Abstract
In this study, a rapid reversed-phase liquid chromatography method for the determination of azithromycin and its related substances was developed and validated on a novel polyfunctional silyl reagent [hexanhexamethyloctadecyltetrasiloxane (HMODTS–C18)] bonded core–shell RP C-18 column (100 × 4.6 mm, 2.6 µm) as per ICH guidelines. A binary gradient elution programme with a mixture of solvent A (phosphate buffer, pH 8.9) and solvent B (ACN: MeOH, 3:1) as mobile phase, at a flow rate of 1.5 mL min−1 and detection at 210 nm was finally optimized for the study. The validation results showed that the method to be specific, selective, highly sensible (0.004 mg mL−1), precise (% RSD ≤ 10), linear and accurate in a concentration range of 0.004–0.032 mg mL−1. Simple SPE method was performed for extraction and checked for repeatability. The result showed that the method is precise (%RSD < 2). The validated chromatographic method was applied to the solid-phase extracted samples of azithromycin and its four major related substances. Also, docking study was carried out using AutoDock Tools to find out the binding affinities, number of hydrogen bonds and residues involved in hydrogen bonds for azithromycin and its four major related impurities with the human plasma proteins. The results confirmed the applicability of the proposed method on the extracted human plasma samples. Finally, the study demonstrates that the impurities of azithromycin have strong binding affinities with the plasma proteins compared to that of azithromycin and half life of these impurities may be higher than azithromycin which can cause major health risk on repeated doses of azithromycin.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Various guidelines and stringent specifications imposed by regulative authorities, such as the International Conference on Harmonization (ICH), the United States Food and Drug administration (FDA), European Medical agency (EMA), Therapeutic Governance Authority (TGA) and Canadian Drug and Health Agency (CDHA) accentuate the purity of the drug substances and identification of the related substances in active pharmaceutical ingredients (API) and finished products. These related substances can be inorganic, organic and volatile solvents. A small amount of these may manipulate the efficacy and potency of the drug substances and also could cause some toxicological problems.
Azithromycin (AZM), a 15-atom lactone macrolide ring compound is a commonly prescribed antibiotic, highly active against Chlamydia and moderately active against M avium complex and T gondii. Pharmacokinetic properties of AZM reveal its low serum concentrations of approximately 400 mg mL -1 on a daily dose of 500 mg for adults. Penetration of AZM into most tissues and phagocytic cells (except the cerebrospinal fluid) are rapid followed by a slow release from the tissues. Due to its longer half life, AZM is prescribed as a single dose of 500 mg/day for three consecutive days. Cytochrome P450 enzymes are not activated by AZM, so it does not cause any drug interactions as that of other macrolides [1].
There are many reports on the estimation of azithromycin and its related substances in bulk pharmaceuticals and finished dosage forms [2]. Also, some reported its analysis in blood plasma but none paid an attention to the impurities that binds to the blood plasma and the half life’s of those impurities. Investigations after short-term administration (3 days) showed accumulation of AZM in white blood cells [3, 4]. United States Pharmacopoeia monograph for azithromycin reports 13 known impurities. Although, specific limits of individual impurities have been specified in various pharmacopoeias, still these impurities could be genotoxic to the human cells and their binding affinity studies need to be carried out.
Currently, core–shell columns bridged the gap between pressure and time by supplementing chromatography with selective short run time methods. The size distribution of a core–shell particle is narrower than a conventional porous particle; therefore, the space between the particle is reduced which increases the efficiency by decreasing eddy diffusion, longitudinal diffusion and diffusion path [5]. Our proposed study was optimized on a core–shell particle column which is bonded with a novel polyfunctional silyl reagent called hexamethyloctadecyltetrasiloxane (HMODTS–C18) which adds more stability to the column for the analysis of certain basic drugs. Highlighting its bonding chemistries, the column is also finally end-capped with trimethylchlorosilane and base deactivated to mask the residual silanol groups. Its HMODTS–C18 group is a short chain which acts as a shield to cover up the residual silanol groups (if any) after base deactivation and end-capping, resulting in increasing the stability and life of the column. The column’s reduced particle size has advantages of resolving the acidic and basic compounds, in an UFLC under reduced pressure, low solvent consumption, shorter run time of analysis, shorter equilibration time, stability in operating temperature in excess of 80 °C for hours which increases its column life and finally good resolution [6].
United States Pharmacopoeia, British Pharmacopoeia, European Pharmacopoeia and Indian Pharmacopoeia allow a change in the column length by ± 70%, flow rate by ± 50%, reduction in the particle size of the column by 50% and column diameter with ± 25%, without changing the solvent system of the method [7,8,9,10,11]. The proposed method for organic impurities in azithromycin stated in United States Pharmacopoeia was carried out on a SunShell RP-C18 (100 × 4.6 mm, 2.6 µ) column. The gradient method was transferred to this short core–shell column by calculating the runtime using the gradient method transfer software [12]. High stability (temperature resistant up to 80 °C) and pH range (1.5–10) and low bleeding made the column suitable for the proposed study [13, 14].
Hence, this study aims to develop a rapid, specific, accurate and precise HPLC method using core–shell column for the determination of AZM and its major related substances (Fig. 1) in bulk, dosage forms and human plasma, with reference to maximum allowable changes as per United States Pharmacopoeia, British Pharmacopoeia, European Pharmacopoeia and Indian Pharmacopoeia on a related substance method of azithromycin mentioned in USP monograph [15]. To the best of authors’ knowledge, such a method has never been reported earlier. It can be predicted that the proposed method could be validated and applied on quality control laboratories for routine analysis. The docking of the molecules gives a sheer picture and comparison of the binding affinities of AZM and its major impurities with human plasma proteins.
Materials and Methods
Materials
Azithromycin reference standard, azaerythromycin A (IMP-A), azithromycin N-oxide, desosaminylazithromycin, 3′-N-demethylazithromycin impurity reference standards were purchased from United States Pharmacopeial convention. Methanol, acetonitrile, ortho-phosphoric acid of HPLC grade were purchased from Merck; anhydrous dibasic sodium phosphate and monobasic ammonium phosphate of chromatographic grade were purchased from Merck; sodium hydroxide of AR grade was purchased from Merck and ammonium hydroxide of AR grade was purchased from Rankem. Azithromycin tablet formulation was purchased from the market.
Instrumentation
The chromatograph consisted of a Shimadzu LC-2010CHT system model equipped with binary pump LC-20AD, autosampler, degasser, column oven, and UV detector. The analysis, collection of data and integration of data’s were carried out using LC solution software. Solid-phase extraction assembly of Varian USA was used and 1-ml Sep-Pac C18 cartridges (1.0 mL Waters, Milky Way, USA) were used for carrying out solid-phase extraction.
Chromatographic Conditions
The chromatographic separation was performed on a ChromaNik Technologies Inc., Japan, SunShell RP-C18 column (core–shell column), 100 mm × 4.6 mm, 2.6 μm, purchased from Prochrome India, Mumbai. The separation was achieved with a binary gradient program for solution A (phosphate buffer, pH 8.9) and solution B (ACN: MeOH, 3:1), as shown in Table 1, with a flow rate of 1.5 mL min−1, injection volume of 20 μL, column oven temperature maintained at 60 °C, sample oven temperature maintained at 4 °C and detection wavelength of 210 nm.
Preparation of Solutions
Preparation of Solution A
Anhydrous dibasic sodium phosphate was weighed and transferred to prepare a solution of 1.8 mg mL−1 in water. The pH of the solution was adjusted to 8.9 with 1 N sodium hydroxide or 10% ortho-phosphoric acid. The solution was filtered using 0.45-µ membrane filter and degassed.
Preparation of Solution B
A mixture of acetonitrile and methanol was prepared in the ratio of 3:1.
Preparation of Solution C
Monobasic ammonium phosphate was weighed and transferred to prepare a solution of 1.73 mg mL−1 in water. The pH of the solution was adjusted to 10.0 ± 0.05 with ammonium hydroxide. The solution was filtered using 0.45-µ membrane filter and degassed.
Preparation of Solution D
A mixture of methanol, acetonitrile and solution C was prepared in the ratio of 7:6:7.
Preparation of Solution E
A mixture of methanol and solution C was prepared in the ratio of 1:1.
Preparation of Standard Stock Solution
Azithromycin standard solution was prepared by dissolving azithromycin in solution D using about 75% of the final volume. Sonicated to dissolve and diluted to volume with solution D. Mixed properly to obtain a solution having a known concentration of 0.4 mg mL−1 of azithromycin.
Preparation of Standard Solution
0.02 mg mL−1 of azithromycin was prepared from the standard stock solution in solution D.
Preparation of System Suitability or Resolution Stock Solution
0.2 mg mL−1 of azaerythromycin A (IMP-A) was prepared in acetonitrile. It was sonicated to dissolve.
Preparation of System Suitability or Resolution Solution
A mixed solution of 0.02 mg mL−1 of azaerythromycin A (IMP-A) was prepared from the system suit stock solution and 0.02 mg mL−1 of azithromycin from the standard stock solution in solution D.
Preparation of System Sensitivity Solution
0.004 mg mL−1 of azithromycin was prepared from standard solution in solution D.
Preparation of Blank Solution
Solution D is used as the blank solution.
Preparation of Placebo Stock Solution
The density of azithromycin suspension placebo was calculated. Accurately weighed suspension placebo equivalent to formulation sample containing 1335 mg of azithromycin (excluding active substance) was transferred into a 100-mL volumetric flask. 75 mL of acetonitrile was added, shaken and sonicated for not less than 15 min. The mixtures were shaken by mechanical means for not less than 15 min. The mixtures were allowed to equilibrate to room temperature and the volume was made up to the mark with acetonitrile and mixed. The mixtures were filtered through GF/C filter paper.
Preparation of Placebo Solution
An aliquot of the placebo stock solutions was centrifuged at 700 RCF for 10–15 min and 3.0 mL of the supernatant was transferred into a 10-mL volumetric flask and diluted with solution E to volume and mixed. The mixture was filtered through GF/C filter paper.
Preparation of Sample Stock Solution
The density of azithromycin suspension was calculated. Accurately weighed suspension equivalent to formulation sample containing 1335 mg of azithromycin were transferred into a 100-mL volumetric flask. 75 mL of acetonitrile was added, shook and sonicated for not less than 15 min. The mixtures were shaken by mechanical means for not less than 15 min. The mixtures were allowed to equilibrate to room temperature and the volume was made up to the mark with acetonitrile and mixed. The mixtures were filtered through GF/C filter paper.
Preparation of Sample Solution
An aliquot of the sample stock solutions was centrifuged at 700 RCF for 10–15 min and 3.0 mL of the supernatant was transferred into a 10-mL volumetric flask, and diluted with solution E to volume and mixed. The mixture was filtered through GF/C filter paper.
Extraction of the Drugs from Plasma by Solid-Phase Extraction (SPE)
To determine azithromycin and its related impurity concentration in human body, SPE was carried out. Sample solution was prepared by taking 1.0 mL (0.02 mg mL−1) of the standard impurities (azithromycin 3′-N-oxide, desosaminylazithromycin, 3′-N-demethylazithromycin, azaerythromycin A) and standard azithromycin (2.0 mg mL−1), mixed with 5.0 mL fresh frozen human plasma. The sample solution was kept in an incubator at 37 °C for 24 h. For extraction, 15.0 mL of acetone was mixed with sample and kept for 30 min. The sample solution was centrifuged at 10,000 rpm (11,180g) for 10.0 min to separate the supernatant. The supernatant was evaporated to dryness under vacuum. The residue was re-dissolved in 10.0 mL phosphate buffer (25 mM, pH 8.9). Sep-Pac C18 cartridges (1.0 mL Waters, Milky Way, USA) were pre-conditioned with 2.0 mL methanol and 5.0 mL Millipore water, separately. The buffer containing the drugs were passed through the cartridge at a flow rate of 0.1 mL min−1, followed by washing of the cartridge with 2.0 mL Millipore water at the same flow rate. The cartridge was dried by passing hot air followed by elution of the drugs using 10.0 mL methanol at a flow rate of 0.1 mL min−1. The eluted methanol solution of the drugs was concentrated under vacuum to 0.5 mL, separately. The sample was further used for HPLC analysis.
Results and Discussion
Optimization of Chromatographic Conditions
For the successful separation of AZM and its related impurities, different types of column and mobile phase have been tried. But best results were obtained in core–shell RP-C18 column (100 × 4.6 mm, 2.6 µm) using binary gradient elution with mixture of solvent A (phosphate buffer, pH 8.9) and solvent B (ACN:MeOH, 3:1) as mobile phase at a flow rate of 1.5 mL min−1 and detection at 210 nm.
Method Validation
Method validation was performed in accordance with ICH (2005) guidelines [16,17,18,19]. The results were evaluated using standard statistical packages for Windows and Graph Pad Prism 4.0 (Graph Pad Software Inc., USA).
System Sensitivity Solution
System sensitivity solution was injected and the peak response was recorded. The acceptance criteria as per United States Pharmacopoeia are the signal-to-noise ratio for the azithromycin peak should not be less than 10. The limit of quantification (LOQ) of this method was determined as a signal-to-noise ratio of 10 for LOQ. The signal-to-noise ratio for LOQ concentration of 4 µg/mL was found to be 12.22 for azithromycin.
System Suitability Test or Resolution Test
System suitability solution was injected and the resolution of azaerythromycin A (IMP-A) and azithromycin was evaluated (Fig. 2). The acceptance criteria as per United States Pharmacopoeia are not less than 2.5. The resolution between azaerythromycin A (IMP-A) and azithromycin was determined to be 17.388.
Specificity
A study was conducted for the interference of diluent and placebo with azithromycin and its related impurities. Samples were prepared by taking the placebo equivalent to about the weight in portion of test preparation as per the test method. Azithromycin spiked in the placebo of suspension was prepared and injected into the HPLC system (Figs. 3, 4). The results are summarized in Table 2. No peaks are eluted at the retention time of azithromycin and its related impurities.
Linearity
The calibration curve for AZM was plotted with different concentrations from the LOQ level, that is, 4–32 µg/mL. The detector response was linear. The linearity was assessed by calculating the slope, y-intercepts and coefficient of determination (r2) using a least squares regression equation. The detailed descriptions of regression curves are depicted in Table 3, which shows good linearity (coefficient of determination r2 = 0.997) for azithromycin in the examined concentration range. The acceptance criteria for the correlation coefficient (r) are NLT 0.995 and the regression coefficient (r2) is NLT 0.99. Y-intercept, slope of regression line should be reported. %Y-intercept at 100% target concentration should be NMT ± 5.0%0.28% and the selected wavelength for all the three markers was 254 nm.
Precision
Precision of the test method was determined by injecting test preparation and tested through the complete analytical procedure from sample preparation to final result. Repeatability assessed using a minimum of six determinations and calculated % relative standard deviation of impurities was calculated. The acceptance criteria for precision are % RSD for all the reported impurities of azithromycin of all the six replicate sample injections are ≤ 10. The test results meet the acceptance criteria. The % RSD of all the reported impurities is shown in Table 4.
Accuracy
The accuracy of the related substances test procedure was determined by spiking of azithromycin on placebo of suspension and injecting triplicate samples of LOQ, 50%, 100% and 150% in triplicate at the target concentration of 0.02 mg mL−1 of azithromycin. The % recovery was calculated. The acceptance criterion is the mean recovery of the impurities at each level should be between 85.0% and 115.0% and the test results meet the acceptance criterion. The details of the % recovery results are shown in Table 5.
Robustness
When the chromatographic conditions were deliberately varied (flow), the system suitability parameter meets the acceptance criteria and there is no change in the relative retention time for the impurities of azithromycin.
Calculation of Related Compounds
The percentage of each related compound in the portion of suspension is given by the formula:
The relative response factor (RRF) values are listed in Table 6, according to the United States Pharmacopoeia 34- NF 29 monograph of azithromycin and azithromycin tablets. The known, unknown and the total impurities should meet the limits listed in Table 6. The peaks obtained in sample solution that correspond to peaks in blank and placebo solutions should be disregarded and the peaks having a response of less than 0.1% are to be disregarded. Peaks eluting before azithromycin 3′-N-oxide and after azithromycin B should be disregarded as per USP azithromycin specification.
Core–Shell Versus Fully Porous Particle
There are many reports which provide the comparison data between a core–shell column and fully porous columns. Core–shell particle supersedes fully porous particle in respect of column efficiency, selectivity as well as reproducibility with advantages of fast analysis, sharp peak shape and increased resolution. The advantage lies in the particle size, giving a backpressure equivalent to fully porous particle of higher micron. To elaborate a 2.6-µ particle size in core–shell provides a back pressure equivalent to a 5-µ fully porous particle. To elaborate it more specifically, the morphology of core–shell particle needs to be understood on a molecular level. It is the morphology of the core–shell particle that improves the efficacy of a column. This is in accordance with the van Deemter equation:
where h is the reduced plate height, v the reduced velocity, and A, B, and C numerical coefficients related to the parameters of the column.
The A term (eddy dispersion term) of the van Deemter equation provides a major clue to the performance of core–shell column. This parameter is completely dependent on the particle size which increases the packing efficiency of the core–shell column than of fully porous particle. Fully porous media has smoother surface compared to solid core–media due to which shear stress which can be applied is more during packing of the column. Therefore, this minimizes the chance of channeling and band broadening [20].
The B term of the van Deemter equation states the diffusion model or longitudinal diffusion occurring within the column. The major factor for using solid core materials is the reduction of the dead volume of the column. A fully porous material packed into a column occupies about one-third of the column volume whereas a solid core–particle occupies about 20–30% more volume than a fully porous material [21]. This causes decrease in column volume of a packed core–shell column which results in less longitudinal diffusion occurring within the column.
According to van Deemter equation, the final term is the C term or resistance to mass transfer. The mass transfer effect is due to the different flow velocities within the mobile phase and not just within the stagnant regions of the pore structure. So, it depends on the molecular weight of the molecule. The porous layer has little effect on the overall h value for small molecules whereas for larger molecules there is increased effect. Although there is difference in the C term for small molecule and large molecule, the overall dispersion is very minimal.
In our current study, for the organic impurities of azithromycin molecule on core–shell column, it can be inferred that due to the advantage of the core–shell particle discussed for the A, B and C terms of the van Deemter equation, the efficiency was better than the data published on fully porous particle by various authors. Also the resolution was much more than the limit published on the USP monograph.
Applicability of the Method to SPE Samples
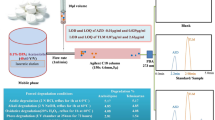
Solid-phase extraction (SPE) method was used to separate the drug mixture and any other new molecules from plasma. To determine the efficiency of the reported SPE method, the percentage recovery of azithromycin and some of its major related impurities was calculated. The percentage recovery of azithromycin and its major related impurities was determined by running the blank experiment (Figs. 5, 6). The calculated percentage recovery of azithromycin and its major related impurities from plasma is given in Table 7. A perusal of this table indicates that the value of the percentage recovery of azithromycin was 35.63% and its related impurities were 20.31%, 32.18%, 30.92% and 28.36% for azithromycin 3′-N-oxide, desosaminylazithromycin, 3′-N-demethylazithromycin and azaerythromycin A, respectively. The remaining amount of the drug binds to plasma protein. The optimization of SPE was achieved by varying different experimental conditions such as pH of phosphate buffer, the flow rates of plasma samples and eluting solvents. Various eluting solvents such as methanol, acetonitrile and combinations of methanol:acetonitrile (50:50, 40:60, 30:70) were also used. As a result of exhaustive experimentation, the best eluting solvent found to be was methanol:acetonitrile in the ratio of 30:70. The maximum percentage recovery of azithromycin and its related impurities was achieved using phosphate buffer (25.0 mM, pH 7.0), at 0.1 mL min−1 flow rate. The values of % RSD for six repeated numbers of injections of standard solution and % recovery of six samples show the method to be specific, precise and accurate. The results are shown in Table 8.
Docking of Azithromycin and Its Major Related Impurities
Molecular docking is a good tool for predicting the interactions of drugs with various macromolecules at supramolecular level. The docking study was performed for azithromycin and its major related impurities (azithromycin 3′-N-oxide, desosaminylazithromycin, 3′-N-demethylazithromycin and azaerythromycin A) with human serum albumin protein (PDB ID: 2BXG) [22] using AutoDock 4.2, AutoDock Vina and further visualized by PyMol software [23]. The binding affinities of the compounds was found in the order of azithromycin 3′-N-oxide > azaerythromycin A > 3′-N-demethylazithromycin > azithromycin > desosaminylazithromycin and the hydrogen bonds involved are 3, 7, 3, 2, 4 for azithromycin 3′-N-oxide, desosaminylazithromycin, 3′-N-demethylazithromycin, azaerythromycin A and azithromycin, respectively. The docking parameters are shown in Table 9. The docked models of azithromycin 3′-N-oxide, desosaminylazithromycin, 3′-N-demethylazithromycin, azaerythromycin A and azithromycin are shown (Figs. 7, 8).
Conclusion
This proposed study uses a short length core–shell column for the routine analysis of azithromycin and its major related impurities in bulk, finished dosage forms and human plasma. The method was rapid and selective as all the impurities resolute well within 30 min. Validation of the method was performed as per ICH guidelines. Applicability of the method was confirmed on a marketed formulation of azithromycin and on human plasma by solid-phase extraction. The results defined themselves to be within the acceptance criteria. Also percentage plasma protein binding of azithromycin and its major impurities were calculated. It can be concluded that the work presented a clear picture for the binding affinities of azithromycin and its major impurities. The risk of repeated dose of azithromycin may increase the level of the impurities in human body and could be genotoxic. Also the method can be applicable for the analysis of related substances of azithromycin and can be used for routine analysis in human plasma samples, bulk and individual dosage forms.
References
Ripa S, Ferrante L, Prenna M (1996) A linear model for the pharmacokinetics of azithromycin in healthy volunteers. Chemotherapy 42:402–409
The United States Pharmacopoeia 36-NF 31 (2013) Azithromycin Tablets, United States Pharmacopeia Convention, Rockville, pp 2580–2582
Dunn CJ, Barradell LB (1996) Azithromycin. A review of its pharmacological properties and use as 3-day therapy in respiratory tract infections. Drugs 51:483–505
Wildfeuer A, Laufen H, Zimmermann T (1994) Distribution of orally administered azithromycin in various blood compartments. Int J Clin Pharm Ther 32:356–360
Tsukamoto T, Nagae N, Silver S (2013) Usability of a core shell column using high performance liquid chromatograph for a routine analysis
Nagae N, Tsukamoto T, Grahn A, Jutvik R, Gaitonde VD (2011) A novel bonding technique using a polyfunctional silyl-reagent for reversed-phase liquid chromatography—a NEW APPROCH
The United States Pharmacopoeia 34-NF 29 (2011) Chapter < 621 > –chromatography. United States Pharmacopeia Convention, Rockville
Neue UD, McCabe D, Ramesh V, Pappa H, DeMuth J (2009) Transfer of HPLC procedures to suitable columns of reduced dimensions and particle sizes, The United States pharmacopoeia. Pharmacop Forum 35:1622
European Pharmacopoeia 7.0 (2010) Chromatographic Separation Techniques 2.2.46: 4412
British Pharmacopoeia (2009) Adjustment to chromatographic conditions, pp 9
Pharmacopoeia Indian (2007) Liquid Chromatogr 2.4.14:128
http://www.prochrome.net/Method%20Development%20Guide/gradient.htmL
Nagae N, Enami T, Doshi S (2002) The retention behavior of reversed-phase HPLC columns with 100% aqueous mobile phase. LC/GC 20:964–972
Enami T, Nagae N (2004) Wetting property and capillarity of stationary phase for reversed phase liquid chromatography. Bunseki Kagaku 53:1309–1313
The United States Pharmacopoeia 34-NF 29 monographs (2011) United States pharmacopeia convention. Rockville 1998–2003:2006–2008
ICH Guidelines (2005) Validation of analytical procedures: text and methodology Q2(R1). In: International conference on harmonisation of technical requirements for registration of pharmaceuticals for human use, pp 1–13
Babu VVS, Sudhakar V, Murthy TEGK (2014) J Chromatogr Sep Tech 5:209
Verma S, Verma RK, Sahoo D, Srivastava SK (2015) Reverse-phase HPLC method for the quantification of two antihyperglycemic glycolipids in Oplismenus burmannii. Biomed Chromatogr 1:1. https://doi.org/10.1002/bmc.3478
Sahoo DR, Jain S (2016) A rapid and validated RP-HPLC method for the simultaneous quantification of benzoic acid, metronidazole and miconazole nitrate in vaginal formulations. J Chromatogr Sci 54:1613–1618
Gritti F, Guiochon G (2011) Theoretical investigation of diffusion along columns packed with fully and superficially porous particles. J Chromatogr A 1218:3476–3488
Hayes R, Ahmed A, Edge T, Zhang H (2014) Core–shell particles: preparation, fundamentals and applications in high performance liquid chromatography. J Chromatogr A 1357:36–52
Trott O, Olson A (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comp Chem 31:455–461
Acknowledgements
The authors are highly grateful to ImpScience Pvt. Ltd, Hyderabad, Telangana, India, for providing the necessary facilities during the research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sahoo, D.R., Sahoo, S. Development and Validation of a Rapid Solid-Phase Extraction: Ultrafast Liquid Chromatographic Method for the Estimation of Azithromycin and Its Major Related Substances in Human Plasma and Dosage Forms Using a Novel Polyfunctional Silyl Reagent-Bonded Core–Shell Column. Chromatographia 82, 1489–1500 (2019). https://doi.org/10.1007/s10337-019-03768-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-019-03768-z