Abstract
Understanding the use of space is essential to promote effective conservation actions, particularly to xenarthran species that are threatened by habitat loss, poaching, and other environment degradation. Information regarding the use of space by the giant anteater (Myrmecophaga tridactyla), a large and vulnerable Neotropical mammal, is sparse and sometimes inconsistent in the scientific literature. Our aims are (1) to present an updated overview of giant anteater use of space; (2) to identify knowledge gaps; and (3) to recommend the next steps to advance this knowledge. We compiled information from 53 publications regarding the key aspects of space use: movement patterns, home range size and overlap, and habitat use. We identified the following research priorities: (a) the need of studies on different populations throughout the species’ range, including continuous native forest areas, and different spatial scales; (b) to develop research on dispersal by the individuals; (c) to understand movement patterns based on trajectory-related methods; (d) to comprehend home range overlap and territoriality ecological meaning; and (e) habitat selection and movement patterns on human-modified landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
We are witnessing a global wave of habitat fragmentation and degradation, mostly driven by intensive human activities (Fahrig 2003; Allen and Singh 2016; Abrahms et al. 2017; McGowan et al. 2017). This scenario contributes to increasing the risk of species extinction (Butchart et al. 2010; Young et al. 2016). Among terrestrial vertebrates, more than 300 species have become extinct since 1500, and many of the remaining species are experiencing a decline in abundance (Dirzo et al. 2014) and are also losing suitable habitats (De Marco et al. 2018). For instance, 173 mammal species have already lost over 50% of their original range (Ceballos and Ehrlich 2002). For giant anteaters (Myrmecophaga tridactyla, Mammalia, Pilosa), the population loss is estimated at 30% in the past 26 years, mostly due to the decline in their range (Miranda et al. 2014; Miranda et al. 2015).
The giant anteaters’ original distribution was broad, ranging from Belize to the south of South America, excluding the Andes. However, the species is considered locally uncommon to rare, and the populations from the northern and extreme southern reaches of this distribution are considered extinct (Miranda et al. 2014). Most populations are facing one or more threats, including habitat loss, wildfires, poaching, conflicts with dogs, and roadkills (Miranda et al. 2015). These threats, coupled with life history traits such as a low reproductive rate and a long period of parental care (Rodrigues et al. 2008), increase their vulnerability.
The species is listed as Vulnerable in the International Union for Conservation of Nature (IUCN) Red List of Threatened Species (Miranda et al. 2014), and currently the giant anteater is the target species of research and conservation programs such as Instituto de Pesquisa e Conservação de Tamanduás do Brasil - Projeto Tamanduá (http://www.tamandua.org/), Brazil; Anteaters & Highways (www.tamanduabandeira.org), Brazil; Proyecto Iberá – Oso Hormiguero (http://www.proyectoibera.org), Argentina; and Proyecto Hormigueros (https://www.cunaguaro.co/proyecto-hormigueros), Colombia. However, there is still a shortage of information about the giant anteater in regard to space use patterns, such as home range size and the extent of home range overlap between individuals, habitat selection, and movement patterns in different localities (Rodrigues et al. 2008; Diniz and Brito 2012). These aspects of space use are fundamental for the definition of strategic conservation plans and actions, particularly to ensure their long-term conservation in the Anthropocene.
Giant anteaters have been recently studied in some localities within their area of distribution. Most of these data are available as gray literature or book chapters, but are sparse in peer-reviewed literature. Therefore, here we ask, what do we already know about the space use and movement patterns of this species? We aim with this review to provide an updated overview of what we know about the use of space by the giant anteater, point out new avenues to fill knowledge gaps, and recommend the next steps to advance this knowledge.
Methods
To collect the references related to space use patterns of the giant anteater, a search was conducted using all databases of Thomson Reuters™ Web of Science™ (available at http://apps.webofknowledge.com), Edentata, the annual publication of the IUCN/SSC Anteater, Sloth and Armadillo Specialist Group (http://xenarthrans.org/newsletter), complemented by a search in Google Scholar (www.scholar.google.com) and A.B. personal bibliographic list until 2019 September, which include theses and dissertations, books, and book chapters. We did not delineate a specific year so that the search would return all possible indexed articles. We used the scientific name (Myrmecophaga tridactyla) and the English, Portuguese, and Spanish common names (“giant anteater”, “tamanduá-bandeira”, “oso hormiguero”) respectively. Then we filtered the result of the search using the keywords: movement, home range, and habitat. We applied following combination of keywords in Google Scholar: “Myrmecophaga tridactyla AND giant anteater AND* OR oso hormiguero AND* OR tamanduá-bandeira AND XXXX”. XXXX was replaced by each one of the three keywords. Our dataset was organized, regarding the main contents of the literature compiled, into the following groups: (a) movement patterns, (b) home range size and overlap, (c) habitat use, and (d) the proximity to human-modified landscapes.
To provide an overview of the most recurrent words that appeared on our review, all titles of the literature on giant anteater were plotted in a word cloud (http://www.wordclouds.com). The program transforms the frequency of the words appearance in different sizes. To be more informative, we eliminated all the prepositions, scientific, and popular names.
For each study included in our dataset, where available, we noted the home range size estimation method used (Minimum Convex Polygon [MCP], Kernel, or Brownian Bridge [BB]) and separate home range size (km2) estimates for males and females, the distance individuals moved per day (m/day), and the speed of movement (km/h; Table 1). In addition, we provide a brief history of monitoring methods of giant anteaters (Online Resource 1).
Results
Our bibliographic search retrieved 192 references in Web of Science™ and 51 in Edentata. After the filtering process, we retained 19 and 10 references from these sources, respectively. Additionally, we included 24 references from Google Scholar and A.B. personal bibliographic list. Our review is based on 53 publications (Table 2).
The word cloud highlighted the most common words registered on the titles of the publications retrieved in our review (Fig. 1). “Brazil”, “Pantanal”, “Cerrado”, “Habitat”, “Mammal”, “Colombia”, “Area”, “Use”, “Selection” and “Conservation” were among the most frequent words in the titles.
One of the remarkable features of most of these references is that they provide data related to a few anteaters restricted to regional areas, and occasionally the studies were from the same study site (for example, Nhumirim Ranch in Brazilian Pantanal; Table 1).
Movement patterns
The average daily distance moved is around 3700 m/day. The shortest distance reported is 1326 m/day and the longest is 11,000 m/day at an average speed of less than 1 km/h (Table 1). No recent study has recorded the movement’s step-turned angles and their associations with habitat types.
Home range size and overlap
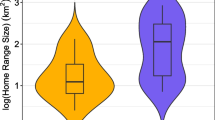
The first home range size estimate for the giant anteater was based on the species’ bio-ecological traits (body mass, terrestrial habit, feeding specialist, low densities) and predicted a size of 9 km2 (Silveira 1969). However, the first field study carried out estimated a home range size three times as large as this (Montgomery and Lubin 1977, Table 1). With the exception of a study that involved reintroduced individuals, subsequent studies in the wild have reported less variation in home range size information (Table 1).
Several studies have confirmed that the home ranges of giant anteaters overlap. The pair-to-pair analysis showed up to 90% overlap at a 1318 km2 reserve in central Brazil (Miranda 2004), a 100% overlap in a 25.9 km2 ranch in an urban/rural area of northern Brazil (Macedo et al. 2010), and 55% at a Brazilian Pantanal ranch of 104 km2 (Medri and Mourão 2005). Overlap can occur within and between sexes, as well as with different ages (Miranda 2004; Medri and Mourão 2005; Macedo et al. 2010), and females can be more tolerant than males with their same-sex neighbors (Shaw et al. 1987).
Habitat use
Habitat use patterns of the giant anteater depend on whether the giant anteater is active or inactive, on the temperature of the environment, and on seasonality (Medri and Mourão 2005; Camilo-Alves and Mourão 2006; Di Blanco et al. 2015; Di Blanco et al. 2016). The species uses vegetation-covered habitats during inactive periods and more open habitats when active (Camilo-Alves and Mourão 2006; Mourão and Medri 2007; Macedo et al. 2010; Di Blanco et al. 2016). It is known that vegetation-covered habitats (e.g., forest, shrublands, and timberlands) act as thermal refuges due to their more stable and milder core temperature (Mourão and Medri 2007), as was also suggested for other xenarthrans (Attias et al. 2018). Giant anteaters used shrub savanna to sleep in the Llanos of Venezuela (Montgomery and Lubin 1977; Montgomery 1985). The same pattern was also reported in central Brazil, even with grassland being the most abundant habitat type (Serra da Canastra National Park; Shaw et al. 1987).
In the Brazilian Pantanal wetland, giant anteater individuals used the available habitats in different proportions depending on whether they were active or sleeping (Medri and Mourão 2005), and forests were positively selected by individuals when inactive (Di Blanco et al. 2017). However, this behavior of preference on different types of habitats in dependency on the type of activity is highly associated with the temperature of the environment (Camilo-Alves and Mourão 2006; Attias et al. 2018). Giant anteaters monitored showed that in the hottest hours of the day, they prefer to be in sheltered habitats, such as forest patches, avoiding body’ overheating and reducing their exposure to the sun’s rays. Nonetheless, as the days became colder, the giant anteaters began and ended their activity progressively earlier and preferred resting in more open areas (Rodrigues et al. 2008).
Giant anteaters have also used anthropogenic areas such as agricultural fields and timber plantations of Pinus sp., Acacia sp., and Eucalyptus sp. (Miranda 2004; Braga 2010; Vynne et al. 2011; Kreutz et al. 2012; Timo et al. 2015). Nevertheless, the species tends to prefer natural habitat composition (Vynne et al. 2011), underutilizing the available plantations and timberlands (Braga 2010; Bertassoni et al. 2017). Other studies have reported that giant anteaters use or cross roads (Macedo et al. 2010; Vynne et al. 2011; Freitas et al. 2014; Versiani 2016; Ciocheti et al. 2017). The use of roads facilitates the individual’s movement due to its free of vegetation space, making ants and termite nests easy targets, especially at the edges (Freitas et al. 2014). A positive relationship between the occupancy of the giant anteater and the dirt roads was observed in a study site in the south-central region of Brazil (Versiani 2016).
In regard to seasonality, only one study conducted in Argentina with a reintroduced population lasted long enough to collect seasonality data. The results showed a shorter activity period in the winter (Di Blanco et al. 2016).
Discussion
The awareness and concern for the maintenance and survival of giant anteater populations were drivers fostering research reflected by words in the cloud such as “Protected”, “Conservation”, “Mortality”, and “Population”. “Brazil” was one of the most frequent words on the cloud showing that most of the research has been done in the country, mainly on “Cerrado” and “Pantanal” biomes (Fig. 1).
Movement patterns
A model developed by Montgomery and Lubin (1977) showed that the general motion pattern of giant anteaters can be characterized as a long series of short stops interspersed with relatively faster movement in a straight line. They also include in the model the tortuosity of the trajectory. The trajectory is a term used in the movement ecology discipline and it is characterized as the curve described by the animal when it moves, taking into account number of discrete “steps” connecting successive relocations of the animal (Calenge et al. 2009). To the species, the rate and angles of the trajectory seem to depend on preys’ characteristics, likely the distribution and abundance of ant and termite nests, foraging time, energy expended in each feeding event, etc. (Montgomery and Lubin 1977; Drumond 1992).
The Montgomery and Lubin model can be improved with research based on movement trajectory, which may better elucidate in detail the speed and step-turned angle of the movement. There is a hypothesis which elucidate that animals will show low movement speed and large turning angles to stay in favorable habitats and the conversely to avoid unprofitable habitats (Turchin 1991); and this idea has been widely tested (Barraquand and Benhamou 2008; Duffy et al. 2011; Da Silveira et al. 2016; Grotta-Neto et al. 2019). Therefore, an important avenue for new research will certainly be studies comparing the trajectory of giant anteaters between well-preserved and human-modified landscapes. Additionally, studies on giant anteater roadkills can use the speed of movement to understand why the species is often a victim of vehicle collisions (Ribeiro et al. 2017; Ascensão et al. 2017; Sistema Urubu - http://cbee.ufla.br/portal/sistema_urubu/urubu-info.php).
Regarding the daily distance traveled, the largest reported value was 11,000 m/day (Montgomery and Lubin 1977, Table 1), which indicates that biologically, the species is able to move long distances. However, the authors attempted to follow individuals on horseback and reported difficulty in tracking the individuals due to their rapid movement. This is unlikely for the species; thus, we assume that the giant anteaters were trying to escape from this approximation, as occurs when the distance is too close and noisy (A.B. comm. pers.). Thereby, we assume an overestimation on this distance traveled per day. The largest known distance traveled is therefore about half of the above value (6000 m/day; Shaw et al. 1987, Table 1). However, since these data about the movement of giant anteaters were obtained in short-term studies (few months or less), some questions arise.
As distance moved influences home range size, and home range and dispersal distance of mammals are related (Bowman et al. 2002; Whitmee and Orme 2013), we ask: Do giant anteaters exhibit natal dispersal—here defined as the movement that an individual makes to establish its own home range? If so, does this dispersal behavior occur equally for both males and females? To date, no study has addressed the dispersal capacity of the individuals and there is a need for studies focusing on the different aspects of it. This can be particularly important for a better understanding of the animal’s resilience to anthropogenic changes. Furthermore, if most giant anteater populations are restricted to small vegetation remnants (Diniz and Brito 2015; Bertassoni et al 2019), the following question emerges: Is it possible that a connected landscape for the species is on the 6000-m spatial scale? One promising avenue for obtaining those answers is real-time animal tracking technology applied in long-term studies (Jønsson et al. 2016).
In addition, based on the fact that a giant anteater cub remains with its mother for about a year (Jerez and Halloy 2003) and that home ranges can overlap, we ask: Do cubs disperse as soon as they gain their independence? Do female cubs remain in areas adjacent to their mothers’ areas? Addressing these questions is challenging since it requires capturing and setting up telemetry devices for both the mother and the cub throughout the cub’s independence, dispersion, and finally, the establishment of its own home range. Thus, studies using noninvasive molecular techniques may represent an alternative to explore those aspects (Smith et al. 2006; Miotto et al. 2012). Clozato et al. (2017) made a first step on this direction studying on the population structure and genetic diversity of giant anteaters.
Home range size
Most studies of home range provide size estimates obtained by the Minimum Convex Polygon (MCP, Mohr 1947) method. Other studies present data obtained from the Kernel estimator (Worton 1989), but the specific method used varied among them (i.e., 95% adaptive Kernel, 90% fixed Kernel, 95% fixed Kernel). However, as the use of GPS technology increases, the new trend is to use estimators that consider the trajectory instead of the MPC and/or Kernel (Benhamou and Riotte-Lambert 2012).
Home range sizes of the giant anteater vary greatly among studies. This variation results from several aspects, such as different monitoring protocols, which home range estimator was used, characteristics of study sites, seasonality, or anthropogenic impact (Rodrigues et al. 2008). Comparisons among different studies have little biological value because it is not possible to exclude the influence of these factors in the biological definition of the individual’s home range. Moreover, home range sizes most likely differ between males and females, and between adults and juveniles. We need more long-term studies in several study sites and regions, using a large number of individuals and a systematic data collection protocol, in order to reach a biological understanding of these differences.
Despite the presence of a few large populations in Brazil (e.g., in Emas and Serra da Canastra National Parks), most of giant anteaters are restricted to small fragments in protected or non-protected areas (Miranda et al. 2014; Diniz and Brito 2015; Bertassoni et al. 2017; 2019). Carrying out studies in different regions, including areas of different sizes, should also examine anthropogenic impacts on the study site. For instance, jaguars present larger home ranges in regions with higher anthropogenic impacts, which may also be true for other large-sized animals, as giant anteaters (Morato et al. 2016; Tucker et al. 2018).
Home range overlap
Giant anteaters seem to be quite tolerant of their intraspecific neighbors, as evidenced by a great deal of home range overlap described in the scientific literature. However, they display a marking behavior that could have a role in territoriality (Braga et al. 2010; Allard et al. 2014), and several studies have reported intraspecific aggression (Shaw et al. 1987; Rocha and Mourão 2006; Kreutz et al. 2009; Miranda Júnior and Bertassoni 2014). More research is needed to resolve this seeming paradox so that we can better understand if territoriality occurs in giant anteaters and, if so, what ecological conditions favor its expression.
Habitat selection
No study has been published to date that focuses on examining giant anteaters in native continuous forest; consequently, the giant anteater is generally considered an open-habitat species that prefer grasslands and areas near swamps. Throughout its distribution, it is also found within forested biomes, such as the Amazonian and Atlantic Forests (Oliveira et al. 2006; Passos et al. 2016; Bertrand and Soares 2018; Santos et al. 2019). Habitat selection studies have highlighted the importance of covered and forested (non-open) habitats within the individual’s core area, which is probably related to the availability of the mosaic of environmental temperatures. For example, the species uses gallery forests for foraging more often than open areas (Shaw et al. 1987); and even when forest habitats are less available, forest habitats can show a high positive selection (Di Blanco et al. 2015). The presence of forested areas that act as a temperature refuge seems to be one of the key factors in determining habitat use of giant anteaters (Camilo-Alves and Mourão 2006; Di Blanco et al. 2015). Timberlands can also be selected instead of native areas if they provide a suitable thermic refuge, as in the Acacia sp. plantations in northern Brazil (Kreutz et al. 2012).
Giant anteaters can tolerate altered habitats to a certain degree, but they depend on the proximity of natural areas (Vynne et al. 2011). In general, the persistence of the species seems to be related to habitat heterogeneity, which may offer a variety of resources, such as food, forest patches for thermic refuge, and corridor areas between habitats and matrix (the non-habitat portion of the landscape), such as sugarcane plantations (Bertassoni et al. 2019). Moreover, giant anteaters’ use of modified areas needs to be addressed in more detail regarding different anthropogenic levels (e.g., rural and urban areas; sugarcane, soy, coffee plantations). Some studies have recorded the occurrence of giant anteaters in exotic pastures (Vynne et al. 2011; Rojano-Bolaño et al. 2015). Contrarily, a study of habitat selection using two different spatial scales showed that giant anteaters, in fact, avoid pasture with cattle high densities (1.3–5.5 livestock/ha) (Di Blanco et al. 2015). The authors pointed out that this avoidance may be due to the “open” characteristics of the pasture that exposes the species to dangerous conditions such as poaching, wildfires, and conflicts with domestic dogs.
The species tends to select natural habitats distant from roads (Vynne et al. 2011). However, in instances when habitat conditions are altered, and then heavily influenced by agricultural areas, individuals prefer to move using roads (Vynne et al. 2011). Overall, roads can act as a passage connector between habitat patches (Vynne et al. 2011; Versiani 2016).
The proximity to human-modified landscapes
The species can change its period of activity depending on the region and the degree of human impact. For example, giant anteaters visited urban areas from 18:00h to 21:00h, synchronizing with the off-peak times of a human population in northern Brazil (Macedo et al. 2010). A similar pattern was found in an intensely modified landscape (41% of sugarcane) in southeast Brazil (Versiani 2016; Paolino et al. 2016). These examples show the versatility of the giant anteater in managing its time and activity in or near human-dominated landscapes.
There is currently a collection of descriptive reports regarding the occurrence of the species in anthropogenic areas (Miranda 2004; Braga 2010; Vynne et al. 2011; Kreutz et al. 2012; Timo et al. 2015). Although descriptive characteristics are a first step toward new knowledge, research emphasizing ecological hypotheses related to the use of anthropogenic areas is essential, such as plastic-foraging strategies (Gilmour et al. 2018) and the effect of the anthropogenic footprint on animal movements (Tucker et al. 2018), especially in this historical moment of great anthropogenic changes (Johnson et al. 2017).
Conclusion
We present updated information about space use by the giant anteater. This information could be used to guide new research and help develop conservation practices. In summary, we highlighted:
There is a need for further knowledge about the giant anteater throughout its geographical range. Most studies have been conducted in Brazil, in the Cerrado and Pantanal wetland biomes;
Studies carried out in other countries and in all existing Brazilian biomes are encouraged and essential to move knowledge forward;
There are no studies focusing on giant anteaters in continuous native forest. Research in these areas will be important to better understand the patterns of landscape use, and how patterns of space use in native forests mimic or differ from those reported from timberlands;
The lack of ecological studies in human-dominated landscapes is also of great concern, and this is a knowledge gap that requires great attention;
The decline of giant anteater populations is predominantly linked to human impacts, but there is evidence of adaptation to human-modified areas where natural resources are preserved. What are these resources? What is the extent of this behavioral plasticity/adaptation?
References
Abrahms B, Sawyer SC, Jordan NR, McNutt JW, Wilson AM, Brashares JS (2017) Does wildlife resource selection accurately inform corridor conservation? J Appl Ecol 54:412–422. https://doi.org/10.1111/1365-2664.12714
Allard SM, Earles JL, DesFosses L (2014) Spatial memory in captive giant anteaters (Myrmecophaga tridactyla). Anim Behav Cogn 1(3):331–340. https://doi.org/10.12966/abc.08.08
Allen AM, Singh NJ (2016) Linking movement ecology with wildlife management and conservation. Front Ecol Evol 3:155–168. https://doi.org/10.3389/fevo.2015.00155
Ascensão F, Desbiez AL, Medici EP, Bager A (2017) Spatial patterns of road mortality of medium–large mammals in Mato Grosso do Sul, Brazil. Wildl Res 44(2):135–146. https://doi.org/10.1071/WR16108
Attias N, Oliveira-Santos LGR, Fagan WF, Mourão G (2018) Effects of air temperature on habitat selection and activity patterns of two tropical imperfect homeotherms. Anim Behav 140:129–140. https://doi.org/10.1016/j.anbehav.2018.04.011
Barraquand F, Benhamou S (2008) Animal movements in heterogeneous landscapes: identifying profitable places and homogeneous movement bouts. Ecol 89:3336–3348. https://doi.org/10.1890/08-0162.1
Benhamou S, Riotte-Lambert L (2012) Beyond the utilization distribution: identifying home range areas that are intensively exploited or repeatedly visited. Ecol Modell 227:112–116. https://doi.org/10.1016/j.ecolmodel.2011.12.015
Bertassoni A (2010) Avaliação da relação entre área de vida, distância média diária percorrida e disponibilidade de energia de tamanduás-bandeira (Myrmecophaga tridactyla) em savanas neotropicais. Dissertation, Universidade Federal de Mato Grosso do Sul, Campo Grande, Brazil
Bertassoni A, Mourão G, Ribeiro RC, Cesário CS, Oliveira JP, Bianchi RC (2017) Movement patterns and space use of the first giant anteater (Myrmecophaga tridactyla) monitored in São Paulo State. Brazil. Stud Neotrop Fauna E. https://doi.org/10.1080/01650521.2016.1272167
Bertrand A, Soares AMVM (2018) First reports of giant anteater (Myrmecophaga tridactyla) and greater naked-tailed armadillo (Cabassous tatouay) for the Iguaçu National Park, Paraná, Brazil, with notes on all xenarthran occurrences. Edentata 19:35–41. https://doi.org/10.2305/IUCN.CH.2018.EDENTATA-19-1.4.en
Bowman J, Jaeger JAG, Fahrig L (2002) Dispersal distance of mammals is proportional to home range size. Ecol 83:2049–2055. https://doi.org/10.1890/0012-9658(2002)083[2049:DDOMIP]2.0.CO;2
Braga FG (2010) Ecologia e Comportamento de Tamanduá-bandeira Myrmecophaga tridactyla Linnaeus, 1758 no município de Jaguariaíva, Paraná. Thesis, Universidade Federal do Paraná, Curitiba
Braga FG, Santos REF, Batista AC (2010) Marking behavior of the giant anteater Myrmecophaga tridactyla (Mammalia: Myrmecophagidae) in Southern Brazil. Zoologia 27:7–12. https://doi.org/10.1590/S1984-46702010000100002
Butchart SH, Walpole M, Collen B, van Strien A, Scharlemann JP, Almond RE et al (2010) Global biodiversity: indicators of recent declines. Science 28:1164–1168. https://doi.org/10.1126/science.1187512
Bertassoni A, Costa RT, Gouvea JA, Bianchi RDC, Ribeiro JW, Vancine MH, Ribeiro MC, (2019) Land-use changes and the expansion of biofuel crops threaten the giant anteater in southeastern Brazil. Journal of Mammalogy 100 (2):435-444
Calenge C, Dray S, Royer-Carenzi M (2009) The concept of animals’ trajectories from a data analysis perspective. Ecol Inform 4:34–41. https://doi.org/10.1016/j.ecoinf.2008.10.002
Camilo-Alves CSP (2003) Adaptações dos tamanduás-bandeira (Myrmecophaga tridactyla Linnaeus, 1758) à variação da temperatura ambiente no Pantanal da Nhecolândia, MS. Thesis, Universidade Federal de Mato Grosso do Sul, Campo Grande, Brazil
Camilo-Alves CSP, Mourão G (2006) Responses of a specialized insectivorous mammal (Myrmecophaga tridactyla) to variation in ambient temperature. Biotropica 38:52–56. https://doi.org/10.1111/j.1744-7429.2006.00106.x
Ceballos G, Ehrlich PR (2002) Mammal population losses and the extinction crisis. Science 296:904–907. https://doi.org/10.1126/science.1069349
Ciocheti G, de Assis JC, Ribeiro JW, Ribeiro MC (2017) Highway widening and underpass effects on vertebrate road mortality. Biotropica 49:765–769. https://doi.org/10.1111/btp.12480
Clozato CL, Miranda FR, Lara-Ruiz P, Collevatti RG, Santos FR (2017) Population structure and genetic diversity of the giant anteater (Myrmecophaga tridactyla: Myrmecophagidae, Pilosa) in Brazil. Genet Mol Biol 40:50–60. https://doi.org/10.1590/1678-4685-GMB-2016-0104
Da Silveira NS, Niebuhr BBS, Muylaert RL, Ribeiro MC, Pizo MA (2016) Effects of land cover on the movement of frugivorous birds in a heterogeneous landscape. Plos One 11:e0156688. https://doi.org/10.1371/journal.pone.0156688
De Marco JP, Villén S, Mendes P, Nóbrega C, Cortes L, Castro T, Souza R (2018) Vulnerability of Cerrado threatened mammals: an integrative landscape and climate modeling approach. Biodivers Conserv.:1–22. https://doi.org/10.1007/s10531-018-1615-x
Di Blanco YE (2015) Patrones de actividad y de uso de hábitat de Osos Hormigueros (Myrmecophaga tridactyla) reintroducidos en Iberá, Corrientes, Argentina. Thesis, Universidad Nacional de Córdoba, Córdoba, Argentina
Di Blanco YE, Jiménez-Perez I, Di Bitetti MS (2015) Habitat selection in reintroduced giant anteaters: the critical role of conservation areas. J Mammal 20:1–12. https://doi.org/10.1093/jmammal/gyv107
Di Blanco YE, Spørring KL, Di Bitetti MS (2016) Daily activity pattern of reintroduced giant anteaters (Myrmecophaga tridactyla): effects of seasonality and experience. Mammalia 81:11–21. https://doi.org/10.1515/mammalia-2015-0088
Di Blanco Y, Desbiez ALJ, Jimenez-Perez I, Kluyber D, Massocato GF, Di Bitetti M (2017) Habitat selection and home-range use by resident and reintroduced giant anteaters in 2 South American wetlands. J Mammal 98:1118–1128. https://doi.org/10.1093/jmammal/gyx019
Diniz MF, Brito D (2012) The charismatic giant anteater (Myrmecophaga tridactyla): a famous John Doe? Edentata 13:76–83. https://doi.org/10.5537/020.013.0108
Diniz MF, Brito D (2015) Protected areas effectiveness in maintaining viable giant anteater (Myrmecophaga tridactyla) populations in an agricultural frontier. Nat Conserv 13:145–151. https://doi.org/10.1016/j.ncon.2015.08.001
Dirzo R, Young HS, Galetti M, Ceballos G, Isaac NJB, Collen B (2014) Defaunation in the Anthropocene. Nature 345(6195):401–406. https://doi.org/10.1126/science.1251817
Drumond MA (1992) Padrões de forrageamento do tamanduá-bandeira (Myrmecophaga tridactyla) no Parque Nacional da Serra da Canastra: dieta, comportamento alimentar e efeito de queimadas. Thesis, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil
Duffy KJ, Dai X, Shannon G, Slotow R, Page B (2011) Movement patterns of African elephants (Loxodonta africana) in different habitat types. S Afr J Wildl Res 41:21–28
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. https://doi.org/10.1146/annurev.ecolsys.34.011802.132419
Freitas CH, Justino CS, Setz EZF (2014) Road-kills of the giant anteater in south-eastern Brazil: 10 years monitoring spatial and temporal determinants. Wild Res 41:673–680. https://doi.org/10.1071/WR14220
Gilmour ME, Castillo-Guerrero JA, Fleishman AB, Hernández-Vázquez S, Young HS, Shaffer SA (2018) Plasticity of foraging behaviors in response to diverse environmental conditions. Ecosphere 9(7):e02301. https://doi.org/10.1002/ecs2.2301
Grotta-Neto F, Peres PHF, Piovezan U, Passos FC, Duarte JMB (2019) Influential factors on gray brocket deer (Mazama gouazoubira) activity and movement in the Pantanal, Brazil. J Mammal 100:454–463. https://doi.org/10.1093/jmammal/gyz056
Jerez S, Halloy M (2003) El oso hormiguero, Myrmecophaga tridactyla: Crecimiento e independizacion de una cria. J Neotrop Mammal 10:323–330
Johnson CN, Balmford A, Brook BW, Buettel JC, Galetti M, Guangchun L, Wilmshurst JM (2017) Biodiversity losses and conservation responses in the Anthropocene. Science 356:270–275. https://doi.org/10.1126/science.aam9317
Jønsson KA, Tøttrup AP, Borregaard MK, Keith SA, Rahbek C, Thorup K (2016) Tracking animal dispersal: from individual movement to community assembly and global range dynamics. Trends Ecol Evol 31:204–214. https://doi.org/10.1016/j.tree.2016.01.003
Kreutz K, Fischer F, Linsenmair KE (2009) Observations of intraspecific aggression in giant anteaters (Myrmecophaga tridactyla). Edentata 8:6–7. https://doi.org/10.1896/020.010.0107
Kreutz K, Fischer F, Linsenmair KE (2012) Timber plantations as favourite habitat for giant anteaters. Mammalia 76:137–142. https://doi.org/10.1515/mammalia-2011-0049
Macedo LSM, Azevedo RB, Pinto F (2010) Área de vida, uso do habitat e padrão de atividade do tamanduá-bandeira na savana de Boa Vista, Roraima. In: Barbosa RI, Melo VF (eds) Roraima: Homem, Ambiente e Ecologia. FEMACT, Boa Vista, pp 585–602
McGowan J, Beger M, Lewison RL, Harcourt R, Campbell H, Priest M et al (2017) Integrating research using animal-borne telemetry with the needs of conservation management. J Appl Ecol 54:423–429. https://doi.org/10.1111/1365-2664.12755
Medri IM, Mourão G (2005) Home range of giant anteaters (Myrmecophaga tridactyla) in the Pantanal wetland, Brazil. J Zool 266:365–375. https://doi.org/10.1017/S0952836905007004
Miotto RA, Cervini M, Begotti RA, Galetti PM Jr (2012) Monitoring a puma (Puma concolor) population in a fragmented landscape in southeast Brazil. Biotropica 44:98–104. https://doi.org/10.1111/j.1744-7429.2011.00772.x
Miranda GHB (2004) Ecologia e conservação do tamanduá-bandeira (Myrmecophaga tridactyla, Linnaeus, 1789) no Parque Nacional das Emas. Thesis, Universidade de Brasília, Brasília, Brazil
Miranda Júnior JF, Bertassoni A (2014) Potential agonistic courtship and mating behavior between two adult giant anteaters (Myrmecophaga tridactyla). Edentata 15:69–72. https://doi.org/10.5537/020.015.0105
Miranda F, Bertassoni A, Abba AM (2014) Myrmecophaga tridactyla. The IUCN Red List of Threatened Species 2014: e.T14224A47441961. https://doi.org/10.2305/IUCN.UK.2014-1.RLTS.T14224A47441961.en. Downloaded on 27 November 2016
Miranda FR, Chiarello AG, Röhe F, Braga FG, Mourão GM, Miranda GHB (2015) Avaliação do risco de extinção de Myrmecophaga tridactyla Linnaeus 1758 no Brasil. In: ICMBio (ed) Avaliação do Risco de Extinção dos Xenartros Brasileiros. ICMBio, Brasília, pp 89–105
Mohr CO (1947) Table of equivalent populations of North American mammals. Am Midl Nat 37:223–249
Montgomery GG (1985) Movements, foraging and food habitats of four extant species of neotropical Vermilinguas (Mammalia; Myrmecophagidade). In: Montgomery GG (ed) The evolution and ecology of armadillos, sloths and vermilinguas. Smithsonian Institution, Washington, pp 365–377
Montgomery GG, Lubin YD (1977) Prey influences on movements of Neotropical anteaters. In: Phillips RL, Jonkel C (eds) Proceedings of the 1975 Predator Symposium. University of Montana, Montana, pp 103–131
Morato RG, Stabach JA, Fleming CH, Calabrese JM, De Paula RC, Ferraz KMPM et al (2016) Space use and movement of a neotropical top predator: the endangered jaguar. Plos One 11:e0168176. https://doi.org/10.1371/journal.pone.0168176
Mourão G, Medri IM (2007) Activity of a specialized insectivorous mammal (Myrmecophaga tridactyla) in the Pantanal of Brazil. J Zool 271:187–192. https://doi.org/10.1111/j.1469-7998.2006.00198.x
Oliveira LC, Mendel SM, Loretto D, Júnior JSS, Fernandes GW (2006) Edentates of the Saracá-Taquera National Forest, Pará, Brazil. Edentata 7:3–7. https://doi.org/10.1896/1413-4411.7.1.3
Passos F, Mello M, Isasi-Catalá E, Mello R, Bernardi I, Varzinczak L et al (2016) The vulnerable giant anteater Myrmecophaga tridactyla: new records from the Atlantic Forest highlands and an overview of its occurrence in protected areas in Brazil. Oryx 51:564–566. https://doi.org/10.1017/S0030605316000740
Paolino RM, Versiani NF, Pasqualotto N, Rodrigues TF, Krepschi VG, Chiarello AG, (2016) Buffer zone use by mammals in a Cerrado protected area. Biota Neotropica 16 (2)
Ribeiro P, Miranda JES, Araújo DR (2017) The effect of roadkills on the persistence of xenarthran populations: the case of the Brazilian Cerrado. Edentata 18:51–61. https://doi.org/10.2305/IUCN.CH.2017.Edentata-18-1.7.en
Rocha FL, Mourão G (2006) An agonistic encounter between two giant anteater (Myrmecophaga tridactyla). Edentata 7:50–51. https://doi.org/10.1896/1413-4411.7.1.50
Rodrigues FHG, Medri IM, Miranda GHB, Camilo-Alves C, Mourão G (2008) Anteater behavior and ecology. In: Vizcaíno SF, Loughry WJ (eds) The biology of the Xenarthra. University Press of Florida, Gainesville, pp 257–268
Rojano-Bolaño C, Giraldo MEL, Miranda-Cortés L, Avilán RA (2015) Área de vida y uso de hábitats de dos individuos de oso palmero (Myrmecophaga tridactyla) en Pore, Casanare, Colombia. Edentata 16:37–45
Shaw JH, Carter TS, Machado-Neto JC (1985) Ecology of the giant anteater Myrmecophaga tridactyla in Serra da Canastra, Minas Gerais, Brazil: a pilot study. In: Montgomery GG (ed) The evolution and ecology of armadillos, sloths and vermilinguas. Smithsonian Institution, Washington, pp 379–384
Shaw JH, Machado-Neto JC, Carter TS (1987) Behavior of free-living giant anteaters (Myrmecophaga tridactyla). Biotropica 19:255–259. https://doi.org/10.2307/2388344
Silveira EKP (1969) História natural do tamanduá-bandeira Myrmecophaga tridactyla Linn. 1758, Myrmecophagidae. Vellozia 7:34–43
Smith DA, Ralls K, Hurt A, Adams B, Parker M, Maldonado JE (2006) Assessing reliability of microsatellite genotypes from kit fox faecal samples using genetic and GIS analyses. Mol Ecol 15:387–406. https://doi.org/10.1111/j.1365-294X.2005.02841.x
Santos PS, Bocchiglieri A, Chiarello AG, Paglia AP, Moreira A, Souza AC, Abba AM, Paviolo A, Gatica A, Medeiro AZ, Costa AN, Gallina AG, Yanosky AA, Jesus A, Bertassoni A, Rocha A, Bovo AAA, Bager A, Mol CA, Martensen AC, Faustino AC, Lopes AMC, Percequillo AR, Vogliotti A, Keuroghlian A, Colina MA, Devlin AL, García‐Olaechea A, Sánchez A, Srbek‐Araujo AC, Ochoa AC, Oliveira ACM, Lacerda ACR, Campelo AKN, Paschoal AMO, Costa ARC, Meiga AYY, Jesus AS, Feijó A, Hirsch A, Silva ALF, Botelho ALM, Regolin AL, Lanna AM, Nunes AV, Kindel A, Moraes AM, Gatti A, Noss AJ, Nobre AB, Montanarin A, Deffaci ÂC, Albuquerque ACF, Oliveira AK, Mangione AM, Pontes ARM, Bertoldi AT, Calouro AM, Desbiez AJL, Fernandes A, Ferreguetti AC, Silva MAA, Zimbres B, Luciano BFL, Thoisy B, Niebuhr BBS, Papi B, Gómez‐Valencia B, Santos BA, Lima BC, Oliveira BG, Santos BS, Campos BATP, Leles B, França BRA, Lim B, Oliveira CT, Cantagallo C, Lara CC, Lima CS, Gestich CC, Melo‐Soares CD, Peres CA, Kasper CB, Candia‐Gallardo C, Angelo CD, Fragoso CE, Freitas CH, Salvador CH, Brocardo CR, Melo CD, Leuchtenberger C, Braga C, Sánchez‐Lalinde C, Bueno C, Luna CL, Rojano C, Hurtado CM, Santos CC, Tellaeche C, Rosa C, Campos CB, Silva CR, Kanda CZ, Jenkins CN, McDonough C, Trinca CT, Cunha CJ, Widmer CE, Santos C, Buscariol D, Carreira DC, Carvalho DR, Ferraz DS, Casali D, Thornton D, Vasconcellos DR, Barcelos D, Brown D, Ramos DL, Moreira DO, Yogui DR, Faria D, Sana DA, Mattia DL, Henz DJ, Friedeberg DB, Carvalho DLKP, Astúa D, Queirolo D, Varela DM, Eaton DP, Dias DM, Rivadeneira EF, Rocha EC, Abreu‐Júnior EF, Carrano E, Santos EM, Setz EZF, Carvalho EAR, Chiquito EA, Cardoso EM, Mendonça EN, D'Bastiani E, Vieira EM, Ramalho EE, Guijosa‐Guadarrama E, González E, Maggiorini EV, Fischer E, Aguiar EF, Castro ÉF, Peña‐Cuéllar EL, Viveiros Castro EB, Brítez EB, Vanderhoeven EA, Pedó E, Rocha FL, Girardi F, Roque FO, Mazim FD, Barros FM, Martello F, Fantacini FM, Pedrosa F, Peters FB, Abra FD, Azevedo FC, Santos FS, Silva FG, Teixeira FZ, Perini FA, Passos FC, Carvalho F, Azevedo FCC, Pinho FF, Gonçalves F, Lima F, Contreras‐Moreno FM, Pedroni F, Tortato FR, Santos FPR, Caruso F, Tirelli FP, Miranda FR, Rodrigues FHG, Ubaid FK, Palmeira FBL, Silva FA, Grotta‐Neto F, Souza FL, Costa FE, Pérez‐Garduza F, Delsuc F, Lemos F, Pinto FR, Boaglio GI, Massocato GF, Preuss G, Hofmann GS, Aguiar GL, Oliveira GS, Duarte GT, Beca G, Giné GAF, Batista GO, Gil GE, Gonsioroski G, Secco H, Medeiros HR, Coelho IP, Franceschi IC, Bernardi I, Torre JA, Zocche JJ, Seibert JB, Falcão JCF, Dias JHM, Nodari JZ, Oliveira JA, Giovanelli JGR, Favoretti JPP, Polisar J, Sponchiado J, Cherem JJ, Ramírez JFM, Toledo JJ, Duarte JMB, Matos JB, Arrabal JP, Oshima JEF, Ribeiro JF, Bogoni JA, Pacheco JJC, Schuchmann KL, Ferraz KMPMB, Everton LS, Bailey LL, Gonçalves LO, Cullen L, Andrade LR, Trevelin LC, Bonjorne L, Rodrigues LA, Leuzinger L, Perillo LN, Araújo LS, Hufnagel L, Ribeiro LO, Bernardo LRR, Oliveira‐Santos LGR, Varzinczak LH, Borges LHM, Guimarães LN, Möcklinghoff L, Oliveira MA, Magioli M, Jardim MMA, Oliveira ML, Tortato MA, Dums M, Iezzi ME, Pereira MJP, Jorge ML, Morini MSC, Landis MB, Xavier MS, Barros MAS, Silva ML, Rivero M, Zanin M, Marques MI, Alves H, Bitetti MSD, Alvarez MR, Graipel ME, Godoi MN, Benedetti MA, Beltrão MG, Monteiro MCM, Paula MJ, Perilli MLL, Silva MP, Villar N, Albuquerque NMD, Canassa NF, Filho NM, Oliveira NR, Pasqualotto N, Cáceres NC, Attias N, Favarini MO, Ribeiro OS, Gonçalves PR, Rocha PA, Condé PA, Akkawi P, Cruz P, Lira PK, Ferreira PM, Arroyo‐Gerala P, Hartmann PA, Antas PTZ, Marinho PH, Peres PHF, Peña‐Mondragón JL, Lombardi PM, Laurindo RS, Alves RSC, Grangeiro RDP, Silva RL, Beltrão‐Mendes R, Bonikowski RTR, Reppucci J, Arrais RC, Sampaio R, Sartorello R, Bovendorp RS, McNab R, Hack ROE, Magalhães RA, Araújo RC, Nobre RA, Pérez RRL, Massara RL, Paula RC, Anleu RG, Marques RV, Dornas R, Rolim SG, Cavalcanti SMC, Lima SR, Ballari SA, Santamaría SB, Silva SM, Age SG, Godim T, Sobral‐Souza T, Maccarini TB, Rodrigues TF, Piovezan U, Tavares VDC, Quiroga VA, Krepschi VG, Filho VP, Bastazini VAG, Gasparotto VPO, Orsini VA, Layme VMG, Hannibal W, Dáttilo W, Carvalho WD, Loughry WJ, Blanco YED, Núñez‐Regueiro MM, Giubbina MF, Passamani M, Querido LCA, Toledo GAC, Ribeiro IK, Quintilham L, Bustos S, Maza J, Neto JFL, Silva KVKA, Sartorello L, Rampim LE, Marás LA, Camino M, Freitas‐Junior M, Perovic PG, Paolino RM, Ferreira SD, Towns V, Esperandio IB, Aximoff I, Beduschi J, Guenther M, Bianchi MC, Keuroghlian‐Eaton S, Mendes SL, Cunha LF, Cirignoli S, Ciocheti G, Prado HA, Fernandes‐Ferreira H, Sena LMM, Yamane MH, Brennand PGG, Silva RD, Escobar S, Endo W, Hurtado RR, Gontijo NRC, Marsh LK, Severo MM, Pardo JM, Costa SA, Melo GL, Santana GG, Mourão GM, Gaspari GG, Duarte H, Cabral H, Silva LH, Mendonça L, Barbosa LL, Santos MV, Moraes MFD, Gordo M, Versiani NF, Cantero N, Pays O, Guedes PG, Colas‐Rosas PF, Ribeiro P, Renaud PC, Hoogesteijn RJ, Ayala R, Cunha RGT, Schaub R, Laurito S, Betkowski SE, Cortez S, Silva SSP, Oliveira TG, Spironello WR, Gengler N, Hidalgo MM, Juárez R, Iglesias JA, Anacleto TC, Fialho MS, Cavicchioli G, Beccato MAB, Silva MD, Neto OC, Lopes KGD, Godoy LP, Luiz MR, Bonzi VBR, Ferreira GB, Oliveira MJR, Hinojosa J, Oliveira LFB, Nagy‐Reis MB, Ramirez SF, Concone HVB, Mourthe I, Martínez‐Lanfranco JA, Zanoni JB, Moreira TC, Guarderas ZV, Bazilio S, Cervini M, Pinheiro MS, Morato RG, Peroni N, Trigo TC, Machado RB, Gaspari F, Koenemann JG, Rudolf JC, Benchimol M, Vieira MV, Retta LM, Santiago PGF, Ciccia PG, Estrela PC, Carvalho S, Esbérard CEL, Cruz YBL, Castro‐Prieto J, Braga RM, Cartes JL, Andrade‐Núñez MJ, Denkiewicz NM, Falconi N, Pezzuti JCB, Cordero HFC, Sousa LC, Gaspari Júnior RL, Santos‐Filho M, Almeida JS, Thompson JJ, Santos JS, Pereira‐Ribeiro J, Burs K, Silva KFM, Velilla M, Silva MX, Sancha NU, Pinheiro PF, Castilho PV, Bercê W, Assis JC, Tonetti VR, Alves‐Eigenheer M, Chinem S, Honda LK, Bergallo HG, Alberici V, Wallace R, Krauer JMC, Ribeiro MC, Galetti M, (2019) NEOTROPICAL XENARTHRANS: a dataset of occurrence of xenarthran species in the Neotropics. Ecology 100 (7)
Timo TPC, Lyra-Jorge MC, Gheler-Costa C, Verdade LM (2015) Effect of the plantation age on the use of Eucalyptus stands by medium to large-sized wild mammals in south-eastern Brazil. iForest 8:108–113. https://doi.org/10.3832/ifor1237-008
Tucker MA, Böhning-Gaese K, Fagan WF, Fryxell JM, Van Moorter B, Alberts SC et al (2018) Moving in the Anthropocene: global reductions in terrestrial mammalian movements. Science 359:466–469. https://doi.org/10.1126/science.aam9712
Turchin P (1991) Translating foraging movements in heterogeneous environments into the spatial distribution of foragers. Ecology 72:1253–1266. https://doi.org/10.2307/1941099
Versiani NF (2016) O tamanduá-bandeira (Myrmecophaga tridactyla) em áreas protegidas e seus entornos no Cerrado do nordeste do estado de São Paulo. Thesis, Universidade de São Paulo, Ribeirão Preto, Brazil
Vynne C, Keim JL, Machado RB, Marinho-Filho J, Silveira L, Groom MJ, Wasser SK (2011) Resource selection and its implications for wide-ranging mammals of the Brazilian Cerrado. Plos One 6:e28939. https://doi.org/10.1371/journal.pone.0028939
Worton BJ (1989) Kernel methods for estimating the utilization distribution in home range studies. Ecology 70:164–168. https://doi.org/10.2307/1938423
Whitmee S, Orme CDL, Humphries H, (2013) Predicting dispersal distance in mammals: a trait-based approach. Journal of Animal Ecology 82 (1):211-221
Young HS, McCauley DJ, Galetti M, Dirzo R (2016) Patterns, causes, and consequences of Anthropocene defaunation. Annu Rev Ecol Evol Syst 47:333–358. https://doi.org/10.1146/annurev-ecolsys-112414-054142
Acknowledgments
We would like to thank all the contributions to improve the manuscript, and the text formatting. We specially thank Christina Lorimer for revising the language and Ana Luíza de Faria Peres for helping in the editing process.
Funding
AB and MCR thank the São Paulo Research Foundation - FAPESP (2013/04957-8) and (2013/50421-2), respectively. MCR also thanks Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq (process no. 312045/2013-1; no. 312292/2016-3) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES (PROCAD project no. 88881.068425/2014-01) for their financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 33 kb)
Rights and permissions
About this article
Cite this article
Bertassoni, A., Ribeiro, M.C. Space use by the giant anteater (Myrmecophaga tridactyla): a review and key directions for future research. Eur J Wildl Res 65, 93 (2019). https://doi.org/10.1007/s10344-019-1334-y
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-019-1334-y