Abstract
Terminal heat stress caused by a late cultivation date, is one of the most important factors limiting the crop yield. This study was conducted to investigate the ameliorative effects of paclobutrazol (PBZ) on growth, physiological, and photosynthetic pigments in three wheat cultivars under various planting dates. This experiment was conducted in a split-factorial arrangement based on a randomized completely block design (RCBD) with three replicates over two years (2020–21 and 2021–22). Planting date was determined as main plots (including November 16, December 11, and January 5). Three wheat cultivars (Karim, Mehrgan, and Chamran-2) and application of PBZ at three concentrations (0 mg L−1, 60 mg L−1, and 120 mg L−1) were also established as two subplots. Our results revealed although late cultivation reduced grain yield, PBZ foliar spraying (120 mg L−1) improved grain yield of Chamran‑2 by 36%, Mehrgan by 32%, and Karim by 20%. Moreover, a decreasing trend in stem length was observed with increased PBZ concentration, which may be attributed to the ability of this growth modifier to remobilize assimilates on grains rather than on stems. Moreover, maximum specific weight, and assimilate remobilization increased with the application of PBZ. The reducing effect of terminal heat stress on photosynthetic pigments was also alleviated. Overall, our results showed that Chamran‑2 outperformed others especially in heat stress conditions. It can be concluded that PBZ foliar application can be an effective way to combat adverse effects of late cultivation wheat.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Abiotic stresses always increase the risks associated with sustainable food production. All over the world crop plants are subjected to various abiotic stresses (El-Bially et al. 2018; Saudy et al. 2020a, 2023a; Abd El-Mageed et al. 2022; El-Metwally et al. 2022a). Hence, reduction in crop growth, yield and quality associated the environmental stresses (El-Metwally and Saudy 2021; Salem et al. 2021; Abd-Elrahman et al. 2022; Saudy et al. 2023b). one of the most significant stress affecting crop productivity if the heat stress, especially in arid zones (El-Bially et al. 2022a; Saudy et al. 2020b). In this concern, global crop production is threatened by high-temperature stress, primarily at the reproductive phase (Nagar et al. 2021). According to Zhao et al. (2017), every 1 °C increase in global mean temperatures will reduce wheat yields by 6.0%, rice yields by 3.2%, maize yields by 7.4%, and soybean yields by 3.1%. Wheat (Triticum aestivum L.) is an essential crop for human consumption growing during winter. Various weather conditions within seasons devastate yield potential (Murungu and Madanzi 2010; Gandjaeva 2019). A major wheat production region in Iran is Khuzestan province. While heat stress for late sowing dates in the mentioned area limits crop productivity. One method for assisting a plant avoid poor environmental conditions is to alter the cropping pattern in that area (Mombeini et al. 2014). Sowing at the right time to coincide with the cultivar maturity length and the growing season is essential for yield optimization (Andarzian et al. 2008, 2015; Mousavi et al. 2021). Inadequate rainfall and high temperatures during grain filling at the end of the season are two significant factors limiting wheat grain production in this region.
It is crucial to consider the appropriate sowing date when managing grain yield in such an environment (Sun et al. 2013; Andarzian et al. 2015; Mousavi et al. 2021), as it affects the crop growth, development, germination percentage, and seedlings establishment (Gandjaeva 2019). Shirinzadeh et al. (2017) stated that winter wheat yield increases with proper sowing dates. It has been reported that a two-week delay in sowing date significantly reduces cereal grain yield by 15%, while a four-week delay in sowing reduces crop yield by 30% (Podolska and Wyzińska 2011). Based on the weather conditions and cultivation technology level during the vegetation period, Oleksiak (2014) found that delayed sowing can negatively impact yield.
In the context of current and future climate change scenarios, terminal heat stress in wheat poses a challenge to the scientific community (Nagar et al. 2021). During all phases of wheat’s growth, the abnormal delay in sowing exposes it to unsuitable photothermal regimes (Nagar et al. 2021). As a result of terminal heat stress during anthesis and grain filling, flowers are aborted, pollen viability is reduced, photosynthates are transported to the developing kernel, and starch synthesis and deposition occur within the kernel. This results in a reduction in grain quality, grain number, and grain weight (Farooq et al. 2011; Reynolds et al. 2012). Additionally, elevated temperatures increase the availability of free electrons in chloroplasts and mitochondria (Modarresi et al. 2010), thereby generating reactive oxygen species (ROS) (O −2, H2O2, OH−, •OH) (Caverzan et al. 2016; Ghadirnezhad Shiade et al. 2022a). Essentially, ROS damage lipids, nucleic acids, and proteins, reducing the plant’s ability to perform physiological processes (Mathur and Jajoo 2013; Zhao et al. 2023). Further, chlorophyll pigment degradation leads to significant reductions in photosynthesis, reduced activity of rubisco and rubisco activase, and structural damage to the thylakoids and oxygen-evolving complex of photosystem II under stressed conditions (Farooq et al. 2011).
Another effective agricultural strategy that has gained plenty of attention is the application of environmental stress modifiers. Several chemical compounds have been exploited as alleviators to cope the environmental stress impacts (El-Metwally et al. 2021; Mubarak et al. 2021; El-Bially et al. 2022b; Salem et al. 2022). A major physiological effect of plant growth regulators is their ability to increase plant tolerance to environmental stress (Desta and Amare 2021; El-Metwally et al. 2022b). In the plant, paclobutrazol (PBZ), a triazole derivative, inhibits kaurene oxidase and blocks the oxidation of kaurene into kaurenoic acid (Nagar et al. 2021). In addition, this compound can increase resistance to cold, heat, drought, and salt in various plants (Nagar et al. 2021). The triazole-mediated stress protection is attributed to changes in plant hormones such as increased cytokinins, abscisic acid, and ethylene levels (Soumya et al. 2017). Besides reducing electrolyte leakage and maintaining relative water content, it enhances the levels of osmolytes, antioxidants, and endogenous hormones in plants so that they can adapt to stress. As a result, photosynthetic activity contributes to plant growth and yield (Desta and Amare 2021). Furthermore, PBZ application increases stress tolerance, promotes tillering, and enhance spike numbers and yields (Peng et al. 2014; Dwivedi et al. 2017), and grain yield. This activity of PBZ could be attributed to enhancing osmolytes, endogenous hormones, and antioxidant activities under adverse environmental conditions (Soumya et al. 2017; Kamran et al. 2018), as well as maintaining physiological function and root activity.
Although there are many studies investigating the use of PBZ to increase crop yields, few studies have focused on how the combination of planting date and PBZ application can result in higher wheat yields by improving remobilization potential and terminal heat tolerance. Hence, it was hypothesized that a strategic combination of planting date and PBZ application will positively influence wheat crops, resulting in higher yields. In other words, the hypothesis suggests that the appropriate timing of planting and PBZ treatment will lead to enhanced growth indicators, improved physiological responses, increased photosynthetic pigment concentrations in wheat cultivars, superior remobilization potential, and better tolerance to terminal heat stress conditions, ultimately contributing to higher wheat yields. This study has two key aims, firstly, to explore the combined impact of planting date and PBZ application on wheat cultivars, with a specific focus on assessing their effects on growth indicators, physiological characteristics, and photosynthetic pigments. Secondly, to address the existing research gap by investigating how the interaction of planting date and PBZ can lead to enhanced wheat yields through improved remobilization potential and increased tolerance to terminal heat stress conditions.
Material and Methods
Experimental Design
This experiment was conducted in a split-factorial arrangement based on a completely randomized design (CRD) with three replicates over two years (2020–21 and 2021–22). Planting date at 25-day intervals was laid out as main plots (including November 16, December 11, and January 5). Three wheat cultivars (Karim, Mehrgan, and Chamran-2) and application of PBZ at three concentrations (0 mg L−1, 60 mg L−1, and 120 mg L−1) were also established as two subplots.
Growth Conditions and Plant Material
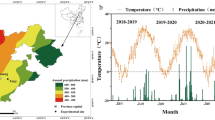
The field soil was deeply plowed and leveled with two vertical disks last fall. Then, the plots were laid out so that, 10 planting rows were established in each plot (with a length of 4 m) with a spacing of 20 cm between seedlings. The planting density was set at 400 plants m−2. According to the physicochemical characteristics of the soil test in the study area (Table 1), phosphorus (270 kg ha−1 with triple superphosphate, corresponding to 124.2 kg ha−1 P2O5) and potassium (230 kg ha−1 with potassium sulfate, corresponding to 115 kg ha−1 k2O) were applied as starting fertilizers. In addition, urea fertilizer (360 kg ha−1 corresponding to 165.6 kg ha−1 nitrogen) was applied at three growth stages (after seedling emergence, at mid-tillage, and at the beginning of flowering) as a top dressing. The study area was located in Nitrogen village, Izeh district, Khuzestan province, Iran (31° 52′ North, 49° 45′ East, with an elevation of 835 m above sea level). The climatic data of the study area were presented (Fig. 1). Three wheat cultivars (Karim, Mehrgan, and Chamran2) were provided from Khuzestan agricultural and natural resources research center. The plants were foliar sprayed at desired levels (0, 60, and 120 mg L−1) at early stem elongation stage. The control plants were exposed with distilled water at the same concentrations.
Measurements
At milk development phase on approximately 90 days after sowing (DAS), three plants were randomly selected from each plot at different treatment levels and their flag leaves were separated. Then, the samples were quickly frozen in liquid nitrogen and transferred to the laboratory to measure the physiological traits. Chlorophyll (chl a and chl b), total chlorophyll and carotenoids content were evaluated in accordance with (Ghadirnezhad Shiade et al. 2022b). A sample of the leaf (0.3 g) was mixed well with 8 mL of methanol and allowed to stand in the dark at room temperature for 48 h. Then the solution was extracted, and its absorbance was measured using a spectrophotometer at a wavelength of 665, 652 and 470 nm, respectively. Then, dried sample were used to calculate maximum stem specific weight (MSSW), dry matter remobilization (DMT), and remobilization efficiency (RE) according to Eqs. 1, 2 and 3 modified by (Vosoghi Rad et al. 2022);
Where Wmax is the maximum stem weight, L is stem length; Wmat refers to plant weight at physiological maturity, and RE (%) shows the percentage of plant remobilization efficiency.
At harvesting stage, when the plants were physiologically matured (120 DAS), samples were taken from each plot. The harvest index (HI) was calculated by dividing the grain yield by the biological yield. To determine the Thousand-grain weight (TGW), two samples of 500 seeds from each plot were counted and measured after 48 h in the oven at 75 °C. After making sure that the standard deviation was less than 4%, its accuracy was confirmed, and its total was reported as the TGW. Then the grain yield was corrected based on 14% moisture (in kg ha−1). Stem length was measured using ruler.
Data Analysis
The Shapiro-Wilk test was used to analyze the normality of the data. According to the results of Bartlett’s test, the variances of the years were not significant; therefore, a composite analysis was used (Sobral et al. 2019). In addition, the means of the data were compared using the LSD test. All data were analyzed using SAS software (version 9.1).
Results
Photosynthetic Pigments
Chlorophyll a
According to analysis of variance results, the triple effects planting date × cultivar × PBZ application considerably affected Chlorophyll a (Table 2). The highest chlorophyll a content was observed on the planting date of November in the Chamran‑2 cultivar and with 120 mg L−1 PBZ (2.07 mg g−1) (Table 3). In contrast, the lowest content was recorded in the Karim cultivar (0.73 mg g−1) under the late cultivation date with no PBZ application. In all three planting dates, this photosynthetic content increased at higher PBZ consumption (Table 3). The amount of chlorophyll a on the delayed planting date, at the level of PBZ consumption of 60 to 120 mg L−1, was higher in Chamran‑2 than the other two Mehregan and Karim cultivars (Table 3).
Chlorophyll b
Based on the analysis of variance results, planting date, cultivar, and PBZ application significantly influenced Chlorophyll b (Table 2). Chlorophyll b levels were highest on the planting date of November when the Chamran‑2 cultivar was treated with 60 mgL−1 PBZ (0.71 mg g−1) (Table 3). The Karim cultivar had the lowest chlorophyll b content (0.29 mg g−1) when not treated with PBZ on the planting date of January (Table 3). Chlorophyll b levels increased in all three planting dates with PBZ consumption of 60 to 120 mg L−1 (Table 3). At delayed planting date, Chamran‑2 cultivar performed better than others in Chlorophyll b content (Table 3).
Total Chlorophyll
The triple effect of planting date, cultivar, and PBZ application greatly affected total chlorophyll (Table 2). Total chlorophyll levels were highest (2.7 mg g−1) in the Chamran‑2 cultivar in November when 120 mg L−1 PBZ was applied (Table 3). On the other hand, the Karim cultivar also had the lowest total chlorophyll content (1 mg g−1) when not using PBZ on the late planting date in January (Table 2). Chamran 2 cultivar had more significant total chlorophyll under PBZ application conditions in all three planting dates (Table 3). The total chlorophyll content differed significantly between cultivars at the 60 to 120 mg L−1 of PBZ treatment on the delayed planting date. Based on the results, total chlorophyll increased with the use of PBZ (Table 3).
Carotenoids
Carotenoids was greatly affected by the triple effect of planting date, cultivar, and PBZ application, according to analysis of variance results (Table 2). On the November 16 planting date in the Mehrgan cultivar, the highest carotenoids level were observed (1.48 mg g−1) with 120 mg L−1 PBZ application. However, the highest value was observed for the Karim cultivar in January 5 cultivation without PBZ treatment (0.52 mg g−1) (Table 3). The results revealed that at higher PBZ concentrations, carotenoids content enhanced on the November 16 planting date. In addition, this pigment content increased with the consumption of 60 mg L−1 for the Karim cultivar and the planting date of December 11. In contrast, the carotenoids level decreased with the higher PBZ consumption (120 mg L−1 PBZ). In general, carotenoids level increased with PBZ consumption on the delayed planting date in all cultivars (Table 3).
Morphological and Traits Related to Yield
Analysis of variance results for the studied parameters of wheat plants treated with year × planting date × cultivars × PBZ were presented in Table 4.
TGW was influenced by the quadruple effect of year × planting date × cultivar × PBZ. The results showed that in both years, the highest amount of TGW was observed on the planting date of November 16 in the Karim cultivar at the treatment level of 120 mg L−1 PBZ (46.34 and 46.23 g) (Table 5). The lowest amount of TGW in the first year was also observed in the Chamran‑2 cultivar (35.73 g) under the conditions of consumption of 60 and 120 mg L−1 PBZ on January 5 planting date (Table 5). On the delayed planting date, a statistically significant difference was observed at the consumption level of 120 mg L−1 of PBZ between Chamran‑2 and Mehrgan cultivars (Table 5). The results showed that in the first year with a delay in planting in Chamran‑2 and Mehrgan cultivars with different levels of PBZ TGW, although there was a significant difference, but compared to the November 16 planting date, TGW was much lower. But in the second year of TGW on the delayed planting date and consumption of 60 mg L−1 in Mehrgan cultivar, the lowest TGW of 35 g was obtained (Table 5).
Grain yield was influenced by the triple effect of planting date × cultivar × PBZ. The highest grain yield was obtained with Chamran‑2 cultivar planted in November 16 and December 11 that treated with 60 mg L−1 PBZ (6940.58 and 6779.47 kg ha−1, respectively). On the other hand, the cultivar Mehrgan, planted in January 5 (late planting date) without PBZ application had the lowest grain yield (3829.93 kg ha−1) (Table 6). At all planting dates, Karim cultivar recorded lower grain yield compared to the other two cultivars. Although no significant difference was observed between Chamran‑2 and Mehrgan at late planting date along with 120 mg L−1 PBZ application, Chamran‑2 performed better than another cultivar (5844.01 kg ha−1).
HI was influenced by the quadruple effect of year × planting date × cultivar × PBZ. The results showed that in the first year on the planting date of November 16, the Mehrgan cultivar obtained the highest HI (39.59%) at the level of 120 mg L−1 paclobetrazol application. After that, Chamran‑2 cultivar was obtained at the level of 120 mg L−1 paclobetrazol application (38.46%). In the second year, on the planting date of December 11, at the level of 60 mg L−1 paclobetrazol application, the highest HI (37.96%) was obtained. After that, on the planting date of November 16, Karim cultivar at the level of 120 mg L−1 PBZ application (37.84%) obtained the highest HI. In the first and second year, on the planting date of January 5, the Chamran‑2 cultivar had the lowest HI in the absence of paclobetrazol application (Table 5).
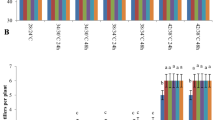
Furthermore, application of PBZ at the highest concentration at the same planting date improved grain yield of Chamran‑2 by 36%, Mehrgan by 32%, and Karim by 20%. Moreover, stem length was affected by double interaction of cultivar × PBZ (Table 4). The highest stem length was recorded by Mehrgan cultivar with no PBZ application (86.23 cm), which increased by 21% compared with Chamran‑2 treated with PBZ at 120 mg L−1 (Fig. 2). In addition, there was a decreasing trend in stem length by increased PBZ concentration. Chamran‑2 exhibited the strongest decreasing trend of stem length compared to the other cultivars (with a slope of 0.088, R2: 0.99%).
Remobilization Traits
Analysis of variance results revealed that the maximum stem specific weight was significantly affected by the triple effects planting date × cultivar × PBZ. In contrast, year had no effect on this parameter (Table 4). Karim cultivar treated with 60 mg L−1 PBZ cultivated in December 11, recorded the highest maximum stem specific weight (43.02 mg cm−1) which was 95% higher than that of the late-grown cultivar Chamran‑2 (Table 6). At all planting dates, the application of PBZ to the Karim cultivar improved maximum specific stem weight more than other cultivars. Whereas, at normal cultivation date (November 16), Chamran‑2 increased this parameter by 18% under 120 mg L−1 PBZ compared with control. Karim and Mehrgan also recorded increases of 23.4 and 7.5%, respectively, in the same situation. Furthermore, when plants were cultivated in December 11 and treated with 120 mg L−1 PBZ, maximum specific stem weight improved by 21%, 17.5%, and 15% for Chamran‑2, Mehrgan, and karim, respectively.
Dry matter remobilization was also influenced by the triple effect of planting date × cultivar × PBZ. In contrast, year had no effect on this parameter (Table 4). The highest value for dry matter remobilization was observed at the two planting dates in November and December in the cultivar Karim without PBZ treatment (1.57 g and 1.47 g, respectively) (Table 6). On the other hand, the lowest value of this trait was observed in Chamran‑2 cultivar (0.69 g) without PBZ application in December 11. The results showed that there was no significant difference between the application and non-application of PBZ in Chamran‑2 cultivar at the late planting date (January 5). At all three planting dates, the Karim cultivar had faster dry matter remobilization than the other two cultivars, either with PBZ application or not (Table 6). No statistically significant difference was observed between the two cultivars at the delayed planting date of Chamran‑2 and Mehrgan under PBZ consumption of 120 mg L−1, but the dry matter remobilization of Mehrgan was higher than that of Chamran‑2 (Table 6).
Dry matter remobilization efficiency was affected by the triple effects planting date × cultivar × PBZ (Table 4). Year had no effect on this parameter. There was a decreasing trend response with increased PBZ concentration for all cultivars at three cultivation dates (Figs. 3, 4 and 5). Although, a linear regression was found in November cultivation (Fig. 3), both December 11 and January 5 planting dates exhibited quadratic regulation (Figs. 4 and 5). In November 16 cultivation, Chamran‑2 (slope: −0.038, R2: 0.86%) responded more slowly than others with 1.5 and 10% reduction at 60 and 120 mg L−1 PBZ concentrations compared with control. By contrast, the dry matter remobilization efficiency of the cultivar Karim decreased faster (slope: −0.108, R2: 0.95%) by 18 and 27% at the same treatments. Moreover, in the both middle and delay cultivations (December 11 and January 5), cultivars behaved differently. Interestingly, R2 = 1 was obtained for all cultivars and treatments. At both cultivation dates (December 11 and January 5), Chamran‑2 exhibited slower response (slope: 0.0002, 0.0007, respectively) to dry matter remobilization efficiency. The cultivar Karim, on the other hand, responded faster and its dry matter remobilization efficiency decreased more than the other two cultivars in the same treatments (slope: 0.0004). Accordingly, its remobilization efficiency reduced by 16.5 and 26.5% in December, and 9.4 and 11.0% in January at PBZ concentrations of 60 and 120 mg L−1, respectively.
Discussion
This study was conducted to investigate ameliorating effects of PBZ for wheat cultivars planted on various planting dates (November 16, December 11, and January 5).
With the increase in temperature in the final stages of plant growth, the plant growth cycle is disrupted, the plant is unable to use resources properly, and the plant performance decreases. The delay in the planting date and, as a result, the sudden increase in temperature with a negative effect on the reproductive organs and prevention of proper fertility will reduce the TGW and ultimately affect the seed yield. Greening of seedlings is done earlier on the appropriate planting date, and TGW and yield are also higher on the appropriate planting date (Saudy et al. 2021). Mojtabaie Zamani et al. (2015), Modhej et al. (2015), and Al-Otayk (2010) also achieved similar results in their research. A delay in the planting date leads to a decrease in seed production. Still, the reason for this is not precisely known because it may be due to the reduction in grain size, lack of seed formation, disruption of pollination, or post-pollination processes (Modhej et al. 2015; Mojtabaie Zamani et al. 2015). By delaying the planting and encountering the final stages of growth with the heat stress at the end of the season, the plant’s ripening is accelerated, and the period of seed filling and the length of the gestation period until the beginning of seeding are reduced. As a result, it causes the production of seeds with less weight (smaller) and the reduction of TGW. Using PBZ as a growth regulator has a significant role in the TGW of wheat. PBZ strongly increased the length of the growth period, the plant’s phenological stages, and especially the length of the seed-filling period, which caused an increase in TGW. In this regard, the researchers stated that PBZ increases the leaves’ lifespan and, subsequently, the photosynthesizing surface’s durability in the post-pollination stage. Also, increasing the length of the grain filling period increased the production of necessary materials for economic performance, and thus the weight of the seeds also increased (Berova et al. 2002; Nouriyani 2017).
The results revealed that although late sowing date decreased grain yield, PBZ application mitigated the adverse effects of terminal heat stress. The results of this study are consistent with the findings of (Pradhan et al. 2010; Mousavi et al. 2021). Similarly, Peng et al. (2014) found that wheat plants exposed to PBZ at 150 mg L−1 exhibited improved early tillers, panicles number, and grain yield. Elevated air temperatures and rapid growth and development processes can be responsible for the lower grain yield at the late cultivation date (January 5). Moreover, in late planting, low soil temperature and the frostbite of the developed terminal meristem possibility lead to the removal of fertile tillers, consequently, decreased the yield (Nazeri 2017; Yousefi et al. 2018). On the other hand, normal sowing results in a longer vegetative growth period, which allows plants to establish themselves in the soil and build a suitable structure before the cold season begins.
The HI is obtained by dividing the grain yield by the biological yield. Therefore, the higher HI in the first year was considered to increase the seed yield to biological yield due to the allocation of more photosynthetic materials to the seed. Also, the positive role of the use of PBZ on the improvement of the HI was effective in this increase. The delay in planting caused a decrease in vegetative and reproductive growth. However, reproductive growth facing heat stress due to a sharp decrease during the phenological stages after flowering and a decrease in seed weight caused a sharp drop in seed yield, which eventually reduced the HI (Mousavi et al. 2021). Treating wheat cultivars with PBZ increases the leaf surface index and durability strengthens the ability to receive light and carry out photosynthesis in the plant and improves grain yield in these conditions (Nouriyani 2017). Increasing the PBZ levels by improving the photosynthesizing level has prolonged the seed-filling period, which increases the seed yield and HI. Researchers also reported similar results (Baylis and Hutley-Bull 1991; Kraus et al. 1991).
As for stem length, our results revealed that all three cultivars responded to increased application of PBZ with decreasing tendency. The reduced plant growth, especially the stem length, can be attributed to the negative effects of heat stress, the reduction of photosynthesis and consequently the lack of carbohydrates for plant growth. On the other hand, this can be considered as a compensatory response of the plant. That is, under these conditions, the plants no longer use the carbohydrates for longitudinal cell growth but keep them soluble in the cells or transfer them to the grains, increasing the remobilization efficiency. Our results are in agreement with those of Zhao et al. (2023) who reported reduced plant height in peanut. Moreover, the different responses of cultivars to higher concentrations of this growth modifier can be explained by their different genetic structure and diversity. Our results showed that Chamran‑2 exhibited the strongest downward trend, while Karim showed the least. In other words, it can be argued that Karim cultivar is more sensitive to the heat stress caused by the late cultivation date. As this cultivar is slow to respond to the ameliorating effect of PBZ and remobilize photosynthates in the grains. The differences in genetic markup of crop genotypes associated with different yield potential (Saudy et al. 2018, 2020c, 2022).
Our results exhibited that PBZ application improved maximum stem specific weight, particularly at the late planting date. It seems that the positive effect of paclobutraozal is on the expansion and durability of the leaf surface and its effect on photosynthetic pigments, and as a result, increasing the photosynthetic capacity. These can be among the reasons for the accumulation of more carbohydrates in the stem and the increase in maximum stem specific weight in this study. It has been shown that PBZ improves the senescence process of leaves and increases chlorophyll, which decreases plant height while increasing dry matter accumulation in the stem and roots (Pan et al. 2013). Furthermore, the increased specific weight under terminal drought stress conditions can be attributed to the enhanced maximum dry matter accumulation relative to their length (Vosoghi Rad et al. 2022).
Dry matter remobilization exhibited an increasing trend by PBZ foliar spray. During terminal heat stress, the process of assimilative remobilization is relatively important as a supporting mechanism because of the relative limitation of assimilate synthesis in the photosynthetic process. This phenomenon may compensate yield decline caused by late heat (Vosoghi Rad et al. 2022). Wheat grain yield is the final stage of photosynthetic production and consumption. Thus, its level depends on the sink-source balance, which may be limited by either factor. Maintaining flag leaf photosynthesis under environmental stress conditions increases seed yield due to the strong correlation between grain yield and flag leaf photosynthesis (Nouriyani 2015). Spraying of PBZ has been reported to increase photosynthesis and assimilate redistribution rate in wheat (Nouriyani 2015). The results show that Karim cultivar has a higher share in dry matter remobilization than the other two cultivars. It seems that genetics plays a big role in dry matter remobilization trait. For this reason, the use of PBZ did not have much effect on this trait. In this regard, the researchers reported that climatic factors mostly affect the grain filling period. This is while the seed filling speed is determined by the genetic structure of the plant and by its physiological and anatomical characteristics, including photosynthetic potential, redistribution of reserve materials, and also the efficiency of the vascular system is controlled (Nouriyani 2015).
Regarding photosynthetic pigments, the application of PBZ increased the content of chlorophyll a, chlorophyll b, total chlorophyll, and carotenoids, especially at delayed cultivation date. Moreover, the concentration of these pigments was higher in November 16 sowing than in others. A high-temperature effect on the photosystem electron transport chain reduces the efficacy of rubisco and rubisco activation, thereby reducing the rate of photosynthesis (Nagar et al. 2021). In the study of Modhej et al. (2011), the delay in planting caused a significant decrease in the amount of chlorophyll in the flag leaf. In this research, the chlorophyll of the flag leaves decreased by 10% in delayed cultivation compared to the cultivation in the suitable planting date of the region. A decrease in chlorophyll can be due to the reduction in chlorophyll synthesis or an increase in its decomposition (Razavizadeh et al. 2013). The delay in planting and the occurrence of end-of-season stresses increase the production of ROS, which leads to the peroxidation of membrane lipids, chloroplast destruction, and the breakdown of pigments, and finally to the reduction of photosynthesis (Modhej et al. 2011).
The ameliorative effect of PBZ can be explained by the fact that it directs photosynthetic products to the sinks, and increases leaf area index and leaf surface durability, resulting in improved light reception and strengthening of the plant photosynthetic system (Moneruzzam et al. 2020). Similarly, Zhao et al. (2022) found that PBZ improved light transmission and relative chlorophyll content. Furthermore, Abd and Abdel-Rrazik (2015) noted that chlorophyll a and b content decreased at both early and late cultivation dates of wheat plants. Other researchers have also reported increased chlorophyll and carotenoid levels in plants using PBZ in non-stressed and stressed environments (Razavizadeh et al. 2013; Rahimi et al. 2023). The increase in the concentration of chlorophyll in the leaves of plants treated with PBZ is related to the increase in the concentration of chlorophyll in each chloroplast, the increase in the number of chloroplasts in each leaf cell (Razavizadeh et al. 2013).
Conclusion
This study was conducted to assess the effects of planting date and PBZ on growth indicators, physiological and photosynthetic pigments in wheat cultivars. Our results revealed that terminal heat stress caused by a late planting date (January) reduced grain yield, stem length, stem specific weight, remobilization efficiency, and photosynthetic pigments. In contrast, foliar spraying with PBZ mitigated these negative effects. Since heat stress can restrict assimilate synthesis in the photosynthetic process, remobilization may be of great importance as a supporting mechanism to compensate for the reduced yield, although there are limitations since dry matter accumulation and remobilization require a high amount of energy. Therefore, it is recommended that future studies focus on investigating alternative options, such as determining the appropriate planting date and using PBZ as a growth regulator for further photosynthetic processes and traits. The important effect of production factors such as the appropriate planting date and the use of PBZ growth regulator should be considered in order to make more use of the photosynthesis process and increase its contribution to yield.
References
Abd MMEHS, Abdel-Rrazik AFA (2015) Effects of paclobutrazol on mitigation of temperature stress induced by manipulation of sowing date in wheat plant. Egypt J Exp Biol 9:125
Abd El-Mageed TA, Mekdad AAA, Rady MOA, Abdelbaky AS, Saudy HS, Shaaban A (2022) Physio-biochemical and agronomic changes of two sugar beet cultivars grown in saline soil as influenced by potassium fertilizer. J Soil Sci Plant Nutr 22:3636–3654. https://doi.org/10.1007/s42729-022-00916-7
Abd-Elrahman SH, Saudy HS, Abd El-Fattah DA, Hashem FA (2022) Effect of irrigation water and organic fertilizer on reducing nitrate accumulation and boosting lettuce productivity. J Soil Sci Plant Nutr 22:2144–2155. https://doi.org/10.1007/s42729-022-00799-8
Al-Otayk SM (2010) Performance of yield and stability of wheat genotypes under high stress environments of the central region of Saudi Arabia. Jkau Met Env Arid L Agric Sci 21:81–92
Andarzian B, Bakhshandeh AM, Bannayan M et al (2008) Wheatpot: a simple model for spring wheat yield potential using monthly weather data. Biosyst Eng 99:487–495. https://doi.org/10.1016/j.biosystemseng.2007.12.008
Andarzian B, Hoogenboom G, Bannayan M et al (2015) Determining optimum sowing date of wheat using CSM-CERES-Wheat model. J Saudi Soc Agric Sci 14:189–199. https://doi.org/10.1016/j.jssas.2014.04.004
Baylis AD, Hutley-Bull PD (1991) The effects of a paclobutrazol-based growth regulator on the yield, quality and ease of management of oilseed rape. Ann Appl Biol 118:445–452. https://doi.org/10.1111/j.1744-7348.1991.tb05645.x
Berova M, Zlatev Z, Stoeva N (2002) Effect of paclobutrazol on wheat seedlings under low temperature stress. Bulg J Plant Physiol 28:75–84
Caverzan A, Casassola A, Brammer SP (2016) Antioxidant responses of wheat plants under stress. Genet Mol Biol 39:1–6. https://doi.org/10.1590/1678-4685-GMB-2015-0109
Desta B, Amare G (2021) Paclobutrazol as a plant growth regulator. Chem Biol Technol Agric 8:1–15
Dwivedi SK, Arora A, Kumar S (2017) Paclobutrazol-induced alleviation of water-deficit damage in relation to photosynthetic characteristics and expression of stress markers in contrasting wheat genotypes. Photosynt 55:351–359. https://doi.org/10.1007/s11099-016-0652-5
El-Bially MA, Saudy HS, El-Metwally IM, Shahin MG (2018) Efficacy of ascorbic acid as a cofactor for alleviating water deficit impacts and enhancing sunflower yield and irrigation water–use efficiency. Agric Wat Manag 208:132–139. https://doi.org/10.1016/j.agwat.2018.06.016
El-Bially MA, Saudy HS, El-Metwally IM, Shahin MG (2022a) Sunflower response to application of L–ascorbate under thermal stress associated with different sowing dates. Gesunde Pflanzen 74:87–96. https://doi.org/10.1007/s10343-021-00590-2
El-Bially MA, Saudy HS, Hashem FA, El-Gabry YA, Shahin MG (2022b) Salicylic acid as a tolerance inducer of drought stress on sunflower grown in sandy soil. Gesunde Pflanzen 74:603–613. https://doi.org/10.1007/s10343-022-00635-0
El-Metwally IM, Saudy HS (2021) Interactional impacts of drought and weed stresses on nutritional status of seeds and water use efficiency of peanut plants grown in arid conditions. Gesunde Pflanzen 73:407–416. https://doi.org/10.1007/s10343-021-00557-3
El-Metwally IM, Saudy HS, Abdelhamid MT (2021) Efficacy of benzyladenine for compensating the reduction in soybean productivity under low water supply. Ital J Agromet 2:81–90. https://doi.org/10.36253/ijam-872
El-Metwally IM, Geries L, Saudy HS (2022a) Interactive effect of soil mulching and irrigation regime on yield, irrigation water use efficiency and weeds of trickle–irrigated onion. Arch Agron Soil Sci 68:1103–1116. https://doi.org/10.1080/03650340.2020.1869723
El-Metwally IM, Sadak MS, Saudy HS (2022b) Stimulation effects of glutamic and 5‑Aminolevulinic acids on photosynthetic pigments, physio-biochemical constituents, antioxidant activity, and yield of peanut. Gesunde Pflanzen 74:915–924. https://doi.org/10.1007/s10343-022-00663-w
Farooq M, Bramley H, Palta JA, Siddique KHM (2011) Heat stress in wheat during reproductive and grain-filling phases. CRC Crit Rev Plant Sci 30:491–507
Gandjaeva L (2019) Effect of sowing date on yield of winter wheat cultivars Grom, Asr and Kuma in Khorezm region. Bulg J Agric Sci 25:474–479
Ghadirnezhad Shiade SR, Fathi A, Taghavi Ghasemkheili F et al (2022a) Plants’ responses under drought stress conditions: effects of strategic management approaches—a review. J Plant Nutr. https://doi.org/10.1080/01904167.2022.2105720
Ghadirnezhad Shiade SR, Pirdashti H, Esmaeili MA, Nematzade GA (2022b) Biochemical and physiological characteristics of mutant genotypes in rice (Oryza sativa L.) contributing to salinity tolerance indices. Gesunde Pflanz. https://doi.org/10.1007/s10343-022-00701-7
Kamran M, Wennan S, Ahmad I et al (2018) Application of paclobutrazol affect maize grain yield by regulating root morphological and physiological characteristics under a semi-arid region. Sci Rep 8:4818. https://doi.org/10.1038/s41598-018-23166-z
Kraus TE, Murr DP, Fletcher RA (1991) Uniconazole inhibits stress-induced ethylene in wheat and soybean seedlings. J Plant Growth Regul 10:229–234. https://doi.org/10.1007/BF02279340
Mathur S, Jajoo A (2013) Effects of heat stress on growth and crop yield of wheat (Triticum aestivum). In: Physiological mechanisms and adaptation strategies in plants under changing environment, vol 1. Springer, New York, pp 163–191
Modarresi M, Mohammadi V, Zali A, Mardi M (2010) Response of wheat yield and yield related traits to high temperature. Cereal Res Commun 38:23–31. https://doi.org/10.1556/CRC.38.2010.1.3
Modhej A, Emam Y, Aynehband A (2011) Effect of nitrogen levels on source restriction and the pattern of assimilate redistribution to grains in wheat genotypes under post-anthesis heat stress conditions. Iran J F Crop Res 9:474–485
Modhej A, Farhoudi R, Afrous A (2015) Effect of post-anthesis heat stress on grain yield of barley, durum and bread wheat genotypes. In: Proceedings of the 10th International Barley Genetics Symposium Alexandria, Egypt, 5–10 Apr 2008
Mojtabaie Zamani M, Nabipour M, Meskarbashee M (2015) Responses of bread wheat genotypes to heat stress during grain filling period under Ahvaz conditions. Plant Prod 37:119–130
Mombeini M, Siadat SA, Lack S et al (2014) Physiological responses to heat stress in rice (Oryza sativa. L): I. nitrogen status, chlorophyll content and Cell Membrane Thermal Stability (CMTS) of flag leaf. Adv Environ Biol 8(5):1420–1431
Moneruzzam M, Syafiq M, Dogara Abd M et al (2020) Influence of Paclobutrazol on growth, yield and quality of eggplant (Solanum melongena). Asian J Plant Sci 19:361–371. https://doi.org/10.3923/ajps.2020.361.371
Mousavi F, Siahpoosh MR, Sorkheh K (2021) Influence of sowing date and terminal heat stress on phonological features and yield components of bread wheat genotypes. J Plant Prod 44(2):157–170
Mubarak M, Salem EMM, Kenawey MKM, Saudy HS (2021) Changes in calcareous soil activity, nutrient availability, and corn productivity due to the integrated effect of straw mulch and irrigation regimes. J Soil Sci Plant Nutr 21:2020–2031. https://doi.org/10.1007/s42729-021-00498-w
Murungu FS, Madanzi T (2010) Seed priming, genotype and sowing date effects on emergence, growth and yield of wheat in a tropical low altitude area of Zimbabwe. Afr J Agric Res 5(17):8
Nagar S, Singh VP, Arora A et al (2021) Understanding the role of gibberellic acid and paclobutrazol in terminal heat stress tolerance in wheat. Front Plant Sci 12:692252. https://doi.org/10.3389/fpls.2021.692252
Nazeri SM (2017) Study on bread wheat cultivar developmental stages and canopy temperature depression changing at environmental conditions due to different sowing dates. Appl F Crop Res 30:18–32
Nouriyani H (2015) Effect of paclobutrazol on the amount of redistribution of assimilates to the grain of three cultivars of wheat (Triticum aestivum L.) under heat tension conditions. Crop Physiol J 7:89–104
Nouriyani H (2017) Effect of paclobutrazol levels on grain growth process and yield of three cultivars of wheat (Triticum aestivum L.) under post-anthesis heat stress conditions. Environ Stress Crop Sci 9:407–415. https://doi.org/10.22077/escs.2017.468
Oleksiak T (2014) Effect of sowing date on winter wheat yields in Poland. J Cent Eur Agric 15:0
Pan S, Rasul F, Li W et al (2013) Roles of plant growth regulators on yield, grain qualities and antioxidant enzyme activities in super hybrid rice (Oryza sativa L.). Rice 6:9. https://doi.org/10.1186/1939-8433-6-9
Peng D, Chen X, Yin Y et al (2014) Lodging resistance of winter wheat (Triticum aestivum L.): Lignin accumulation and its related enzymes activities due to the application of paclobutrazol or gibberellin acid. F Crop Res 157:1–7. https://doi.org/10.1016/j.fcr.2013.11.015
Podolska G, Wyzińska M (2011) The response of new winter wheat cultivars to density and sowing date. Polish J Agron 6:44–51
Pradhan GP, Prasad PVV, Fritz AK, Kirkham MB, Gill BS (2010) High temperature tolerance in Aegilops species and its potential transfer to wheat. Crop Sci. https://doi.org/10.2135/cropsci2011.04.0186
Rahimi R, Paknejad F, Sadeghi Shoae M et al (2023) Changes in chlorophyll content and fluorescence indices and some physiological traits of wheat under the influence of paclobutrazol and growth-promoting bacteria at different levels of irrigation. J Plant Process Funct 11:1–19
Razavizadeh R, Kazemzadeh M, Enteshari S (2013) The effect of paclobutrazole on some physiological parameters of rapeseed seedlings under saline stress. Crop Physiol J 5:35–48
Reynolds M, Foulkes J, Furbank R et al (2012) Achieving yield gains in wheat. Plant Cell Environ 35:1799–1823. https://doi.org/10.1111/j.1365-3040.2012.02588.x
Salem EMM, Kenawey MKM, Saudy HS, Mubarak M (2021) Soil mulching and deficit irrigation effect on sustainability of nutrients availability and uptake, and productivity of maize grown in calcareous soils. Commun Soil Sci Plant Anal 52:1745–1761. https://doi.org/10.1080/00103624.2021.1892733
Salem EMM, Kenawey MKM, Saudy HS, Mubarak M (2022) Influence of silicon forms on nutrient accumulation and grain yield of wheat under water deficit conditions. Gesunde Pflanzen 74:539–548. https://doi.org/10.1007/s10343-022-00629-y
Saudy HS, Abd El-Momen WR, El-khouly NS (2018) Diversified nitrogen rates influence nitrogen agronomic efficiency and seed yield response index of sesame (Sesamum indicum, L.) cultivars. Commun Soil Sci Plant Anal 49:2387–2395. https://doi.org/10.1080/00103624.2018.1510949
Saudy HS, El-Metwally IM, Abd El-Samad GA (2020a) Physio-biochemical and nutrient constituents of peanut plants under bentazone herbicide for broad-leaved weed control and water regimes in dry land areas. J Arid Land 12(4):630–639. https://doi.org/10.1007/s40333-020-0020-y
Saudy HS, Hamed MF, Abd El-Momen WR, Hussein H (2020b) Nitrogen use rationalization and boosting wheat productivity by applying packages of humic, amino acids and microorganisms. Commun Soil Sci Plant Anal 51:1036–1047. https://doi.org/10.1080/00103624.2020.1744631
Saudy HS, Noureldin NA, Mubarak M, Fares W, Elsayed M (2020c) Cultivar selection as a tool for managing soil phosphorus and faba bean yield sustainability. Arch Agron Soil Sci 66:414–425. https://doi.org/10.1080/03650340.2019.1619078
Saudy HS, El-Bially MA, El-Metwally IM, Shahin MG (2021) Physio-biochemical and agronomic response of ascorbic acid-treated sunflower (Helianthus annuus) grown at different sowing dates and under various irrigation regimes. Gesunde Pflanzen 73:169–179. https://doi.org/10.1007/s10343-020-00535-1
Saudy HS, Abd El-Samad GA, El-Temsah ME, El-Gabry YA (2022) Effect of iron, zinc and manganese nano-form mixture on the micronutrient recovery efficiency and seed yield response index of sesame genotypes. J Soil Sci Plant Nutr 22:732–742. https://doi.org/10.1007/s42729-021-00681-z
Saudy HS, El-Bially MA, Hashem FA, Shahin MG, El-Gabry YA (2023a) The changes in yield response factor, water use efficiency, and physiology of sunflower owing to ascorbic and citric acids application under mild deficit irrigation. Gesunde Pflanz. https://doi.org/10.1007/s10343-022-00736-w
Saudy HS, Salem EMM, Abd El-Momen WR (2023b) Effect of potassium silicate and irrigation on grain nutrient uptake and water use efficiency of wheat under calcareous soils. Gesunde Pflanz. https://doi.org/10.1007/s10343-022-00729-9
Shirinzadeh A, Heidari H, Abad S, Nourmohammadi G (2017) Effect of planting date on growth periods , yield , and yield components of some bread wheat cultivars in Parsabad Moghan. Int J Farming Allied Sci 6:109–119
Sobral BS, de Oliveira-Júnior JF, de Gois G et al (2019) Drought characterization for the state of Rio de Janeiro based on the annual SPI index: trends, statistical tests and its relation with ENSO. Atmos Res 220:141–154. https://doi.org/10.1016/j.atmosres.2019.01.003
Soumya PR, Kumar P, Pal M (2017) Paclobutrazol: a novel plant growth regulator and multi-stress ameliorant. Indian J Plant Physiol 22:267–278. https://doi.org/10.1007/s40502-017-0316-x
Sun H, Shao L, Chen S et al (2013) Effects of sowing time and rate on crop growth and radiation use efficiency of winter wheat in the North China Plain. Int J Plant Prod 7:117–138
Vosoghi Rad M, Jami Moeini M, Taherian M, Armin M (2022) Accumulation and remobilization of assimilates in different genotypes of durum wheat under terminal drought stress. J Crop Sci Biotechnol 25:199–214. https://doi.org/10.1007/s12892-021-00123-3
Yousefi R, Bannayan Aval M, Khorramdel S, Nassiri Mahallati M (2018) Comparison of old and new dryland wheat cultivars in response to different planting dates. Appl F Crop Res 31:46–72
Zhao C, Liu B, Piao S et al (2017) Temperature increase reduces global yields of major crops in four independent estimates. Proc Natl Acad Sci USA 114:9326–9331. https://doi.org/10.1073/pnas.1701762114
Zhao J, Lai H, Bi C et al (2023) Effects of paclobutrazol application on plant architecture, lodging resistance, photosynthetic characteristics, and peanut yield at different single-seed precise sowing densities. Crop J 11:301–310. https://doi.org/10.1016/j.cj.2022.05.012
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
N. Bakhtiyarinejad, M. Mombeini, A. Bahrani and M. Mojtabaie Zamani declare that they have no competing interests.
Rights and permissions
Springer Nature oder sein Lizenzgeber (z.B. eine Gesellschaft oder ein*e andere*r Vertragspartner*in) hält die ausschließlichen Nutzungsrechte an diesem Artikel kraft eines Verlagsvertrags mit dem/den Autor*in(nen) oder anderen Rechteinhaber*in(nen); die Selbstarchivierung der akzeptierten Manuskriptversion dieses Artikels durch Autor*in(nen) unterliegt ausschließlich den Bedingungen dieses Verlagsvertrags und dem geltenden Recht.
About this article
Cite this article
Bakhtiyarinejad, N., Mombeini, M., Bahrani, A. et al. Paclobutrazol Application Improves Growth and Physiological Traits of Wheat Cultivars Under Heat Stress Imposed by Late Planting Date. Gesunde Pflanzen 75, 2491–2503 (2023). https://doi.org/10.1007/s10343-023-00924-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10343-023-00924-2