Abstract
The invasive Asian chestnut gall wasp, Dryocosmus kuriphilus (Hymenoptera: Cynipidae), is one of the most economically important pests of chestnut forest systems. Control is either based on the use of pesticides or the release of the parasitoid Torymus sinensis (Torymidae) for classical biological control of the pest. Both approaches require frequent monitoring of wasp emergence patterns, usually by recording adult emergence from samples of galls stored in dark boxes. Here, we have improved this monitoring method by developing and evaluating a microcontroller-based device. Cages in which galls were placed were modified by insertion of a turntable, which automatically separates wasps emerging at different times. We used this device to monitor the seasonal emergence of D. kuriphilus, its coevolved parasitoid T. sinensis and newly associated native parasitoids from fresh or overwintered chestnut galls formed in 2013 and 2014. In summer, native parasitoids (3.6 and 5.8% of 2-year parasitism) associated with current-year chestnut galls started emerging before the gall wasp and only partially overlapped with D. kuriphilus emergence. After overwintering, in the spring of the following year, T. sinensis (4.3 and 5.5% of 2-year parasitism from overwintered galls) emerged earlier than indigenous parasitoids (1.6 and 4.8%) and indicated potential for D. kuriphilus control. Overall, 14 species of native parasitoids belonging to five families were recorded from fresh and overwintered galls. We provide the schematic and the programming codes for this new device and propose that this automatic device be used to better track the flight periods of D. kuriphilus and its parasitoids.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Key message
-
Integrated pest management strategies, including classical biological control with T. sinensis, are needed to control D. kuriphilus in chestnut.
-
We developed and evaluated a microcontroller-based rearing cage to better measure the seasonal emergence of D. kuriphilus and its parasitoids in a chestnut plantation in central Italy.
-
Over a 2-year period, total parasitism increased from 9.5 to 16.1%. Torymus sinensis prevalence was higher than any of the indigenous parasitoids, confirming its potential for biological control of this pest.
Introduction
The Asian chestnut gall wasp Dryocosmus kuriphilus Yasumatsu (Hymenoptera: Cynipidae) is one of the most important pests of chestnut trees (EFSA Panel on Plant Health 2010). This wasp is indigenous to China (Yasumatsu 1951) and became established in Japan, Korea and the USA in the twentieth century (Paine et al. 2004; Rieske 2007). In Europe, D. kuriphilus was first recorded in 2002 at a chestnut tree nursery in northern Italy (Brussino et al. 2002; Graziosi and Santi 2008), likely from a small introduction (only a few founders) (Bonal et al. 2018). Dryocosmus kuriphilus spread quickly and was soon detected in other parts of Europe: Slovenia (2004), France (2005), the Netherlands (2008), Switzerland (2009), Croatia (2010), the Czech Republic, Germany and Spain (2012), Austria and Hungary (2013), Greece, Portugal and Turkey (2014), Belgium and the UK (2015) (EPPO 2017). The life cycle of the Asian chestnut gall wasp in Europe has been studied by several authors (Romani et al. 2010; Maltoni et al. 2012; Bernardo et al. 2013; Graziosi and Rieske 2014; Reale et al. 2016). Parthenogenetic females oviposit from mid-June to mid-July in the dormant buds of chestnut trees. Eggs hatch after 30–40 days, and the larvae develop slowly in the buds and overwinter. The following spring, developing galls are visible on chestnut leaves, sprouts and inflorescences, and in June adult wasps (all females) emerge. Galls may be unilocular or, more often, multilocular, since several larvae might be present at the same time inside each individual gall (Stone and Schönrogge 2003; Warmund 2013). The formation of galls causes severe damage (Battisti et al. 2014), reducing photosynthesis and nut production and reducing tree vigor (Kato and Hijii 1997; Ugolini et al. 2014).
Control with insecticides (kaolin or pyrethroids) targets adults, as other stages are inside structures that protect the insect from contact with pesticides. For biological control, native natural enemies of D. kuriphilus, while of interest, have had little impact (Aebi et al. 2006, 2007; Quacchia et al. 2012; Colombari and Battisti 2016) as is often the case with newly invasive insects (Keane and Crawley 2002; Comont et al. 2014; Martorana et al. 2017; Rondoni et al. 2017). Therefore, a classical biological control program has been developed to control this invasive pest (Aebi et al. 2006; Panzavolta et al. 2011), based on the release of the parasitoid Torymus sinensis Kamijo (Quacchia et al. 2008; Gibbs et al. 2011), from the pest’s native range. Females of T. sinensis lay their eggs in spring in the newly formed galls, where the developed larvae later overwinter. Adult parasitoids only emerge the following spring from dry, overwintered galls (Gibbs et al. 2011). A prolonged diapause has been also observed in this parasitoid (Ferracini et al. 2015b; Picciau et al. 2017).
In this system, both chemical and biological control methods require knowledge of the pest’s population density and the timing of emergence of adult D. kuriphilus and its introduced and native parasitoids. A conventional method to track the emergence of gall-associated wasps is based on the collection of branches bearing galls that are then held in dark cardboard boxes sealed with tape and provided with clear extractable vials positioned on the vertical sides of the cage (Cooper and Rieske 2007; Quacchia et al. 2008; Santi and Maini 2011; Quacchia et al. 2012). Emerging wasps, which are positively phototactic, are collected periodically (i.e., once per day) from the vials. Here, we report a modification of this rearing system that uses electronic mechanisms to automatically separate emerged wasps at predefined time intervals. The proposed method was used to investigate the seasonal emergence of D. kuriphilus and its associated parasitoids from galls collected in central Italy. We present data from a 2-year sample period from one chestnut plantation, where we evaluated the new device. Previous studies in ecology and entomology have used microcontrollers to control data loggers (Wickert 2014), pitfall traps (Buchholz 2009), mimic-signal broadcast devices (Rohde et al. 2013) and drive acoustic devices (Hill et al. 2018), demonstrating high potential for large-scale applications due to the reproducibility of the methods and the low cost of the components.
Our method allows collecting newly emerged parasitoids and tracking parasitoid emergence without the need for daily inspections, which are otherwise necessary with the conventional method. In addition, our method makes it easier to monitor the D. kuriphilus flight period, allowing better timing of pesticide applications wherever chemical control is used.
Materials and methods
Design of the rearing cage
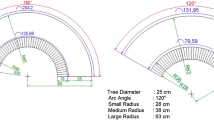
The device consisted of a wooden cage (30 cm length × 20 cm high × 20 cm wide) with an opening (dia 2.8 cm) in the front and a set of ten plastic 50 ml Falcon™ tubes (2.8 cm internal diameter) (BD Biosciences, Bedford, Massachusetts), equidistantly screwed into a plastic turntable (dia 19 cm) affixed to the vertical front side of the cage (Designs in Fig. 1). The turntable was bolted to a 6 V unipolar stepper motor (0.8 A, 1.8 degrees/step), which rotated the turntable of 36° (20 steps) at each predefined event (in our case daily at 17:00 h) moving new plastic tubes into the collecting position daily. A stepper motor was connected to the motor driver (L293D, Texas Instrument Inc., Dallas, Texas) and to a microcontroller (ATmega 328p, Atmel Corp., San Jose, California) for motor direction, steps and speed control. An external real-time clock (Chronodot v2.1 based on DS3231 fully equipped with resistors and 3 V coin cell battery, Macetech LLC, Foster City, California) offered accurate time information to the microcontroller. In addition, an SD memory card was used as a data logger for the step events. The data files were saved in text format. The power supply was provided by two 6 V 4.5 Ah sealed lead acid batteries, and two voltage regulators (S7V7F5 and D24V3F3, Pololu Corporation, Las Vegas, Nevada) ensured constant 5 V to the microcontroller and 3.3 V to the SD memory card. Circuit diagrams (Figure S1) were designed using Fritzing software (http://fritzing.org). The Arduino Integrated Development Environment (IDE) software based on the Processing IDE (http://processing.org) was used to program the device. We used this software because our original prototype included the Arduino Uno shield (Wheat 2012), but we later simplified the system by using a stand-alone ATmega 328p. Indeed, a fully preassembled commercial board would have been easier to use but is not practical in chestnut plantations where electricity is not available. Therefore, a stand-alone version with only the bare essentials was needed to ensure a minimum rate of energy use. Two batteries powered the device for up to 3 weeks. The programming code is available in the Supporting Information file.
Designs of the proposed device, which consisted of a wooden cage with an opening in the front and a set of ten plastic catch vials, equidistantly screwed into a plastic turntable affixed to the vertical front side of the cage. Galls were stored inside the cage. The turntable automatically separated D. kuriphilus and its associated parasitoids that emerged at different times
Emergence pattern of D. kuriphilus and associated parasitoids
Galls were randomly collected on two sample dates from the same chestnut forest (coordinates: 42.650°; 12.355°) located in the chestnut production district in the Umbria region, Central Italy, where chestnut trees were heavily infested by D. kuriphilus. Each year from 2011 to 2014, across the whole district, female and male (2:1 ratio) T. sinensis were released soon after chestnut budding. Newly formed galls were randomly collected in June 2013 and 2014 to analyze the emergence pattern of D. kuriphilus and those associated native parasitoids that emerge from galls in the summer. In addition, dry, overwintered galls were collected in March 2014 and 2015 to measure the emergence pattern of T. sinensis, as well as that of those native parasitoids that overwintered inside the galls. For each sampling date, two subsamples of 200 galls each were randomly selected. The surrounding leaf tissue was removed, and the galls were then placed in the cages. To test whether microclimate in the wooden cage may affect the emergence pattern of the females, two different cages were used. One sample of galls was stored in a ventilated fine-mesh bag (30 cm length × 20 cm high × 20 cm wide) provided with an opening window on the front side, while the second sample was placed in the wooden cage we designed. Both cages were placed outdoors (summer: ~ 15 h: 9 h L:D; 15–34 °C; winter/spring: ~ 13 h: 11 h L:D; 9–24 °C) and were protected from direct sun or rain. The emergence of D. kuriphilus and its associated parasitoids was monitored daily, counting the number of adults found on the walls of the mesh bag or that entered the catch vials for the wooden cage. Chalcidoid parasitoids were identified using an unpublished key produced by R.R. Askew and C. Thúroczy (similar to Kos et al. 2015) and by comparison with specimens, deposited at the Department of Agricultural, Food and Environmental Sciences (University of Perugia), that had previously been identified by Dr. Petr Jansta (Charles University, Prague). After emergence ceased, all new or overwintered galls in our samples were dissected, cutting through the outer parenchyma layer to reveal an inner vacuolated parenchyma where larval gall wasp chambers were located, delimited by a few layers of lignified cells (Stone and Schönrogge 2003; Reale et al. 2016). The numbers of dead D. kuriphilus adults and pupae and the number of parasitoid larvae potentially present inside the gall chambers, as well as the number of empty gall chambers, were recorded.
The numbers of D. kuriphilus or parasitoid adults that emerged in the mesh bag versus the automatic device in the wooden cage were compared using the Chi-squared test (R Core Team 2014). Similarly, we compared the numbers of dead D. kuriphilus, parasitoid larvae and empty gall chambers that were found in dissection between the mesh bag and the wooden cage. We also compared D. kuriphilus mortality in new galls versus overwintered galls, as well as the number of emerged parasitoids. Raw emergence numbers were also reported as percentages of outcomes relative to all the gall chambers recorded. To confirm the ability of gall wasp and parasitoid adults to move out of the rearing cage and enter the catch vials, 20 newly emerged D. kuriphilus females or 20 T. sinensis males or 20 females were placed inside the rearing cage and the number captured determined. All insects successfully entered the vials within 24 h.
Results
The percentage of D. kuriphilus wasps that emerged from fresh galls in the mesh cage and were trapped in collection vials did not differ significantly from the percentage that emerged in the wooden cage, in 2013 (Figs. 2, 3 and Table 1, Chi-squared test, P = 0.65) or 2014 (P = 0.86). Overall, 11 species of native parasitoids belonging to five families were collected from fresh galls (Table 1). The emergence of native parasitoids (all species combined) did not differ significantly between mesh bag and wooden cage in either sample year (2013: P = 1; 2014: P = 0.19). Also, there were no significant differences in the number of dead gall wasps inside the gall chambers between mesh bag and our wooden rearing box (2013: P = 0.70; 2014: P = 0.17). Nor were there significant differences in the number of parasitoid larvae inside the gall chambers (2013: P = 0.51; 2014: P = 0.53) or in the number of empty gall chambers (2013: P = 0.59; 2014: P = 0.61) between treatments.
From overwintered galls, there were no significant differences in the number of T. sinensis emerging in the mesh bag compared to the wooden cage, in 2014 (galls formed in 2013: P = 0.80) or 2015 (galls formed in 2014: P = 0.63). In total, five species of native parasitoids belonging to three families were recorded from overwintered galls (Table 1). No differences between treatments were detected in the emergence of overwintered parasitoids (galls formed in 2013: P = 1; galls formed in 2014: P = 0.40), in the occurrence of dead gall wasp individuals found via dissection inside gall chambers (2013: P = 0.50; 2014: P = 0.62), or in the percentage of empty gall chambers (2013: P = 0.58; 2014: P = 0.38).
Overall, higher D. kuriphilus mortality was detected in the fresh galls compared to the overwintered galls in 2013 (χ2 = 197.05, df = 1, P < 0.001) or 2014 (χ2 = 182.44, df = 1, P < 0.001), and fewer parasitoids emerged from fresh galls compared to overwintered galls, formed in 2013 (χ2 = 6.48, df = 1, P = 0.011) or 2014 (χ2 = 15.96, df = 1, P < 0.001).
Discussion
Classical biological control based on the use of the parasitoid T. sinensis is a viable option for managing the chestnut gall wasp (Moriya et al. 2002; Gibbs et al. 2011; reviewed by Csóka et al. 2017), and our results confirm the potential of this parasitoid to be used in Central Italy. Indeed, within 3 years from its release, T. sinensis caused parasitism levels (4.3% in 2013 and 5.5% in 2014) higher than that caused by the local native parasitoids in Italy. Among the native parasitoids, Eupelmus urozonus Dalman was one of the most abundant species, as in other parts of Italy (Quacchia et al. 2012; Francati et al. 2015) and Slovenia (Kos et al. 2015). However, this species is a facultative hyperparasitoid of other D. kuriphilus parasitoids, and its ecological role is still undetermined. Interestingly, we detected a higher number of empty gall chambers in new galls compared to the number of gall inhabitants. Similarly, Cooper and Rieske (2010) found 25% empty gall chambers in Kentucky, USA. This high rate of empty chambers may be due to unknown physiological causes (Cooper and Rieske 2010) or host feeding by adult parasitoids (Murakami and Tokuhisa 1985). Kato and Hijii (1999) observed the presence of empty gall chambers in Japanese chestnut galls ranging from 0.4 to 40.7% and concluded that their presence might be due to host feeding by Torymus beneficus Yasumatsu. Indeed, the presence of feeding tubes inside larval gall chambers was observed in Umbria, Italy, at a rate of 0–12% (authors’ unpublished data). Another possible explanation supposes that empty gall chambers could represent an evolved defensive strategy toward the reduction of parasitoid attacks and, consequently, of gall wasp mortality (Stone and Schönrogge 2003). Alternatively, early mortality from plant resistance might exist (similar to Rondoni et al. 2018), resulting in more empty galls (Cooper and Rieske 2010).
The dissection of overwintered galls showed no evidence of T. sinensis larvae that did not emerge. This might confirm that individuals with a prolonged diapause appear only at a high field density of the parasitoid or at a low host density (Picciau et al. 2017). Intriguingly, a higher number of dead D. kuriphilus was found inside the fresh galls, which was only partially confirmed by the dissection of overwintered galls. A possible explanation is that the collection of the fresh galls accelerated the maturation phase (sensu Stone et al. 2002); therefore, the inner tissues may harden, making it difficult for the gall wasps that are located in the deeper tissues to emerge. However, this possibility deserves future investigation.
In Italy, a protocol for the rearing of T. sinensis has been released by the Ministry of Agricultural, Food and Forestry Policies. This protocol requires the daily collection of T. sinensis emerging from samples of galls and the pairing of parasitoids of similar age. In this respect, biological control practitioners could use our modified rearing cage to efficiently separate individuals by day of emergence and pair them by date. However, in spite of the parasitoid’s release, a risk assessment procedure for its use is still under development (Aebi et al. 2011; Gibbs et al. 2011; Ferracini et al. 2015a, 2017). Hence, the introduction of the parasitoid is still underevaluation in those chestnut plantations that are located in specially protected areas, recognized by the European Union as ‘Natura 2000.’
Alternative strategies may be necessary against D. kuriphilus. Chemical treatments with systemic insecticides intended to kill both young and mature larvae did not provide effective control, because of the histological morphology of the galls, which protects the developing larvae from pesticide exposure (Bosio et al. 2009; Reale et al. 2016). Treatments with entomopathogenic fungi have also been proposed (Tosi et al. 2015), but their effectiveness under field conditions is still underevaluation. Instead, treatments targeted to adults with kaolin or pyrethroids (although only locally authorized in chestnut ecosystems) seem to cause high female mortality and contribute to a lower infestation of the buds (Bosio et al. 2009). However, to effectively protect chestnut buds, repeated applications timed to the presence of D. kuriphilus adults are needed (Bosio et al. 2009). Repeated treatments are needed because female gall wasps are short-lived (few days according to Romani et al. 2010) but their emergence from galls extends for up to 5 weeks (Bernardo et al. 2013). Moreover, our results confirm that the emergence of D. kuriphilus partly overlaps with that of its indigenous parasitoids (Bernardo et al. 2013), which start to emerge from the galls before D. kuriphilus adults (Santi and Maini 2011; Panzavolta et al. 2013; Francati et al. 2015) and then migrate to other hosts (e.g., gall wasps developing on oak trees). In this respect, to avoid insecticide side effects on parasitoids, the emergence of both D. kuriphilus adults and parasitoids should be carefully monitored to selectively time chemical treatments.
Dark rearing boxes have been successfully used to track the emergence of herbivorous insects from excised plants and of their associated parasitoids. Such rearing boxes have been used for species of cerambycid beetles (Millar et al. 2002; Paine et al. 2004), leafhoppers (Corbett and Rosenheim 1996), the moth Tuta absoluta (Meyrick) (Ferracini et al. 2012), the exotic leaf miner Cameraria ohridella Deschka and Dimic (Girardoz et al. 2006), the oak gall wasp Synergus itoensis Abe, Ide and Wachi (Abe et al. 2011) and the Asian chestnut gall wasp, D. kuriphilus (Cooper and Rieske 2007; Quacchia et al. 2008; Santi and Maini 2011; Quacchia et al. 2012). The effect of the microclimate inside the rearing cage on the development of the gall-forming midge Rhopalomyia californica Felt has been previously investigated (Boukili et al. 2007). Although the cages slightly altered the microclimate, this had a negligible effect on the timing of insect emergence, confirming that such cages can be used to predict field event (Boukili et al. 2007). In our case, the phenology and rates of emergence of D. kuriphilus and its parasitoids from mesh versus wooden cages were very similar. Other methods can be used for monitoring the gall wasp populations, although likely with less accuracy. For example, yellow sticky traps were shown to be an effective method to detect the pest presence (Böckmann et al. 2015), and more specifically to assess the emergence of D. kuriphilus adults, and could potentially be used to define the correct timing of pesticide applications (Bernardo et al. 2013). However, because yellow sticky traps might attract all flying insects, they are not suitable to distinguish the parasitoids that emerge directly from the galls from those that have emerged from other hosts in the surrounding environment and then immigrated into the chestnut plantation.
Using our open-source microcontroller-based device, we were able to track the daily emergence of the invasive gall wasp D. kuriphilus and its associated parasitoids, thus providing a simple method, which would be useful, both for research and implementation purposes.
Author Contribution Statement
GR and EC wrote the paper. GR and CR collected the data. GR designed the device and analyzed the data. All authors conceived the research, read and reviewed the paper.
References
Abe Y, Ide T, Wachi N (2011) Discovery of a new gall-inducing species in the inquiline tribe Synergini (Hymenoptera: Cynipidae): inconsistent implications from biology and morphology. Ann Entomol Soc Am 104:115–120
Aebi A, Schönrogge K, Melika G, Alma A, Bosio G, Quacchia A et al (2006) Parasitoid recruitment to the globally invasive chestnut gall wasp Dryocosmus kuriphilus. In: Ozaki K, Yukawa J, Ohgushi T, Price PW (eds) Ecology and evolution of galling arthropods and their associates. Springer, Tokyo, pp 103–121
Aebi A, Schonrogge K, Melika G, Quacchia A, Alma A, Stone GN (2007) Native and introduced parasitoids attacking the invasive chestnut gall wasp Dryocosmus kuriphilus. IOBC WPRS Bull 37:166–171
Aebi A, Schoenenberger N, Bigler F (2011) Evaluating the use of Torymus sinensis against the chestnut gall wasp Dryocosmus kuriphilus in the Canton Ticino, Switzerland. Agroscope Reckenholz-Tänikon, Zürich
Battisti A, Benvegnù I, Colombari F, Haack RA (2014) Invasion by the chestnut gall wasp in Italy causes significant yield loss in Castanea sativa nut production. Agric For Entomol 16:75–79
Bernardo U, Iodice L, Sasso R, Tutore VA, Cascone P, Guerrieri E (2013) Biology and monitoring of Dryocosmus kuriphilus on Castanea sativa in Southern Italy. Agric For Entomol 15:65–76
Böckmann E, Hommes M, Meyhöfer R (2015) Yellow traps reloaded: What is the benefit for decision making in practice? J Pest Sci 88:439–449
Bonal R, Vargas-Osuna E, Mena JD, Aparicio JM, Santoro M, Martín A (2018) Looking for variable molecular markers in the chestnut gall wasp Dryocosmus kuriphilus: first comparison across genes. Sci Rep 8:5631
Bosio G, Gerbaudo C, Piazza E (2009) Dryocosmus kuriphilus Yasumatsu: an outline seven years after the first report in Piedmont (Italy). Acta Hortic 866:341–348
Boukili VKS, Hoopes MF, Briggs CJ (2007) Effect of microenvironment on development of a gall midge. Environ Entomol 36:441–450
Brussino G, Bosio G, Baudino M, Giordano R, Ramello F, Melika G (2002) A dangerous exotic insect threatening European chestnut. Inf Agrar 58:59–61 (Italian)
Buchholz S (2009) Design of a time-sorting pitfall trap for surface-active arthropods. Entomol Exp Appl 133:100–103
Colombari F, Battisti A (2016) Native and introduced parasitoids in the biocontrol of Dryocosmus kuriphilus in Veneto (Italy). EPPO Bull 46:275–285
Comont RF, Purse BV, Phillips W, Kunin WE, Hanson M, Lewis OT et al (2014) Escape from parasitism by the invasive alien ladybird, Harmonia axyridis. Insect Conserv Divers 7:334–342
Cooper WR, Rieske LK (2007) Community associates of an exotic gallmaker, Dryocosmus kuriphilus (Hymenoptera: Cynipidae), in eastern North America. Ann Entomol Soc Am 100:236–244
Cooper WR, Rieske LK (2010) Gall structure affects ecological associations of Dryocosmus kuriphilus (Hymenoptera: Cynipidae). Environ Entomol 39:787–797
Corbett A, Rosenheim JA (1996) Quantifying movement of a minute parasitoid, Anagrus epos (Hymenoptera: Mymaridae), using fluorescent dust marking and recapture. Biol Control 6:35–44
Csóka G, Stone G, Melika G (2017) Non-native gall-inducing insects on forest trees: a global review. Biol Invasions 19:3161–3181
EFSA Panel on Plant Health (2010) Risk assessment of the oriental chestnut gall wasp, Dryocosmus kuriphilus for the EU territory and identification and evaluation of risk management options. EFSA J 8:1–114
EPPO (2017) PQR—EPPO database on quarantine pests. http://eppo.int. Accessed 28 Dec 2017
Ferracini C, Ingegno BL, Navone P, Ferrari E, Mosti M, Tavella L, Alma A (2012) Adaptation of indigenous larval parasitoids to Tuta absoluta (Lepidoptera: Gelechiidae) in Italy. J Econ Entomol 105:1311–1319
Ferracini C, Ferrari E, Saladini MA, Pontini M, Corradetti M, Alma A (2015a) Non-target host risk assessment for the parasitoid Torymus sinensis. Biocontrol 60:583–594
Ferracini C, Gonella E, Ferrari E, Saladini MA, Picciau L, Tota F et al (2015b) Novel insight in the life cycle of Torymus sinensis, biocontrol agent of the chestnut gall wasp. Biocontrol 60:169–177
Ferracini C, Ferrari E, Pontini M, Nova LKH, Saladini MA, Alma A (2017) Post-release evaluation of non-target effects of Torymus sinensis, the biological control agent of Dryocosmus kuriphilus in Italy. BioControl 62:445–456
Francati S, Alma A, Ferracini C, Pollini A, Dindo ML (2015) Indigenous parasitoids associated with Dryocosmus kuriphilus in a chestnut production area of Emilia Romagna (Italy). Bull Insectol 68:127–134
Gibbs M, Schonrogge K, Alma A, Melika G, Quacchia A, Stone GN, Aebi A (2011) Torymus sinensis: A viable management option for the biological control of Dryocosmus kuriphilus in Europe? Biocontrol 56:527–538
Girardoz S, Kenis M, Quicke DL (2006) Recruitment of native parasitoids by an exotic leaf miner, Cameraria ohridella: host-parasitoid synchronization and influence of the environment. Agric For Entomol 8:49–56
Graziosi I, Rieske LK (2014) Potential fecundity of a highly invasive gall maker, Dryocosmus kuriphilus (Hymenoptera: Cynipidae). Environ Entomol 43:1053–1058
Graziosi I, Santi F (2008) Chestnut gall wasp (Dryocosmus kuriphilus): spreading in Italy and new records in Bologna province. Bull Insectol 61:343–348
Hill AP, Prince P, Covarrubias EP, Doncaster CP, Snaddon JL, Rogers A (2018) AudioMoth: evaluation of a smart open acoustic device for monitoring biodiversity and the environment. Methods Ecol Evol 9:1199–1211
Kato K, Hijii N (1997) Effects of gall formation by Dryocosmus kuriphilus Yasumatsu (Hym., Cynipidae) on the growth of chestnut trees. J Appl Entomol 121:9–15
Kato K, Hijii N (1999) Mortality factors of the chestnut gall-wasp, Dryocosmus kuriphilus Yasumatsu (Hymenoptera: Cynipidae) after gall formation. Entomol Sci 2:483–491
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17:164–170
Kos K, Kriston E, Melika G (2015) Invasive chestnut gall wasp Dryocosmus kuriphilus (Hymenoptera: Cynipidae), its native parasitoid community and association with oak gall wasps in Slovenia. Eur J Entomol 112:698–704
Maltoni A, Mariotti B, Tani A (2012) Case study of a new method for the classification and analysis of Dryocosmus kuriphilus Yasumatsu damage to young chestnut sprouts. iForest 5:50–59
Martorana L, Foti MC, Rondoni G, Conti E, Colazza S, Peri E (2017) An invasive insect herbivore disrupts plant volatile-mediated tritrophic signalling. J Pest Sci 90:1079–1085
Millar J, Paine T, Campbell C, Hanks L (2002) Methods for rearing Syngaster lepidus and Jarra phoracantha (Hymenoptera: Braconidae), larval parasitoids of the phloem-colonizing longhorned beetles Phoracantha semipunctata and P. recurva (Coleoptera: Cerambycidae). Bull Entomol Res 92:141–146
Moriya S, Masakazu S, Ishizue A (2002) Classical biological control of the chestnut gall wasp in Japan. In: 1st international symposium on biological control of arthropods, Waikiki, Japan, pp 407–415
Murakami Y, Tokuhisa E (1985) Behavioural sequences of oviposition and host-feeding of Torymus (Syntomaspis) beneficus Yasumatsu et Kamijo (Hymenoptera: Torymidae), a native parasitoid of Dryocosmus kuriphilus Yasumatsu (Hymenoptera: Cynipidae). Appl Entomol Zool 20:43–49
Paine TD, Joyce AL, Millar JG, Hanks LM (2004) Effect of variation in host size on sex ratio, size, and survival of Syngaster lepidus, a parasitoid of Eucalyptus longhorned beetles (Phoracantha spp.): II. Biol Control 30:374–381
Panzavolta T, Bracalini M, Croci F, Campani C, Bartoletti T, Miniati G et al (2011) Asian chestnut gall wasp in Tuscany: gall characteristics, egg distribution and chestnut cultivar susceptibility. Agric For Entomol 14:139–145
Panzavolta T, Bernardo U, Bracalini M, Cascone P, Croci F, Gebiola M et al (2013) Native parasitoids associated with Dryocosmus kuriphilus in Tuscany, Italy. Bull Insectol 66:195–201
Picciau L, Ferracini C, Alma A (2017) Reproductive traits in Torymus sinensis, biocontrol agent of the Asian chestnut gall wasp: implications for biological control success. Bull Insectol 70:49–55
Quacchia A, Moriya S, Bosio G, Scapin I, Alma A (2008) Rearing, release and settlement prospect in Italy of Torymus sinensis, the biological control agent of the chestnut gall wasp Dryocosmus kuriphilus. Biocontrol 53:829–839
Quacchia A, Ferracini C, Nicholls JA, Piazza E, Saladini MA, Tota F et al (2012) Chalcid parasitoid community associated with the invading pest Dryocosmus kuriphilus in north-western Italy. Insect Conserv Divers 6:114–123
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Reale L, Tedeschini E, Rondoni G, Ricci C, Bin F, Frenguelli G, Ferranti F (2016) Histological investigation on gall development induced by a worldwide invasive pest, Dryocosmus kuriphilus, on Castanea sativa. Plant Biosyst 150:35–42
Rieske LK (2007) Success of an exotic gallmaker, Dryocosmus kuriphilus, on chestnut in the USA: a historical account. IOBC WPRS Bull 37:172–174
Rohde B, Paris T, Heatherington E, Hall D, Mankin R (2013) Responses of Diaphorina citri (Hemiptera: Psyllidae) to conspecific vibrational signals and synthetic mimics. Ann Entomol Soc Am 106:392–399
Romani R, Rondoni G, Gragnoli L, Pergolari P, Santinelli C, Rossi Stacconi MV, Ricci C (2010) Bio-ethological and morphological investigations on Dryocosmus kuriphilus Yasumatsu. Atti Accad Naz Italiana Entomol Rend 58:97–104 (in Italian, English abstract)
Rondoni G, Bertoldi V, Malek R, Foti MC, Peri E, Maistrello L et al (2017) Native egg parasitoids recorded from the invasive Halyomorpha halys successfully exploit volatiles emitted by the plant–herbivore complex. J Pest Sci 80:1087–1095
Rondoni G, Bertoldi V, Malek R, Djelouah K, Moretti C, Buonaurio R, Conti E (2018) Vicia faba plants respond to oviposition by invasive Halyomorpha halys activating direct defences against offspring. J Pest Sci 91:671–679
Santi F, Maini S (2011) New association between Dryocosmus kuriphilus and Torymus flavipes in chestnut trees in the Bologna area (Italy): first results. Bull Insectol 64:275–278
Stone GN, Schönrogge K (2003) The adaptive significance of insect gall morphology. Trends Ecol Evol 18:512–522
Stone GN, Schönrogge K, Atkinson RJ, Bellido D, Pujade-Villar J (2002) The population biology of oak gall wasps (Hymenoptera: Cynipidae). Annu Rev Entomol 47:633–668
Tosi L, Beccari G, Rondoni G, Covarelli L, Ricci C (2015) Natural occurrence of Fusarium proliferatum on chestnut in Italy and its potential entomopathogenicity against the Asian chestnut gall wasp Dryocosmus kuriphilus. J Pest Sci 88:369–381
Ugolini F, Massetti L, Pedrazzoli F, Tognetti R, Vecchione A, Zulini L, Maresi G (2014) Ecophysiological responses and vulnerability to other pathologies in European chestnut coppices, heavily infested by the Asian chestnut gall wasp. For Ecol Manag 314:38–49
Warmund MR (2013) Dryocosmus kuriphilus-induced chestnut galls and their inhabitants. HortScience 48:969–974
Wheat D (2012) Arduino internals. Apress, New York
Wickert AD (2014) The ALog: inexpensive, open-source, automated data collection in the field. Bull Ecol Soc Am 95:166–176
Yasumatsu K (1951) A new Dryocosmus injurious to chestnut trees in Japan (Hym., Cynipidae). Mushi 22:89–93
Acknowledgements
Authors would like to thank Davide Pettorossi, Marida Corradetti, Quirino Santini, Daniela Fortini and Luciana Bartoli for assistance and technical support.
Funding
Personal funding to GR was provided by ‘Cassa di Risparmio di Perugia’ Foundation and by PSR UMBRIA 2014–2020, mis. 19.2, ‘PAL GAL Media Valle del Tevere’. Work was supported by PSR UMBRIA 2007-2013, mis. 1.2.4 project ‘CoBiot.’
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Human and animal rights
This article does not contain any studies with human participants or animals (vertebrates) performed by any of the authors.
Additional information
Communicated by J. J. Duan.
Special Issue on Invasive Pests of Forests and Urban Trees.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rondoni, G., Ricci, C. & Conti, E. Tracking seasonal emergence dynamics of an invasive gall wasp and its associated parasitoids with an open-source, microcontroller-based device. J Pest Sci 92, 361–369 (2019). https://doi.org/10.1007/s10340-018-1037-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-018-1037-2