Abstract
Watercore is a typical physiological disorder of apples and can normally induce the development of related internal disorders during storage. The aim of this study was to investigate the disappearance of watercore, the development of internal storage disorders (ISD) and the maintenance of quality traits of ‘Cox Orange Pippin’ apple during 6 months storage at 3 °C (±0.3 °C) under the following storage conditions: Regular air (RA); rapid storage at 1.5 kPa O2 plus 1.5 kPa CO2; 20 d pre-storage in air at 10 °C followed by storage at 1.5 kPa O2 plus 1.5 kPa CO2; 20 d delayed in the pull down of oxygen partial pressure (p) at 3 °C followed by storage at 1.5 kPa O2 plus 1.5 kPa CO2; and the rapid storage at 3.0 kPa O2 plus <0.2 kPa CO2. At storage end, watercore disappeared faster in RA-stored fruit; however, these fruit were softer, yellower and lower in acidity and showed the highest ISD index (40.0). The rapid storage under 1.5 kPa O2 plus 1.5 kPa CO2 resulted in fruit without ISD, higher firmness, higher acidity, greener skin colour, and a watercore index of 7.5 at storage end. Delayed CA-stored fruit did not keep satisfactory quality traits, and resulted in high watercore (7.6) and ISD indexes (6.9). A similar behaviour to the delayed CA-stored fruit, was observed in apple rapid stored at 3.0 kPa O2 plus <0.2 kPa CO2 and in fruit subjected to 20 d pre-storage in air at 10 °C. The fruit specific weight decreased from 0.89 at harvest time to 0.82 at storage end, but no significant differences were observed between treatments. A positive correlation (r = 0.91) was found between the fruit specific weight and the severity of watercore. In conclusion, the rapid storage of severe watercored ‘Cox Orange Pippin’ apple at 1.5 kPa O2 plus 1.5 kPa CO2 maintained higher firmness, higher acidity and greener skin colour, without occurrence of ISD after 6 months’ storage.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Apples severely affected by watercore are rejected during market quality control because the fruit can develop alcoholic off-flavour and internal storage disorders (Neuwald et al. 2012). Watercore is a physiological disorder especially of apples, which generally appears only in physiologically mature fruit attached to the tree, and in susceptible cultivars, it has been associated with high source to sink relationship (Marlow and Loescher 1984; Yamada et al. 1994; Gao et al. 2005). This disorder is characterized by a translucent water soaked appearance of the fruit flesh and it is attributed to the accumulation of sorbitol in the intercellular spaces, initially associated with the vascular bundles of the core line, but which can extend from the core region to the skin surface in severe affected fruit (Faust et al. 1969; Marlow and Loescher 1984; Melado-Herreros et al. 2013).
Structurally, the intercellular spaces which normally occupy between 15 and 23% of the total volume of an apple (Dražeta et al. 2004), have become waterlogged due to high sorbitol accumulation in the apoplast. The specific weight of the affected areas therefore approaches the 1.1 of the cellular tissue, instead of the characteristic 0.85 of a healthy apple fruit (Marlow and Loescher 1984). An affected apple, therefore, has a specific weight somewhere between these two values depending on the severity of the disorder (Fidler et al. 1973). The affected tissue is normally lower in acid contents, particularly malic acid, and the accumulations of ethanol and acetaldehyde have been reported (Marlow and Loescher 1984). Recently, it was observed higher contents of dihydrochalcone and higher peroxidase activity in watercored apple fruit (Zupan et al. 2016). In high quantities, both ethanol and acetaldehyde are toxic to apple tissue and may be involved in the development internal storage disorders during long-term storage (Meheriuk et al. 1994; Brackmann et al. 2001; Saquet and Streif 2006; Neuwald et al. 2012).
The accumulation of sorbitol in the intercellular spaces of affected fruit suggests an interruption of normal metabolism, because in healthy tissues sorbitol is converted to fructose by the enzyme sorbitol dehydrogenase before its utilization by cell respiration or starch synthesis. In the same apple, it has been observed that healthy tissues carry out this conversion, whereas affected tissue with watercore does not (Bangerth 1973; Fidler et al. 1973). Faust et al. (1969) suggested that the failure of the tissue to convert sorbitol to fructose might be due to the absence or inhibition of the enzyme sorbitol dehydrogenase, and/or to the unavailability of its cofactor nicotinamide adenine dinucleotide. However, Marlow and Loescher (1985) showed no relationship between susceptibility to watercore and the activity of sorbitol dehydrogenase. Investigation of Gao et al. (2005) studying the gene expression of membrane transporters, verified that the sorbitol transporter expression was evident in all sink tissues tested with the exception of watercore-affected fruit tissues. Sorbitol accumulation in apple sink tissues thus was shown to involve an apoplasmic active membrane transport step and that watercore results from a defect in that process. The sorbitol transporter expression was also quite low in watercored apple skin and cortical tissues and especially when compared to healthy fruit (Gao et al. 2005).
Watercore becomes usually less severe during storage and can, according to its severity and storage conditions, in fact disappear entirely (Fidler et al. 1973; Meheriuk et al. 1994; Brackmann et al. 2001; Neuwald et al. 2012). It is also important in that it can be a precursor of the abnormality known as watercore-breakdown, which is a true storage disorder in the sense that it is a permanent injury observed during and after storage (Fidler et al. 1973). Flesh browning and internal breakdown, however, are likely to develop in severely affected fruit depending on the storage conditions and duration (Bangerth 1973; Argenta et al. 2002). However, it has been observed, that keeping fruit at warm temperatures after harvest, before storage, hastens the disappearance of watercore, but it also accelerates ripening and the possibility of senescent breakdown incidence may increase (Fidler et al. 1973; Meheriuk et al. 1994).
‘Cox Orange Pippin’ apple is grown commercially especially in West Europa, especially in England and in Germany. However, it shows frequently high incidence of watercore depending on climate conditions, seasons and other pre-harvest factors such as plant nutrition, crop load, fruit thinning, etc. This situation requires a special attention and management at harvest time and a carefully decision of producers and storage managers. They have to decide if fruit are able or not to store, according to the incidence of this physiological disorder at harvest.
Therefore, the main objectives of this study were: a) to monitor the disappearance of watercore symptoms during six months’ storage of ‘Cox Orange Pippin’ apple; b) to verify if watercored apple fruit develop internal storage disorders and; c) to evaluate changes in flesh firmness, titratable acidity, total soluble solids and skin colour during storage period.
Material and Methods
Fruit Material, Storage Procedure and Treatments
Fruit of ‘Cox Orange Pippin’ apple were obtained from experimental orchards of the Competence Center for Fruit Science Bavendorf, Ravensburg, Germany. The apple fruit were harvested from external limbs on the sun side in the middle portion of the apple trees on either east or west exposition to get a greater number of affected fruit. For each storage treatment 200 fruits were used, which were separated in 4 batches of 50 fruit each for analyses during storage. At harvest, all fruit were individually identified and analyzed for specific weight before storage. Three replicates of 15 fruit each were used for analyses of flesh firmness, titratable acidity, skin colour and total soluble solids. Fruit were then stored at 3 °C (±0.3 °C) during 6 months considering the following conditions: a) Regular air (RA), b) rapid storage at 1.5 kPa O2 + 1.5 kPa CO2; c) rapid storage at 3.0 kPa O2 + <0.2 kPa CO2; d) 20 d pre-storage in air at 10 °C with subsequently storage at 1.5 kPa O2 + 1.5 kPa CO2; e) and 20 d delayed pull down of pO2 with subsequently storage at 1.5 kPa O2 + 1.5 kPa CO2. Samples of pre-storage was accomplished by keeping the fruit under air conditions at 10 °C (±0.3 °C) for 20 d before CA-storage. Delayed storage was accomplished by keeping the fruit under air conditions at 3 °C (±0.3 °C) for 20 d before storage under 1.5 kPa O2 plus 1.5 kPa CO2. The pull down of pO2 was carried out in within 20 h in all storage conditions. Storage procedures are described in Saquet et al. (2000). The evaluations were carried out at the first, second, fourth and sixth month of storage.
Measurement of Fruit Specific Weight
At harvest and at each storage sampling interval during storage, fruit mass and specific weight were determined as follows: A 500 mL volume beaker containing distilled water was placed on a balance and tared. Each apple was then placed into the beaker and its mass recorded. The apple was then submerged using a probe composed by a support equipped with a fine wire net and fruit mass recorded again. Immediately after this non-destructive measurement of fruit specific weight, all fruit were destructively assessed for quality traits and watercore incidence.
Measurements of Quality Attributes
The skin colour (SC) was measured in CIE L*a* + b* colour space with a tri-stimulus CR-300 colorimeter (Konica Minolta Inc., Tokyo, Japan). Measurements were made on the widest and greenest part of the equatorial region of each fruit in three replicate batches of 15 fruit each. Results were expressed as L a + b.
Flesh firmness (FF) was measured after skin removal, with a penetrometer equipped with a 10 mm probe. The maximum force to insert the probe 10 mm into the fruit flesh was recorded. FF was measured twice in each fruit, in opposite sides at the equatorial region, in 3 replicated samples of 15 fruit each.
For the soluble solids (SS) and the titratable acidity (TA) analyses, same fruit samples used for skin colour and flesh firmness were cut transversely at the equatorial region and a disc of 10 mm thick was used for juice extraction. SS were measured directly in the juice with a digital refractometer (Atago PR 1, Bellevue, USA) and values given in percent. For TA measurements, an aliquot of 10 mL juice was diluted in 90 mL distilled water and the solution titrated with 0.1 M NaOH until pH 8.1. Titratable acidity was expressed as mval/100 mL. The pH change was monitored with WTC pH meter (Weilheim, Germany).
Watercore Assessment
After determining fruit specific weight and quality parameters, fruit were cut transversely several times at the equatorial region and visually scored for watercore severity according to the following scale: 0% = watercore absent (healthy fruit); up to 10% of the fruit flesh affected = slight incidence; between 11 and 30% = moderate incidence; from 31 to 60% = severe incidence; and higher than 60% of the affected fruit flesh was considered a very severe incidence.
Results and Discussion
Watercore Disappearance During Storage and the ISD Occurrence
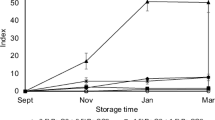
The disappearance of watercore during storage of ‘Cox Orange Pippin’ apple is shown in Fig. 1. The faster and largest differences in watercore disappearance occurred during the first 60 d of storage. Similar behaviour was observed by Neuwald et al. (2012) with severe watercored ‘Fuji’ apple in which watercore disappeared very fast during the first 51 d of storage. Although the fruit rapid stored at 3.0 kPa O2 plus 0.2 kPa CO2 showed the faster disappearance of watercore symptoms, those fruit which were rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 had the slower decreasing rate during the same period. From the 60th to the 90th d of storage, the rate of watercore disappearance further decreased, but with minor differences between treatments. After six months of storage, fruit from all treatments showed very similar and low indexes of watercore ranging from 4.4 to 7.5. These final values corresponded to 10.6 and 18.1%, respectively, of the watercore indexes observed at harvest.
From the indexes shown in Fig. 1 were calculated the rates of watercore disappearance (WD) during the storage period. The rapid storage of apple fruit at 3.0 kPa O2 plus 0.2 kPa CO2 allowed the faster disappearance of watercore during the first 60 d of storage period at a rate of 0.43 WD d−1, followed by fruit kept at 1.5 kPa O2 plus 1.5 kPa CO2 subjected to 20 d delayed CA, RA-stored apple and those fruit kept at 1.5 kPa O2 plus 1.5 kPa CO2 subjected to the pre-storage of 20 d in air at 10 °C, at rates of 0.30, 0.24 and 0.14 WD d−1, respectively. The slower watercore disappearance was observed in fruit rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 at a rate of only 0.03 WD d−1 during the first 60 d of storage period. However, from the 60th to the 90th d storage period, the apple fruit rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 showed a pronounced decrease in the watercore symptoms by a rate of 0.60 WD d−1 and coming close to the other storage conditions.
The slowest rate of watercore disappearance measured in fruit rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 coincided with the lowest incidence of ISD at storage end i.e. 100% of healthy fruit. The fruit subjected to 20 d pre-storage in air at 10 °C and those rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 showed the slower rates of watercore disappearance and the lowest indexes of affected fruit by ISD of 3.2 and 0.0 after six months’ storage period, respectively. It was remarkable, that the two above storage conditions, which could better regulate the apple fruit metabolism regarding the disappearance of watercore related to the occurrence of ISD were: 1) The rapid storage under 1.5 kPa O2 plus 1.5 kPa CO2, and 2) the pre-storage of fruit for 20 d in air at 10 °C. The first condition allowed 100% healthy fruit and the second an ISD index of 3.2.
Low levels of watercore may disappear during low temperature storage (Ferguson et al. 1999), but the most apple cultivars affected by watercore may develop internal disorders and alcoholic off-flavor during mid to long-term CA-storage (Marlow and Loescher 1984; Ferguson et al. 1999; Argenta et al. 2002; Dart and Newman 2005). Some apple cultivars such as ‘Red Delicious’ moderate or severe affected by watercore at harvest time can develop internal browning within three months in CA-storage (Kupferman 2002). One of the proposed mechanism in watercore inducing internal disorders during CA-storage is the lower intercellular air space volume, the reduced permeance to gas diffusion and the increased internal pCO2 caused by the accumulation of sorbitol within the apple fruit (Argenta et al. 2002).
In the present investigation, severe watercored ‘Cox Orange Pippin’ apple at harvest, showed a very different behaviour, that all above reported situations and various apple cultivars, which normally are difficult to keep under long-term CA-storage due to the development of internal storage disorders associated (or not) to the presence of watercore within the fruit. The present results are instead near to those reported by Neuwald et al. (2012) with ‘Fuji’ apple cultivated in Southern Germany, which watercore did not increase the occurrence of other internal storage disorders during long-term CA-storage.
Fruit flesh was found to exert a significant resistance to molecular O2 diffusion resulting in measurable O2 gradients between tissues immediately beneath the skin and those at the apple fruit core (Rajapakse et al. 1990). Gradients of oxygen and carbon dioxide exist in apple fruit and depend not only on anatomic fruit diffusion properties, but also on respiration rates of the different apple cultivars, temperature and other factors (Rajapakse et al. 1990; Ho et al. 2010).
‘Cox Orange Pippin’ apple in the present investigation seems to have the ability to metabolize high amounts of sorbitol, measured at harvest time, even when subjected to rapid storage at 1.5 kPa O2 plus 1.5 kPa CO2. Under this CA-condition was measured the slowest rate of watercore disappearance at the beginning of storage (first 60 d), however decreasing faster from the 60th to the 90th d of storage. This suggests a possible change in gas diffusion properties within the fruit during storage, allowing apple tissues to become more adapted from the middle to late stages of storage. Park et al. (1993) investigating the diffusion properties during storage of ‘McIntosh’ apple observed that the longer the ‘McIntosh’ apple were held in CA-storage, the lower was the fruit resistance to gas diffusion indicating an increase in the fruit ability to carry out the gas exchange even under CA-conditions. ‘Cox Orange Pippin’ apple behaved beyond the general role observed in apples affected by watercore during CA-storage.
The Occurrence of Internal Storage Disorders (ISD)
The browning of the fruit flesh was the ISD identified after storage of ‘Cox Orange Pippin’ apple. The rapid establishment of CA conditions at 1.5 kPa O2 plus 1.5 kPa CO2 was the best storage condition, maintaining 100% healthy fruit after six months of storage (Fig. 2). Apples maintained in RA-storage showed the highest ISD index of 40.0. Fruit rapid stored at 3.0 kPa O2 plus <0.2 kPa CO2 and those under 1.5 kPa O2 plus 1.5 kPa CO2 subjected to 20 d delayed CA developed the same severity ISD index of 6.9. A promising condition for ‘Cox Orange Pippin’ apple regarding disorders occurrence was the pre-storage of fruit during 20 d in air at 10 °C before CA-storage, resulting in low ISD index of 3.2 after six month of storage (Fig. 2). However, fruit from this treatment could not keep satisfactory quality attributes after six months’ storage period (discussed in item 3.4.).
‘Cox Orange Pippin’ apple in this study behaved different of many other apple cultivars, regarding the occurrence of ISD. The majority of apple cultivars frequently develop internal disorders during CA-storage, when fruit are moderate or severe affected by watercore at harvest (Ferguson et al. 1999; Argenta et al. 2002; Kupferman 2002; Dart and Newman 2005). A relative positive, but not remarkable effect of 20 d delaying the pull down of pO2 or even the pre-storage of ‘Cox Orange Pippin’ apple for 20 d in air at 10 °C prior to controlled atmospheres establishment was observed in alleviating the occurrence of ISD after storage period, but these conditions were not able to keep satisfactory quality traits. A similar behaviour has been reported by Fidler et al. (1973) and Meheriuk et al. (1994), who reported that keeping fruit at warm temperatures after harvest, before storage, hastens the disappearance of watercore, but it also accelerates ripening and the possibility of senescent breakdown incidence may increase. A 21 d delayed CA has shown very positive effect in reducing ISD in apples (Streif and Saquet 2003; Saquet et al. 2003a; Castro et al. 2007; DeEll and Ehsani-Moghaddam 2012) and in ‘Conference’ pear (Höhn et al. 1996; Saquet et al. 2003b), but was not effective in the present research with watercored ‘Cox Orange Pippin’ apple.
Changes in Apple Fruit Specific Weight During Storage and its Correlation with Watercore
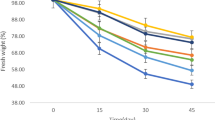
The specific weight of ‘Cox Orange Pippin’ apple decreased from 0.89 at harvest to 0.83 and 0.85 in fruit rapid stored at 3.0 kPa O2 plus 0.2 kPa CO2 and in RA-stored apple, which were the lowest and highest values, respectively, but statistically not different (Fig. 3). In general, it was verified the capacity of the apple fruit to metabolize the sorbitol during storage period even under low temperature and CA. Furthermore, in fruit, which were rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 the sorbitol in the fruit flesh disappeared slowly, but continuously during storage, resulting in 100% healthy fruit after six months of storage (Fig. 2). It seems that rapid establishment of CA-storage could help regulating and adjusting sorbitol metabolism to ideal levels, inhibiting the mechanisms leading to other disorders development in the fruit during storage.
For the correlation study (Fig. 4), the apple fruit were separated in 5 classes to generate the data: the class 1 was composed by the fruit absent of watercore (healthy fruit); the class 2 included fruit slightly affected by watercore (0 to 10% of affected flesh); the class 3 were the fruit moderate affected (10 to 30% of affected flesh); the class 4 those fruit severe affected by the disorder (30 to 60% affected flesh); and the class 5 included the fruit, which were very severe affected by watercore with more than 60% of the fruit flesh affected by the disorder at harvest time. In the latter class, where fruit were very severe affected by the disorder, it was possible to visualize the damage on the fruit skin with external water soaked appearance. The lowest specific weight of 0.83 to 0.85 were measured in fruit of the class 1, which were the healthy fruit of ‘Cox Orange Pippin’, while the highest values were measured in very severe affected fruit with values ranging from 0.93 to 0.98 in specific weight.
Quality Attributes of Apple Fruit During Storage Period
Changes in flesh firmness of ‘Cox Orange Pippin’ apple during storage period are shown in Fig. 5. The flesh firmness of ‘Cox Orange Pippin’ apple at harvest was 91.7 N and decreased to 41.2 N in RA-stored apple fruit after six months of storage. One factor, which contributed to this high flesh firmness of apples at harvest, was the relatively small size of fruit (55–65 mm diameter). Intermediary firmness values were measured in fruit, which were rapid stored at 3.0 kPa O2 plus 0.2 kPa CO2 and those under 1.5 kPa O2 plus 1.5 kPa CO2 subjected to 20 d delayed CA, with values ranging from 49.5 to 54.2 N at storage end. Apple fruit rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 were firmer than fruit from all treatments keeping the flesh firmness around 67.0 N at storage end. The beneficial effect of the rapid establishment of CA-conditions on quality maintenance of apples has been frequently reported (Lau et al. 1983; Lau 1985; Little and Peggie 1987; Brackmann and Saquet 1999). Furthermore, in combination with the rapid establishment of controlled atmospheres, the pO2 in the range of 1.5 kPa or lower was very effective in firmness retention in various apple cultivars (Lau 1990; Streif and Saquet 2003; Wright et al. 2015; Saquet 2016).
The skin colour of ‘Cox Orange Pippin’ apple yellowed faster in RA-stored fruit (Fig. 6), in which the value of 26.2 (CIE Lab a + b) measured at harvest time increased continuously to 46.8 after six months’ storage period. Fruit from all other CA-conditions yellowed slower and remained in the range from 34.0 to 38.4 at storage end, however, statistically not different among them. Slightly greener, but statistically not different, was the skin colour of apple fruit rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2. The effect of low or ultra-low pO2 in delaying the yellowing in ‘Golden Delicious’ (Lau 1990), ‘Granny Smith’ (Zanella 2003) and ‘Jonagold’ (Saquet 2016) apples was shown.
The titratable acidity of ‘Cox Orange Pippin’ apple, at harvest, was 14.5 mval/100 mL (Fig. 7), which may be considered high comparable to various other apple cultivars. The acidity of apple fruit decreased during storage period to the half contents of those measured at harvest time in all storage conditions, except in fruit rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2, which maintained higher acidity than all other treatments. Ultra-low oxygen kept higher acidity in ‘Golden Delicious’ apple (Lau 1990). Other apple cultivars respond positively to low pO2 during CA-storage keeping higher acidity (Wright et al. 2015). However, some exceptions are reported as was the case of ‘McIntosh’ and ‘Delicious’ apples, which the ultra-low pO2 as low as 0.5 kPa did not influence significantly the juice acidity during CA-storage (Sitton and Patterson 1992).
At harvest time, ‘Cox Orange Pippin’ apple has 11.4% total soluble solids (Fig. 8). TSS increased to a maximum of 14.1% in fruit rapid stored at 1.5 kPa O2 plus 1.5 kPa CO2 at the fifth month storage period. After this time in storage, TSS remained stable in fruit from all CA-conditions until storage end, except the RA-stored fruit, which decreased to 13.2% at storage end. The increase in TSS of ‘Cox Orange Pippin’ apple fruit was relatively sharp, but no significant differences were found between CA-treatments. Only the RA-stored apple fruit begun to decrease the TSS from the fifth to the sixth month storage, certainly associated to an accelerated metabolism and fruit senescence. TSS are normally not affected by CA-conditions during storage of apples. Sitton and Patterson (1992) observed, that low pO2 as low as 0.5 kPa or above did not influence significantly the TSS of ‘McIntosh’, ‘Delicious’ and ‘Golden Delicious’ apples. Harb et al. (2013) could not measure significant differences in TSS during long-term CA-storage of ‘Cameo’ apple fruit.
Conclusions
-
The rapid pull down in pO2 with subsequently storage of ‘Cox Orange Pippin’ apple at 1.5 kPa O2 plus 1.5 kPa CO2 allowed the successful storage of severely affected apple fruit for six months without ISD occurrence;
-
The rapid storage of ‘Cox Orange Pippin’ apple at 1.5 kPa O2 plus 1.5 kPa CO2 allowed higher flesh firmness, higher titratable acidity and greener skin colour of fruit after six months’ storage period;
-
The increase in the fruit specific weight was positive correlated with the severity of watercore incidence;
References
Argenta LC, Fan X, Mattheis J (2002) Impact of watercore on gas permeance and incidence of internal disorders in ‘Fuji’ apples. Postharvest Biol Technol 24:113–122
Bangerth F (1973) Investigations upon Ca related physiological disorders. Phytopathol Z 77:20–37
Brackmann A, Saquet AA (1999) Low ethylene and rapid CA storage of ‘Gala‘ apples. Acta Hortic 485:79–83
Brackmann A, Neuwald DA, Steffens CA (2001) Storage of ‘Fuji’ apples with watercore incidence. Rev Bras Frutic 23:526–531
Castro E, Biasi B, Mitcham E, Tustin S, Tanner D, Jobling J (2007) Carbon dioxide-induced flesh browning in ‘Pink Lady’ apples. J Am Soc Hortic Sci 132:713–719
Dart JA, Newman SM (2005) Watercore of apples. Primefact 49:1–2
DeEll JR, Ehsani-Moghaddam B (2012) Delayed controlled atmosphere storage affects storage disorders of ‘Empire’ apples. Postharvest Biol Technol 67:167–171
Dražeta L, Lang A, Hall AJ, Volz RK, Jameson PE (2004) Air volume measurement of `Braeburn’ apple fruit. J Exp Bot 55(399):1061–1069
Faust M, Shear CB, Williams MW (1969) Disorders of carbohydrate metabolism of apples (watercore, internal breakdown, low temperature and carbon dioxide injuries). Bot Rev 35:168–194
Ferguson I, Volz R, Woolf A (1999) Preharvest factors affecting physiological disorders of fruit. Postharvest Biol Technol 15:255–262
Fidler JC, Wilkinson BG, Edney KL, Sharples RO (1973) The biology of apple and pear storage. Commonwealth Agricultural Bureaux, East Malling, Maidstone, Kent
Gao Z, Jayanty S, Beaudry R, Loescher WH (2005) Sorbitol transporter expression in apple sink tissues: implications for fruit sugar accumulation and watercore development. J Am Soc Hortic Sci 130(2):261–268
Harb J, Kittemann D, Neuwald DA, Hoffmann T, Schwab W (2013) Correlation between changes in polyphenol composition of peels and incidence of CO2 skin burning of ‘Cameo’ apples as influenced by controlled atmosphere storage. J Agric Food Chem 61:3624–3630
Ho QT, Verboven P, Verlinden BE, Schenk A, Delele MA, Rolletschek H, Vercammen J, Nicolai BM (2010) Genotype effects on internal gas gradients in apple fruit. J Exp Bot 61(10):2745–2755
Höhn E, Jampen M, Dätwyler D (1996) Kavernenbildung in ‘Conférence‘—Risikoverminderung. Schweiz Z Obst Weinbau 7:180–181
Kupferman E (2002) Observations on harvest maturity and storage of apples and pears. Postharvest Information Network, Washington State University, Wenatchee, pp 1–8
Lau OL (1985) Storage procedures, low oxygen, and low carbon dioxide atmospheres on storage quality of ‘Golden Delicious’ and ‘Delicious’ apples. J Am Soc Hortic Sci 110:541–547
Lau OL (1990) Tolerance of three apple cultivars to ultra-low levels of oxygen. HortScience 25(11):1412–1414
Lau OL, Meheriuk M, Olsen KL (1983) Effects of rapid CA, high CO2, and CaCl2 treatment on storage behaviour of ‘Golden Delicious’ apples. J Am Soc Hortic Sci 108:230–233
Little CR, Peggie ID (1987) Storage injury of pome fruit caused by stress levels of oxygen, carbon dioxide, temperature, and ethylene. HortScience 22:783–790
Marlow GC, Loescher WH (1984) Watercore. Hortic Rev 6:189–251
Marlow GC, Loescher WH (1985) Sorbitol metabolism, the climacteric and watercore in apples. J Am Soc Hortic Sci 110:676–680
Meheriuk M, Prange RK, Lidster PD, Porritt SW (1994) Postharvest disorders of apples and pears. Publication 1737/E. Agriculture and Agri-Food Canada, Ottawa
Melado-Herreros A, Muñoz-García MA, Blanco A, Val J, Fernández-Valle ME, Barreiro P (2013) Assessment of watercore development in apples with MRI: Effect of fruit location in the canopy. Postharvest Biol Technol 86:125–133
Neuwald D, Kittemann D, Streif J, Andrade CAW (2012) Watercore dissipation in ‘Fuji’ apples by postharvest temperature conditioning treatments. Acta Hortic 934:1097–1102
Park YM, Blanpied GD, Joswiak Z, Liu FW (1993) Postharvest studies of resistance to gas diffusion in ‘McIntosh’ apples. Postharvest Biol Technol 2(4):329–339
Rajapakse NC, Banks NH, Hewett EW, Cleland DJ (1990) Development of oxygen concentration gradients in flesh tissues of bulky plant organs. J Am Soc Hortic Sci 115(5):793–797
Saquet AA (2016) Storability of ‘Jonagold’ apple under extreme controlled atmosphere conditions. J Agric Sci Technol B 6:262–268
Saquet AA, Streif J (2006) Fermentative metabolism of ‘Conference’ pear under various storage conditions. J Hortic Sci Biotechnol 81(5):910–914
Saquet AA, Streif J, Bangerth F (2000) Changes in ATP, ADP and pyridine nucleotides levels related to the incidence of physiological disorders in ‘Conference’ pears and ‘Jonagold’ apples during controlled atmosphere storage. J Hortic Sci Biotechnol 75(2):243–249
Saquet AA, Streif J, Bangerth F (2003a) Reducing internal browning disorders in ‘Braeburn’ apples by delayed controlled atmosphere storage, and some related physiological and biochemical changes. Acta Hortic 628:453–458
Saquet AA, Streif J, Bangerth F (2003b) Energy metabolism and membrane lipid alterations in relation to brown heart development in ‘Conference’ pears during delayed controlled atmosphere storage. Postharvest Biol Technol 30:123–132
Sitton JW, Patterson ME (1992) Effect of high-carbon dioxide and low-oxygen controlled atmospheres on postharvest decay of apples. Plant Dis 76(10):992–995
Streif J, Saquet AA (2003) Internal flesh browning of ‘Elstar‘ apples as influenced by pre- and postharvest factors. Acta Hortic 599:523–527
Wright AH, Delong JM, Arul J, Prange RK (2015) The trend toward lower oxygen levels during apple (Malus x domestica Borkh) storage—A review. J Hortic Sci Biotechnol 90(1):1–13
Yamada H, Ohmura H, Arai C, Terui M (1994) Effect of preharvest fruit temperature on ripening, sugars, and watercore occurrence in apples. J Am Soc Hortic Sci 119(6):1208–1214
Zanella A (2003) Control of apple superficial scald and ripening—a comparison between 1‑methylcyclopropene and diphenylamine postharvest treatments, initial low oxygen stress and ultra low oxygen storage. Postharvest Biol Technol 27(1):69–78
Zupan A, Mikulic-Petkovsek M, Stamparand F, Veberic R (2016) Sugar and phenol content in apple with or without watercore. J Sci Food Agric 96:2845–2850
Acknowledgements
The author thanks Dr. S.T. de Freitas (Brazilian Agricultural Research Corporation—Embrapa, Petrolina, PE, Brazil) for reading the text and helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A.A. Saquet declares that he has no competing interests.
Rights and permissions
About this article
Cite this article
Saquet, A.A. Storage of ‘Cox Orange Pippin’ Apple Severely Affected by Watercore. Erwerbs-Obstbau 62, 391–398 (2020). https://doi.org/10.1007/s10341-020-00520-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10341-020-00520-y