Abstract
We analyzed vein and stomatal traits (vein density and vein volume per leaf area, stomatal density, stomatal pore length and pore index) and their relationships with other morphological traits [leaf area, leaf thickness and leaf mass per unit area (LMA)] of three co-occurring Mediterranean tree species with contrasting leaf habits [Quercus faginea Lam., Q. suber L. and Q. ilex L. subsp. ballota (Desf.) Samp.]. The results showed that leaf size, thickness and LMA varied among the species in parallel with the differences in leaf longevity. By contrast, the traits most related to water use showed inconsistent differences among the three species. Stomatal pore index was lowest in the species with intermediate leaf life span. The species with longest leaf longevity had highest vein density but minimum vein volume per area. Vein and stomatal traits also varied independently from other leaf traits within each species. The absence of association between LMA and vein volume suggests that a large LMA is the result of the accumulation of other tissues, and not necessarily veins. We concluded that in contrast with most economic leaf traits that tend to vary in parallel with leaf life span, traits related to water use varied inconsistently with leaf duration in the three species studied. Stomatal and vein traits, in addition, were not related to the maximum stomatal conductance of the different species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Leaves are the most important organs for plant growth and, as a consequence, leaf traits, which determine leaf performance, exhibit dramatic changes among different species and in response to environmental gradients (Roth-Nebelsick et al. 2001; Brodribb et al. 2010; Zhang et al. 2018). Amid the most important leaf traits, besides form and size, are vein patterns, which are responsible for at least two essential functions. Veins allow the transport of water for photosynthesis and transpiration, as well as nutrients, photosynthate and molecular signals to the rest of the plant (Brodribb et al. 2007, 2010; Sack et al. 2012; Sack and Scoffoni 2013). Additionally, leaf veins provide mechanical support, allowing the plant to display its leaves toward light, and play a key role in defensive functions against herbivores and environmental stressors (Katifori et al. 2010; Méndez-Alonzo et al. 2013; Sack and Scoffoni 2013). Some traits within the leaf vein system have essential roles, which is the case of vein length per unit leaf area (also known as vein density) that contributes to determine water transport capacity and, therefore, photosynthesis and tolerance to drought (Brodribb et al. 2007, 2010; Scoffoni et al. 2011). Therefore, identifying the venation characteristics of different species is crucial for understanding differences in productivity, competitive success and distribution patterns (Brodribb et al. 2005; Hao et al. 2008). Given their importance for maintaining leaf water status and conditioning tolerance to drought (Roth-Nebelsick et al. 2001; Sack and Holbrook 2006), leaf vein traits may also be fundamental in the responses of different species to climate change. It is not surprising, therefore, that the interest in describing leaf venation characteristics and the consequences they have in different plant species and environments has considerably grown in recent years (Brodribb et al. 2007; Pérez-Harguindeguy et al. 2013; Sack and Scoffoni 2013).
In Mediterranean-climate regions, two types of oaks have been recognized: the evergreen sclerophyllous type and the winter-deciduous malacophyllous type. The evergreen type has been commonly associated to the archetype of the Mediterranean-type climate (Gil-Pelegrín et al. 2017), but in fact, the number of deciduous species present in regions with Mediterranean climates in the Northern Hemisphere is almost as high as that of evergreens (Escudero et al. 2017). Between evergreen and deciduous Mediterranean oak species, there are significant differences in leaf chemistry, morphology and physiology (Escudero et al. 2017; Gil-Pelegrín et al. 2017). One of the most relevant differences between both leaf habits is that stomatal conductance and transpiration rates tend to be much higher in deciduous species, which, under the water-limited conditions typical of Mediterranean regions, may constitute a fundamental determinant of the distribution and coexistence of both oak types. We should expect that the higher transpiration rates in deciduous species would be accompanied by parallel changes in leaf hydraulic traits, such as stomatal patterns and the venation system needed to guarantee water supply to the transpiring leaves (Gil-Pelegrín et al. 2017). However, to our knowledge, no studies have addressed the interspecific differences in venation and stomatal characteristics among Mediterranean oak species under the same environmental conditions, despite the importance of these traits for productivity and leaf survival under drought-stressed conditions.
In recent years, there has been much debate about the role of leaf venation in relation to the “leaf economics spectrum” (LES), a series of correlations between diverse leaf traits (including the rate of photosynthesis, foliar nitrogen concentration, leaf dry mass per area (LMA) and leaf longevity) that control carbon, nitrogen and water usage (Wright et al. 2004). Since both leaf economic and hydraulic traits are crucial for proper leaf functioning, a linkage between these two suites of traits has been postulated by different authors (Blonder et al. 2011; Reich 2014). However, most of the experimental evidence available does not support this, since no significant correlations of vein traits with economic traits across species have been reported in several studies (Dunbar-Co et al. 2009; Nardini et al. 2012; Sack et al. 2013; Kawai and Okada 2020). Accordingly, Sack et al. (2013) proposed the so-called “flux trait network” hypothesis, according to which vein density has important influences on hydraulic conductance, stomatal conductance and photosynthetic rate independently of LMA (Sack et al. 2003; Sack and Holbrook 2006; Hao et al. 2010). Given the linkage between the patterns of water use and the differences in leaf life span in Mediterranean oaks, the independence between economic and hydraulic traits seen by other authors in other sets of species seems counterintuitive and should be better investigated for the oak species typical of Mediterranean environments.
A basic assumption of comparative plant ecology is that the between-species differences in functional traits are larger than intraspecific differences (Jung et al. 2010). Consequently, most studies examining correlations between leaf traits have assigned a single value to each trait of each species, thereby ignoring intraspecific variation (Siefert et al. 2014). However, recent studies have revealed that intraspecific variation is a major component of trait variation, both within and among communities (Richardson et al. 2013), and that intraspecific trade-offs may be different from interspecific trade-offs (Albert et al. 2011). Some of the trends in the relationships between vein traits and other leaf traits seen across different species are not clear when different individuals of one population are compared (Uhl and Mosbrugger 1999; Roth-Nebelsick et al. 2001). Furthermore, the intraspecific relationships seem to be species specific (Uhl and Mosbrugger 1999). Considering intraspecific variability may be especially important in species-poor communities, such as Mediterranean woodlands, where intraspecific variability may account for a large proportion of total community functional diversity (Siefert et al. 2015).
In the present work, we analyze the veins, stomatal and other morphological traits of the leaves of three Mediterranean Quercus species [Quercus faginea Lam., Q. suber L. and Q. ilex L. subsp ballota (Desf.) Samp.] coexisting in the same environment. The three species are among the oaks most widely distributed in the Mediterranean contour and exhibit contrasting leaf habits and leaf life spans. Numerous works have described important differences among these species in various aspects of foliar morphology and physiology associated with their differences in leaf life span (Mediavilla and Escudero 2003a; Peguero-Pina et al. 2009; Vaz et al. 2010), which may be considered as a manifestation of the trade-offs involved in the LES (Mediavilla and Escudero 2003a). Alternatively, the differences between deciduous and evergreen Mediterranean species have been frequently interpreted as different strategies to cope with water limitation (Gil-Pelegrín et al. 2017), which should involve differences in the traits most related to water flux between both leaf habits. Our hypothesis was that the differences in maximum stomatal conductances among Mediterranean tree species differing in leaf life span (Mediavilla and Escudero 2003b) should be associated with differences in stomatal and vein traits. As a secondary objective, we intend to verify whether the relationships between leaf traits at the interspecific level are also observed among individuals of a single species and whether the same intraspecific tendencies are shared by our three Quercus species.
Materials and methods
Study species and area
Quercus suber L. and Q. ilex L. subsp ballota (Desf.) Samp. are evergreen trees with 15 and 24 months of mean leaf life span, respectively (data taken from Mediavilla and Escudero 2003a), whereas Q. faginea Lam. is a deciduous tree with a mean leaf longevity of around 7 months. The three species were selected on a site (41°08′49.02″ N, 5°47′17.38″ W) situated near the town of Salamanca, at an altitude of 830 m above sea level. The site is a savannah-like formation constituted by an open tree layer dominated by different Quercus species and some accompanying shrubs. The climate in the study area is cold Mediterranean [Csb, according to the classification of Köppen (Peel et al. 2007)]. The mean annual temperature ranges around 11–13 °C with an average annual rainfall around 500–600 mm. A summer drought period is always present. The soils, dystric Cambisols, are poor in organic matter and nutrients, with a low pH and medium/low water retention capacity (Dorronsoro 1992).
Morpho-anatomical leaf traits
Several branches with sun leaves were taken from each of ten individuals per species at mid-height at the periphery of the crown. The sampling was carried out in mid-July 2017. The samples were immediately taken to the laboratory, and in the two evergreen species, the branches were separated into annual segments of different age classes. Only leaves of the current year were included in the study. The list of traits analyzed is reported in Table 1.
The morphological traits were analyzed in 50 leaves randomly selected from each species (5 leaves per tree). Leaf thickness (LT, μm) was measured with a digital micrometer (Digimatic micrometer, Mitutoyo, Japan) as a mean of three measurements taken at random positions on each leaf, avoiding the main ribs. The projected leaf area (LA, cm2) was determined using a leaf area meter (Delta-T Devices LTD, Cambridge, UK). The samples were oven-dried at 70 °C to constant mass, and the dry mass per leaf was determined. From the data obtained, the leaf dry mass per area (LMA, gm−2) was calculated.
An additional subsample (two leaves per tree, 20 leaves per species) was used for measuring stomatal traits. Only the abaxial epidermis was analyzed, since no stomata were found on the adaxial side. The leaf surface was shaved using a razor blade and sectioned into 0.5 cm2 vein-free portions that were then fixed in glutaraldehyde 2.5% in 0.1 M phosphate buffer and pH 7.4. At the Microscopy Service of the University of Salamanca, the samples were dehydrated and metallized, and then some photographs were taken with a scanning electron microscope (SEM EVO HD25, Carl Zeiss Microscopy, Germany) in two fields for each leaf fraction. To guarantee that stomatal density estimates were comparable for the different species, we verified that the samples for SEM examination were prepared by the Microscopy Service to minimize structural damage and sample distortion. A secondary fixation step was omitted, since usual secondary fixatives can extract protein components that compromise structural integrity. Also, the dehydration of the sample included a step of critical point drying to preserve morphology and assure a consistent preservation of tissue dimensions and structure between samples. From the images obtained, we calculated stomatal density (SD, number mm−2). Stomatal pore length (SPL, μm) was measured as the length in micrometers between the junctions of the guard cells at each end of the stomata (Maherali et al. 2002). Finally, stomatal pore index (SPI, %) was calculated as stomatal density × the square of stomatal pore length × 10−4 (Sack et al. 2003; Tian et al. 2016).
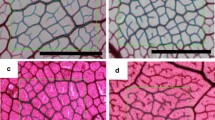
Vein traits were measured in two additional leaves per tree. The samples were fixed for 5 h in FAA (formalin: acetic acid: 50% ethanol, 5:5:90) and rinsed in 50% ethanol and distilled water. Leaves were subsequently cleared with 2 M NaOH and washed with a bleach solution and 50% and 70% ethanol (2 h each). Finally, the samples were stained with a 0.01% (w/v) safranine solution in 70% ethanol for 3 min and extensively rinsed in water. Images obtained with a Leica M205 FA stereo microscope equipped with a Leica DFC495 camera (Leica Microsystems, Germany) were analyzed with ImageJ software (http://rsb.info.nih.gov/ij/; Abràmoff et al. 2004). We measured the total length of the mid-vein (first order). The density of the second-, third- and minor-order veins was averaged for three subsampled regions, taken centrally in the top, middle and bottom third of the leaf. We included the smallest secondary veins branching from the mid-vein as part of the third-order vein category, since the diameters of both were similar, thus using the vein diameter at the point of origin of the vein as the basic criterion in determining the vein order (Leaf Architecture Working Group 1999; Roth-Nebelsick et al. 2001). Vein density was calculated as the length measured for each vein order divided by leaf area. The total major vein density (VD maj) was the sum of the first-, second- and third-order vein densities, the rest corresponding to the density of minor veins (VD min). Vein diameters (measured halfway along their length, perpendicular to the vein) were measured for each vein order by averaging six measurements (two taken centrally in the top, middle and bottom third of the leaf). Vein volume per leaf area (TVV) was determined by multiplying the vein density by π × the square of half the diameter (Sack et al. 2012). For each trait, a value for each tree was obtained as an average of the data corresponding to the two leaves analyzed in each case.
Interveinal distances and vertical distance between veins and the abaxial epidermis were measured in transverse sections of four leaves taken from five trees per species. Square pieces (5 × 5 mm), taken from the middle part of the leaves, were fixed in formaldehyde, acetic acid and ethanol (FAA) and dehydrated in an ethanol series. The material was then embedded in methacrylate liquid for 72 h. Transverse leaf sections (5–6 μm thick) were made on a HM 350S Rotary Microtome (Microm International GmbH, Germany) and mounted on glass slides. Images of each sample were obtained using a digital camera (Nikon Sight DS-smc, Nikon Instruments INC, USA) mounted on a microscope (Nikon Eclipse 90i, Nikon Instruments INC, USA).
Data analysis
A mean value for each trait in each species was obtained as an average of the data corresponding to the ten trees analyzed in each case. Data were expressed as mean ± standard error. One-way analysis of variance was used to establish significant differences among species, followed by post hoc Student–Newman–Keuls test. Normality was checked with Kolmogorov–Smirnov tests, and homoscedasticity was confirmed with the Levene test. The relationships between leaf traits at intraspecific level were explored by correlation analyses using the mean values obtained for each tree as data points. To analyze the data, we used the SPSS version 23 statistical package (SPSS Inc., Chicago, IL).
Results
Interspecific differences in leaf traits
The deciduous Q. faginea was the species with largest leaf size and lowest LMA and thickness, while the opposite was observed for Q. ilex (Fig. 1). The interspecific differences in vertical distance between veins and epidermis were parallel to the differences in leaf thickness, with Q. ilex exhibiting the greatest separation. Q. ilex was the species with the lowest SD, but larger SPL, which resulted in SPI values intermediate between the two other species, without significant differences with any of them (Fig. 1). Between Q. faginea and Q. suber, there were no significant differences in SD, but SPL was lower in Q. suber. Thus, Q. faginea reached the highest SPI, while Q. suber reached the lowest value, with significant differences between both species (Fig. 1).
The smaller size of the leaves of Q. ilex was accompanied by significantly lower mean diameters of all types of veins analyzed (Fig. 2), whereas the length per unit leaf area, and therefore TVD, was significantly higher than that of the two other Quercus (Fig. 3). Between Q. suber and Q. faginea no differences were observed for the average diameter of the second-, third- and minor-order veins. However, there were differences in the first-order veins, up to 22% thicker in Q. suber than in the deciduous species and by more than 50% in Q. suber with respect to the leaves of Q. ilex (Fig. 2). There were no differences in the density of any of the types of veins between Q. faginea and Q. suber (Fig. 3). The reduction in the vein diameter in Q. ilex with respect to the two other species was much higher than the increase in vein density. Therefore, finally TVV reached significantly lower values in the species with greatest leaf longevity (Fig. 3). Between Q. faginea and Q. suber, the differences were limited to the TVV, with significantly higher values in the latter species.
Intraspecific leaf traits correlations
Among the different leaf traits, LA and SPI exhibited strong intraspecific variability in the three species (Table 2), with coefficients of variation around 20–40% for the mean values of the different individuals. LMA and LT were the traits exhibiting minimum intraspecific variability (less than 10% in the three species). At the intraspecific level, no correlation was observed between the LMA (or thickness, not shown) and stomatal features or vein traits in any of the study species (Table 3). Among the morphological features, only leaf size correlated with some of the vein traits analyzed (Table 3), but not with the stomatal traits. A larger leaf size was associated with greater diameter and lower density (not shown) of the first-order veins, but finally, there was no correlation between leaf size and the TVD, or with the TVV (Table 3).
No relationship between the density of major and minor veins or between the volume of both types of veins was observed in the leaves of our species. Total density was determined mainly by minor veins, while total volume was determined by vein diameter and therefore was most conditioned by the major veins (Table 3). However, among the leaves of a single species, the differences in the TVV depended on the differences in the diameters of the first-order veins. In Q. faginea, the diameter of the three types of major veins correlated positively with TVV, because in this species the diameters of the three types of veins were correlated. In fact, the effect of the diameter of the second- and third-order veins on total volume disappeared in multiple regression for a constant diameter of the first-order veins (data not shown).
Discussion
Interspecific trends in vein and stomatal traits
Among our three study species, we did not find a definite trend in the stomatal and venation traits associated with the differences in leaf life span or leaf habit, in contrast with the changes in morphological traits, such as leaf size, thickness or LMA. Although Q. ilex, the species with longest leaf life span, showed the highest TVD, the evergreen Q. suber shared a large number of vein traits with the deciduous Q. faginea, whereas it showed venation characteristics different to those of Q. ilex. Something similar happened with stomata, whose density or pore index changed independently from the changes in leaf habit and leaf life span. These results would be in line with other studies that found no significant correlation across species of TVD with LMA (Dunbar-Co et al. 2009; Nardini et al. 2012; Sack et al. 2013; Kawai and Okada 2020) or stomatal density and stomatal pore index with structural leaf traits (Beerling and Kelly 1996; Zhang et al. 2012). By contrast, in agreement with other studies, vein density was significantly greater in the species with smallest individual leaf area, a trend probably due to developmental patterns during leaf growth (Sack et al. 2012).
At the interspecific level, our results do not support, therefore, the presumed association of a high vein density with large LMA (Blonder et al. 2011). The assertions of Blonder et al. (2011) seem to assume that veins make a positive contribution to leaf density (Sack et al. 2013), such that a high volume of veins in the leaf would contribute to increasing LMA. In this study, however, although Q. ilex maintained the highest length of veins per unit leaf area, it also had significantly lower mean vein diameters than the two other species, which resulted in the lowest TVV. Accordingly, the proportion of the leaf area occupied by veins tended to be lower in the species with largest LMA, which suggests that a large LMA is the result of the accumulation of other tissues, and not necessarily veins (Sack et al. 2013; John et al. 2017). By contrast, the leaves of Q. suber showed the highest TVV among the three species, despite having a lower TVD than in Q. ilex, which reveals that, as other authors have pointed out (Sack et al. 2013), the total volume of veins per unit of leaf area depends more on vein thickness than on vein density. In any case, the lack of any correspondence between vein volume per unit of leaf area and LMA of the different species suggests that the contribution of vascular tissues to increasing LMA is less than previously supposed. Several authors have associated a higher density of veins with a higher lignin content (Mooney and Gulmon 1982; Lambers and Poorter 1992), due to the participation of lignin in the construction of the vascular tissues. For the same species, we did not observe any relationship between lignin concentration and leaf life span (Mediavilla et al. 2008), which also supports the lack of correspondence between leaf life span and the fraction of leaf area occupied by veins found in this study.
The lack of consistent differences in vein and stomatal traits among our species was unexpected in view of the clear differences in the patterns of water use among them. Maximum stomatal conductance and stomatal sensitivity to vapor pressure deficit changed in a consistent manner among the three species (Mediavilla and Escudero 2003b), with Q. faginea sustaining relatively high stomatal conductances under conditions of strong evaporative demand, while Q. ilex exhibited the opposite behavior. We expected that the vein system and the stomatal traits of Q. faginea would exhibit significant differences with respect to the two evergreen species, since numerous studies have found significant correlations of stomatal conductance with vein density and with stomatal density or SPI (Sack et al. 2013). By contrast, the high vein density of Q. ilex leaves and the absence of significant differences between Q. faginea and Q. ilex in SPI imply that the leaf traits most related to water movements showed inconsistent changes with respect to the gas-exchange rates of the different species, which at first sight seems counterintuitive. Different authors (Brodribb et al. 2007; Rockwell and Holbrook 2017) have emphasized the limiting role of diffusion out of the vascular tissues for water movements. The pathlength for water movements through the mesophyll should depend on interveinal distance (IVD), which decreases with increasing vein density, but also on the vertical vein-to-epidermis distance (see Eq. 5 in the paper by Rockwell and Holbrook 2017). Mean IVD values, as measured in the leaf transverse sections, were 204.4, 205.4 and 176.3 µm for Q. faginea, Q. suber and Q. ilex, respectively. By contrast, the distance between veins and epidermis was larger in Q. ilex than in the two other species (Fig. 1). Applying Eq. 5 of Rockwell and Holbrook (2017) to our data, we estimated an effective length for water transport from the vein system to the abaxial epidermis of 71.7, 75.5 and 78.6 µm, respectively, for Q. faginea, Q. suber and Q. ilex. Accordingly, the smaller leaf thickness of Q. faginea more than compensated the relatively large interveinal distance in this species, thus probably reducing the resistance to water movements through the mesophyll. Similarly, for Q. ilex, the larger effective distance for water transport out of the vascular tissues, combined with the smaller stomatal density in this species, may be more in agreement with its smaller maximum stomatal conductance.
We postulate that the independence between the veins and other leaf traits, in association with the differences in leaf phenology among our three oak species, may facilitate coexistence in a Mediterranean environment. The leaves of Q. ilex reach an average life span of approximately 2 years, which may explain their strong reinforcement with a large thickness and LMA. Both traits imply costs in terms of a lower instantaneous photosynthesis per unit leaf mass, which would be compensated by a long leaf duration (van Ommen Kloeke et al. 2012). The high temperatures and the drought stress during summer are important factors for the growth and survival of tree species under Mediterranean climate conditions (Flexas and Medrano 2002). A large vein density and reduced leaf size may play a key role in reducing the hydraulic vulnerability and increasing leaf drought tolerance (Sack et al. 2008; Scoffoni et al. 2011), which is especially important for Q. ilex leaves that have to endure at least two dry seasons. In addition, a high density of veins is also advantageous to reduce the damages due to herbivore attacks (Roth-Nebelsick et al. 2001; Sack et al. 2008). Leaf venation patterns contribute to determine leaf mechanical strength (Roth-Nebelsick et al. 2001; Méndez-Alonzo et al. 2013; Sack and Scoffoni 2013), which in turn affects herbivory levels (Coley 1983; Pérez-Harguindeguy et al. 2003). Additionally, reticulation can enhance damage resilience to herbivory or environmental stressors by providing alternate flow pathways that maintain hydraulic functioning (Sack et al. 2008; Katifori et al. 2010). Therefore, the higher TVD and smaller size of the leaves of Q. ilex with respect to the two other species would be crucial for increasing leaf life span and pay-back time. However, investment in veins also involves a substantial cost in terms of carbon and nutrients, since veins have higher construction costs than other leaf tissues (Lambers and Poorter 1992; Sack et al. 2013). In addition, the vascular tissues contribute to displace other tissues with greater photosynthetic activity (Niinemets 1999). According to our results, in Q. ilex, the large total length of veins per unit leaf area is compensated by a low vein diameter, causing the proportion of the leaf area occupied by veins to be in fact lower than that of the other species. This should contribute to reducing the costs associated with the investment in vascular tissues.
Quercus suber is, like Q. ilex, an evergreen species. However, in Q. suber, leaf mortality occurs massively coinciding with the development of a new leaf cohort during spring (Del Río-García et al. 2015), so that the average duration of the leaves is just around 14 months (Mediavilla and Escudero 2003a). This may explain why Q. suber presents venation characteristics more similar to those of the deciduous species than to those of Q. ilex. Although a lower TVD would contribute to reducing tolerance to drought, the leaves of Q. suber only have to cope with one single period of summer drought, since most of the leaves of this species are shed at the end of spring of the year following their emergence, before a new summer period. In addition, Q. suber is the species, among the three studied, that had stomata of smaller size and in greater number per unit area. A large stomatal density combined with small stomata allow more rapid changes in stomatal conductances in response to changes in atmospheric conditions, which results in greater water use efficiency (Aasamaa et al. 2001; Franks et al. 2009). The thick central vein of Q. suber also facilitates leaf flexion that would allow reduce transpiration (Cooley et al. 2004). Additionally, a reinforced midrib contribute to increase protection and to compensate the relatively low first-order vein density of Q. suber leaves (Sack et al. 2008). Despite the lack of significant differences in TVD, herbivory rates were significantly lower in Q. suber than in Q. faginea, according to a previous work carried out at the same site (Mediavilla et al. 2018), which might reflect the importance of a reinforced midrib besides other physical and chemical attributes to avoid herbivory injury (Delaney and Higley 2006). In conclusion, venation and stomatal traits seem to be more responsive to differences in phenology in our set of species than to differences in leaf habit or in leaf longevity.
Intraspecific variation in leaf traits
In most studies of leaf traits, species are described by mean trait values, which implies that intraspecific trait variability can be neglected compared to interspecific variability (Albert et al. 2011). In view of our results, the extent of intraspecific variability is considerably different for the different traits analyzed. This has been a common finding in previous studies (Siefert et al. 2015), but the ranking of the different traits with respect to the extent of intraspecific variability differ according to different studies. LMA has been reported as one leaf trait exhibiting strong intraspecific variability (Siefert et al. 2015; Messier et al. 2017) in contrast with the similar values observed in the present study for the different individuals. Probably, the strong variation in LMA observed in other studies is an effect of differences in irradiance gradients within the canopy (Messier et al. 2017). However, when measured in sun leaves, LMA seems to be a “robust” trait (Albert et al. 2011), which allows characterizing the different species. In contrast, in our set of data, individual leaf area was an extremely variable trait, since it varied more than 2-fold across the different individuals of a single species. This result is at variance with respect to the trends reported by Siefert et al. (2015), which found a low intraspecific variability for leaf area. The remainder traits analyzed in the present study also exhibited relatively low levels of intraspecific variability, with the exception of SPI, which showed levels of variation similar to those of LA. At the intraspecific level, then, the vein traits appear to be quite conservative, at least within a single population, possibly because of constraints derived from developmental or functional dependence with respect to other important leaf features (Sack et al. 2008).
Within a single species, no significant correlations were seen between stomatal and vein traits with respect to other leaf traits. In particular, the intraspecific differences in LMA and thickness were not related to changes in the vein or stomatal traits, which would confirm that also at the intraspecific level both types of traits can vary independently. Similarly, venation and stomatal traits varied independently. Again, this result seems counterintuitive and contrasts with the numerous studies that report a coordination between vein and stomatal traits (Brodribb et al. 2013; Sack et al. 2013). Larger leaf size was accompanied by increases in the thickness of the central veins in the three species, which may be explained by developmental trends, since the diameter of first-order veins can increase during most of the leaf expansion (Sack et al. 2012). However, despite the strong intraspecific variation in leaf area, variations in TVD were independent from leaf size, corroborating the results obtained by Uhl and Mosbrugger (1999) in Q. petraea. Similarly, stomatal traits varied independently of the variations in leaf size. One of the most important ecological implications of leaf size is that smaller leaves tend to have a thinner boundary layer, which enables more rapid convective cooling (Scoffoni et al. 2011). According to our results, leaf area exhibit strong intraspecific variations while changing independently from the variation in vein and stomatal traits. This means that different individual trees may experience strong differences in leaf temperature and maximum potential transpiration rates, depending on the combination of leaf size and stomatal traits characteristic of each individual, which probably is a response of each tree to high finescale soil heterogeneity (Jung et al. 2014). The independence among different traits allows for greater plasticity in performance of each plant individual in a particular set of micro-environmental conditions (Li et al. 2015).
Conclusions
According to our results, the leaf economics and the vein and stomatal traits can vary independently both across and within different species. Different authors have proposed that correlations between traits often vary between groups of species or environments (Yin et al. 2018; Liu et al. 2019) and that it is precisely the fact that more functional leaf traits vary independently which may allow alternative strategies, thus facilitating species coexistence (Adler et al. 2013; Li et al. 2015). Our study shows that three species of the same genus exhibit a different combination of traits related to water and carbon economy, as well as differences in leaf life span and leaf phenology. These different combinations of traits provide the species with a balance of costs-benefits that allows them to coexist under the same environmental conditions.
References
Aasamaa K, Sober A, Rahi M (2001) Leaf anatomical characteristics associated with shoot hydraulic conductance, stomatal conductance and stomatal sensitivity to changes of leaf water status in temperate deciduous trees. Aust J Plant Physiol 28:765–774
Abràmoff M, Magalhães P, Ram S (2004) Image processing with ImageJ. Biophoton Intern 11:36–42
Adler PB, Fajardo A, Kleinhesselink AR, Kraft NJ (2013) Trait-based tests of coexistence mechanisms. Ecol Lett 16:1294–1306
Albert CH, Grassein F, Schurr FM, Vieilledent G, Violle C (2011) When and how should intraspecific variability be considered in trait-based plant ecology? Perspect Plant Ecology 13:217–225
Beerling DJ, Kelly CK (1996) Evolutionary comparative analyses of the relationship between leaf structure and function. New Phytol 134:35–51
Blonder B, Violle C, Bentley LP, Enquist BJ (2011) Venation networks and the origin of the leaf economics spectrum. Ecol Lett 14:91–100
Brodribb TJ, Holbrook NM, Zwieniecki MA, Palma B (2005) Leaf hydraulic capacity in ferns, conifers and angiosperms: impacts on photosynthetic maxima. New Phytol 165:839–846
Brodribb T, Feild T, Jordan G (2007) Leaf maximum photosynthetic rate and venation are linked by hydraulics. Plant Physiol 144:1890–1898
Brodribb TJ, Feild TS, Sack L (2010) Viewing leaf structure and evolution from a hydraulic perspective. Funct Plant Biol 37:1–11
Brodribb TJ, Jordan GJ, Carpenter RJ (2013) Unified changes in cell size permit coordinated leaf evolution. New Phytol 199:559–570
Coley PD (1983) Herbivory and defensive characteristics of tree species in a lowland tropical forest. Ecol Monogr 53:209–234
Cooley AM, Reich A, Rundel P (2004) Leaf support biomechanics of neotropical understory herbs. Am J Bot 91:573–581
Del Río-García T, Mediavilla S, Silla F, Escudero A (2015) Differences in the environmental control of leaf senescence of four Quercus species coexisting in a Mediterranean environment. Forest Syst 24(2):e027
Delaney KJ, Higley LG (2006) An insect countermeasure impacts plant physiology: midrib vein cutting, defoliation and leaf photosynthesis. Plant, Cell Environ 29:1245–1258
Dorronsoro F (1992) El medio físico-químico: suelos. In: Gómez-Gutiérrez JM (ed) El Libro de las Dehesas Salmantinas. Junta de Castilla y León, Salamanca, pp 71–124
Dunbar-Co S, Sporck MJ, Sack L (2009) Leaf trait diversification and design in seven rare taxa of the Hawaiian Plantago radiation. Int J Plant Sci 170:61–75
Escudero A, Mediavilla S, Olmo M, Villar R, Merino J (2017) Coexistence of deciduous and evergreen oak species in Mediterranean environments: costs associated with the leaf and root traits of both habits. In: Gil-Pelegrín E, Peguero-Pina JJ, Sancho-Knapik D (eds) Oaks physiological ecology. Exploring the functional diversity of genus Quercus L. Springer, Cham, pp 195–237
Flexas J, Medrano H (2002) Photosynthetic responses of C3 plants to drought. In: Hemantaranjan A (ed) Advances in plant physiology IV. Scientific Publishers, Jodhpur, pp 1–56
Franks PJ, Drake PL, Beerling DJ (2009) Plasticity in maximum stomatal conductance constrained by negative correlation between stomatal size and density: an analysis using Eucalyptus globulus. Plant, Cell Environ 32:1737–1748
Gil-Pelegrín E, Saz MA, Cuadrat JM, Peguero-Pina JJ, Sancho-Knapik D (2017) Oaks under Mediterranean-type climates: functional response to summer aridity. In: Gil-Pelegrín E, Peguero-Pina JJ, Sancho-Knapik D (eds) Oaks physiological ecology. Exploring the functional diversity of genus Quercus L. Springer, Cham, pp 137–193
Hao GY, Hoffmann WA, Scholz FG, Bucci SJ, Meinzer FC, Franco AC, Cao KF, Goldstein G (2008) Stem and leaf hydraulics of congeneric tree species from adjacent tropical savanna and forest ecosystems. Oecologia 155:405–415
Hao GY, Sack L, Wang AY, Cao KF, Goldstein G (2010) Differentiation of leaf water flux and drought tolerance traits in hemiepiphytic and non-hemiepiphytic Ficus tree species. Funct Ecol 24:731–740
John GP, Scoffoni C, Buckley TN, Villar R, Poorter H, Sack L (2017) The anatomical and compositional basis of leaf mass per area. Ecol Lett 20:412–425
Jung V, Violle C, Mondy C, Hoffmann L, Muller S (2010) Intraspecific variability and trait-based community assembly. J Ecol 98:1134–1140
Jung V, Albert CH, Violle C, Kunstler G, Loucougaray G, Spiegelberger T (2014) Intraspecific trait variability mediates the response of subalpine grassland communities to extreme drought events. J Ecol 102:45–53
Katifori E, Szollosi GJ, Magnasco MO (2010) Damage and fluctuations induce loops in optimal transport networks. Phys Rev Lett 104:048704
Kawai K, Okada N (2020) Leaf vascular architecture in temperate dicotyledons: correlations and link to functional traits. Planta 251:17. https://doi.org/10.1007/s00425-019-03295-z
Lambers H, Poorter H (1992) Inherent variation in growth rate between higher plants: a search for physiological causes and ecological consequences. Adv Ecol Res 23:187–261
Leaf Architecture Working Group (1999) Manual of leaf architecture: morphological description and categorization of dicotyledonous and net-veined monocotyledonous angiosperms. Smithsonian Institution, Washington
Li L, McCormack ML, Ma CH, Kong D, Zhang Q, Chen X, Zeng H, Niinemets U, Gua D (2015) Leaf economics and hydraulic traits are decoupled in five species-rich tropical-subtropical forests. Ecol Lett 18:899–906
Liu C, Li Y, Xu L, Chen Z, He N (2019) Variation in leaf morphological, stomatal, and anatomical traits and their relationships in temperate and subtropical forests. Sci Rep 9:5803
Maherali H, Reid CD, Polley HW, Johnson HB, Jachson RB (2002) Stomatal acclimation over a subambient to elevated CO2 gradient in a C3/C4 grassland. Plant, Cell Environ 25:557–566
Mediavilla S, Escudero A (2003a) Photosynthetic capacity, integrated over the lifetime of a leaf, is predicted to be independent of leaf longevity in some tree species. New Phytol 159:203–211
Mediavilla S, Escudero A (2003b) Stomatal responses to drought at a Mediterranean site: a comparative study of co-occurring woody species differing in leaf longevity. Tree Physiol 23:987–996
Mediavilla S, García-Ciudad A, García-Criado B, Escudero A (2008) Testing the correlations between leaf life span and leaf structural reinforcement in 13 species of European Mediterranean woody plants. Funct Ecol 22:787–793
Mediavilla S, Babiano J, Martínez-Ortega MM, Escudero A (2018) Ontogenetic changes in anti-herbivore defensive traits in leaves of four Mediterranean co-occurring Quercus species. Ecol Res 33:1093–1102
Méndez-Alonzo R, Ewers FW, Sack L (2013) Ecological variation in leaf biomechanics and its scaling with tissue structure across three Mediterranean climate plant communities. Funct Ecol 27:544–554
Messier J, McGill BJ, Enquist BJ, Lechowicz MJ (2017) Trait variation and integration across scales: is the leaf economic spectrum present at local scales? Ecography 40:685–697
Mooney HA, Gulmon L (1982) Constraints on leaf structure and function in reference to herbivory. Bioscience 32:198–206
Nardini A, Peda G, La Rocca N (2012) Trade-offs between leaf hydraulic capacity and drought vulnerability: morpho-anatomical bases, carbon costs and ecological consequences. New Phytol 196:788–798
Niinemets Ü (1999) Differences in chemical composition relative to functional differentiation between petioles and laminas of Fraxinus excelsior. Tree Physiol 19:39–45
Peel MC, Finlayson BL, McMahon TA (2007) Updated world map of Köppen-Geiger climate classification. Hydrol Earth Syst Sci 11:1633–1644
Peguero-Pina JJ, Sancho-Knapik D, Morales F, Flexas J, Gil-Pelegrín E (2009) Differential photosynthetic performance and photoprotection mechanisms of three Mediterranean evergreen oaks under severe drought stress. Funct Plant Biol 35:453–462
Pérez-Harguindeguy N, Diaz S, Garnier E et al (2013) New handbook for standardised measurement of plant functional traits worldwide. Aust J Bot 61:167–234
Reich PB (2014) The world-wide ‘fast–slow’ plant economics spectrum: a traits manifesto. J Ecol 102:275–301
Richardson SJ, Allen RB, Buxton RP, Easdale TA, Hurst JM, Morse CW, Smissen RD, Peltzer DA (2013) Intraspecific relationships among wood density, leaf structural traits and environment in four co-occurring species of Nothofagus in New Zealand. PLoS ONE 8(3):e58878
Rockwell FE, Holbrook NM (2017) Leaf hydraulic architecture and stomatal conductance: a functional perspective. Plant Physiol 174:1996–2007
Roth-Nebelsick A, Uhl D, Mosbrugger V, Kerp H (2001) Evolution and function of leaf venation architecture: a review. Ann Bot 87:553–566
Sack L, Holbrook NM (2006) Leaf hydraulics. Annu Rev Plant Biol 57:361–381
Sack L, Scoffoni C (2013) Leaf venation: structure, function, development, evolution, ecology and applications in the past, present and future. New Phytol 198:983–1000
Sack L, Cowan PD, Jaikumar N, Holbrook NM (2003) The ‘hydrology’ of leaves: co-ordination of structure and function in temperate woody species. Plant, Cell Environ 26:1343–1356
Sack L, Dietrich EM, Streeter CM, Sanchez-Gomez D, Holbrook NM (2008) Leaf palmate venation and vascular redundancy confer tolerance of hydraulic disruption. Proc Natl Acad Sci USA 105:1567–1572
Sack L, Scoffoni C, McKown AD, Frole K, Rawls M, Havran JC, Tran T (2012) Developmentally based scaling of leaf venation architecture explains global ecological patterns. Nat Commun 3:1–10
Sack L, Scoffoni C, John GP, Poorter H, Mason CM, Mendez-Alonso R, Donovan LA (2013) How do leaf veins influence the worldwide leaf economic spectrum? Review and synthesis. J Exp Bot 64:4053–4080
Scoffoni C, Rawls M, McKown A, Cochard H, Sack L (2011) Decline of leaf hydraulic conductance with dehydration: relationship to leaf size and venation architecture. Plant Physiol 156:832–843
Siefert A, Fridley JD, Ritchie ME (2014) Community functional responses to soil and climate at multiple spatial scales: when does intraspecific variation matter? PLoS ONE 9(10):e111189
Siefert A, Violle C, Chalmandrier L, Albert CH, Taudiere A, Fajardo A et al (2015) A global meta-analysis of the relative extent of intraspecific trait variation in plant communities. Ecol Lett 18:1406–1419
Tian M, Yu GR, He NP, Hou JH (2016) Leaf morphological and anatomical traits from tropical to temperate coniferous forests: mechanisms and influencing factors. Sci Rep 6:19703
Uhl D, Mosbrugger V (1999) Leaf venation density as a climate environmental proxy: a critical review and new data. Palaeogeogr Palaeoclimatol Palaeoecol 149:15–26
van Ommen Kloeke AEE, Douma JC, Ordoñez JC, Reich PB, van Bodegom PM (2012) Global quantification of contrasting leaf life span strategies for deciduous and evergreen species in response to environmental conditions. Global Ecol Biogeogr 21:224–235
Vaz M, Pereira JS, Gazarini LC, David TS, David JS, Rodrigues A, Maroco J, Chaves MM (2010) Drought-induced photosynthetic inhibition and autumn recovery in two Mediterranean oak species (Quercus ilex and Quercus suber). Tree Physiol 30:946–956
Wright I, Reich P, Westoby M, Ackerly D, Baruch Z, Bongers F et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827
Yin Q, Wang L, Lei M, Dang H, Quan J, Tian T, Chai Y, Yue M (2018) The relationships between leaf economics and hydraulic traits of woody plants depend on water availability. Sci Total Environ 621:245–252
Zhang SB, Guan ZJ, Sun M, Zhang JJ, Cao KF, Hu H (2012) Evolutionary association of stomatal traits with leaf vein density in Paphiopedilum. Orchidaceae. PLoS ONE 7(6):e40080
Zhang L, Yang J, Huang Y, Jia Z, Fang Y (2018) Leaf venation variation and phenotypic plasticity in response to environmental heterogeneity in Parrotia subaequalis (H. T. Chang) R. M. Hao et H. T. Wei, an endemic and endangered tree species from China. Forests 9:247
Acknowledgements
This article has received financial support from the Spanish Ministerio de Ciencia e Innovación—EU-FEDER (Project No. CGL2016-79861-P). We thank to Marta Ortiz and to the Electron Microscopy Facilities-NUCLEUS of the University of Salamanca for his help in the analysis of stomata density.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Judy Simon.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mediavilla, S., Martín, I. & Escudero, A. Vein and stomatal traits in leaves of three co-occurring Quercus species differing in leaf life span. Eur J Forest Res 139, 829–840 (2020). https://doi.org/10.1007/s10342-020-01290-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-020-01290-8