Abstract
Human activities affect large carnivores worldwide by increasing mortality due to destruction and fragmentation of habitats, decrease of prey availability, and hunting pressure. The jaguar (Panthera onca) is a large carnivore strongly influenced by these threats, with poaching of predators being primarily a retaliatory response that is motivated by depredation of domestic animals. We investigate the distribution of jaguars and cattle depredation in 21 sampling units located in Central Brazil. We consider native and domestic prey availability as well as landscape configuration and composition as possible contributory factors. We removed correlated variables and conducted a set of logistic regressions in a step-wise approach. We used the difference between χ2 of the newest and the previous model to evaluate if the addition of a variable increased the explanatory power of the newest model. Jaguar occurrence was influenced by prey richness, which is correlated with habitat aggregation in the landscape, revealing the attributes that can act as proxies for environmental quality for jaguar. The relationship between jaguar and prey richness also suggests that jaguar can act as a surrogate for the presence of other species. Jaguar occurrence is also related to the availability of non-native prey such as cattle, but not cattle depredation, suggesting that cattle depredation could be an opportunistic event. Patterns of cattle depredation warrant further study because human-wildlife conflict is one of the greatest threats to jaguar conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Large carnivores are currently one of the most threatened groups of species (Ripple et al. 2014). Their large body size demands large areas of suitable habitat and prey availability, resulting in their occurrence in less disturbed habitat. In this way, human-derived landscape changes are the main threats for large carnivores worldwide (Ripple et al. 2014) because they destroy and fragment habitats (Lord and Norton 1990; Ritters et al. 2000; Villard and Metzger 2014), decrease food availability (O’Brien et al. 2003; Ryall and Fahrig 2006), and increase hunting pressure (White and Lowe 2008; Inskip and Zimmermann 2009).
Jaguar (Panthera onca), the largest American feline and an important top predator, is an example of a large carnivore affected by these threats. Human-derived landscape changes affect jaguar distributions by reducing and subdividing the available habitat (Hatten et al. 2005; Zanin et al. 2014), altering the colonization rates of patches (Zanin et al. 2015), and modifying the land cover matrix (Zeilhofer et al. 2014). Reductions of natural habitat can also reduce the availability of prey for jaguar (Tews et al. 2004; Ahumada et al. 2011), further compounding the negative impacts of human landscape changes. To make matters worse, livestock ranching is one of the principal factors of human-driven land-use change within the jaguar’s distribution, and livestock may be an important component of their diet (Polisar et al. 2003; Azevedo 2008; Cavalcanti and Gese 2010). Livestock depredation results in retaliatory killing and intensifies human-wildlife conflict (Azevedo and Murray 2007b; White and Lowe 2008), which is one of the main threats for jaguar conservation (Altrichter et al. 2006; Mazzolli 2009).
Therefore, human impacts on landscapes act in several ways on jaguar occurrence and should be investigated to increase the effectiveness of conservation strategies (Woodroffe and Ginsberg 1998; Dickman et al. 2011). The jaguar has already lost almost half of its original range distribution (Zeller 2007) and has been listed among the top 20 large-bodied species with the highest anthropogenically driven range contractions (Morrison et al. 2007), highlighting the need for studies investigating these impacts on jaguar distribution.
Here, we investigate the ecological patterns of jaguar occurrence and livestock depredation in light of the two main anthropogenic changes to landscapes, habitat conversion, and decreased prey base. This study was conducted at a landscape scale in a set of 21 landscapes (our sampling units), using interviews with local residents as source information. This landscape-scale (sensu McGarigal and Cushman 2002) approach is uncommon for jaguar studies (Zanin et al. 2014); instead, local (Azevedo and Murray 2007a; Zeilhofer et al. 2014), patch (Michalski and Peres 2007; Calaça et al. 2010), or macroecological approaches are more typical (Hatten et al. 2005; Inskip and Zimmermann 2009; Tôrres et al. 2012), making our investigation both challenging and innovative for determining the ecological attributes contributing to the species’ occurrence. Interviews have been widely used to obtain information about the presence of large mammals in tropical forests, especially in areas where human populations have been present for a long time (Barthem et al. 1991; Cesar and Azevedo 2003; Zimmermann et al. 2005; Bagchi and Mishra 2006; Bisi et al. 2007; Zeller et al. 2011; Petracca et al. 2014a, b), and are increasingly recognized as a reliable method for our type of scaled approach.
We expected a higher occurrence probability of jaguar in less disturbed landscapes because the jaguar has extensive ecological requirements and should occur only in less anthropogenically influenced landscapes (Zeilhofer et al. 2014). Therefore, we expected to see a positive relationship between jaguar occurrence and high prey availability and low configuration complexity according to landscape metrics, which is a property of less disturbed landscapes (Li et al. 2005). Consequently, we also predicted a negative relationship between jaguar occurrence and human pressures on the environment, which were represented by matrix use variables. The probability of cattle depredation by jaguar should be related to low environmental quality (i.e., landscapes with a high complexity, anthropogenic matrix use, and low prey availability) because under such conditions, jaguars could be forced to search for non-native prey, thereby increasing livestock depredation (Polisar et al. 2003; Bagchi and Mishra 2006).
Methods
Study area
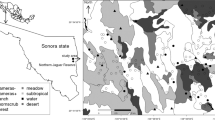
This study was carried out in Central Brazil along the Araguaia River, located between 3° 25′ 13″ (S) and 18° 15′ 40″ (S) latitude and 53° 26′ 26″ and 47° 53′ 07″ longitude (Fig. 1). The Araguaia River extends for 1982.5 km, with a source near Emas National Park in Goiás state and empties into the Tocantins River in Pará state. Our study area comprised a 20-km buffer strip along each side of the river and continued along a stretch of 248.7 km of the Tocantins River to the Tucuruí hydroelectric dam. This area is considered a potential corridor for species dispersion between Brazil’s two major biomes: the Amazon rainforest and Cerrado savanna (http://www.jaguar.org.br/en/projects/araguaia-corridor/index.html).
Many types of native vegetation cover, which are characteristic of Amazon and Cerrado biomes, can be observed along the Araguaia River. However, the native vegetation cover does not impact directly on jaguar distribution because it is a species with considerable environmental plasticity in terms of this attribute (Conde et al. 2010; Colchero et al. 2011). In contrast, anthropogenic land cover is less diverse and can influence the distribution of jaguar (Zeilhofer et al. 2014). The main anthropogenic land uses in the study area are agricultural production of soy, corn, rice, and pasture for livestock.
Sampling design
We divided the study area into a grid of 10 km × 10 km. This grid cell size was chosen because it is an acceptable approximation of jaguar home range (Cullen Jr. 2006; Cavalcanti and Gese 2009) and since the scale should be meaningful to the species under study (Fahrig 2003). Thirty cells were randomly chosen as the sampling units, but nine of them had to be discarded due to the logistical difficulties in reaching the areas (Fig. 1).
Interview protocol and species occurrence data
We first conducted test interviews with 13 local residents of different age classes and from different professions to determine interview criteria in order to ensure the reliability of the collected data. Based on these initial tests, we established the following set of criteria for the final interviewees: (i) they must be at least 15 years old; (ii) must have lived in the area for more than 1 year; (iii) must not have any close relation with another interviewee of the same sampling unit (i.e., first-degree relation, such as husband and wife or father and son). We conducted the interviews between January and April 2009, using a standard questionnaire (Appendix A—standard questionnaire), following a standard protocol to guarantee the veracity of information (Fig. 2). We asked residents about the occurrence of jaguars, cattle depredation by jaguars, and the presence of potential jaguar prey species within the last 3 years. To guarantee the veracity of information, we asked the interviewees to describe the species that they knew to occur in the sampling area, or we tested their ability to recognize intentionally incorrect information provided by us. If they had some additional evidence, such as the skins or skulls, we asked to see it. We only considered interviews for analysis where species identification was correctly assigned by the interviewee on the first hand.
Standard protocol used to survey the sampling units and to obtain information about cattle predation, and jaguar and prey species occurrence. An account of species occurrence was accepted by the interviewer when the interviewee provided a coherent description of the species or if they provided irrefutable evidence such as the skin or skull, thereby guaranteeing the veracity of the information
We conducted five interviews in each landscape to increase the detectability of species. To maximize reliability of the true presence data, we adopted the threshold of at least two independent identifications of an event (e.g., species occurrence or cattle depredation) to assign a positive occurrence to the sampling unit. Therefore, the five interviews conducted in each sampling unit were condensed into one response (presence or absence) for an investigated event.
Landscape characterization
The landscapes were characterized through the metrics of the configuration and composition of native vegetation. We homogenized the composition of landscapes, considering all native land cover categories as equal. This level of generalization was adopted because jaguars show significant environmental plasticity in terms of their use of vegetation types (Conde et al. 2010; Colchero et al. 2011), making this an acceptable generalization for jaguar habitat use at a broad environmental scale. The GLOBCOVER map was adopted to define land use in the study landscapes, which has a resolution of 1 km (Bontemps et al. 2011).
Landscape metrics were calculated to represent the composition and configuration of sampling units using the Fragstats software (McGarigal et al. 2012). Some metrics expressed the degree of native vegetation cover (total area of native vegetation cover, average patch size, and largest patch index), and others were selected to capture landscape heterogeneity (patch number, standard deviation of patch area, aggregation index, and landscape shape index) (Table 1).
We chose indirect metrics to represent anthropogenic landscape use because the spatial configuration of anthropogenic cover and native cover are correlated (Riitters et al. 1995; Li et al. 2005). Therefore, these indirect matrix metrics were more informative for our analyses. The descriptors of human pressure in the landscapes were human density, cattle density, and agricultural productivity. The values for these variables were derived from the Brazilian Institute of Statistics and Geography (IBGE—Instituto Brasileiro de Geografia e Estatística 2007, 2008a, b). The minimum scale for data compilation by the IBGE is at the municipal level; so, for our analyses, we considered the municipality of a given sampling unit; for sampling units divided between more than one municipality, the IBGE values were corrected for the area of sampling unit in each municipality.
Native jaguar prey was represented by richness of native prey (S) occurring in the landscape according to the results of interviews (described above). However, cattle in the landscape could also consist of an important component of jaguar diets; so, cattle density can also be interpreted as a component of prey availability. To account for this, we created a variable that represented the total jaguar prey (native and livestock) through a statistical interaction of cattle density and native prey richness (corrected by the maximum native prey richness observed among all the sampling units).
Data analysis
One important concern regarding the evaluation of environmental variables affecting predator distributions is spatial autocorrelation. Many studies have demonstrated the difficulty of obtaining the spatially independent observations that are required for reliable testing of ecological hypotheses (Legendre and Legendre 1998; Wagner and Fortin 2005; Mortelliti et al. 2010). Our data on jaguar and cattle depredation occurrences showed this kind of spatial structure (Table 2). Therefore, we used a statistical approach that explicitly factors in spatial autocorrelation to counteract this problem and to measure its effect on the analyses.
We used spatial filters to control spatial autocorrelation, which is a method that includes space as a covariate. This method permitted us to capture the orthogonal variation in spatial structure through different scales without inserting redundant parameters (Diniz-Filho and Bini 2005; Patuelli et al. 2010; Dray 2011). Spatial filters are eigenvectors from principal coordinates of neighbor matrices (Borcard and Legendre 2002), which we generated using the Spatial Analysis Macroecology (SAM) software (Rangel et al. 2010) based on the geographical distance between sampling units.
Landscape variables and spatial filters can capture the same ecological processes, generating a problem of over-representation in statistical tests; so, we performed a factor analysis (FA) to determine the number of descriptors and avoid this problem. We used the Varimax normalized method to rotate axes (factors) and eigenvalues greater than one as the criterion to select factors. We selected all noncorrelated variables plus one from each group of correlated variables. We used the variables, as opposed to factors as is common in ecological studies, because our intention was to directly measure the effects of variables to make inferences on the impact of landscape properties on jaguar occurrence and cattle depredation.
The effects of selected variables on jaguar presence and cattle depredation were evaluated through logistic regression. We conducted a set of logistic regressions, starting with one variable and including new variables in a step-wise approach. We used the difference between χ2 of the newest and the previous model to evaluate if the addition of a variable increased the explanatory power of the newest model. This method uses maximum likelihood estimates of the loss function to permit selection of the most parsimonious model describing the ecological process in question (Hosmer et al. 2013). For cattle depredation models, we only selected sampling units where the jaguar was present.
The residuals of the fitted models were evaluated to identify any possible remaining spatial autocorrelation and to evaluate the efficiency of our analysis to control spatial autocorrelation. We calculated the standardized Moran’s I index (Moran 1950) for four distance classes, which were defined based on the number of nearest neighbors of sampling units. The standardized Moran’s I index can vary between −1.0 and 1.0 for the maximum negative and positive autocorrelation, respectively, with a value of zero indicative of no autocorrelation.
Results
Interview data characterization
We conducted 105 independent interviews across the 21 sampling units. The interviewees had an average of 18.7 years (SD = 16.09) of residency in the sampled area, and most of them (76.3 %) lived/worked in rural areas within the study region. We identified 15 landscapes with jaguar occurrence and 10 with cattle depredation covering both biomes (Fig. S1). Regarding potential prey species, prey richness varied from eight to 16 species, but only the collared peccary (Pecari tajacu) and the agouti (Dasyprocta sp.) were found in all sampling units. Five prey species were only found in a few locations: greater long-nosed armadillo (Dasypus kappleri—four locations), giant armadillo (Priodontes maximus—seven locations), pampas deer (Ozotoceros bezoarticus—eight locations), and marsh deer (Blastocerus dichotomus—nine locations) (Fig. S1).
Variable selection
The relationship among sampling unit distances generated two spatial filters, and these were incorporated into the factor analysis. We selected four factors with eigenvalues greater than one, which explained 80.6 % of the variation and included four sets of correlated variables and one uncorrelated variable (Table S1), which was automatically selected (standard variation of patch size).
Total native vegetation area, large patch index, and mean patch area were correlated in the first factor. Among this set of variables, we chose the total native vegetation area, which totaled 695 km2 for all sampling units, but with landscapes varying from nearly no native vegetation whatsoever to almost complete native vegetation cover (mean = 33 km2, SD = 30 km2).
The second group of variables (factor two) included prey richness, aggregation index, agricultural density, and spatial filter two. Agricultural productivity was negatively correlated with native prey richness and the aggregation index, suggesting that intensification of agricultural activity decreases habitat aggregation and native prey availability. We selected the native prey richness to represent this set of variables.
For factor 3, we identified a correlation between human density, spatial filter one, cattle density, and the interaction between cattle density and native prey richness. We selected cattle density because cattle frequently feature in the diet of jaguar and it is an important factor relating to conflict with the species so can influence jaguar occurrence.
Factor 4 showed a correlation between the number of patches and the landscape shape index (Table S1), which together captured heterogeneity in the landscape. We selected number of patches for simplicity and because landscape heterogeneity is probably best defined by that parameter. The number of patches varied from one to eight (mean = 2.14; standard deviation = 3.76).
Matrix variables did not correlate with any of the structural metrics used here, supporting our decision to include anthropogenic variables as independent explanatory variables of the land cover matrix in place of structural metrics. Additionally, we observed that the spatial filters did not contribute new information to the models because the structure could already be captured by our variables.
Occurrence of jaguars and cattle depredation
Jaguar occurrence was clearly revealed as being a response to prey base because the most parsimonious model among those evaluated was composed of the additive effect between prey richness and cattle density. This also suggests that landscape configuration does not affect jaguar occurrence (Table 3). Even so, the occurrence of jaguar throughout our study area showed a spatially structured pattern. However, the residuals of the final model did not evidence spatial autocorrelation, showing that the variables were efficient in capturing the spatial pattern present in the data (Table 4). Our set of variables was not able to explain patterns of cattle depredation (Table 3).
Discussion
Landscape-scale approaches for scientific studies on jaguar are uncommon (Zanin et al. 2014); so, we could expect differences in the patterns observed in our study from those remarked on in previous literature because different results are frequently observed at different scales of analysis in ecological studies (Lyra-Jorge et al. 2010; Červinka et al. 2013; Wong and Linkie 2013). Therefore, our findings on the ecological patterns for jaguar are novel, with some surprising outcomes for jaguar conservation.
The first interesting result concerns the methodological approach to solve the spatial autocorrelation of our data. As a preventive strategy, we inserted artificial variables (spatial filters) to control and measure the effect of space on the potentially observed patterns. However, these variables were unnecessary in our models because they were correlated with other variables. This was a good outcome for our models because “space” is not explanatory in itself; so, the best way to control spatial autocorrelation is through utilization of true variables to reduce the variation explained by “space,” such as we did.
Regarding the biological results, we observed the inability of landscape configuration or composition variables to explain the probability of jaguar occurrence. Other studies have shown correlations between jaguar occurrence and landscape configuration or composition, but they were conducted at a patch level or with fewer landscape types and did not include prey base information (Ortega-Huerta and Medley 1999; Michalski and Peres 2007; Calaça et al. 2010). Therefore, it raises the question if configuration or composition variables can strongly correlate with prey availability, such as appears to be the case with some variables in our study. It is probable that habitat conversion reduces native prey richness and availability, as observed for species in general (Fahrig 2003; Campos et al. 2013), thereby it is indirectly and/or synergistically affecting jaguar occurrence.
Therefore, it is reasonable to assume that prey base is a surrogate of environmental quality for jaguar. Thus, jaguar could be interpreted as being a bioindicator of other important mammal species in Cerrado-Amazonia landscapes. The presence of jaguar is frequently deemed to be a suitable proxy for environmental quality due to their broad ecological requirements, which involve large home ranges (Cullen Jr. 2006; Cavalcanti and Gese 2009), native habitat (Conde et al. 2010; Colchero et al. 2011), and extensive prey base (Azevedo and Murray 2007b). Therefore, the ecological requirements of jaguar potentially overlap those of many sympatric species. This reveals an important conservation issue because some of the native prey species evaluated in this study are considered threatened (Priodontes maximus—VU; Tapirus terrestris—VU; Myrmecophaga tridactyla—VU; Blastocerus dichotomus—VU), near threatened (Tolypeutes matacus—NT; Ozotoceros bezoarticus—NT; Tayassu pecari—NT), or may be susceptible to local extinction in the future given agriculture expansion in this region (IUCN 2013).
Interestingly, the livestock variable was included in the function that explained jaguar occurrence, but did not have predictive ability for the occurrence of cattle depredation. The absence of a model able to explain cattle depredation could be due to the low statistical power (only 15 sampling units with jaguar occurrence), or the explanatory variables selected were insufficient, despite our clear predictions of the effects our variables would have on the tested patterns. The literature suggests that the common pattern for felines is higher cattle depredation rates in environments with low availability of native prey (Polisar et al. 2003; Bagchi and Mishra 2006). However, there is some evidence that non-native prey selection by jaguars is not associated with the absence of native prey in the Pantanal biome (Azevedo and Murray 2007a). Therefore, patterns of cattle depredation across different scales seem complex and remain somewhat difficult to decipher, but we suggest that cattle depredation at a meson scale is an opportunistic event related to jaguar presence and not with the native prey base or other variables tested here.
In a conservation context, strategies to guarantee long-term jaguar persistence must consider more than a simple increase in jaguar populations. It can be complicated to maintain populations of a large predator in areas associated with livestock farming within natural ecosystems due to increased human-wildlife conflict (Inskip and Zimmermann 2009). Any effort to increase jaguar populations could intensify cattle depredation and decrease regional public acceptance for conservation efforts. Public acceptance is fundamental for the conservation of jaguars—or indeed any other species—since it is human activity that in many places has led to the species’ local extinction (Altrichter et al. 2006; Morrison et al. 2007; Mazzolli 2009). In this way, the design of jaguar conservation strategies is probably dependent on our comprehension of cattle depredation patterns; so, much greater effort must be designated to understanding this topic, particularly at different scales of analysis.
References
Ahumada JA, Silva CEF, Gajapersad K et al (2011) Community structure and diversity of tropical forest mammals: data from a global camera trap network. Philos Trans R Soc Lond 366:2703–11. doi:10.1098/rstb.2011.0115
Altrichter M, Boaglio G, Perovic P (2006) The decline of jaguars Panthera onca in the Argentine Chaco. Oryx 40:302–309. doi:10.1017/S0030605306000731
Azevedo FCC (2008) Food habits and livestock depredation of sympatric jaguars and pumas in the Iguaçu National Park Area, South Brazil. Biotropica 40:494–500. doi:10.1111/j.1744-7429.2008.00404.x
Azevedo FCC, Murray DL (2007a) Evaluation of potential factors predisposing livestock to predation by jaguars. J Wildl Manag 71:2379–2386. doi:10.2193/2006-520
Azevedo FCC, Murray DL (2007b) Spatial organization and food habits of jaguars (Panthera onca) in a floodplain forest. Biol Conserv 137:391–402. doi:10.1016/j.biocon.2007.02.022
Bagchi S, Mishra C (2006) Living with large carnivores: Predation on livestock by the snow leopard (Uncia uncia). J Zool 268:217–224. doi:10.1111/j.1469-7998.2005.00030.x
Barthem RB, de Brito Ribeiro MCL, Petrere M (1991) Life strategies of some long-distance migratory catfish in relation to hydroelectric dams in the Amazon Basin. Biol Conserv 55:339–345. doi:10.1016/0006-3207(91)90037-A
Bisi J, Kurki S, Svensberg M, Liukkonen T (2007) Human dimensions of wolf (Canis lupus) conflicts in Finland. Eur J Wildl Res 53:304–314. doi:10.1007/s10344-007-0092-4
Bontemps S, Defourny P, Bogaert E Van, et al (2011) Globcover 2009. 10
Borcard D, Legendre P (2002) All-scale spatial analysis of ecological data by means of principal coordinates of neighbour matrices. Ecol Modell 153:51–68. doi:10.1016/S0304-3800(01)00501-4
Calaça A, Melo F, De Marco Junior P et al (2010) A influência da fragmentação sobre a distribuição de carnívoros em uma paisagem de cerrado. Neotrop Biol Conserv 5:31–38. doi:10.4013/nbc.2010.51.05
Campos PR, Rosas A, de Oliveira VM, Gomes MAF (2013) Effect of landscape structure on species diversity. PLoS One 8:e66495. doi:10.1371/journal.pone.0066495
Cavalcanti SMC, Gese EM (2009) Spatial ecology and social interactions of jaguars (Panthera onca) in the Southern Pantanal, Brazil. J Mammal 90:935–945. doi:10.1644/08-MAMM-A-188.1
Cavalcanti SMC, Gese EM (2010) Kill rates and predation patterns of jaguars (Panthera onca) in the southern Pantanal, Brazil. J Mammal 91:722–736. doi:10.1644/09-MAMM-A-171.1.Key
Červinka J, Šálek M, Padyšáková E, Šmilauer P (2013) The effects of local and landscape-scale habitat characteristics and prey availability on corridor use by carnivores: a comparison of two contrasting farmlands. J Nat Conserv 21:105–113. doi:10.1016/j.jnc.2012.11.004
Cesar F, Azevedo C (2003) Local perceptions of jaguars (Panthera onca) and pumas (Puma concolor) in the Iguaçu National Park area, south Brazil. Biol Conserv 111:215–221
Colchero F, Conde DA, Manterola C et al (2011) Jaguars on the move: modeling movement to mitigate fragmentation from road expansion in the Mayan Forest. Anim Conserv 14:158–166. doi:10.1111/j.1469-1795.2010.00406.x
Conde DA, Colchero F, Zarza H et al (2010) Sex matters: Modeling male and female habitat differences for jaguar conservation. Biol Conserv 143:1980–1988. doi:10.1016/j.biocon.2010.04.049
Cullen Jr. L (2006) Jaguars as landscape detectives for the conservation of Atlantic Forests in Brazil. Thesis, University of Kent
Dickman AJ, Macdonald E, Macdonald DW (2011) A review of financial instruments to pay for predator conservation and encourage human-carnivore coexistence. Proc Natl Acad Sci U S A 108:13937–13944. doi:10.1073/pnas.1012972108
Diniz-Filho JAF, Bini LM (2005) Modelling geographical patterns in species richness using eigenvector-based spatial filters. Glob Ecol Biogeogr 14:177–185. doi:10.1111/j.1466-822X.2005.00147.x
Dray S (2011) A new perspective about Moran’s coefficient : spatial autocorrelation as a linear regression problem. Geogr Anal 43:127–141
IBGE – Instituto Brasileiro de Geografia e Estatística (2007) Contagem da população 2007. 1–311
IBGE – Instituto Brasileiro de Geografia e Estatística (2008) Produção agrícola municipal: cereais, leguminosas e oleaginosas (2007). 1–56
IBGE – Instituto Brasileiro de Geografia e Estatística (2008) Produção da pecuária municipal (2007). 1–62
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515. doi:10.1146/annurev.ecolsys.34.011802.132419
Hatten JR, Averill-Murray A, van Pelt WE (2005) A spatial model of potential jaguar habitat in Arizona. J Wildl Manag 69:1024–1033. doi:10.2193/0022-541X(2005)069[1024:ASMOPJ]2.0.CO;2
Hosmer DW, Lemeshow S, Sturdivant RX (2013) Applied Logistic Regression, 3rd ed. 508
Inskip C, Zimmermann A (2009) Human-felid conflict: a review of patterns and priorities worldwide. Oryx 43:18–34. doi:10.1017/S003060530899030X
IUCN (2013) The IUCN red list of threatened species. http://www.iucnredlist.org/ . Accessed 1 Dec 2013
Legendre P, Legendre L (1998) Numerical ecology, Second ed. 870
Li X, He HS, Bu R et al (2005) The adequacy of different landscape metrics for various landscape patterns. Pattern Recogn 38:2626–2638. doi:10.1016/j.patcog.2005.05.009
Lord JM, Norton DA (1990) Scale and the spatial concept of fragmentation. Conserv Biol 4:197–202. doi:10.1111/j.1523-1739.1990.tb00109.x
Lyra-Jorge MC, Ribeiro MC, Ciocheti G et al (2010) Influence of multi-scale landscape structure on the occurrence of carnivorous mammals in a human-modified savanna, Brazil. Eur J Wildl Res 56:359–368. doi:10.1007/s10344-009-0324-x
Mazzolli M (2009) Loss of historical range of jaguars in southern Brazil. Biodivers Conserv 18:1715–1717. doi:10.1007/s10531-008-9552-8
McGarigal K, Cushman SA (2002) Comparative evalution of experimental approaches to the study of habitat fragmentation effects. Ecol Appl 12:335–345. doi:10.1890/1051-0761(2002)012[0335:CEOEAT]2.0.CO;2
McGarigal K., Cushman SA, Ene E (2012) FRAGSTATS v4: Spatial pattern analysis program for categorical and continuous maps. Computer software program available at the following web site: http://www.umass.edu/landeco/research/fragstats/fragstats.html. Accessed 2 June 2015
Michalski F, Peres CA (2007) Disturbance-mediated mammal persistence and abundance-area relationships in Amazonian forest fragments. Conserv Biol 21:1626–1640. doi:10.1111/j.1523-1739.2007.00797.x
Moran PAP (1950) Notes on continuous stochastic phenomena. Biometrika 37:17–23
Morrison JC, Sechrest W, Dinerstein E et al (2007) Persistence of large mammal faunas as indicators of global human impacts. J Mammal 88:1363–1380. doi:10.1644/06-MAMM-A-124R2.1
Mortelliti A, Amori G, Capizzi D et al (2010) Experimental design and taxonomic scope of fragmentation studies on European mammals: Current status and future priorities. Mamm Rev 40:125–154. doi:10.1111/j.1365-2907.2009.00157.x
O’Brien TG, Kinnaird MF, Wibisono HT (2003) Crouching tigers, hidden prey: Sumatran tiger and prey populations in a tropical forest landscape. Anim Conserv 6:131–139. doi:10.1017/S1367943003003172
Ortega-Huerta MA, Medley KE (1999) Landscape analysis of jaguar (Panthera onca) habitat using sighting records in the Sierra de Tamaulipas, Mexico. Environ Conserv 26:257–269
Patuelli R, Griffith DA, Tiefelsdorf M, Nijkamp P (2010) Spatial filtering and eigenvector stability: space-time models for German unemployment data. Int Reg Sci Rev 34:253–280. doi:10.1177/0160017610386482
Petracca LA, Hernández-Potosme S et al (2014a) Agricultural encroachment and lack of enforcement threaten connectivity of range-wide jaguar (Panthera onca) corridor. J Nat Conserv 22:436–444. doi:10.1016/j.jnc.2014.04.002
Petracca LS, Ramíres-Bravo O, Hernández-Santín L (2014b) Occupancy estimation of jaguar Panthera onca to assess the value of east-central Mexico as a jaguar corridor. Oryx 48:133–140. doi:10.1017/S0030605313000069
Polisar J, Maxit I, Scognamillo D et al (2003) Jaguars, pumas, their prey base, and cattle ranching: Ecological interpretations of a management problem. Biol Conserv 109:297–310. doi:10.1016/S0006-3207(02)00157-X
Rangel TF, Diniz-Filho JAF, Bini LM (2010) SAM: a comprehensive application for Spatial Analysis in Macroecology. Ecography 33:46–50. doi:10.1111/j.1600-0587.2009.06299.x (Cop)
Riitters KH, Oneill RV, Hunsaker CT et al (1995) A factor-analysis of landscape pattern and structure metrics. Landsc Ecol 10:23–39
Ripple WJ, Estes JA, Beschta RL et al (2014) Status and ecological effects of the world’s largest carnivores. Science 343:1241484–1241484. doi:10.1126/science.1241484, 80-
Ritters K, Wickham J, O’Neill R, et al (2000) Global-scale patterns of forest fragmentation. Conserv. Ecol. 4: http://www.ecologyandsociety.org/vol4/iss2/art3/. Accessed 2 June 2015
Ryall KL, Fahrig L (2006) Response of predators to loss and fragmentation of prey habitat: a review of theory. Ecology 87:1086–1093
Sokal RR, Oden NL (1978) Spatial autocorrelation in biology. Biol J Linn Soc 10:199–228
Tews J, Brose U, Grimm V et al (2004) Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. J Biogeogr 31:79–92. doi:10.1046/j.0305-0270.2003.00994.x
Tôrres NM, De Marco P, Santos T et al (2012) Can species distribution modelling provide estimates of population densities? A case study with jaguars in the Neotropics. Divers Distrib 18:615–627. doi:10.1111/j.1472-4642.2012.00892.x
Villard M-A, Metzger JP (2014) Beyond the fragmentation debate: a conceptual model to predict when habitat configuration really matters. J Appl Ecol 51:309–318. doi:10.1111/1365-2664.12190
Wagner HH, Fortin M-J (2005) Spatial analysis of landscapes: concepts and statistics. Ecology 86:1975–1987
White PCL, Lowe P (2008) Wild mammals and the human food chain. Mamm Rev 38:117–122. doi:10.1111/j.1365-2907.2008.00121.x
Wong W-M, Linkie M (2013) Managing sun bears in a changing tropical landscape. Divers Distrib 19:700–709. doi:10.1111/ddi.12020
Woodroffe R, Ginsberg JJR (1998) Edge effects and the extinction of populations inside protected areas. Science 280:2126–2128. doi:10.1126/science.280.5372.2126, 80-
Zanin M, Palomares F, Brito D (2014) What we (don’t) know about the effects of habitat loss and fragmentation on felids. Oryx 49:96–106. doi:10.1017/S0030605313001609
Zanin M, Palomares F, Brito D (2015) The jaguar’s patches: viability of jaguar populations in fragmented landscapes. J Nat Conserv 23:90–97. doi:10.1016/j.jnc.2014.06.003
Zeilhofer P, Cezar A, Torres NM et al (2014) Jaguar Panthera onca habitat modeling in landscapes facing high land-use transformation pressure — findings from Mato Grosso, Brazil. Biotropica 46:98–105
Zeller K (2007) Jaguars in the new millennium data set update: the state of the jaguar in 2006. 82
Zeller KA, Nijhawan S et al (2011) Integrating occupancy modeling and interview data for corridor identification: a case study for jaguar in Nicaragua. Biol Conserv 144:892–901. doi:10.1016/j.biocon.2010.12.003
Zimmermann A, Walpole MJ, Leader-Williams N (2005) Cattle ranchers’ attitudes to conflicts with jaguar Panthera onca in the Pantanal of Brazil. Oryx 39:406. doi:10.1017/S0030605305000992
Acknowledgments
We wish to thank Dr. Daniel Brito and Dr. Francisco Palomares for their valued contributions to this manuscript.
Compliance with ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Acevedo
Rights and permissions
About this article
Cite this article
Zanin, M., Sollmann, R., Tôrres, N.M. et al. Landscapes attributes and their consequences on jaguar Panthera onca and cattle depredation occurrence. Eur J Wildl Res 61, 529–537 (2015). https://doi.org/10.1007/s10344-015-0924-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10344-015-0924-6