Abstract
Natural regeneration potential is an important indicator for any forest ecosystem and Andaman Padauk (Pterocarpus dalbergioides) is the principal timber tree endemic to Andaman & Nicobar Islands. This study analyzed the natural regeneration potential of Andaman Padauk under different disturbance regimes across its distribution range with regard to population dynamics, growth pattern, population structure, population status and allometry. Contrary to the perception that it is a poor regenerator, the species showed a luxuriant growth. While ephemerals, seedlings, saplings, poles and total regenerates are significantly high at the leading edge of its habitat, trees are significantly high at the trailing edge. Their heights are mostly positively skewed and normally distributed. With a reverse-J shaped demography, the species is in its most dominant form in the stand. Though its population status is declining, the prospects of population stabilization are high. There exists a strong proportionality between DBH and height of regenerates. Hence, the species should be assisted through considered disturbance to realize its natural regeneration potential. As all the species under the pantropical Pterocarpus genus share a number of growth and reproductive traits, the path breaking findings of this study emanated from refined methodologies will help assess the natural regeneration potential of other species whose existence is threatened by either unstable or declining population in their habitat.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
World has 3999 million ha of forests wherein 93% are natural forests and the rest are planted forests (FAO 2015). While natural forests conserve the genotypic diversity and maintain the natural tree species composition, structure and ecological dynamics; planted forests are established for the purpose of industrial timber production. But, together they provide a bulk of timber and non-timber products like sawn wood, round wood, fire wood, charcoal, paper, fodder, medicinal & aromatic plants, gums & resins, wild fruits & vegetables, nuts and seeds (FAO 2018). For the same reason, forests are being exploited indiscriminately oblivious of their ecological and environmental services like water purification, erosion control and carbon sequestration.
Recognizing the crucial role of forests in eradicating poverty, ending hunger, achieving inclusive growth, narrowing inequalities, mitigating climate change and managing natural resources, Sustainable Development Goal (SDG) 15.2 of United Nations has explicitly called for sustainable management of all types of forests, to halt deforestation, to restore degraded forests and to substantially increase afforestation and reforestation globally (FAO 2018). Going by a conservative estimate of 2000 USD per ha for artificial regeneration, the cost of achieving restoration is quite high (Löf et al. 2019) which risks Official Development Assistance (ODA) and public expenditure on conservation and sustainable use of forests as envisaged by SDG 15.B (FAO 2018).
Natural regeneration, the spontaneous recovery of native tree species that colonize and establish in abandoned fields or natural disturbances (Crouzeilles et al. 2017) offers a cost-effective option for large-scale forest restoration (Chazdon and Guariguata 2016). A Naturally regenerating forest will emerge as a complex adaptive system that possesses eight key properties, viz. heterogeneity, hierarchy, self-organization, openness, adaptation, memory, nonlinearity and uncertainty (Filotas et al. 2014) which confer resilience on the system following disturbances or major shocks (Cumming 2011). In addition to enhancing resilience and supplying multiple ecosystem goods and services, natural regeneration can be highly effective for recovering local biodiversity, species interactions and movement within landscapes (Rahman et al. 2011). However, it relies on the inherent regeneration potential of the native tree species in terms of seed production, dispersal, germination, seedling establishment and growth of the seedlings under varied environmental conditions like climate, soil characteristics, disturbance regimes and seed bank composition (Bawa and Seidler 1998; Luo et al. 2017; Sharma et al. 2018).
Pterocarpus is a pantropical genus of dalbergioid legumes (Lavin et al. 2001) having an estimated 32 species distributed across tropical Africa, Neotropics and Indomalaya (Saslis-Lagoudakis et al. 2011). While India shares the habitat of P. marsupium with other Asian countries like Srilanka, Nepal, Bangladesh and Taiwan, P. santalinus and P. dalbergioides are endemic to Eastern Ghats of Peninsular India and Andaman Islands, respectively (IUCN 2020). Apart from a few sporadic studies on P. dalbergioides (Prasad et al. 2008), P. santalinus (Ankalaiah et al. 2017), P. angolensis (Gonçalves et al. 2018) and P. soyauxii (Mokake et al. 2018), the natural regeneration potential of these pantropical but yet endemic species is still unexplored. Therefore, scrutinizing the natural regeneration potential of these heavily traded timber species gains significance in the light of unsustainably declining population (Winfield et al. 2016) leaving them near threatened to endangered in their natural habitat (Barstow 2018).
Andaman & Nicobar Islands is one of the nine Union Territories of India, strategically located in the south-eastern Bay of Bengal 1200 km away from the mainland as a gateway to ASEAN countries. Aligned in north–south direction between 92.12° and 93.57° E longitude; and between 6.45o and 13.41° N latitude with 10oN channel separating Andaman group from Nicobar group of Islands, the archipelago comprises of 836 islands, islets and rocks (DoEF 2017) covering a geographical area of 8249 sq.km, accommodating 0.38 million population including 28,530 notified tribes (MoHA 2011). Apart from the rapidly urbanizing South Andaman district where the capital town of Port Blair is located, the Islands is predominantly rural with dispersed settlements.
Staying true to its biodiversity hotspot tag, the ecosystem of this tropical Islands is highly diversified with innumerable endemic species. The land use pattern is highly conservative as 86% of area is legally preserved under 12 forest types (Champion and Seth 1968). Andaman Padauk (Pterocarpus dalbergioides Roxb.) is the principal timber tree endemic to Andaman group of Islands forming an important component of moist deciduous and semi-evergreen forests (Rao 2000).
In moist deciduous forest, the species occurs in the top canopy along with Terminalia bialata, T. citrina, T. procera, Canarium euphyllum, Pterocymbium tinctorium, Tetrameles nudiflora, Albizzia lebbek, and Lagerstroemia hypoleuca; the middle story is occupied by Lannea coromandelica, Adenanthera microsperma, Dillenia andamanica, Diospyros marmorata, Cratoxylum formosum, Semecarpus kurzii, Pterospermum aceroides, Murraya paniculata, and Atalantia monophylla; and the lower story species are Canthium gracilipes, Rinorea bengalensis, Glycosmis pentaphylla, and Tabernaemontana alternifolia. In semi-evergreen forest, it occurs in the top canopy along with Dipterocarpus grandiflorus, D. gracilis, Pterygota alata, Pterocymbium tinctorium, terminalia bialata, T. procera, Albizzia chinensis, A. lebbek, Artocarpus lacucha, and A. chama; the middle canopy is dominated by Lagerstroemia hypoleuca, Dillenia andamanica, Pometia pinnata, Horsifieldia irya, Xanthophyllum andamanicum, Fagraea racemosa, Sageraea elliptica, Aporosa villosa and Maesa andamanica; and the under story consists of Licuala peltata, Caryota mitis, Areca triandra, Leea indica, and Clerodendrum infortunatum (Alappatt 2017).
The slow growing tree takes about 35–40 years for the extraction of good quality timber (Rao 2000) and reaches a height of above 30 m attaining a girth of over 3 m, often with very large buttresses (Ganapathy and Rangarajan 1964). Owing to good polishing and termite resistance, the moderately hard crimson wood is used in high-class joinery, furniture, cabinetry, fancy turnery, carving and sculpting, inlay, flooring, knife handles, veneer and tool handles, boat building, paddles, oars, agricultural implements, billiard tables, decorative woods and in dye making (Prasad et al. 2008).
Like any other tropical forests of the world, Andaman forests are also subjected to deforestation leading to fragmentation of the forest system, resulting in extinction of valuable species from their native habitats. This problem is a significant issue for Andaman Padauk whose population is affected by legal and illegal logging on the one hand and scanty natural reproduction on the other hand (Prasad et al. 2008; Ganapathy and Rangarajan 1964). Therefore, the present study was undertaken during April 2019 to examine the natural regeneration potential of Andaman Paduak under various physiographic conditions of Andaman Islands in terms of population dynamics, growth pattern, population structure, population status and allometry.
Materials and methods
Study area
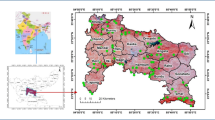
In accordance with the objective of the study, four locations, viz. Mohanpur and Lamiya Bay in North Andaman, Prolobjig in Middle Andaman and Hut Bay in South Andaman were purposively selected to cover the whole range of species distribution from 10o39′N to 13o15′N covering an area of 6408 sq km. (Fig. 1).
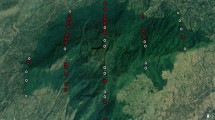
All these sites falling under protected forests are slopy lands that have undergone disturbance in one way or other. While a dead bamboo grove has disrupted the canopy cover at Mohanpur, a road cutting across has broken the canopy contiguity at Lamiya Bay. Even as Prolobjig is an erstwhile selective logging plot, Hut Bay is an abandoned quarry (Fig. 2).
Sampling design
At the outset, the study area was transected to have a general idea about the species composition wherein Padauk regenerates had a dominant presence over Terminalia bialata, Canarium euphyllum, Albizzia lebbek, Lagerstroemia hypoleuca, Dillenia andamanica, Diospyros marmorata, Semecarpus kurzii, Lannea coromandelica, Calamus viminalis, Gigantochloa nigrociliata, and Sageraea elliptica. Following a stratified random quadrat method with a sampling design similar to Tesfaye et al. (2002), Malik et al. (2012), Gao et al. (2017), Gonçalves et al. (2018), Rahman et al. (2020), and Maua et al. (2020) two quadrats of size 20 m × 20 m (400 m2) were laid each at a distance of 10 m uphill and downhill from a padauk tree. Within each of these quadrats, 10 random sub-plots of size 2 m × 2 m (4 m2) were laid from which relevant data were collected. Thus, the total sample size amounted to 80 plots of 4 m2 with 20 at each of the four locations.
Data
The information on number of padauk regenerates, their height and Diameter at Breast Height (DBH) were collected from each of the sub-plots of the sampling quadrats. While the number of regenerates was enumerated manually, their height and DBH were calculated using measuring tape and Digital Vernier Caliper, respectively.
Methodology
Population dynamics, growth pattern, population structure, regeneration status and allometry were analyzed to compare and contrast the natural regeneration potential of Andaman Padauk among different locations across Andaman Islands and between uphill and downhill of a padauk tree in slopy lands.
Population dynamics
The presence of sufficient number of seedlings, saplings and young trees in a given population provides valuable information about the population dynamics and indicates a successful regeneration of forest species (Saxena and Singh 1984; Dang et al. 2010; Dolanc et al. 2013). Different studies have applied different yardsticks to classify the regenerates either on the basis of height or diameter of the stem; or both in tandem. To mention a few, Malik et al. (2012) classified the regenerates as recruits that are current year’s seedlings, unestablished with height < 2 m and established > 2 m; Juřička et al. (2019) categorized as seedlings of height < 137 cm and saplings of 137–200 cm with further division of seedlings into four sub-categories; Nerlekar et al. (2019) considered plants having a DBH ≥ 10 cm as trees, 1–10 cm as saplings and < 1 cm as seedlings; and de Carvalho et al. (2017) divided the plants into seedlings 0.5–1 m tall, small saplings 1–3 m tall, large saplings > 3 m tall and ≤ 5 m DBH; and poles 5–10 cm DBH.
Considering the morphology of padauk regenerates in the sampling plots, the plants were categorized as ephemerals of height < 30 cm, seedlings 30–100 cm, saplings 100–200 cm, poles 200–300 cm and trees > 300 cm for the study. The number of individuals under each of these categories in 10 sub-plots of a quadrat was enumerated separately, averaged and represented as:
Subsequently, the plant density of regenerates was examined for its significant difference among the four study locations using One-way ANOVA and Tukey’s Post Hoc Test (Do et al. 2016; Hallsby et al. 2015).
Growth pattern
The height of enumerated regenerates was analyzed for its mean, median, mode, range and inter-quartile range through Box Plot technique to compare and contrast the growth pattern of padauk regenerates among the study locations.
Population structure
Population structure is defined as the proportion of plants under different life stages of a species per unit area (Endels et al. 2004). It indicates the regeneration process at a certain moment in time (Poorter et al. 1996). Height/DBH-class (Gao et al. 2017; Khan et al. 1987) and height-class (Tesfaye et al. 2002) distribution that work well in treelets (Condit et al. 1998) are often used to represent the population structure of forests.
A reverse-J shaped distribution with plenty of individuals in the small size classes indicates good regeneration (Khamyong et al. 2003), while a J-shaped distribution with relatively few individuals in the small size classes indicates poor regeneration and probably declining population (Kumar et al. 2016). Similarly, a bell-shaped distribution with many saplings between few seedlings and adults is attributed to disturbed forest in which regeneration is hampered (Saxena et al. 1984), while an inverted-bell shaped distribution illustrates a recovering population that experienced a period of very poor recruitment (Hall and Bawa 1993). The size-class distribution diagrams are further tested for Pearson Correlation, wherein a negative correlation indicates the reverse-J shaped curve, a positive correlation indicates the J-shaped curve and no correlation indicates haphazard population distribution (Poorter et al. 1996).
Accordingly, the calculated plant density was proportionalized among different categories of regenerates and plotted as height-class distribution in a Line Graph to elucidate the population structure of padauk regenerates in uphill, downhill and whole of the four locations, followed by correlation analysis.
Population status
Successful regeneration of a species depends on its ability to produce new seedlings and the ability of seedlings and saplings to survive and grow (Good and Good 1972). Thus, the population status of a species is determined either by the proportion of seedlings and saplings in total regenerates (Bhuyan et al. 2003), the relative proportion of all life stages of regenerates (Shankar 2001) or by the presence of certain minimum quantity of plants under each life stage of regenerates (Juřička et al. 2019).
In the current study, the population status of padauk was determined based on the proportion of seedlings in total regenerates excluding ephemerals (Baboo et al. 2017; Sagar and Singh 2004): (i) 0% was considered as conservation focused species, (ii) 1–24% was considered as severely declining species, (iii) 25–49% was considered as declining species, (iv) 50–66% was considered as relatively stable species, (v) 67–99% was considered as potentially expanding species, and (vi) 100% was considered as newly recruited species from neighboring areas. The results have been illustrated as Percentage Bar Graph (Paul et al. 2019).
Allometry
Forests play a significant role in mitigating climate change by sequestering atmospheric carbon (Nogueira Junior et al. 2014; Istrefi et al. 2019). Hence, accurate estimates of forest biomass are crucial for quantifying the restoration level of degraded forests in view of emerging carbon credit market opportunities under Reducing Emission from Deforestation and Forest Degradation (REDD +) (Djomo et al. 2016; Mugasha et al. 2016). While it is practically easy to measure DBH of all trees within the sample plot, height, wood density, crown length, crown width, crown area, crown diameter, height-to-diameter ratio, age, etc. are measured only for a few trees during forest inventories (Ahmadi et al. 2013; Li et al. 2015; En et al. 2019). As these tree attributes are functionally related, allometric equations are worked out among them to indirectly estimate the forest biomass (Chave et al. 2015).
Here, the height and DBH of poles and trees above 2 m height in the study areas were examined for their allometric relationship through Scatter Plots and Pearson Correlation (Daba and Soromessa 2019; Jia and Zhou 2018) to enable estimation of biomass of padauk forests.
All analyses were carried out in SPSS software.
Results
Population dynamics of padauk regenerates
Among the four study areas, the density of ephemerals was significantly high at Lamiya Bay on the downhill on a par with Mohanpur, uphill on a par with Hut Bay and combined on a par with Mohanpur (Table 1). On the other hand, there was no significant difference in the density of seedlings among the downhill of study areas, but it was significantly high at Mohanpur on the uphill and combined on a par with Lamiya Bay. While there was no significant difference in the density of saplings among the uphill of study areas, it was significantly high at Mohanpur on the downhill and combined on a par with Prolobjig. Similarly, the density of poles was not significantly different among the uphill of study areas, but it was significantly high at Lamiya Bay on the downhill and combined, both on a par with Mohanpur and Prolobjig. Incidentally, the density of trees was significantly high at Hut Bay on both gradients and combined. Overall, the density of total regenerates was significantly high at Mohanpur on the downhill and combined, both on a par with Lamiya Bay and Hut bay.
Growth pattern of regenerates
Further scrutiny on the growth pattern of enumerated regenerates revealed that the height of ephemerals was normally distributed around 18 to 20 cm with half of them concentrated on the higher end between 15 and 23 cm (Fig. 3). While the height of seedlings at Lamiya Bay and Hut Bay was normally distributed around 61 to 62 cm with half of them concentrated evenly between 41 and 80 cm, the same at Mohanpur and Prolobjig was positively skewed around 43 to 44 cm with half of them concentrated on the lower end between 36 and 62 cm. Similarly, the height of saplings across all the study locations was normally distributed around 143 to 157 cm with half of them spread between 122 and 178 cm.
On the other hand, the height of poles at Mohanpur, Lamiya Bay and Prolobjig was negatively skewed around 264 to 275 cm with half of them spread between 224 and 283 cm, while the same at Hut bay was positively skewed around 275 cm with half of them concentrated on the higher end between 265 and 291 cm. Similarly, the height of trees at Mohanpur and Prolobjit was negatively skewed around 411 cm with half of them spread between 356 and 458 cm while the same at Lamiya Bay and Hut Bay was positively skewed around 375 to 415 cm with half of them spread between 336 and 475 cm. Overall, the height of all regenerates at Mohanpur ranged from 8 to 487 cm, positively skewed around 32 cm with half of them spread between 21 and 128 cm, while the same at Lamiya Bay ranged from 5 to 476 cm, positively skewed around 24 cm with half of them spread between 18 and 96 cm. Even as the height of all regenerates at Prolobjig ranged from 8 to 496 cm, positively skewed around 33 cm with half of them spread between 21 and 144 cm, the same at Hut Bay ranged from 6 to 562 cm, positively skewed around 38 cm with half of them spread between 19 and 178 cm.
Population structure of padauk regenerates
The height-class distribution of padauk regenerates on the downhill, uphill and mean at Mohanpur has revealed a closely plotted and relatively flatter reverse-J shaped curves with significant correlation coefficients of − 0.56, − 0.63 and − 0.60, respectively, while the same at Lamiya Bay has shown overlapping and relatively steeper reverse-J shaped curves with significant correlation coefficients of − 0.55, − 0.54 and − 0.54, respectively (Fig. 4).
Similarly, the distribution at Prolobjig has shown overlapping and relatively flatter reverse-J shaped curves with significant correlation coefficients of − 0.57, − 0.59 and − 0.58, respectively. Strikingly, the distribution at Hut Bay has exhibited closely plotted and relatively steeper reverse-J shaped curves that elongates further to form inverse-bell shaped curves with significant correlation coefficients of − 0.47, − 0.51 and − 0.50, respectively.
Population status of Andaman Padauk
With 38% seedlings, the population on the downhill of Mohanpur is declining, while the same is relatively stable on the uphill with 56% seedlings (Fig. 5). Overall, the population of Andaman Padauk at Mohanpur is declining with 47% seedlings. On the other hand, the population on the downhill, uphill and overall at Lamiya Bay is declining with around 45% seedlings. With 41 to 46% of seedlings, the population on the downhill, uphill and overall at Prolobjig is also declining. In consonance with the population structure, the trees have almost replaced the poles at Hut Bay and their proportion on the downhill has far exceeded that of seedlings and almost equaled on the uphill. With 35 to 39% seedlings, the population at Hut Bay is also declining.
Allometry
The allometric relationship between DBH and height of poles and trees across the study areas shows that the DBH and height are strongly correlated at Mohanpur and Prolobjig with significant correlation coefficients of 0.91 and 0.88, respectively (Fig. 6). Though significantly correlated, the relationship between DBH and height at Lamiya Bay and Hut Bay is weak with correlation coefficients of 0.31 and 0.50, respectively.
Discussion
Natural regeneration potential is an important indicator for any forest ecosystems (Rahman et al. 2011) and Andaman Padauk, the State Tree is a historical legacy to the economy of the Islands. This study for the first time has comprehensively analyzed the natural regeneration potential of Andaman Padauk across its whole habitat from five different perspectives. At the outset, all the study locations were found endowed with all stages of regenerates from recruits to trees in their population signifying a successful regeneration (Saxena and Singh 1984). Contrary to the observation of Prasad et al. 2008 that Andaman Padauk is a poor regenerator owing to absence of regenerates in the padauk bearing semi-evergreen and moist deciduous forests of North Andaman, the extrapolated average population density of 64,219 regenerates/ha across all the study locations comprising 31,656 recruits, 14,094 seedlings, 7688 saplings, 2969 poles and 7813 trees has far exceeded the stipulations laid out under various studies (Juřička et al. 2019; Chacko 1965; Luo et al. 2017), thanks to the disturbance caused by dead bamboo grove, secondary road, selective logging and quarrying. Canopy gap created by such disturbance regimes has increased the light level and soil temperature providing favorable germinating conditions and thus triggering regeneration (Peters 1994).
Moreover, difference in forest structure and physiographic conditions among the study locations that stretch across 3° latitude has led to irregular spatial distribution of the species (Ward et al. 2006). While ephemerals and poles were significantly high at Lamiya Bay; and seedlings and saplings were significantly high at Mohanpur—both at the leading edge of the habitat, trees were significantly high at Hut Bay, the trailing edge (Wei et al. 2015). Overall, the total regenerates were significantly high at Mohanpur, again at the leading edge of Andaman Padauk’s distribution range. The height analysis has confirmed a healthy growth pattern of padauk regenerates.
Though the germination capacity of Andaman Padauk is quite high at 45%, ultimate survival is reduced by half due to mortality in early seedling stage (Ganapathy and Rangarajan 1964). Accordingly, its population structure in all the study locations exhibited a reverse-J shaped distribution indicating that a small fraction of ephemerals survive to become large trees. Nevertheless, the reverse-J shaped distribution with highest number of individuals in ephemeral stage and the number gradually decreasing in seedling, sapling, pole and tree stages shows that Andaman Padauk is in its most dominant form in the stand at present (Malik and Bhatt 2016). Exceptionally, the elongated reverse-J shaped curve to form an inverted-bell shape is a case of past intense harvesting of adults at Hut Bay, wherein the remnant trees grew and began regenerating (Hall and Bawa 1993). Although, the fate of a species cannot be predicted from the shape of size-class distribution curve alone, a constant lack of regeneration gives a good indication that the forest ecosystem is disturbed (Lykke 1998).
The forest wealth depends on the potential regenerative status of species composing the forest stand in space and time (Jones et al. 1994). Except for the uphill of Mohanpur where the population status was relatively stable, all the other study locations and gradients have exhibited a declining population status. However, considering the luxuriant growth of regenerates in all the study locations wherein ephemerals share around 50% of total population with the prospects of at least 25% of them graduating to seedlings, the population status of Andaman Padauk across its distribution range can be conveniently considered as relatively stable.
Tree height together with DBH defines the main structural pattern of forest systems (Nogueira Junior et al. 2014). Among DBH, wood specific gravity, height and forest type that are primarily applied in different combinations to develop biomass regression models (Chave et al. 2005), power equation incorporating the compound variable of DBH and height proved to be the most appropriate model for predicting the biomass of Andaman Padauk (Jaisankar et al. 2020a) as it takes care of heteroscedasticity exhibited by biomass data (Istrefi et al. 2019). By showing a strong proportionality between DBH and height, the growth pattern of regenerates at Mohanpur and Prolobjig has asserted the reliability of these variables for biomass estimation. But a weak proportionality shown by the regenerates at Lamiya Bay and Hut Bay necessitates relating DBH and height to other structural and functional characteristics for accurate biomass estimation of padauk forests in such localities.
Conclusions
An understanding of seed morphology and the processes that affect regeneration of a species is of crucial importance to forest managers to utilize a given forest ecosystem wisely and sustainably (Slik et al. 2003; Pala et al. 2012; Jaisankar et al. 2020b). Luxuriant growth, healthy growth pattern, ideal population structure, prospects of population stabilization and DBH-height proportionality of regenerates under different disturbance regimes across the distribution range assert the importance of considered disturbance in stimulating natural regeneration of Andaman Padauk (Hobbs and Huenneke 1992). But the underlying challenge is not to exceed the little understood or unknown thresholds of ecosystem resilience (Karsten et al. 2014). In this regard, Assisted Natural Regeneration (ANR) which is an inexpensive but effective technique that employs simple methods like weed suppression, fire management, protection against grazing, assisting seed dispersal and tending of seedlings comes handy in restoring the degraded forests (Durst et al. 2011). Moreover, special priority to conserve this gifted timber species while executing the ambitious tourism projects that are underway would further strengthen the value of ecotourism in the Islands. As all the species under Pterocarpus genus share a number of growth and reproductive traits (Winfield et al. 2016), the path breaking findings of this study emanated from categorization of P. dalbergioides regenerates, analysis of their growth pattern, determination of their regeneration status and their delineation for allometry will help assess the natural regeneration potential of other species across the tropics whose existence is threatened by either unstable or declining population in their habitat.
References
Ahmadi K, Alavi SJ, Kouchaksaraei MT, Aertsen W (2013) Non-linear height-diameter models for Oriental Beech (Fagus orientalis Lipsky) in the Hyrcanian forests. Iran Biotechnol Agron Soc Environ 17(3):431–440
Alappatt JP (2017) Common forest plants of Andaman and Nicobar Islands. Department of Environment & Forests, Andaman & Nicobar Administration, Port Blair, India
Ankalaiah C, Mastan T, Reddy MS (2017) A study on the density, population structure and regeneration of Red Sanders Pterocarpus santalinus (Fabales: Fabaceae) in a protected natural habitat - Sri Lankamalleswara Wildlife Sanctuary, Andhra Pradesh, India. J Threat Taxa 9(9):10669–10674. https://doi.org/10.11609/jott.2869.9.9.10669-10674
Baboo B, Sagar R, Bargali SS, Verma H (2017) Tree species composition, regeneration and diversity of an Indian dry tropical forest protected area. Trop Ecol 58(2):409–423
Barstow M (2018) Pterocarpus dalbergioides. The IUCN Red List of Threatened Species 2018: e.T33261A67802958. https://doi.org/10.2305/IUCN.UK.2018-1.RLTS.T33261A67802958.en
Bawa K, Seidler R (1998) Natural forest management and conservation of biodiversity in tropical forests. Conserv Biol 12(1):46–55. https://doi.org/10.1111/j.1523-1739.1998.96480.x
Bhuyan P, Khan ML, Tripathi RS (2003) Tree diversity and population structure in undisturbed and human-impacted stands of tropical wet evergreen forest in Arunachal Pradesh, Eastern Himalayas. India Biodivers Conserv 12(8):1753–1773. https://doi.org/10.1023/A:1023619017786
Chacko VJ (1965) A manual on sampling techniques for forest surveys. Manager of Publications, Delhi
Champion HG, Seth SK (1968) A revised survey of forest types of India. Government of India Publication, New Delhi
Chave J, Andalo C, Brown S et al (2005) Tree allometry and improved estimation of carbon stocks and balance in tropical forests. Oecologia 145(1):87–99. https://doi.org/10.1007/s00442-005-0100-x
Chave J, Réjou-Méchain M, Búrquez A et al (2015) Improved allometric models to estimate the aboveground biomass of tropical trees. Glob Chang Biol 20:3177–3190. https://doi.org/10.1111/gcb.12629
Chazdon RL, Guariguata MR (2016) Natural regeneration as a tool for large-scale forest restoration in the tropics: Prospects and challenges. Biotropica 48(6):716–730. https://doi.org/10.1111/btp.12381
Condit R, Sukumar R, Hubbell SP, Foster RB (1998) Predicting population trends from size distributions: a direct test in a tropical tree community. Am Nat 152(4):495–509. https://doi.org/10.1086/286186
Crouzeilles R, Ferreira MS, Chazdon RL et al (2017) Ecological restoration success is higher for natural regeneration than for active restoration in tropical forests. Sci Adv 3(11):e1701345. https://doi.org/10.1126/sciadv.1701345
Cumming GS (2011) Spatial resilience: integrating landscape ecology, resilience, and sustainability. Landscape Ecol 26:899–909. https://doi.org/10.1007/s10980-011-9623-1
Daba DE, Soromessa T (2019) The accuracy of species-specific allometric equations for estimating aboveground biomass in tropical moist montane forests: Case study of Albizia grandibracteata and Trichilia dregeana. Carbon Balance Manag. https://doi.org/10.1186/s13021-019-0134-8
Dang H, Zhang Y, Zhang K, Jiang M, Zhang Q (2010) Age structure and regeneration of Subalpine Fir (Abies fargesii) forests across an altitudinal range in the Qinling mountains, China. Forest Ecol Manage 259(3):547–554. https://doi.org/10.1016/j.foreco.2009.11.011
de Carvalho AL, d’Oliveira MVN, Putz FE, de Oliveira LC (2017) Natural regeneration of trees in selectively logged forest in western Amazonia. For Ecol Manag 392:36–44. https://doi.org/10.1016/j.foreco.2017.02.049
Djomo AN, Picard N, Fayolle A et al (2016) Tree allometry for estimation of carbon stocks in African tropical forests. Forestry. https://doi.org/10.1093/forestry/cpw025
Do TV, Cam NV, Sato T et al (2016) Post-logging regeneration and growth of commercially valuable tree species in evergreen broadleaf forest, Vietnam. J Trop For Sci 28(4):426–435. https://www.jstor.org/stable/43956809
DoEF (2017) Forest Statistics, Department of Environment & Forests. Andaman & Nicobar Administration, Port Blair.
Dolanc CR, Thorne JH, Safford HD (2013) Widespread shifts in the demographic structure of subalpine forests in the Sierra Nevada, California, 1934 to 2007. Global Ecol Biogeogr 22(3):264–276. https://doi.org/10.1111/j.1466-8238.2011.00748.x
Durst PB, Sajise P, Leslie RN (2011) Forests beneath the grass: Proceedings of the regional workshop on advancing the application of assisted natural regeneration for effective low-cost restoration. Food and Agriculture Organization of the United Nations, Bangkok, Thailand
En L, Chunqian J, Xudong Z et al (2019) Structural characteristics of mixed plantation of Pinus massoniana and Oak in Danjiangkou reservoir area. IOP Conf Ser Earth Environ Sci. https://doi.org/10.1088/1755-1315/371/2/022041
Endels P, Adriaens D, Verheyen K, Hermy M (2004) Population structure and adult plant performance of forest herbs in three contrasting habitats. Ecography 27(2):225–241. https://doi.org/10.1111/j.0906-7590.2004.03731.x
FAO (2015) Global forest resources assessment 2015: how are the world’s forests changing? 2nd edn. Rome
FAO (2018) The State of the World’s Forests 2018 - Forest pathways to sustainable development. Rome.
Filotas E, Parrott L, Burton PJ et al (2014) Viewing forests through the lens of complex systems science. Ecosphere 5(1):1–23. https://doi.org/10.1890/ES13-00182.1
Ganapathy PM, Rangarajan M (1964) A study of phenology and nursery behaviour of Andaman timber species. Indian Forest 90(11):758–766. https://doi.org/10.36808/if%2F1964%2Fv90i11%2F25043
Gao W-Q, Ni Y-Y, Xue Z-M et al (2017) Population structure and regeneration dynamics of Quercus variabilis along latitudinal and longitudinal gradients. Ecosphere 8(4):e01737. https://doi.org/10.1002/ecs2.1737
Gonçalves FMP, Revermann R, Cachissapa MJ et al (2018) Species diversity, population structure and regeneration of woody species in fallows and mature stands of tropical woodlands of southeast Angola. J for Res 29(6):1569–1579. https://doi.org/10.1007/s11676-018-0593-x
Good NF, Good RE (1972) Population dynamics of tree seedlings and saplings in a mature eastern hardwood forest. Bull Torrey Bot Club 99(4):172–178. https://doi.org/10.2307/2484571
Hall P, Bawa K (1993) Methods to assess the impact of extraction of non-timber tropical forest products on plant populations. Econ Bot 47(3):234–247. https://doi.org/10.1007/BF02862289
Hallsby G, Ulvcrona KA, Karlsson A et al (2015) Effects of intensity of forest regeneration measures on stand development in a nationwide Swedish field experiment. Forestry 88:441–453. https://doi.org/10.1093/forestry/cpv010
Hobbs RJ, Huenneke LF (1992) Disturbance, diversity, and invasion: implications for conservation. Conserv Biol 6(3):324–337. https://doi.org/10.1046/j.1523-1739.1992.06030324.x
Istrefi E, Toromani E, Çollaku N, Thaçi B (2019) Allometric biomass equations for young trees of four broadleaved species in Albania. N Z J for Sci. https://doi.org/10.33494/nzjfs492019x51x
IUCN (2020) International Union for Conservation of Nature’s Red List of Threatened Species. https://www.iucnredlist.org. Accessed 27 October 2020
Jaisankar I, Ravi R, Jerard BA et al (2020a) Regional volume table for Andaman Padauk (Pterocarpus dalbergioides). The Indian Forest 146(1):1–6. https://doi.org/10.36808/if%2F2020%2Fv146i1%2F146637
Jaisankar I, Srividhya S, Jerard BA, Moses EM (2020b) Seed microstructures of the Andaman Padauk (Pterocarpus dalbergioides). Curr Sci 119(3):562–566. https://doi.org/10.18520/cs/v119/i3/562-566
Jia B, Zhou G (2018) Growth characteristics of natural and planted Dahurian larch in northeast China. Earth Syst Sci Data 10:893–898. https://doi.org/10.5194/essd-10-893-2018
Jones RH, Sharitz RR, Dixon PM et al (1994) Woody plant regeneration in four floodplain forests. Ecol Monogr 64(3):345–367. https://doi.org/10.2307/2937166
Juřička D, Kusbach A, Pařílková J et al (2019) Evaluation of natural forest regeneration as a part of land restoration in the Khentii massif, Mongolia. J for Res. https://doi.org/10.1007/s11676-019-00962-5
Karsten RJ, Meilby H, Larsen JB (2014) Regeneration and management of lesser known timber species in the Peruvian Amazon following disturbance by logging. For Ecol Manag 327:76–85. https://doi.org/10.1016/j.foreco.2014.04.035
Khamyong S, Lykke AM, Seramethakun D, Barford AS (2003) Species composition and vegetation structure of an upper montane forest at summit of Mt. Doi Inthanon, Thailand. Nord J Bot 23(1):83–97. https://doi.org/10.1111/j.1756-1051.2003.tb00371.x
Khan ML, Rai JPN, Tripathi RS (1987) Population structure of some tree species in disturbed and protected subtropical forests of north-east India. Acta Oecol 8(3):247–255
Kumar R, Shamet GS, Mehta H et al (2016) Regeneration complexities of Pinus gerardiana in dry temperate forests of Indian Himalaya. Environ Sci Pollut Res 23(8):7732–7743. https://doi.org/10.1007/s11356-015-6010-5
Lavin M, Pennington RT, Klitgaard BB et al (2001) The dalbergioid legumes (Fabaceae): delimitation of a pantropical monophyletic clade. Am J Bot 88(3):503–533. https://doi.org/10.2307/2657116
Li Y-Q, Deng X-W, Huang Z-H et al (2015) Development and evaluation of models for the relationship between tree height and diameter at breast height for Chinese-Fir plantations in subtropical China. PLoS ONE 10(4):e0125118. https://doi.org/10.1371/journal.pone.0125118
Löf M, Madsen P, Metslaid M et al (2019) Restoring forests: regeneration and ecosystem function for the future. New for 50:139–151. https://doi.org/10.1007/s11056-019-09713-0
Luo M, Zheng X, Du Y (2017) Natural regeneration of an artificial platycladus orientalis stand in Beijing. Nat Environ Pollut Technol 16(1):287–293
Lykke AM (1998) Assessment of species composition change in savanna vegetation by means of woody plants’ size class distributions and local information. Biodivers Conserv 7(10):1261–1275. https://doi.org/10.1023/A:1008877819286
Malik AR, Shamet GS, Butola JS (2012) Natural regeneration status of Chilgoza Pine (Pinus gerardiana Wall) in Himachal Pradesh, India: an endangered pine of high edible value. Appl Ecol Environ Res 10(3):365–373
Malik ZA, Bhatt AB (2016) Regeneration status of tree species and survival of their seedlings in Kedarnath Wildlife Sanctuary and its adjoining areas in Western Himalaya, India. Trop Ecol 57(4):677–690
Maua JO, MugatsiaTsingalia H, Cheboiwo J, Odee D (2020) Population structure and regeneration status of woody species in a remnant tropical forest: a case study of South Nandi forest. Kenya Glob Ecol Conserv 21:e00820. https://doi.org/10.1016/j.gecco.2019.e00820
MoHA (2011) Census 2011. Office of the Registrar General and Census Commissioner, Ministry of Home Affairs, Government of India. New Delhi.
Mokake SE, Chuyong GB, Egbe AE et al (2018) Natural regeneration of some commercial timber tree species following selective logging in a semi deciduous forest in the east region of Cameroon. J Bio Env Sci 12(1):22–39
Mugasha WA, Mwakalukwa EE, Luoga E et al (2016) Allometric models for estimating tree volume and aboveground biomass in lowland forests of Tanzania. Int J for Res. https://doi.org/10.1155/2016/8076271
Nerlekar AN, Kamath V, Saravanan A, Ganesan R (2019) Successional dynamics of a regenerated forest in a plantation landscape in Southern India. J Trop Ecol 35(2):57–67. https://doi.org/10.1017/S0266467418000445
Nogueira Junior LR, Engel VL, Parrotta JA et al (2014) Allometric equations for estimating tree biomass in restored mixed-species Atlantic Forest stands. Biota Neotrop 14(2):e20130084. https://doi.org/10.1590/1676-06032013008413
Pala NA, Negi AK, Gokhale Y et al (2012) Diversity and regeneration status of Sarkot Van Panchayat in Garhwal Himalaya, India. J for Res 23(3):399–404. https://doi.org/10.1007/s11676-012-0276-y
Paul A, Khan ML, Das AK (2019) Population structure and regeneration status of rhododendrons in temperate mixed broad-leaved forests of western Arunachal Pradesh, India. Geol Ecol Landsc 3(3):168–186. https://doi.org/10.1080/24749508.2018.1525671
Peters CM (1994) Sustainable harvest of non-timber plant resources in tropical moist forest: an ecological primer. Biodiversity Support Programme, Washington DC
Poorter L, Bongers F, van Rompaey RSAR, de Klerk M (1996) Regeneration of canopy tree species at five sites in West African moist forest. For Ecol Manag 84:61–69
Prasad PRC, Reddy CS, Raza SH, Dutt CBS (2008) Population structure, age gradations, and regeneration status of pterocarpus dalbergioides Roxb., an endemic species of Andaman Islands, India. Pacif J Sci Technol 9(2):667–673
Rahman MH, Khan MASA, Roy B, Fardusi MJ (2011) Assessment of natural regeneration status and diversity of tree species in the biodiversity conservation areas of Northeastern Bangladesh. J Res 22(4):551–559. https://doi.org/10.1007/s11676-011-0198-0
Rahman MR, Rahman MM, Chowdhury MA (2020) Assessment of natural regeneration status: the case of Durgapur hill forest, Netrokona, Bangladesh. Geol Ecol Landsc 4(2):121–130. https://doi.org/10.1080/24749508.2019.1600911
Rao PSN (2000) The Andaman red-wood tree Pterocarpus dalbergioides Roxb.—an economic timber plant. Bull. Bot. Surv. India 42(1–4):149–154
Sagar R, Singh JS (2004) Local plant species depletion in a tropical dry deciduous forest of northern India. Environ Conserv 31(1):55–62. https://doi.org/10.1017/S0376892904001031
Saslis-Lagoudakis CH, Klitgaard BB, Forest F et al (2011) The use of phylogeny to interpret cross-cultural patterns in plant use and guide medicinal plant discovery: an example from Pterocarpus (Leguminosae). PLoS ONE 6(7):e22275
Saxena AK, Singh JS (1984) Tree population structure of certain Himalayan forest associations and implications concerning their future composition. Vegetatio 58(2):61–69. https://www.jstor.org/stable/20146141.
Saxena AK, Singh SP, Singh JS (1984) Population structure of forests of Kumaun Himalaya: implications for management. J Environ Manage 19:307–324
Shankar U (2001) A case of high tree diversity in a Sal (Shorea robusta)-dominated lowland forest of Eastern Himalaya: floristic composition, regeneration and conservation. Curr Sci 81(7):776–786
Sharma CM, Mishra AK, Tiwari OP et al (2018) Regeneration patterns of tree species along an elevational gradient in the Garhwal Himalaya. Mt Res Dev 38(3):211–219. https://doi.org/10.1659/MRD-JOURNAL-D-15-00076.1
Slik JWF, Keßler PJA, van Welzen PC (2003) Macaranga and Mallotus species (Euphorbiaceae) as indicators for disturbance in the mixed lowland dipterocarp forest of East Kalimantan (Indonesia). Ecol Indic 2(4):311–324. https://doi.org/10.1016/S1470-160X(02)00057-2
Tesfaye G, Teketay D, Fetene M (2002) Regeneration of fourteen tree species in Harenna forest, southeastern Ethiopia. Flora 197(6):461–474. https://doi.org/10.1078/0367-2530-1210063
Ward JS, Worthley TE, Smallidge PJ, Bennett KP (2006) Northeastern forest regeneration handbook: a guide for forest owners, harvesting practitioners, and public officials. USDA Forest Service, Newton Square, p 19073
Wei X, Wu H, Meng H et al (2015) Regeneration dynamics of Euptelea pleiospermum along latitudinal and altitudinal gradients: trade-offs between seedling and sprout. For Ecol Manag 353:232–239. https://doi.org/10.1016/j.foreco.2015.06.004
Winfield K, Scott M, Grayson C (2016) CITES CoP17 Information Document 48 - Global status of Dalbergia and Pterocarpus rosewood producing species in trade. Technical Report.
Acknowledgements
This study is a part of the project entitled “Regeneration and Molecular Characterization of Andaman Padauk (Pterocarpus dalbergioides)” funded by Department of Biotechnology, Ministry of Science & Technology, Government of India [Grant Number BT/PR29179/FCB/125/7/2018]. Authors are thankful to Department of Environment & Forests, Andaman & Nicobar Administration for necessary support in data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Peter Annighoefer.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jaisankar, I., Varadan, R.J., Jerard, B.A. et al. Natural regeneration potential of Andaman Padauk (Pterocarpus dalbergioides) in sustaining the tropical forests of Andaman & Nicobar Islands, India. Eur J Forest Res 141, 535–546 (2022). https://doi.org/10.1007/s10342-022-01458-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-022-01458-4