Abstract
The effects of substrate-borne vibration with frequencies of 30 and 300 Hz on the number of the greenhouse whitefly Trialeurodes vaporariorum and the number of fruit sets in greenhouse tomatoes were investigated. When tomatoes were intermittently subjected to 300-Hz vibration generated from vibrational exciters installed in a greenhouse, the number of adult and larval T. vaporariorum was significantly reduced compared with non-vibration plots. The substrate-borne 30-Hz vibration generated from different vibrational exciters did not affect the number of T. vaporariorum when smaller acceleration was applied to tomato plants; however, it showed a suppressive effect when greater acceleration was applied. Regarding pollination, the number of fruit sets in tomatoes subjected to 300-Hz vibration was the same as that in non-vibration plots, which was lower than that in plant growth regulation treatment plots. The number of fruit sets in tomatoes subjected to 30-Hz vibration significantly increased compared with that in non-vibration plots, which was comparable to that in plant growth regulation treatment plots. Our findings suggest that applying substrate vibration to tomatoes is effective in suppressing the plant infestation by T. vaporariorum and promoting tomato pollination. Vibrations could disrupt various behaviors associated with infestation and cause repellency in T. vaporariorum. This study focused on the effectiveness of vibrational conditions, including frequency, acceleration, and temporal characteristics, in detail, aiming to incorporate them into integrated pest management programs in greenhouse tomato cultivation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key Message
-
We applied 30- and 300-Hz substrate-borne vibrations to tomato plants in a greenhouse.
-
Applying 30-Hz vibration increased the number of tomato fruit sets.
-
Applying 300-Hz vibration decreased the number of T. vaporariorum.
-
Substrate-borne vibrations could be suitable for both pest control and pollination.
Introduction
Vibrational communication is widespread in the social and ecological interactions among insects (Cocroft 2011; Takanashi et al. 2019; Strauß et al. 2021). A large number of insect species use substrate-borne vibrations as intra- or interspecific signals, and plants are the most widely used substrates for transmitting these vibrations (Cocroft and Rodríguez 2005). Plant-borne vibrations play important roles in various behavioral contexts, such as finding partners, preys, or hosts (Virant-Doberlet and Čokl 2004; Hill 2008), and among them, mating behavior is probably the most studied function of vibrational communication (Polajnar et al. 2015). For instance, the male brown planthopper Nilaparvata lugens (Stål) (Hemiptera: Delphacidae) locates the female brown planthopper that produces vibrations by inserting her stylet into a rice plant stem and vibrates her abdomen in a dorso-ventral direction without tapping the rice plant directly (Ichikawa and Ishii 1974). The mating process of the brown marmorated stink bug, Halyomorpha halys (Stål) (Hemiptera: Pentatomidae), is aided by low-frequency (50–80 Hz) substrate-borne vibrational signals, which are of two types in females and three types in males and are present in various combinations (Polajnar et al. 2016).
Several species of hemipteran families use substrate-borne vibrations for mate location and assessment, communication between sexes (Čokl and Virant-Doberlet 2003; Takanashi et al. 2019). Therefore, various pest control techniques involving mating disruption throughout the period of substrate vibration have been studied with promising results for these insect pests (Krugner and Gordon 2018; Avosani et al. 2020). For example, Laumann et al. (2018) found that pure tone vibrations (75–200 Hz) reduced copulation by 94.2%–100% compared with control pairs, thereby disrupting the reproductive behavior of the neotropical brown stink bug, Euschistus heros Fabricius (Hemiptera: Pentatomidae). Further, Mazzoni et al. (2017) reported that a vibrational signal of 80 Hz was effective in disturbing the mating system of the glassy-winged sharpshooter Homalodisca vitripennis (Germar) (Hemiptera: Cicadellidae). Additionally, Parent et al. (2022) indicated that non-specific substrate-borne vibrations caused reductions in number of some aphid species not known use vibration for communication. These studies demonstrate the potential of mechanical vibration as an integrated pest management (IPM) tool for several hemipteran pests.
The greenhouse whitefly Trialeurodes vaporariorum (Westwood) (Hemiptera: Aleyrodidae) is a severe pest on vegetables and numerous greenhouse plants, attacking more than 800 plant species worldwide (Perring et al. 2018). This insect species damages plants, including the tomato plant Solanum lycopersicum L., via direct as well as indirect methods. It reduces plant vitality and productivity and damages plants by directly extracting the plant sap (Colvin et al. 2006; Prijović et al. 2013), resulting in the growth of sooty mold fungi through the secretion of sticky honeydew on the fruit (Bi et al. 2002). In addition, T. vaporariorum can transmit some viruses (Morales and Jones 2004). In the mating behavior of whiteflies, including T. vaporariorum and Bemisia tabaci (Gennadius), communication through vibrations occurs when males and females oscillate their abdomen up and down (Kanmiya 1996, 2006; Fattoruso et al. 2021).
In greenhouse tomato production, using Bombus bumblebees as pollinators that vibrate the anthers of a flower to extract pollen has become a common practice worldwide (De Luca and Vallejo-Marin 2013). However, the Japanese government has been phasing out the use of non-native bumblebees, such as B. terrestris L., as greenhouse tomato pollinators since 2017 because of their ecological risks (Nishimura 2021). A number of alternative pollination methods have been applied, including plant growth regulation using synthetic plant hormones (hereinafter abbreviated as “PGR”) treatment or plant vibration treatment, such as hitting the wire associated with plants and apparatus that is manually applied to individual flowers (Palma et al. 2008; Dingley et al. 2022). However, these pollination treatments are labor-intensive methods, especially among large-scale growers. Although conventional vibrational pollination is inefficient in terms of labor-intensiveness, vibration generated from vibrational exciters installed in a greenhouse is expected to be effective for pollination.
In this study, we used two different substrate-borne vibrations with pure tone frequencies: 300-Hz vibration, which is present in various signals emitted by male T. vaporariorum (Kanmiya 1996), and 30-Hz vibration, which is effective in pollinating plants (Shimizu and Sato 2018). We conducted two field experiments to investigate how substrate-borne vibrations with these two frequencies would affect the number of T. vaporariorum and pollination in terms of the number of insect species on tomato plants and the number of tomato fruit sets.
Materials and methods
Study site and plant cultivation
Experiments were conducted at Miyagi Prefectural Agriculture and Horticulture Research Center in Natori, Miyagi Prefecture, Japan (38°10′N, 140°51′E, 46 m above the sea level).
For all experiments, the tomato cultivar was “Rinka 409” (Sakata Seed Co., Yokohama, Japan). Tomato seeds were sown in a 72-cell tray and grown in a greenhouse until the seedlings reached the five-leaf stage. Tomato seedlings were transplanted into 0.8-m wide beds covered with a black plastic mulching sheet in greenhouses at a plant-to-plant distance of 40 cm within rows and columns each. Fertilizers were applied at a standard amount for the study region (300 kg N/ha).
Vibrational exciters and measurements
A vibrational exciter using a magnetostrictive FeCo clad steel plate (Yang et al. 2018) cantilever was developed by Tohoku Steel Co., Ltd. (Fig. 1). The bending behavior of the cantilever causes vibrations with a constant frequency on the object connected to the base plate. The output signals were controlled and amplified using a custom-made system to drive vibrational exciters at either 300 or 30 Hz. The vibrational signals were measured using an accelerometer (Type NP-2106; Onosokki, Japan / Type PV-08A; RION, Japan) connected to a charge converter (Type VP40; RION), and the signals were fed into data acquisition hardware (Type SA-A1; RION). The accelerometer was attached to the target position using a plastic tape. Frequencies (Hz) and accelerations (m/s2, zero-to-peak) were measured at each position on the same day. The output signal (300 or 30 Hz) was confirmed in the detected signals through spectral analysis (fast Fourier transform: Hanning, 1 kHz bandwidth, 1600 lines).
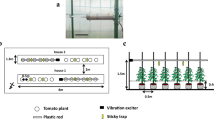
Vibrational exciter installed on a pipe. See Fig. 2 for details
Experiment 1: Influences of substrate-borne vibrations of 300 and 30 Hz on the number of T. vaporariorum
This experiment was conducted from August to September 2018. It was designed to evaluate the effects of substrate-borne vibrations of 300 and 30 Hz on the infestation by T. vaporariorum. The experiment was performed in a greenhouse (20-m long × 5.5-m wide × 3.0-m high) whose openings were covered with a white net (0.8 mm mesh). We constructed a tower in the greenhouse using iron pipes with a diameter of 19 mm (18-m long × 4.2-m wide × 2.4-m high) (Fig. 2a). Vibrational exciters with two different frequencies were established on 1.8-m-high pipes. Vibrational exciters with 300 Hz were installed at two points per pipe, 6.0 and 12.0 m from the edge of the pipe (Fig. 2b). Similarly, those with 30 Hz were installed at two points per pipe, 1.4 and 2.2 m from the edge of the pipes (Fig. 2b).
Schematic of an experiment comparing the incidence of Trialeurodes vaporariorum under conditions with 300- and 30-Hz vibrations in Experiment 1. The greenhouse was 20-m long × 5.5-m wide × 3-m high. A tower of iron pipes (18-m long × 4.2-m wide × 2.4-m high) was built (a). Two parallel strips were created in the greenhouse, and vibrations with the two frequencies were tested in a strip. A plot with both 300- and 30-Hz vibrations contained 20 plants and the plot without vibration contained 10 plants (b). Vibrational exciters were set up on iron pipes at a height of 1.8 m. In the treatment plots with vibration, tomato seedlings were supported by iron wires suspended from the iron pipes. In the treatment plots without vibration, tomato seedlings were supported by iron poles to prevent transmission of the vibration (c)
Tomato seedlings were transplanted into two parallel strips (18-m long × 0.8-m wide, with 1.0-m interval between their edges) in two rows covered with a black plastic mulching sheet on June 14; they were present at an interplant distance of 0.4 m within rows and columns each (Fig. 2b, c). Thus, each strip contained 90 tomato plants. One strip was used for examining 300-Hz vibration, and the other for 30-Hz vibration. Each strip was divided into six plots, and both treatments, i.e., with and without vibrations, were arranged alternately. However, the plot size was different as the treatment with vibration was 4.0-m long and that without vibration was 2.0-m long (Fig. 2b). While a plot with each vibration contained 20 tomato plants, a plot without vibration contained 10 tomato plants. The total number of tomato plants in each treatment was the same, i.e., 60 tomato plants. Thus, the size of the plot without vibration was half that of the plot with vibration. In the treatment with vibration, tomato seedlings were supported by iron wires (2 mm diameter) suspended from 1.8-m or 2.1-m-high iron pipes to transmit vibrations from exciters to tomato seedlings (Fig. 2c). In contrast, in the treatment without vibration, iron poles were installed from the ground to support tomato plants and prevent transmission of the vibration. The 300-Hz vibration was set to apply 1 min of pulsed vibration, which comprised repetitions of 1 s of vibration at an interval of 9 s, at an interval of 14 min from 5 a.m. to 7 p.m. (Supplemental Fig. 1a). This vibrational start and end time of the day was determined by taking into account the time of sunrise and sunset at experimental site during this periods. The stimulation cycle and timing in plots with 300-Hz vibration were based on previous studies to avoid the effects of habituation of vibration (Kishi and Takanashi 2019a; Yanagisawa et al. 2021). The 30-Hz vibration was set to apply 1 min of continuous vibration to ensure a long duration of vibration in the plants at three time points, 9, 10, and 11 a.m. (Supplemental Fig. 1b). The stimulation cycle and timing in plots with 30-Hz vibration were based on our preliminary experiments and previous studies (Hanna 1999, 2004). Both the vibrations were applied every day for 22 days from August 20 to September 10, 2018.
The number of adult and larval T. vaporariorum on two compound leaves located at intervals of three and four leaves from the growing point were counted on August 20 (before providing the vibration). These two leaves were marked at that time and were continuously counted on August 28 and September 3 and 10. These investigations were conducted on four plants located at the center of each plot. After providing vibrations of either 300 or 30 Hz, we measured the accelerations on plant stems at a height of 1.35 m above the ground on August 20 (Supplemental Fig. 2).
Experiment 2: Influences of substrate-borne vibrations of 300 and 30 Hz on the number of T. vaporariorum, number of fruit sets, and growth of tomato plants
The experiment was conducted from November 2018 to March 2019. It was designed to evaluate the effects of substrate-borne vibrations of 300 and 30 Hz on the infestation by T. vaporariorum, number of fruit sets, and height and stem diameter of tomato plants. The experiment was performed in two greenhouses (12-m long × 5.5-m wide × 3.0-m high) whose openings were covered with a white net (0.8 mm mesh). One greenhouse was for examining the 300-Hz vibration, and the other for examining the 30-Hz vibration.
Tomato seedlings were transplanted into three parallel strips (10.0-m long × 0.4-m wide, with 1.0-m interval between their edges) in a single row covered with a black plastic mulching sheet on October 29; they were present at an interplant distance of 0.5 m. Thus, each strip contained 19 tomato plants. Next, iron pipes with a diameter of 19 mm were installed at a height of 1.8 m above the strips, and iron poles were used to suspend them. Tomato seedlings were supported by iron wires (2-mm diameter) that were suspended from iron pipes. The treatments with 300- and 30-Hz vibrations were performed in a greenhouse. Two of the three strips in both the greenhouses were plots with vibration, and the remaining strip was a plot without vibration (Fig. 3a, b). The vibrational exciters were set up on the pipes. Exciters with 300-Hz vibration were installed at one point per pipe, 1.7 m from the edge of the pipe (Fig. 3a). In contrast, those with 30-Hz vibration, which were modified to provide greater acceleration than in Experiment 1, were installed at two points per pipe, 2.2 m and 7.8 m from the edge of the pipe (Fig. 3b). We started providing the vibrations on November 28. The 300-Hz vibration was set to apply 1 min of pulsed vibration, which comprised repetitions of 1 s of vibration at an interval of 9 s, at an interval of 14 min from 6 a.m. to 6 p.m. (Supplemental Fig. 1c). This vibrational start and end time of the day was determined by taking into account the time of sunrise and sunset at experimental site during this periods. The 30-Hz vibration was applied under different conditions for each strip; one was applied as continuous vibration for 1 min at two time points, 9 and 11 a.m. (hereinafter “30 Hz-1”, Supplemental Fig. 1d) and the other was applied as continuous vibration for 1 min, nine times every hour from 9 a.m. to 5 p.m. (hereinafter referred as “30 Hz-2”, Supplemental Fig. 1e). All vibrations were applied every day for 108 days from November 28 to March 15. In the plots without vibration, PGR (30 ppm of 4-chlorophenoxyacetic acid) was applied to every two plants, i.e., nine plants, for comparison of the number of fruit sets.
Greenhouse layout in an experiment comparing the incidence of Trialeurodes vaporariorum and the number of tomato fruit sets under conditions with (a) 300- and (b) 30-Hz vibrations in Experiment 2. The greenhouse was 12-m long × 5.5-m wide × 3-m high. Iron pipes were installed at a height of 1.8 m above the ground using iron poles. Tomato seedlings were contained 19 plants a strip and supported by iron wires suspended from iron pipes. The filled circles represent iron poles for supporting iron pipes
The number of adult and larval T. vaporariorum on tomato compound leaves was counted on November 28 (before providing the vibration), December 7, 14, and 26, January 11 and 18, February 1, 14, and 28, and March 11, but the counting on January 11 was limited to the greenhouse of the 300-Hz vibration experiment. The countings were conducted on four compound leaves located at the middle and lower sections of the stems from November 28 to December 26. Further, the countings were conducted on three compound leaves that were located at the upper, middle, and lower sections of the stems from January 11 to March 11. The height of tomato plant was approximately equivalent of 0.6 m to 1.7 m during this investigation period, therefore, compound leaf located at middle meant about 0.3 to 0.85 m height from the ground. These countings were conducted for every two plants per strip. Thus, 18 tomato plants were investigated in strips with 300-Hz vibration, whereas nine tomato plants were investigated in strips with 30 Hz-1 and 30 Hz-2 and those without either 300- or 30-Hz vibration. For the greenhouse where the 300-Hz vibration experiment was conducted, the number of T. vaporariorum on tomato plants was plotted against the distance from the exciter to examine the correlation between them.
The growth parameters of tomato plants were investigated in terms of the number of fruit sets, plant height, and stem diameter. Meter rulers were used to measure the tomato plant height from the base to the tip of the main shoots, whereas calipers were used to measure the tomato stem diameter at a height below 15 cm from the growing point of the main shoots. These investigations were conducted at two time points on January 21 and March 11. The number of fruit sets was counted for each cluster during harvest. We measured accelerations on plant stems at a height of 1.35 m above the ground on March 15. Further, we measured accelerations on iron pipes to compare the vibration transmissions on November 28 (after providing the vibration) (Supplemental Fig. 3).
Statistical analysis
The effect of treatments on the number of T. vaporariorum was evaluated using a generalized linear mixed model (GLMM) with a linked function of Poisson distribution (Poisson-GLMM). However, based on the dispersion estimator φ = D/(n − p) proposed by Wedderburn (1974), where D represents the deviance of the model, n the sample size, and p the number of parameters for the model, if overdispersion greater than the expected overdispersion was observed, we adopted the negative binomial GLMM (NB-GLMM) with a log-link function for those comparisons. In GLMM, the number of T. vaporariorum is a response variable, and the treatment type, which comprised two levels, is included as an explanatory (fixed) effect. The plots nested within fields and the date of investigation was included in the model as random effects. These GLMM analyses were performed using the R 4.0.5 lme4 package (Bates et al. 2015), and the significance of treatments was evaluated using a likelihood ratio test (LRT) in the R 4.0.5 car package (Fox and Weisberg 2019). The number of fruit sets, height, and stem diameter of tomato plants in Experiment 2 was compared via the Student’s t test or Tukey’s HSD test using R 4.0.5.
Results
Experiment 1: Influences of substrate-borne vibrations of 300 and 30 Hz on the number of T. vaporariorum
The temporal change in the number of adult and larval stage T. vaporariorum is shown in Figs. 4 and 5. In the 300-Hz experimental strip, the number of larval T. vaporariorum in vibration plots was significantly lower than that in non-vibration plots, while a significant difference was not observed in the adult stage (Fig. 4; LRT for the NB-GLMM: df = 1, χ2 = 9.00, p = 0.0027 [larva], LRT for the Poisson-GLMM: df = 1, χ2 = 0.5, p = 0.4933 [adult]). Meanwhile, in the 30-Hz experimental strip, the numbers of both adult and larval T. vaporariorum were similar between vibration and non-vibration plots (Fig. 5; LRT for the NB-GLMM: df = 1, χ2 = 1.79, p = 0.1815 [larva], LRT for the Poisson-GLMM: df = 1, χ2 = 0.0006, p = 0.9804 [adult]).
Temporal change in the number of (a) adult and (b) larval Trialeurodes vaporariorum on tomato plants in the 300-Hz experimental strip in Experiment 1. Circles and diamonds represent the mean number of T. vaporariorum in the treatment with and without 300-Hz vibration, respectively. While the number of adults was not different between treatments (likelihood ratio test (LRT), p > 0.05), the number of larva was significantly different throughout the experimental period (LRT, p < 0.01). The error bars represent standard deviations
Temporal change in the number of (a) adult and (b) larval Trialeurodes vaporariorum on tomato plants in the 30-Hz experimental strip in Experiment 1. Circles and diamonds represent the mean number of T. vaporariorum in the treatment with and without 30-Hz vibration, respectively. The number of adults and larvae did not differ significantly between treatments (likelihood ratio test, p > 0.05). The error bars represent standard deviations
The accelerations in the 300-Hz strip were 10.0 and 4.0 m/s2 at both the measuring positions, but the maximum acceleration in the 30-Hz strip was 1.5 m/s2 (Supplemental Table 1). As the iron pipes in both the frequency strips were interconnected, the 300-Hz vibration was transmitted with maximum acceleration of 1.2 m/s2, even in the 30-Hz strips, and vice versa with maximum acceleration of 0.2 m/s2 in the 300-Hz strips.
Experiment 2: Influences of substrate-borne vibrations of 300 and 30 Hz on the number of T. vaporariorum, number of fruit sets, and growth of tomato plants
The temporal change in the number of adult and larval stage T. vaporariorum is shown in Figs. 6 and 7. In the 300-Hz vibration greenhouse, the number of adult and larval T. vaporariorum in vibration strips was significantly lower than that in non-vibration strips, respectively (Fig. 6; LRT for the NB-GLMM: df = 1, χ2 = 10.0, p = 0.0015 [adult], LRT for the NB-GLMM: df = 1, χ2 = 29.5, p < 0.0001 [larva]). When the number of T. vaporariorum on tomato plants was plotted against the distance from the exciter, no correlation was observed between them (Supplemental Fig. 4). In the 30-Hz vibration greenhouse, the number of adult and larval T. vaporariorum in both the strips, i.e., 30 Hz-1 or 30 Hz-2, was significantly lower than that in non-vibration strips, respectively (Fig. 7; Tukey’s HSD post hoc test: p < 0.05).
Temporal change in the number of each (a) adult and (b) larval Trialeurodes vaporariorum on tomato plants in the 300-Hz experimental house in Experiment 2. Circles and diamonds represent the mean number of T. vaporariorum in the strips with and without 300-Hz vibration, respectively. The numbers of adults and larvae were significantly different throughout the experimental period (likelihood ratio test, p < 0.001). The error bars represent standard deviations
Temporal change in the number of each (a) adult and (b) larval Trialeurodes vaporariorum on tomato plants in the 30-Hz experimental house in Experiment 2. Circles, triangles and diamonds represent the mean number of T. vaporariorum in the strip with 30 Hz-1 (for 1 min at two time points, 9 and 11 a.m.), 30 Hz-2 (for 1 min, nine times every hour from 9 a.m. to 5 p.m.) and without vibrations, respectively. No significant differences were observed in these numbers indicated with the same lowercase letter along lines of treatments (Tukey’s HSD post hoc test, p > 0.05). The error bars represent standard deviations
The number of tomato fruit sets in strips with 300-Hz vibration was similar to that in strips without vibration; however, it was significantly lower than that in PGR treatment without vibration (Fig. 8a; Tukey’s HSD test, p < 0.05). In 300-Hz vibration strips, the number of fruit sets tended to be lower in the first–third clusters than that in PGR treatment without vibration (Supplemental Fig. 5; Tukey’s HSD test, p < 0.05). Conversely, the number of tomato fruit sets in both the 30-Hz vibration plots was significantly higher than that in non-vibration plots but was similar to that in PGR treatment without vibration (Fig. 8b; Tukey’s HSD test, p < 0.05). There was no difference in the number of fruit sets among 30 Hz-1, 30 Hz-2, and PGR treatment in the first–fifth clusters, but the number of fruit sets in the sixth cluster was the highest in 30 Hz-2 plots (Supplemental Fig. 6; Tukey’s HSD test, p < 0.05).
Number of tomato fruit sets in each (a) 300- and (b) 30-Hz experimental greenhouse in Experiment 2. PGR indicates plant growth regulation using synthetic plant hormones. Significant differences in the number of Trialeurodes vaporariorum were not observed among treatments indicated with the same lowercase letter in each greenhouse (Tukey’s HSD post hoc test, p > 0.05). The number of fruit sets summed across first–sixth clusters is shown. The error bars represent standard deviations
Regarding plant height and stem diameter, there was no significant difference in plant height and stem diameter between strips with and without vibration in the 300-Hz experimental greenhouse (Supplemental Table 2; Student’s t test, p > 0.05). Meanwhile, in the 30-Hz experimental greenhouse, the plant stem diameter tended to be larger in the strip with vibration than that without vibration, and this tendency was the strongest in 30 Hz-2 plots. Moreover, the plant height in 30 Hz-2 plots tended to be higher at the survey in Jan. 21, but subsequent growth was restrained (Supplemental Table 3; Tukey’s HSD test, p < 0.05).
At the start of the experiment, the tomato stems that were applied 300-Hz vibration showed accelerations ranging from 2.8 to 59 m/s2 on the iron pipe (Supplemental Table 4). However, the acceleration on the tomato stem at the end of the experiment ranged from 0.8 to 18 m/s2. Regarding 30-Hz vibration, the acceleration on the tomato stem at the end of the experiment ranged from 0.6 to 2.5 m/s2.
Discussion
In this study, we demonstrated that providing substrate-borne vibrations of 300 and 30 Hz to tomato plants reduced the plant infestation by T. vaporariorum and that 30-Hz substrate-borne vibration resulted in the same number of fruit sets as the PGR treatment.
Whiteflies communicate with each other through sexual signals, which vary among species (Kanmiya 2006). Trialeurodes vaporariorum males, the target of this study, generally communicate with T. vaporariorum females by emitting vibrations in the range of 250–375 Hz (Kanmiya 1996). Therefore, applying 300-Hz vibration to tomato plants probably interfered with mating behavior and suppressed the infestation by T. vaporariorum. In this study, the vibrational suppressing effect was investigated against the naturally occurring T. vaporariorum. Therefore, the number of the target pest was small. In particular, significant reduction in the number of adults was observed when 300-Hz vibration was applied in Experiment 2, but the difference was less than one individual (Fig. 6a). It would be necessary to confirm whether the same density suppression effect can be investigated, even under the condition with more T. vaporariorum, including artificial pest release. Further, the 30-Hz substrate-borne vibration reduced the infestation by T. vaporariorum depending on the acceleration, which may have some effect on their behavior, such as a rival male emitting mating disruption signals (Fattoruso et al. 2021). A previous study has shown the use of substrate-borne vibration based on the rivalry signal that functions as a mating disruption signal in the leafhopper Scaphoideus titanus Ball (Hemiptera: Cicadellidae) (Eriksson et al. 2012). Although the rivalry signal does not function sufficiently in the green leafhopper Empoasca vitis (Gothe) (Hemiptera: Cicadellidae), a different signal that focuses on the dominant frequency of this species is successfully used (Nieri and Mazzoni 2019). Further research on substrate-borne communication signals emitted by T. vaporariorum, including the possibility of this rivalry signal, is needed.
Bemisia tabaci is as important as T. vaporariorum among whiteflies damaging horticultural crops. Yanagisawa et al. (2021) focused on a frequency of 100 Hz, which causes a startle response, and confirmed the effectiveness of vibration in suppressing the number of this species in tomatoes. As 100-Hz vibration is also included in the mating signal emitted by male B. tabaci (Kanmiya), it is unknown whether the suppression in the number of B. tabaci by the vibrations is due to the startle response or the disruption of mating (Yanagisawa et al. 2021). Furthermore, in Coleoptera, vibrations of 100–120 Hz are found to induce escape and disrupt behavior other than mating; for example, feeding (Takanashi et al. 2019; Kishi and Takanashi 2019b; Takanashi and Nishino 2022). In the present study, similarly, 300-Hz vibration could cause disrupt various behaviors associated with infestation, in addition to disrupt mating behavior. It is necessary to elucidate the mechanism of behavioral control using vibrations and determine the vibrational properties that can achieve a greater suppression effect among whiteflies. Although there have been several behavioral and anatomical studies on the effect of vibration on the families of Heteroptera and Auchenorrhyncha (Čokl and Virant-Doberlet 2003; Takanashi et al. 2019), this information is still scarce in whiteflies Aleyrodidae of Sternorrhyncha, which are much smaller in size (Kubota et al. 2019). To establish effective control against whiteflies through vibration, vibrational communication and behavior in whiteflies should be studied behaviorally and anatomically in further detail.
The transmission of vibration can be crucial to the practical application of vibrational pest control. Yanagisawa et al. (2021) stated that vibration attenuates as the distance from the exciter increases, and this vibration attenuation was observed in this study (Supplemental Tables 1 and 2). However, regarding 300-Hz vibration, although acceleration was attenuated, there was no correlation between the number of T. vaporariorum and the distance from the exciter (Supplemental Fig. 4), indicating that a range of vibration accelerations obtained in this study suppressed the infestation to some extent. It is necessary to examine the correlation between the vibration accelerations and T. vaporariorum infestation in a larger field and determine the number of vibrational exciters that should be installed. The 300-Hz vibration may have more attenuation than 30-Hz vibration (Supplemental Table 1), and the frequencies of vibrational exciters can affect its performance. Furthermore, it is necessary to consider a more efficient way of transmitting vibration rather than using the iron wires employed in this study.
In this study, the application of 30-Hz vibration showed a positive effect on pollination, whereas that of 300-Hz vibration showed no effect. However, it is difficult to state a general comparison because of different temporal characteristics of vibration applications, i.e., time, duration, and intervals between 30- and 300-Hz treatments in this study. Some buzz-pollinators emit vibrations of approximately 300 Hz (De Luca and Vallejo-Marin 2013), which may promote pollination depending on the conditions. However, we did not investigate the fruit weight and traits in this study even though the 30-Hz vibration was effective in promoting pollination. Considering these factors, it is necessary to conduct more detailed investigations to evaluate their practicality. The plant height and stem diameter were slightly higher and thicker with 30-Hz vibration than without vibration, respectively. However, in this study it was not determined whether a positive or negative effect on tomatoes. Mechanical stimuli, such as shaking, rubbing, touching, and wind blowing, generally inhibit shoot elongation in plants, whereas vibration with a particular frequency promotes shoot elongation in some plants, suggesting that vibration may have a different effect on plants compared with other mechanical stimulations (Takahashi et al. 1991). Additionally, providing wind as a mechanical stimulus, depending on the conditions under which it is provided, enhances plant immunity and inhibits the condensation in plants, thereby suppressing the incidence of diseases (Cipollini 1997; Sekine et al. 2007). Future research should also shed light on various other possibilities, such as plant growth promotion and plant disease control in addition to insect pest control and pollination.
When an IPM system incorporated with vibrational pest control and pollination promotion is established, it is expected to significantly reduce the number of hours spent on pest control and pollination using synthetic chemicals in production fields. While applying the vibration technology to production sites, it would be simpler and inexpensive to apply a single frequency that is effective for both pest control and pollination than to apply two frequencies on tomatoes, i.e., 300 Hz to control pests and 30 Hz to promote pollination, as shown in this study. In future studies, it is necessary to determine effective frequencies, accelerations, and temporal characteristics for applying vibration to agricultural production. Moreover, vibration technology is expected to contribute to the management of resistant pests as well as the environmental impact by reducing the use of pesticides.
Authors' contributions
TT, TS, RO, and TK designed the study; TS, YK, TT, RO, and SA performed the experiments; TS and TO analyzed the data; TS wrote the first draft of the manuscript, and all authors commented on the manuscript. All authors read and approved the manuscript for submission.
Availability of data materials
The datasets during and/or analyzed during the current study available from the corresponding author on reasonable request.
References
Avosani S, Sullivan TE, Ciolli M, Mazzoni V, Suckling DM (2020) Can vibrational playbacks disrupt mating or influence other relevant behaviours in Bactericera cockerelli (Triozidae: Hemiptera)? Insects 11:299. https://doi.org/10.3390/insects11050299
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Soft. https://doi.org/10.18637/jss.v067.i01
Bi JL, Toscano NC, Ballmer GR (2002) Greenhouse and field evaluation of six novel insecticides against the greenhouse whitefly Trialeurodes vaporariorum on strawberries. Crop Prot 21:49–55. https://doi.org/10.1016/S0261-2194(01)00063-1
Cipollini DF (1997) Wind-induced mechanical stimulation increases pest resistance in common bean. Oecologia 111:84–90. https://doi.org/10.1007/s004420050211
Cocroft RB (2011) The public world of insect vibrational communication. Mol Ecol 20:2041–2043. https://doi.org/10.1111/j.1365-294X.2011.05092.x
Cocroft RB, Rodríguez RL (2005) The behavioral ecology of insect vibrational communication. Bioscience 55:323–334. https://doi.org/10.1641/0006-3568(2005)055[0323:TBEOIV]2.0.CO;2
Čokl A, Virant-Doberlet M (2003) Communication with substrate-borne signals in small plant-dwelling insects. Annu Rev Entomol 48:29–50. https://doi.org/10.1146/annurev.ento.48.091801.112605
Colvin J, Omongo CA, Govindappa MR, Stevenson PC, Maruthi MN, Gibson G, Seal SE, Muniyappa V (2006) Host-plant viral infection effects on arthropod-vector population growth, development and behaviour: management and epidemiological implications. Adv Virus Res 67:419–452. https://doi.org/10.1016/S0065-3527(06)67011-5
De Luca PA, Vallejo-Marín M (2013) What’s the ‘buzz’ about? The ecology and evolutionary significance of buzz-pollination. Curr Opin Plant Biol 16:429–435. https://doi.org/10.1016/j.pbi.2013.05.002
Dingley A, Anwar S, Kristiansen P, Warwick NWM, Wang C, Sindel BM, Cazzonelli CI (2022) Precision pollination strategies for advancing horticultural tomato crop production. Agronomy 12:2. https://doi.org/10.3390/agronomy12020518
Eriksson A, Anfora G, Lucchi A, Lanzo F, Virant-Doberlet M, Mazzoni V (2012) Exploitation of insect vibrational signals reveals a new method of pest management. PLoS ONE 7:e32954. https://doi.org/10.1371/journal.pone.0032954
Fattoruso V, Anfora G, Mazzoni V (2021) Vibrational communication and mating behavior of the greenhouse whitefly Trialeurodes vaporariorum (Westwood) (Hemiptera: Aleyrodidae). Sci Rep 11:6543. https://doi.org/10.1038/s41598-021-85904-0
Fox J, Weisberg S (2019) An R companion to applied regression, 3rd edition; 2019. http://socserv.socsci.mcmaster.ca/jfox/Books/Companion. Accessed 17 August 2021
Hanna HY (1999) Assisting natural wind pollination of field tomatoes with an air blower enhances yield. HortScience 34(5):846–847. https://doi.org/10.21273/HORTSCI.34.5.846
Hanna HY (2004) Air blowers are less effective pollinators of greenhouse tomatoes than electric vibrators but cost less to operate. HortTechnology 14(1):104–107. https://doi.org/10.21273/HORTTECH.14.1.0104
Hill PSM (2008) Vibrational communication in animals. Harvard University Press, Cambridge. https://doi.org/10.4159/9780674273825
Ichikawa T, Ishii S (1974) Mating signal of the brown planthopper, Nilaparvata lugens Stål (Homoptera: Delphacidae): vibration of the substrate. Appl Entmol Zool 9:196–198. https://doi.org/10.1303/aez.9.196
Kanmiya K (1996) Discovery of male acoustic signals in the greenhouse whitefly, Trialeurodes vaporariorum (Westwood) (Homoptera: Aleyrodidae). Appl Entomol Zool 31:255–262. https://doi.org/10.1303/aez.31.255
Kanmiya K (2006) Mating behaviour and vibratory signals in whiteflies (Hemiptera: Aleyrodidae). In: Drosopoulos S, Claridge MF (eds) Insect sounds and communication. Taylor & Francis, London, pp 365–379. https://doi.org/10.1201/9781420039337.ch28
Kishi M, Takanashi T (2019a) Tonic immobility and startle responses induced by substrate-borne vibrations in the sap beetle, Phenolia (Lasiodites) picta (Coleoptera: Nitidulidae). Jpn J Appl Entomol Zool 63:13–16. https://doi.org/10.1303/jjaez.2019.13 (in Japanese with English abstract)
Kishi M, Takanashi T (2019b) Escape behavior induced by substrate-borne vibrations in larvae of the Sap Beetle, Phenolia (Lasiodites) picta (Coleoptera: Nitidulidae). Jpn J Appl Entomol Zool 63:150–154. https://doi.org/10.1303/jjaez.2019.150 (in Japanese with English abstract)
Krugner R, Gordon SD (2018) Mating disruption of Homalodisca vitripennis (Germar) (Hemiptera: Cicadellidae) by playback of vibrational signals in vineyard trellis. Pest Manag Sci 74:2013–2019. https://doi.org/10.1002/ps.4930
Kubota K, Mizutani K, Ebihara T, Hayashi Y, Uga H, Ishii M (2019) Towards the management of whiteflies (Hemiptera: Aleyrodidae) in greenhouse by using acoustic and vibrational methods. Jpn J Appl Entomol Zool 63:97–107. https://doi.org/10.1303/jjaez.2019.97 (in Japanese)
Laumann RA, Maccagnan DHB, Čokl A, Blassioli-Moraes MC, Borges M (2018) Substrate-borne vibrations disrupt the mating behaviors of the Neotropical brown stink bug, Euschistus heros: implications for pest management. J Pest Sci 91:995–1004. https://doi.org/10.1007/s10340-018-0961-5
Mazzoni V, Gordon SD, Nieri R, Krugner R (2017) Design of a candidate vibrational signal for mating disruption against the glassy-winged sharpshooter, Homalodisca vitripennis. Pest Manag Sci 73:2328–2333. https://doi.org/10.1002/ps.4619
Morales FJ, Jones PG (2004) The ecology and epidemiology of whitefly-transmitted viruses in Latin America. Virus Res 100:57–65. https://doi.org/10.1016/j.virusres.2003.12.014
Nieri R, Mazzoni V (2019) Vibrational mating disruption of Empoasca vitis by natural or artificial disturbance noises. Pest Manag Sci 75:1065–1073. https://doi.org/10.1002/ps.5216
Nishimura T (2021) The effect of greenhouse pollination methods on consumers’ willingness to pay for tomatoes in Japan. J Agric Appl Econ 53:186–208. https://doi.org/10.1017/aae.2020.33
Palma G, Quezada-Euán JJG, Reyes-Oregel V, Meléndez V, Moo-Valle H (2008) Production of greenhouse tomatoes (Lycopersicon esculentum) using Nannotrigona perilampoides, Bombus impatiens and mechanical vibration (Hym.: Apoidea). J Appl Entomol 132:79–85. https://doi.org/10.1111/j.1439-0418.2007.01246.x
Parent JP, Laidlaw R, Abram PK (2022) Disruptive effects of non-specific airborne and substrate-borne vibrations on aphids. J Pest Sci 95:949–958. https://doi.org/10.1007/s10340-021-01425-9
Perring TM, Stansly PA, Liu TX, Smith HA, Andreason SA (2018) Whiteflies: biology, ecology, and management. In: Wakil W, Brust GE, Perring TM (eds) Sustainable management of arthropod pests of tomato. Academic Press, Cambridge, MA, USA, pp 73–110. https://doi.org/10.1016/B978-0-12-802441-6.00004-8
Polajnar J, Eriksson A, Lucchi A, Anfora G, Virant-Doberlet M, Mazzoni V (2015) Manipulating behaviour with substrate-borne vibrations-potential for insect pest control. Pest Manag Sci 71:15–23. https://doi.org/10.1002/ps.3848
Polajnar J, Maistrello L, Bertarella A, Mazzoni V (2016) Vibrational communication of the brown marmorated stink bug (Halyomorpha halys). Physiol Entomol 41:249–259. https://doi.org/10.1111/phen.12150
Prijović MR, Marčić D, Drobnjaković TM, Medjo IS, Perić P (2013) Life history traits and population growth of greenhouse whitefly (Trialeurodes vaporariorum Westwood) on different tomato genotypes. Pesticidi i Fitomed 28:239–245. https://doi.org/10.2298/PIF1304239P
Sekine T, Aizawa M, Nagano T, Takahashi C (2007) Suppression of gray mold and leaf mold of tomato by ventilation using small fans and analysis of mechanism. Ann Rept Plant Prot North Japan 58:46–53. https://doi.org/10.11455/kitanihon1966.2007.46 (in Japanese)
Shimizu H, Sato T (2018) Development of strawberry pollination system using ultrasonic radiation pressure. IFAC-PapersOnLine 51:57–60. https://doi.org/10.1016/j.ifacol.2018.08.060
Strauß J, Stritih-Peljhan N, Nieri R, Virant-Doberlet M, Mazzoni V (2021) Communication by substrate-borne mechanical waves in insects: from basic to applied biotremology. In: Jurenka R (ed) Advances in Insect Physiology Vol. 61. Academic Press. Cambridge, MA, USA, pp 189-307. https://doi.org/10.1016/bs.aiip.2021.08.002
Takahashi H, Suge H, Kato T (1991) Growth promotion by vibration at 50 Hz in rice and cucumber seedlings. Plant Cell Physiol 32:729–732. https://doi.org/10.1093/oxfordjournals.pcp.a078137
Takanashi T, Nishino H (2022) Exploitation of vibration sensing for pest management in longicorn beetles. In: Hill PSM, Mazzoni V, Stritih Peljhan N, Virant-Doberlet M, Wessel A (eds) Biotremology Physiology ecology and evolution. Springer Nature, Switzerland, pp 495–509. https://doi.org/10.1007/978-3-030-97419-0_20
Takanashi T, Uechi N, Tatsuta H (2019) Vibrations in hemipteran and coleopteran insects: behaviors and application in pest management. Appl Entomol Zool 54:21–29. https://doi.org/10.1007/s13355-018-00603-z
Virant-Doberlet M, Čokl A (2004) Vibrational communication in insects. Neotrop Entomol 33:121–134. https://doi.org/10.1590/S1519-566X2004000200001
Wedderburn RWM (1974) Quasi-likelihood functions, generalized linear models, and the Gauss-Newton method. Biometrika 61:439–447. https://doi.org/10.1093/biomet/61.3.439
Yanagisawa R, Suwa R, Takanashi T, Tatsuta H (2021) Substrate-borne vibrations reduced the density of tobacco whitefly Bemisia tabaci (Hemiptera: Aleyrodidae) infestations on tomato, Solanum lycopersicum: an experimental assessment. Appl Entomol Zool 56:157–163. https://doi.org/10.1007/s13355-020-00711-9
Yang Z, Nakajima K, Onodera R, Tayama T, Chiba D, Narita F (2018) Magnetostrictive clad steel plates for high-performance vibration energy harvesting. Appl Phys Lett. https://doi.org/10.1063/1.5016197
Acknowledgements
We thank Dr. Valerio Mazzoni for his valuable comments on an earlier version of this manuscript and express our appreciation to the members of Vibrational Agricultural Technology Consortium. This work was partly supported by Grants-in-Aid for Scientific Research (19KT0040) from Japan Society for the Promotion of Science and the research program on development of innovative technology grants (JPJ007097) from the Project of the Bio-oriented Technology Research Advancement Institution (BRAIN).
Funding
Funding was provided by Grants-in-Aid for Scientific Research (19KT0040) from the Japan Society for the Promotion of Science and the research program on the development of innovative technology grants (JPJ007097) from the Project of the Bio- oriented Technology Research Advancement Institution (BRAIN).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This article does not contain any studies with human participants or animals (Vertebrates) performed by any of the authors.
Additional information
Communicated by Chris Cutler.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sekine, T., Takanashi, T., Onodera, R. et al. Potential of substrate-borne vibration to control greenhouse whitefly Trialeurodes vaporariorum and increase pollination efficiencies in tomato Solanum lycopersicum. J Pest Sci 96, 599–610 (2023). https://doi.org/10.1007/s10340-022-01564-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-022-01564-7