused by degeneration, taking into consideration the effect of mechanical loading. Moreover, to our knowledge, no studies to date have used ultrasound devices to investigate cartilage deformation in patients with knee OA. The previous review suggested the possibility that ultrasound imaging offers advantages for assessing acute cartilage deformation due to its immediate measurability and greater spatial resolution compared with MRI [18]. Therefore, we might be able to discern the features of cartilage deformation in patients with knee OA using ultrasound devices.
Cartilage echo intensity has recently attracted attention as an ultrasound biomarker of early cartilage degeneration prior to thinning in knee OA [19, 24]. Although the physiological mechanism of enhanced cartilage echo intensity remains unclear, we believe that the decline in cartilage quality, such as a change in the collagen fiber orientation, and an increase in water content are impacted. However, few studies have investigated the acute changes in cartilage echo intensity after mechanical loading. Harkey et al. [10] reported no significant changes in cartilage echo intensity after treadmill walking [10]. However, how cartilage echo intensity is altered after mechanical loading in patients with knee OA remains unclear. This is because Harkey et al.’s study only included healthy young adults. As the cartilage of patients with early-mild knee OA presents with enhanced echo intensity [19], the characteristics of early cartilage degeneration in patients with knee OA are also possibly indicated by the difference in echo intensity after mechanical loading compared with healthy controls.
This study aimed to determine whether the acute cartilage response, assessed by cartilage thickness and echo intensity, differs between patients with early-mild knee OA and healthy controls. We hypothesized that (1) cartilage thickness after mechanical loading is significantly thinner in patients with early-mild knee OA than in healthy controls and (2) cartilage echo intensity after mechanical loading is significantly more enhanced in patients with early-mild knee OA than in healthy controls.
Materials and Methods
Participants
We recruited 56 women aged ≥ 50 years (age, 70.6 ± 7.4 years; height, 153.7 ± 5.2 cm; weight, 51.9 ± 8.2 kg). The participants’ inclusion criteria were as follows: (1) ability to live independently and walk continuously without a cane for more than 15 min and (2) OA diagnosed as Kellgren–Lawrence (KL) grade ≤ 2 for both sides of the knee. The exclusion criteria were as follows: (1) surgical history of the lower limbs, (2) lateral knee OA, (3) rheumatoid disease, (4) cardiovascular and neurological diseases, and (5) knee flexion range of < 130°. In patients with knee OA, all variables were measured in the knee with the more severe side of knee OA. The sample size required for repeated-measures analysis of covariance (RM-ANCOVA) to compare the acute cartilage response between patients with knee OA and healthy controls (α = 0.05, power = 0.80, effect size = 0.25) was calculated using G*Power software (version 3.1; Heinrich Heine University, Düsseldorf, Germany). Accordingly, a minimum of 34 total participants (n = 17 for each group) was determined.
Before the experiments, all participants were informed of the aim and procedure of the study and provided written informed consent. This study was approved by the Ethics Committee of the Kyoto University Graduate School of Medicine and complied with the Declaration of Helsinki (R3014).
Study Design
The measurement procedure is shown in Fig. 1. To minimize the influence of physical activities before the tests, all participants were instructed to avoid engaging in high-intensity exercises before arriving at the laboratory. A radiographic image of the participants, which is an anteroposterior weight-bearing view, was obtained, and the severity of knee OA was determined using the KL grade. In addition, the participants completed a questionnaire regarding knee symptoms, and their gait was assessed using a motion capture system. Subsequently, to suppress the effect of activities of daily living on the pre-walking cartilage assessment, the participants rested on the bed in the supine position for at least 30 min to unload the knee cartilage [8]. To assess the acute cartilage response after loading, the medial femoral cartilage was captured using ultrasonographic B-mode images before and after 15-min treadmill walking.
Assessment of Knee OA Symptoms and Classification of the Groups
The symptom section of the Knee Society Score 2011 Japanese edition (KSS symptom score) was used to assess knee symptoms [27]. The scores range from 25 to 0, with lower scores indicating more severe pain or stiffness. Based on a previous study, we defined a score of 23 or more as asymptomatic [26].
This study defined early-mild knee OA as follows: (1) KL grade ≤ 2 and (2) presence of knee symptoms with patient-reported outcomes [17]. Subjects with KL grade 0 disease who were symptomatic were excluded since their symptoms may have been caused by factors other than knee OA. Based on KL grades and KSS symptom scores, participants were classified into control (KL grade ≤ 1 and asymptomatic) and early-mild knee OA groups (KL grade = 1 and symptomatic or KL grade = 2).
Ultrasound Imaging of Femoral Cartilage
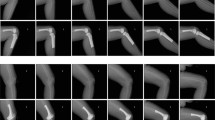
All B-mode images of the femoral cartilage were acquired by one examiner (Sh O) before and after walking using an ultrasound imaging device with a linear probe SL15-4 (Aixplorer, Knee-mode, SuperSonic Imagine, Axi-en-Provence, France). All images were acquired using the following settings: knee mode; depth, 2 cm; focus, 1 cm; gain, 48%; frequency, 44 MHz; and dynamic range, 62 dB. The examiner acquired longitudinal and transverse B-mode images of the femoral cartilage with 130° of knee flexion at each time point (Fig. 2). In a previous study, longitudinal images were used to evaluate the tibiofemoral weight-bearing surface [20] and transverse images above the patella were used to evaluate the femoral trochlea within the patellofemoral joint [11]. We defined the measurement location as follows: longitudinal image, at the midpoint between the medial edge of the patella and the medial epicondyle [25], and transverse image, in line with the medial and lateral femoral condyles above the superior edge of the patella when measuring transverse images above the patella [8]. When acquiring images, the examiner adjusted the tilt angle of the probe to describe the two white bands (synovia–cartilage and cartilage–bone interfaces) on the images. For all participants, the anterior area of cartilage was portrayed on the left side of the longitudinal image, while the medial area of cartilage was portrayed on the left side of the transverse image.
Positioning of ultrasound measurements. Participants were positioned supine on the treatment bed at 130° of knee flexion (a). All ultrasonographic measurement were performed in this position. The ultrasound probe was positioned as follows (b): longitudinal image, at the midpoint between the medial edge of the patella and the medial epicondyle [25]; transverse image, in line with the medial and lateral femoral condyles above the superior edge of the patella [8]
Ultrasonographic Image Analysis
Image J software (https://imagej.nih.gov/) was used to analyze the ultrasonographic images (Fig. 3). An examiner measured cartilage thickness and echo intensity. Regarding the transverse images acquired above the patella, we manually segmented the cartilage based on a previous study [11] and analyzed only the medial area.
Ultrasound image of femoral cartilage. a longitudinal image; b transverse image. ① is the white band of the synovium–cartilage interface; and ② is the white band of the cartilage–bone interface. The region surrounded by the yellow line indicates the area analyzed in this study. The transverse images were manually segmented. We defined the intercondylar compartment, which is the middle 25% of the ultrasonographic image centering on the deepest part of the intercondylar notch and decided the medial femoral compartment as the left side of the intercondylar compartment. We analyzed only the medial compartment in this study
The cartilage thickness was measured as the distance between the synovia–cartilage and cartilage–bone interfaces at three points in each image [25]. We calculated the average thickness from three images at each time point. To assess the cartilage echo intensity, the region of interest (ROI) was manually set as large as possible in the cartilage band, excluding the cartilage–bone interface, soft tissue–cartilage interface, and blurry area in the cartilage band. The signal intensity was then expressed as a 256-point value ranging from 0 (black) to 255 (white) in the ROI, and the mean signal intensity was calculated as the echo intensity [21]. At each time point, the average value of the three images was used as the echo intensity. The values of each indicator were calculated from the longitudinal and transverse images, and the amount of acute cartilage response after treadmill walking assessed by cartilage thickness and echo intensity–delta (Δ) values–were calculated by subtracting the pre-walking value from the post-walking value.
Gait Analysis
Gait analysis was conducted using a three-dimensional motion capture system (Vicon Nexus; Vicon Motion Systems, Ltd., Oxford, England), consisting of eight cameras sampled at 200 Hz and two force plates (Kistler Japan Co., Ltd., Tokyo, Japan) sampled at 1000 Hz. To capture the gait motion at the same speed as treadmill walking, participants were instructed to walk along a straight walkway at 3.3 km/h (0.91 m/s) and practiced a few times to adapt the designated walking speed before measurements. The marker data were processed using a fourth-order low-pass Butterworth filter at 6 Hz, and the ground reaction force data were processed using a fourth-order low-pass Butterworth filter at 20 Hz. Markers were placed based on the Plug-in-Gait full-body model marker placement protocol. The kinetic and kinematic variables were calculated from the ground reaction force and marker location data. The time integrals of the positive values of the knee adduction moment (KAM-impulse) and knee flexion moment (KFM-impulse) during the stance phase of the gait were computed. The average values of the three trials in which the participants could walk at a designated speed were used for further analysis.
Treadmill Walking Protocol
After the pre-walking assessment of the femoral cartilage, the participants were positioned barefoot on a treadmill (MyRun, Technogym, Cesena, Italy) besides the bed. All participants wore a pedometer with a three-axis accelerometer (EX-500; YAMASA, Tokyo, Japan) to measure their steps during treadmill walking. While walking, the participants were instructed to lightly hold the treadmill handrails for their own safety. Following the 2-minutes practice to adapt walking on the treadmill, the participants walked at 3.3 km/h (0.91 m/s) for 15 minutes. The gait speed was set slower than the general gait speed of patients with knee OA (1.1 m/s) [30]. This is because prior research reported that the gait speed on the treadmill is 17.2% faster than that on the ground [3]. The physical therapist monitored the participants during each session. If a participant’s physical condition or knee symptoms worsened, the measurement was immediately stopped. After the walking session, the participants returned to bed as soon as possible to undergo their femoral cartilage assessment using an ultrasound imaging device; all post-walking measurements were completed within 5 min. The time required to perform post-exercise ultrasonographic imaging was sufficiently minimized, compared with the recovery time required to return to the same thickness as that before loading [6].
Statistical Analysis
To clarify whether changes in cartilage thickness or echo intensity exceeded the measurement error, we calculated the minimal detectable change (MDC) using the standard error of measurement (SEM). Based on formulas (1) and (2), we calculated the SEMs of the cartilage thickness and echo intensity using B-mode images for all participants captured at the pre-walking phase. To calculate the SEM, SD(d) was calculated using the measured values from the first and second images of the three consecutive images acquired at the pre-walking phase, uniformly obtained across all participants. SD(d) is defined as the standard deviation of the difference in measured values between two images and represents the variability of measurements. The MDC, which is the value beyond the error of ultrasonographic measurement in this study, was calculated using SEM (equation [2]).
Unpaired t-tests were used to confirm differences in pre-walking participant characteristics between the groups. In addition, paired t-tests were performed to confirm whether acute cartilage response was found after 15 min of walking in each group. Finally, a 2 × 2 (groups × repeated measures factor) RM-ANCOVA was conducted to investigate whether acute cartilage responses differed between control and early-mild OA groups, while statistically controlling for potential confounding variables among each participant as covariates. The analysis was performed with cartilage values (thickness or echo intensity) as dependent variables; pre- and post-walking phases as repeated measures factors; and age, external knee moment impulse, steps during treadmill walking, and cartilage values at the pre-walking phase as covariates. The interaction of this analysis indicates whether the acute cartilage response in the early mild OA group differed from that of the control group. In this study, the interaction indicates the difference in cartilage values at the post-walking phase between the groups because the cartilage values at the pre-walking phase were adjusted through RM-ANCOVA. For the external knee moment, the KAM-impulse was used to adjust the load at the medial tibiofemoral joint, while the KFM-impulse was used to adjust the load at the patellofemoral joint. These indicators reportedly reflected the mechanical loading of the knee joint in previous studies [28, 30]. All statistical analyses were performed using IBM SPSS Statistics version 22 (IBM Corp., Armonk, NY, USA), and the statistical significance level was set at 5%.
Results
Participant Characteristics
The characteristics of each group are presented in Table 1. The weight in the early-mild OA group was significantly higher than that in the control group (p = 0.004). The KSS symptom score in the early-mild OA group was significantly lower than that in the control group (p < 0.001). No significant differences were found in the age, height, external knee moment impulse, and steps during treadmill walking between the two groups.
Repeatability and MDC of Ultrasonographic Cartilage Measurement
The reliability of the measurement was verified in 10 participants randomly selected from this study (age, 68.2 ± 9.3 years; height, 154.1 ± 4.3 cm; weight, 50.0 ± 5.8 kg; and KL grade 0, n = 3; grade 1, n = 4; and grade 2, n = 3). Three B-mode images of the femoral cartilage were acquired using the same methods as those in the experiment before walking. During each measurement, the probe was removed from the knee and readjusted. Intraclass correlation coefficients (ICC [1,3]) were calculated for cartilage thickness and echo intensity on the longitudinal and transverse B mode images [14]. The ICCs (1,3) of cartilage thickness were 0.99 and 0.97 on the longitudinal and transverse images, respectively, while those of cartilage echo intensity were 0.99 on the longitudinal and transverse images.
The MDC of the ultrasonographic cartilage measurements was calculated using the values obtained before loading. The MDCs of cartilage thickness were 0.051 and 0.059 mm on the longitudinal and transverse images, respectively, and those of cartilage echo intensity were 4.83 and 3.57 a.u. on the longitudinal and transverse images, respectively (Table 2).
Comparison of Acute Cartilage Response After Loading
Before loading, no significant difference was found in the cartilage thickness between the groups (longitudinal images, p = 0.619; transverse images, p = 0.100). The cartilage echo intensity in the early-mild OA group was more enhanced than that in the control group on the longitudinal images (p = 0.038); however, no significant difference was found between the groups on the transverse images (p = 0.118).
The paired t-test indicated that cartilage thickness was significantly decreased after walking in both groups based on the longitudinal images (control: p < 0.001, early-mild OA: p < 0.001) and transverse images (control: p = 0.002, early-mild OA: p = 0.003). However, no significant changes were identified regarding echo intensity, except in the early-mild OA group based on the transverse images (longitudinal, control: p = 0.711, early-mild OA: p = 0.194; transverse, control: p = 0.441, early-mild OA: p = 0.002). The results of the RM-ANCOVA comparing the acute cartilage response after loading between the two groups are shown in Table 2. With regard to cartilage thickness, a significant interaction was found on the longitudinal images (p = 0.002), and the cartilage thickness in the early-mild OA group after walking was significantly decreased compared with that in the control group. On the transverse images, a significant main effect of time (p < 0.001) was observed, but no interaction was noted (p = 0.802). Regarding the cartilage echo intensity, no significant main effects or interactions were found on the longitudinal images (p = 0.273 and p = 0.295, respectively). Transverse imaging revealed a significant main effect of time (p = 0.007), and the cartilage echo intensity after walking was enhanced compared to that before walking in both groups. However, no significant interaction was observed (p = 0.063). In addition, a change after walking greater than the MDC was found only in cartilage thickness on the longitudinal images.
Discussion
The most important findings of the present study were that at the tibiofemoral weight-bearing surface assessed using longitudinal images, femoral cartilage deformation after walking was significantly greater in patients with early-mild knee OA than in healthy controls. However, at the patellofemoral joint above the patella, as assessed using transverse images, no significant differences in acute cartilage deformation were observed between the groups. Additionally, no changes in the cartilage echo intensity above the MDC were detected after walking, either at the tibiofemoral weight-bearing surface or at the patellofemoral joint above the patella.
The cartilage thickness at the tibiofemoral weight-bearing surface was significantly decreased after walking in the early-mild knee OA group compared to the control group, as assessed on the longitudinal images, supporting our hypothesis. Our results align with those of a previous study that found greater acute deformation after loading in young participants with cartilage damage than in healthy controls [7]. Healthy cartilage generally possesses abundant proteoglycans, which have a high affinity for water, allowing it to withstand mechanical stress [1]. However, OA cartilage tends to have increased permeability to water because of proteoglycan loss caused by cartilage degeneration [2]; this qualitative change occurs in the early-mild stage of knee OA before cartilage volume loss occurs [22]. This difference in cartilage condition may be associated with cartilage deformation after mechanical loading, as acute cartilage deformation depends on fluid dynamics. In contrast to our study, Ho et al. [12] reported no significant differences in cartilage deformation after treadmill walking between patients with knee OA and healthy controls [12]. The discrepancy between the findings of our study and the previous study [12] may be attributed to differences in participant characteristics or sample size. Ho et al. included only nine patients with medial and/or lateral knee OA, whereas we included 29 patients with medial knee OA only. In addition, our results emphasize that greater acute deformation characterizes the cartilage of patients with knee OA, as indicated by the statistical adjustment for mechanical loading, such as knee moment and number of steps. Therefore, our results suggest that greater acute deformation after walking might be a feature found in patients with early-mild knee OA. It is generally difficult to detect differences in cartilage condition, as cartilage volume is not decreased significantly in the early-mild stage of knee OA [22]. However, our finding, indicating the mechanical loading ability has already decreased in patients with early-mild knee OA and could be detected by ultrasonographic assessment, is novel and may be valuable in the clinical setting.
At the patellofemoral joint, as assessed by transverse images [11], no significant difference was noted between the two groups. A previous simulation study suggested that the contact force in the patellofemoral joint during gait is smaller than that in the tibiofemoral joint [23]. Considering that the amount of acute cartilage deformation after walking in both groups was less than the MDC, the mechanical stress on the patellofemoral joint during the 15-min treadmill walking may not have been sufficient to deform the cartilage. In addition, the mechanical properties of cartilage based on their modulus vary widely among joints and anatomical regions [15, 29], which may have accounted for the different results on the longitudinal images.
No changes in the cartilage echo intensity were detected after walking in either group, contradicting our hypothesis. A previous study reported that cartilage echo intensity remains unchanged after treadmill walking in healthy young participants [10]. Considering this previous study and our results, cartilage echo intensity may not be suitable for assessing the acute cartilage response to mechanical loading, which relies on fluid dynamics. However, the mechanism underlying the altered cartilage echo intensity remains unknown. To accurately explain the acute response of echo intensity after loading, further studies are warranted to confirm echo intensity alterations under various conditions, such as higher loading intensities or in participants with different characteristics.
This study had several limitations. First, the external knee moment impulse, which was used for adjustment in the statistical analysis, was not measured during treadmill walking but during walking on the ground at a speed similar to treadmill walking. Therefore, we could not strictly adjust mechanical loading on the knee joint during the task. However, considering that few studies have taken into account the effect of mechanical loading when comparing acute cartilage responses, we can contend that acute deformation is not solely attributable to differences in the amount of mechanical loading, at least when estimated using commonly used indices. Second, we could not clarify whether the difference in acute cartilage response was caused by cartilage degeneration because femoral cartilage was not assessed by MRI or histology. However, it was apparent that the cartilage deformation of the medial tibiofemoral joint underwent significant changes in patients with early-mild knee OA than in healthy controls. Finally, the participants were middle-aged and older women with medial knee OA. Therefore, additional research is needed to determine whether the results of this study can be generalized to men or patients with lateral knee OA.
In conclusion, during the acute cartilage response after walking, the thickness of the medial tibiofemoral joint was more significantly reduced in the early-mild knee OA group than in the control group, but that cartilage echo intensity may not be altered following mechanical loading. These findings suggest that greater acute deformation after walking might be a feature found in patients with early-mild knee OA.
Data Availability
Data are not available due to ethical restrictions.
References
Bank, R. A., M. T. Bayliss, F. P. J. G. Lafeber, A. Maroudas, and J. M. Tekoppele. Ageing and zonal variation in post-translational modification of collagen in normal human articular cartilage: the age-related increase in non-enzymatic glycation affects biomechanical properties of cartilage. Biochemical Journal. 330:345–351, 1998.
Choi, J. A., and G. E. Gold. MR imaging of articular cartilage physiology. Magn Reson Imaging Clin N Am. 19:249–282, 2011.
Dal, U., T. Erdogan, B. Resitoglu, and H. Beydagi. Determination of preferred walking speed on treadmill may lead to high oxygen cost on treadmill walking. Gait Posture. 31:366–369, 2010.
Eckstein, F., M. Hudelmaier, and R. Putz. The effects of exercise on human articular cartilage. J Anat. 208:491–512, 2006.
Eckstein, F., B. Lemberger, C. Gratzke, M. Hudelmaier, C. Glaser, K.-H. Englmeier, and M. Reiser. In vivo cartilage deformation after different types of activity and its dependence on physical training status. Ann Rheum Dis. 64:291–295, 2005.
Eckstein, F., M. Tieschky, S. Faber, K. H. Englmeier, and M. Reiser. Functional analysis of articular cartilage deformation, recovery, and fluid flow following dynamic exercise in vivo. Anat Embryol (Berl). 200:419–424, 1999.
Van Ginckel, A., P. Verdonk, J. Victor, and E. Witvrouw. Cartilage status in relation to return to sports after anterior cruciate ligament reconstruction. American Journal of Sports Medicine. 41:550–559, 2013.
Harkey, M. S., J. T. Blackburn, H. Davis, L. Sierra-Arévalo, D. Nissman, and B. Pietrosimone. Ultrasonographic assessment of medial femoral cartilage deformation acutely following walking and running. Osteoarthritis Cartilage. 25:907–913, 2017.
Harkey, M. S., J. T. Blackburn, H. Davis, L. Sierra-Arévalo, D. Nissman, and B. Pietrosimone. The association between habitual walking speed and medial femoral cartilage deformation following 30 min of walking. Gait Posture. 59:128–133, 2018.
Harkey, M. S., J. T. Blackburn, A. C. Hackney, M. D. Lewek, R. J. Schmitz, D. Nissman, and B. Pietrosimone. Comprehensively assessing the acute femoral cartilage response and recovery after walking and drop-landing: an ultrasonographic study. Ultrasound Med Biol. 44:311–320, 2018.
Harkey, M. S., E. Little, M. Thompson, M. Zhang, J. B. Driban, and M. J. Salzler. Femoral cartilage ultrasound echo intensity associates with arthroscopic cartilage damage. Ultrasound Med Biol. 47:43–50, 2021.
Ho, K. Y., J. McClaren, and S. Sudweeks. Acute effects of walking on the deformation of femoral articular cartilage in older adults. Journal of Geriatric Physical Therapy. 42:E35–E41, 2019.
Hudelmaier, M., C. Glaser, J. Hohe, K. H. Englmeier, M. Reiser, R. Putz, and F. Eckstein. Age-related changes in the morphology and deformational behavior of knee joint cartilage. Arthritis Rheum. 44:2556–2561, 2001.
Koo, T. K., and M. Y. Li. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 15:155–163, 2016.
Li, H., J. Li, S. Yu, C. Wu, and W. Zhang. The mechanical properties of tibiofemoral and patellofemoral articular cartilage in compression depend on anatomical regions. Sci Rep. 2021. https://doi.org/10.1038/s41598-021-85716-2.
Liess, C., S. Lüsse, N. Karger, M. Heller, and C. G. Glüer. Detection of changes in cartilage water content using MRI T2-mapping in vivo. Osteoarthritis Cartilage. 10:907–913, 2002.
Luyten, F. P., S. Bierma-Zeinstra, F. Dell’Accio, V. B. Kraus, K. Nakata, I. Sekiya, N. K. Arden, and L. S. Lohmander. Toward classification criteria for early osteoarthritis of the knee. Semin Arthritis Rheum. 47:457–463, 2018.
Neu, C. P. Functional imaging in OA: role of imaging in the evaluation of tissue biomechanics. Osteoarthritis Cartilage. 22:1349–1359, 2014.
Okada, S., M. Taniguchi, M. Yagi, Y. Motomura, S. Okada, Y. Fukumoto, M. Kobayashi, K. Kanemitsu, and N. Ichihashi. Ultrasonographic echo intensity in the medial femoral cartilage is enhanced prior to cartilage thinning in women with early mild knee osteoarthritis. Knee Surg Sports Traumatol Arthrosc. 2023. https://doi.org/10.1007/s00167-023-07440-w.
Okano, T., E. Filippucci, M. Di Carlo, A. Draghessi, M. Carotti, F. Salaffi, G. Wright, and W. Grassi. Ultrasonographic evaluation of joint damage in knee osteoarthritis: feature-specific comparisons with conventional radiography. Rheumatology (United Kingdom). 55:2040–2049, 2016.
Pamukoff, D. N., M. N. Vakula, S. C. Holmes, E. J. Shumski, and S. A. Garcia. Body mass index moderates the association between gait kinetics, body composition, and femoral knee cartilage characteristics. Journal of Orthopaedic Research. 38:2685–2695, 2020.
Reichenbach, S., M. Yang, F. Eckstein, J. Niu, D. J. Hunter, C. E. McLennan, A. Guermazi, F. Roemer, M. Hudelmaier, P. Aliabadi, and D. T. Felson. Does cartilage volume or thickness distinguish knees with and without mild radiographic osteoarthritis? The Framingham Study. Ann Rheum Dis. 69:143–149, 2010.
Van Rossom, S., C. R. Smith, D. G. Thelen, B. Vanwanseele, D. Van Assche, and I. Jonkers. Knee joint loading in healthy adults during functional exercises: Implications for rehabilitation guidelines. Journal of Orthopaedic and Sports Physical Therapy. 48:162–173, 2018.
Saarakkala, S., P. Waris, V. Waris, I. Tarkiainen, E. Karvanen, J. Aarnio, and J. M. Koski. Diagnostic performance of knee ultrasonography for detecting degenerative changes of articular cartilage. Osteoarthritis Cartilage. 20:376–381, 2012.
Schmitz, R. J., H. M. Wang, D. R. Polprasert, R. A. Kraft, and B. G. Pietrosimone. Evaluation of knee cartilage thickness: A comparison between ultrasound and magnetic resonance imaging methods. Knee. 24:217–223, 2017.
Taniguchi, M., T. Ikezoe, T. Kamitani, T. Tsuboyama, H. Ito, S. Matsuda, Y. Tabara, F. Matsuda, N. Ichihashi, Nagahama Study. Extracellular-to-intracellular water ratios are associated with functional disability levels in patients with knee osteoarthritis: results from the Nagahama Study. Clin Rheumatol. 40:2889–2896, 2021.
Taniguchi, N., S. Matsuda, T. Kawaguchi, Y. Tabara, T. Ikezoe, T. Tsuboyama, N. Ichihashi, T. Nakayama, F. Matsuda, and H. Ito. The KSS 2011 reflects symptoms, physical activities, and radiographic grades in a Japanese population. Clin Orthop Relat Res. 473:70–75, 2015.
Teng, H. L., N. E. Calixto, T. D. MacLeod, L. Nardo, T. M. Link, S. Majumdar, and R. B. Souza. Associations between patellofemoral joint cartilage T1ρ and T2 and knee flexion moment and impulse during gait in individuals with and without patellofemoral joint osteoarthritis. Osteoarthritis Cartilage. 24:1554–1564, 2016.
Wong, B. L., and R. L. Sah. Mechanical asymmetry during articulation of tibial and femoral cartilages: Local and overall compressive and shear deformation and properties. J Biomech. 43:1689–1695, 2010.
Yamagata, M., M. Taniguchi, H. Tateuchi, M. Kobayashi, and N. Ichihashi. The effects of knee pain on knee contact force and external knee adduction moment in patients with knee osteoarthritis. J Biomech.123:110538, 2021.
Acknowledgements
All authors acknowledge all the participants in this study and Editage (www.editage.jp) for English language editing.
Funding
This study was supported by the Japan Society for the Promotion of Science (grant numbers: 18H03164 and 20K19376).
Author information
Authors and Affiliations
Contributions
All authors have made substantial contributions to (1) the conception and design of the study (2) revising it critically for important intellectual content, (3) final approval of the version to be submitted. The specific contributions of each author are as follows: 1. Analysis and interpretation of the data: ShO, MT, MY, and NI. 2. Drafting of the article: ShO, MT, MY, and NI.
Corresponding author
Ethics declarations
Competing Interests
All authors declare no competing interest.
Ethical Approval
This study was approved by the Ethics Committee of the Kyoto University Graduate School of Medicine and complied with the Declaration of Helsinki (R3014).
Informed Consent
Before the experiments, all participants were informed of the aim and procedure of the study and provided written informed consent.
Additional information
Associate Editor Eiji Tanaka oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
We recognize the bias in citation practices such that papers from women and other minority scholars are undercited relative to the number of papers and have worked to ensure that we are referencing appropriate papers with fair gender and racial author inclusion.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Okada, S., Taniguchi, M., Yagi, M. et al. Characteristics of Acute Cartilage Response After Mechanical Loading in Patients with Early-Mild Knee Osteoarthritis. Ann Biomed Eng 52, 1326–1334 (2024). https://doi.org/10.1007/s10439-024-03456-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-024-03456-6