Abstract
The most significant single event in the study of alien bird invasions occurred in 1981, with the publication of John L. Long’s seminal book “ Introduced birds of the world” (full title: Introduced birds of the world: The worldwide history, distribution and influence of birds introduced to new environments”; David & Charles Ltd., Newton Abbot, UK). The significance of this book derives not just from its content, but also from its timing, coincident with the 1982 Scientific Committee on Problems of the Environment (SCOPE) programme on the ecology of biological invasions. It was not long before studies started to appear that exploited the data in Long’s book to address the questions posed by SCOPE regarding alien invasions. As a result, we arguably have a more complete understanding of the invasion process for birds than for any other taxon. Nevertheless, there are still some key issues in the study of bird invasions where understanding is not all it should be. The aim of this review is to highlight four of these issues by arguing that (1) we do not know half so much about bird invasions as we think; (2) propagule pressure promotes invasions; (3) colonization pressure matters; (4) there is no evidence that escape from parasites promotes alien (bird) invasions. We expect some of the views expressed to be controversial, and others less so, but either way we hope this paper will stimulate others to provide better evidence for—or against—our propositions.
Zusammenfassung
Der wichtigste Moment in der Erforschung der Vogelinvasionen war das Erscheinen John L. Long’s Buches „Eingeführte Vögel der Welt“ 1981 [Titel in Englisch: Introduced birds of the world: The worldwide history, distribution and influence of birds introduced to new environments”; David & Charles Ltd., Newton Abbot, UK]. Die Bedeutsamkeit dieses Buches ist nicht nur dem Inhalt geschuldet, sondern auch dem Zeitpunkt. 1982 begann der wissenschaftliche Beirat der Umweltprobleme [Englisch: Scientific Committee on Problems of the Environment (SCOPE)] mit seinem Programm über die Ökologie biologischer Invasionen. Kurz danach erschienen die ersten Studien, die Long‘s Daten nutzten um einige der Fragen zu biologischen Invasionen aus dem SCOPE Programm zu beantworten. Auch resultierend daraus haben wir heute unbestreitbar ein weitaus tieferes Verständnis der Invasionsprozesse von Vögeln als für irgendeine andere Artengruppe. Trotz allem gibt es immer noch Lücken im Verständnis von Vogelinvasionen. Das Ziel dieser Übersichtsarbeit ist es vier dieser Lücken zu adressieren, indem wir argumentieren, dass (1) nur halb so viel über Vogelinvasionen wissen, als wir denken; (2) Propagulendruck Invasionen unterstützt; (3) Kolonisierungsdruck eine Rolle spielt; (4) es keine Beweise gibt, dass die Abwesenheit von Parasiten(Vogel-)Invasionen unterstützt. Wir hoffen, dass unsere mehr oder weniger kontroversen Ansichten andere Forscher anregen weitere Beweise für oder gegen unsere Behauptungen zu liefern.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The most significant single event in the study of alien bird invasions to date occurred in 1981, with the publication of John Long’s (1981) seminal book “Introduced birds of the world”. The book is subtitled “The worldwide history, distribution and influence of birds introduced to new environments”, but it is the second of these three components that really sets this book apart. The bulk of Long’s book consists of an astonishing catalogue of occasions where bird species have been deliberately or accidentally liberated into areas beyond the limits of their native geographic ranges by human activities. The introductions (sensu Blackburn et al. 2011a) of more than 400 bird species are recorded in this catalogue, of which more than 200 species were considered to have established self-sustaining alien populations. Long (1981) mapped the native and alien distributions of these species and provided a wealth of supplementary information on the circumstances of the introductions. The information provided by Long (1981) was about as ideal a dataset for the study of human-mediated biological invasions as it would have been possible to conceive of at the time.
The significance of Long’s book (1981) derives not just from its content, but also from its timing. While many people identify the publication of Charles Elton’s seminal monograph “The ecology of invasions by animals and plants” (1958) as the founding point of the modern study of human-mediated biological invasions, in reality a greater influence was probably a programme on this topic initiated in 1982 by the Scientific Committee on Problems of the Environment (SCOPE; an arm of the International Council of Scientific Unions). The request to SCOPE to fund a programme on the ecology of biological invasions grew out of observations by some of the world’s most influential ecologists on the impacts that alien species could have on natural ecosystems (Simberloff 2011). The aim of the SCOPE programme was to answer three main questions (Williamson et al. 1986; Drake et al. 1989): (1) What factors determine whether a species becomes an invader or not? (2) What site properties determine whether an ecological system will resist or be prone to invasions? (3) How should management systems be developed to best advantage given the knowledge gained from studying questions 1 and 2? The SCOPE programme led to a large number of national initiatives, spawned several influential edited volumes and introduced many senior and young scientists to the field of invasion biology (Simberloff 2011). It therefore prompted scientists to think about questions for which a large dataset on alien species introductions would have been particularly useful, at exactly the moment when just such a dataset (Long 1981) was published.
It was not long before studies started to appear that exploited the data in Long’s (1981) book to answer questions about alien invasions. The first of which we are aware was by Moulton and Pimm (1983), who analysed data reported in Long (1981) and elsewhere to test for evidence of interspecific competition. These authors found that per species extinction rates increased as more alien species were introduced to islands in the Hawaiian archipelago, which was consistent with an influence of interspecific competition on the probability of species persistence. Long (1981) has therefore provided the basis for a wide range of studies on facets of human-mediated bird invasions, including on the influence of competition (Moulton 1993), numbers of birds introduced (Cassey et al. 2004), life history traits (Cassey 2002; Sol et al. 2005) and location of introduction (Blackburn and Duncan 2001) on establishment success, on the types of species introduced (Lockwood 1999) and the reasons for introduction (Blackburn et al. 2009a). The data in Long’s book (1981) have been particularly effective in advancing our understanding of biological invasions since it was realized that the invasion process was best studied as a sequential series of stages, with success at each stage filtering the identities and characteristics of species entering the subsequent stage (e.g. Williamson 1996; Richardson et al. 2000; Blackburn et al. 2011a).
Arguably, we have a more complete understanding of the invasion process for birds than for any other taxon (Blackburn et al. 2009b), and much of the credit for this must go to John Long (Long himself was always surprised by what people made of his book; Mawson 2003). Nevertheless, more than 30 years on from the publication of Long’s influential tome, there are still some key issues in the study of bird invasions where understanding is not all it should be. Our aim in this review is to highlight four of these issues. Two of the issues we discuss are issues that we believe we do know a lot about, but which we do not think are widely enough appreciated, or about which debate continues to rumble on in the literature. The other two issues are issues that we believe we do not know as much about as we would like to think. We expect some of the views we express to be controversial, and we hope this will stimulate others to provide better evidence either for or against our positions. We would like to think that some of the uncertainties will be put to rest by our comments below, but we are not holding our collective breath.
We do not know half so much about bird invasions as we think
Long (1981) catalogued 426 bird species recorded as having been introduced somewhere in the world as a result of human activities. Many of these were deliberate introductions carried out in the nineteenth and early twentieth centuries by Acclimatization Societies—organizations specifically formed to promote the establishment of alien species (McDowall 1994)—and concerned species introduced for hunting or ornamental purposes. As a result, there is a preponderance of gamebirds (order Galliformes: 64 species), wildfowl (Anseriformes: 27 species) and pigeons (Columbiformes: 31 species) on Long’s list, relative to the species richness of these groups (Blackburn and Duncan 2001), and a preponderance of species in these taxa also amongst successful introductions (Lockwood 1999). Long’s list also documents relatively large numbers of parrots (Psittaciformes: 51 species) and sparrows and their allies (Passeridae: 56 species) (Lockwood 1999; Blackburn and Duncan 2001). The species in these last two families were primarily moved beyond their natural range limits by the cage-bird trade—for example, 20 % of all traded birds are parrots (FAO 2011)—and the large numbers of them catalogued by Long (1981) reflects the fact that cages are permeable barriers to incursions by alien species.
As noted above, Long’s (1981) catalogue has been a hugely influential source of information for studying the invasion process, yet it is now more than 30 years old. In the absence of an updated list, it would be easy to fall into the trap of thinking that bird introductions have become a thing of the past. The realignment of social (or at least scientific and political) attitudes against alien introductions has effectively killed the Acclimatization Societies, with the last of these dying out in New Zealand in the 1980s. More recently, fears about the cage-bird trade as a route by which human pathogenic strains of influenza might be imported into Europe have led to bans on this trade into European Union countries (Cooney and Jepson 2006). Both of these developments will undoubtedly have led to fewer species being moved beyond their natural range limits, and the global trade in wild birds declined from an estimated 7.5 million birds a year in 1975 to around 1.5 million in 2007 (Leader-Williams and Tibanyenda 1996; http://www.cites.org/eng/news/pr/2007/070111_EU_bird_ban.shtml). Unfortunately, new evidence suggests that these developments have not stopped incursions by alien bird species.
Dyer and Blackburn (unpublished data) have brought Long’s (1981) list up to date by compiling a spatially and temporally explicit database on the distributions of exotic birds, called the global avian invasions atlas (GAVIA). As of March 2014, the GAVIA database comprised 27,741 distribution records for exotic bird species. It is based on almost 700 published references and substantial unpublished information derived from consultation with more than 600 organizations and experts worldwide. This database is, as far as we are aware, the most comprehensive resource on the global distribution of exotic species in any major taxon and allows the spatial and temporal dynamics of exotic bird population spread to be examined. GAVIA includes records of introduction for 973 bird species, of which 420 have established apparently viable populations (Dyer and Blackburn, unpublished data). Thus, GAVIA more than doubles the number of known introduced bird species, relative to the information reported by Long (1981), and increases the number of established species known by a similar percentage (Long lists just over 200 species as definitely or probably established).
GAVIA records the dates of first introduction for species, when available, which allows us to explore temporal trends in introductions. The first known introduction dates for species listed in Long (1981) span the range 500–1993 A.D. (some dates are after the publication of Long (1981) because the first known introduction date for a species in Long is for a population introduced after Long was published), with a median date of 1898 (N = 395). For species in GAVIA but not in Long (1981), the dates span the range 1750–2010, with a median of 1979 (N = 347). A graph of bird introductions versus time for species with known first introduction dates shows that the rate at which species are first introduced is not slowing down (Fig. 1), even though the pool of species available for first introduction is decreasing.
A graph of bird introductions over time for species with known first introduction dates. Dates are grouped into 50-year intervals starting from 1511 A.D. The few dated introductions before 1510 A.D. are excluded. Black portions of the bars represent species catalogued in Long (1981), grey portions represent species not catalogued in Long (1981). The lines show the best-fit piecewise regression lines, with the split point (lowest mean square error) at 1860 coinciding with the appearance of Acclimatization Societies (see text)
It should not really come as a surprise that bird introductions are an ongoing, and indeed increasing, phenomenon. First of all, there is abundant evidence from a variety of other taxa and spatial scales that the rate of invasions is not slowing down. For example, marine species invasions into San Francisco Bay classically show an accelerating rate of increase in the period 1850–1990 (Cohen and Carlton 1998). The number of new mammal species introduced to Europe shows exponential growth since 1500 A.D., as does the number of new species per year (Genovesi et al. 2009). The rate at which alien terrestrial invertebrate species establish in Europe also continues to grow, and in the period 2000–2007 the rate was double that of 1950–1974 (Roques et al. 2009). In fact, the number of new bird species introduced into Europe has grown relatively modestly in this context (see Fig. 2, with an exponential model fitting no better than a linear one). Worldwide, however, the temporal change in bird species introductions is well modelled by an exponential increase (R 2 = 0.98, F 3,6 = 190.3, P < 0.001; Fig. 1). However, it is better modelled by a piecewise regression with a break point at 1860 (R 2 = 0.996, F 4,5 = 560, P < 0.001): the rate at which bird species introductions occur takes off rapidly at almost exactly the same time that the first Acclimatization Societies were founded (McDowall 1994), and it never looks back.
The rate at which alien bird species have been introduced to Europe (number of species introduced per decade). From data in Kark et al. (2009)
Second, the data catalogued by Long (1981) present a very Eurocentric view of the process of bird introductions. It is undoubtedly true that much of the history of bird introductions, in terms of quantity if not length, has been a tale of European endeavours deliberately to naturalize species (Crosby 1993), but the focus has shifted dramatically in recent years. Nowadays, most bird introductions are accidental, primarily the result of escaping pets (Lever 2005). The data in Long (1981) already identify this shift in composition (Blackburn et al. 2009a). Further, the most important correlate of variation in the number of invasive alien species across countries is the volume of merchandise imports (Westphal et al. 2008), suggesting that international trade is now the primary driver of species introductions. This is likely to be a result of higher colonization pressure (Lockwood et al. 2009; see also below). To understand modern patterns of bird introductions, we should be looking to regions with cultures of bird-keeping and high levels of international (and within biogeographically diverse countries, national) trade. In these regards, East and South-East Asia stand out.
Bird-keeping is deeply rooted in East Asian culture and includes such activities as bird-walking and bird competitions (e.g. comparing birds on condition or song quality; Jepson and Ladle 2005). Eastern religions also practice prayer animal release (e.g. Environment and Animal Society of Taiwan 2009), and adherents buy and liberate enormous numbers of animals, including birds, for the purpose of accruing karma. These activities support an extensive bird trade, moving thousands of birds of hundreds of species beyond their natural range limits. A good example in these respects is provided by Taiwan. In a recent survey of a sample of Taiwanese bird shops, Su et al. (2014; Su et al. unpublished data) recorded more than 26,000 individual birds of more than 240 species for sale (see also Shieh et al. 2006). The total market value of these birds was over US$ 0.75 million. Over two-thirds of the species for sale were alien, and there is clear potential for the deliberate and accidental release of such birds to contribute to avian invasions. Indeed, at least 90 alien bird species have gone wild in Taiwan, and breeding records are available there for about 35 alien species (Shieh et al. 2006; Su et al., unpublished data). Nine of these species with breeding records are not listed in Long (1981) at all. Alien bird species have already proven to be a conservation threat in Taiwan through actual and potential hybridization with native bird species (Severinghaus 2007; Yao 2007; Li et al. 2010), and so the ongoing trade in cage birds is likely to be of significant conservation concern. This will be true not only in Taiwan, but also in other countries with active and developing markets in wild birds, such as China (Li and Jiang 2014) and Brazil (Regueira and Bernard 2012).
Overall, it is worrying that while scientists and policy-makers know well the dangers posed by alien species (see Roy et al. 2014), the range of bird species (and species in most other taxa) with recorded introductions continues to grow, with no sign of the rate slowing down (Fig. 1). It is also worrying that most introductions nowadays stem from the pet bird trade: this trade is likely to grow further worldwide because keeping birds is related to prosperity in developing regions (e.g. Jepson and Ladle 2005). It would be easy to imagine that bird introductions are no longer a problem because the cultures responsible for the “golden age” of acclimatization have abandoned this practice. Unfortunately, nothing could be further from the truth. The fact that these ongoing invasions provide new opportunities and questions for scientists studying the invasion process might be viewed as scant consolation for the problems that they are likely to cause.
Propagule pressure promotes invasions
The data in Long (1981) and GAVIA (Dyer and Blackburn, unpublished data) identify a large number of established alien bird populations, but also show that most introductions do not result in establishment. Understanding why some introductions succeed while others fail was a primary motivation for the first two questions in the SCOPE programme on the ecology of biological invasions. However, it turns out that a key element of the answer relates neither to the characteristics of the species (question 1) nor to characteristics of the location (question 2).
A basic fact of population biology is that populations comprising fewer individuals are more likely to go extinct in any given period of time than populations with more individuals; in other words, extinction probability is negatively related to population size. The reasons for this are well known: small populations are more likely to suffer from effects of demographic and genetic stochasticity, more likely to be extirpated by environmental stochasticity and more likely to suffer from the Allee effect. Small population size is a major concern for conservation biologists, and the lower the number of individuals of a species, the higher the category of threat on the International Union for the Conservation of Nature (IUCN) Red List to which it is assigned (IUCN 2014). It makes sense to assume that alien species released in lower numbers would also be more likely to go extinct, and hence be less likely to establish a persistent population at the location of introduction. Numerous empirical analyses, including many on the data on introductions of alien birds compiled in Long (1981) and elsewhere (e.g. Thomson 1922), are consistent with this expectation (e.g. Dawson 1984; Cassey et al. 2004; Lockwood et al. 2005; Sol et al. 2005, 2012; Colautti et al. 2006; Hayes and Barry 2008; Blackburn et al. 2009a, 2011b, 2013; Simberloff 2009; Duncan et al. 2014).
Given that a positive relationship between establishment success and number of animals released (we term this the “propagule pressure effect”) is expected on theoretical grounds, is observed in data and has been verified in experimental releases (albeit in taxa other than birds), one would not imagine that it could be controversial. Indeed, arguably it is not! Yet, there has recently been an attempt to make it so by a small group of collaborators working on historical bird introductions who argue that propagule pressure does not drive establishment success (Moulton et al. 2011, 2012a, b, 2013).
Moulton and colleagues have used several lines of argument to promote this view, mostly based around the data on intentional bird introductions to New Zealand. First, they attempted to undermine the propagule pressure effect (Moulton et al. 2010, 2012a), arguing that historical data have a number of uncertainties and errors that render the conclusions of previous analyses of them insecure—“all that can be garnered from the historical record is a list of the species that were introduced, and some indication of when the individuals were released” (Moulton and Cropper 2014). This statement is incorrect, as recent detailed examination of the historical data demonstrates (Pipek et al. 2015), but even were it not, it would be interesting that the uncertainties and errors all apparently favour the propagule pressure effect. It is also interesting that Moulton and colleagues are happy enough to use these data, showing that the smallest releases for species successfully introduced to New Zealand were smaller than the largest releases for failed introductions, which they argue shows that release size does not determine success. However, this conclusion assumes that success is a deterministic function of release size (it clearly is not because some failed introductions are larger than those for some successes in many, if not most, species), and Moulton et al.’s analysis (2010, 2012a) cannot recover a propagule pressure effect in simulation models where the effect is explicitly specified (Blackburn et al. 2011b). A robust re-analysis of Moulton et al.’s data on release sizes showed that the propagule pressure effect is indeed supported (Blackburn et al. 2011b).
Second, Moulton et al. argued that the propagule pressure effect arises because establishment success encourages people to release more animals, rather than because releasing more animals increases the likelihood of establishment. Blackburn et al. (2013) countered this by identifying six predictions of the hypothesis that establishment success drives propagule pressure and demonstrating that all six predictions could be falsified. For example, if success is unrelated to the number of birds released, and successful populations had larger releases because their initial success led to more releases, then there should be no relationship between establishment success and propagule pressure, controlling for the number of releases. Yet, there is. Moulton and Cropper’s (2014) response, that this could be because both numbers introduced and number of introductions could be higher if undertaken by organizations with more resources, is no more than special pleading.
Moulton and Cropper (2014) subsequently shifted their position on the propagule pressure effect, arguing that “there is a somewhat increased probability of failure…for very small numbers introduced”, but that in many cases, more individual birds were introduced than needed to guarantee establishment. That claim may be true, but is impossible to prove, and beside the point. For a population to have established, at least one individual within that population must have descendants surviving at a location after some period of time (Caswell 2001; Fox 2005). The probability that at least one individual leaves a surviving lineage depends on the birth and death rates of individuals in the population, and hence on features of the species and environment concerned, but inevitably increases with the number of individuals initially present (Leung et al. 2004; Jerde and Lewis 2007; Duncan et al. 2014). It is hard to conceive of realistic situations where the introduction of more birds would lead to lower per capita establishment probabilities, as would be necessary for success not to be a positive function of propagule pressure, at least for the release sizes we tend to see in birds (typically in the range 10–500, median of 50 for deliberate releases; Blackburn et al. 2009a).
Ultimately, therefore, logic dictates that propagule pressure must enhance the likelihood of establishment success and that success is indeed consistently higher for bird populations where larger numbers have been introduced, within species, within countries, across regions and globally (Blackburn et al. 2009a, 2011b). Moulton and Cropper (2014) argue that “That tendency explains little of the historical pattern of establishment outcomes for passeriform birds”. Propagule pressure does indeed explain only a fraction of the variance in establishment success, and characteristics of the species and environment clearly do matter (Duncan et al. 2014), but propagule pressure is nevertheless the most robust and consistent predictor of establishment success that we know (Lockwood et al. 2005).
Colonization pressure matters
While propagule pressure is the most consistent predictor of alien bird establishment success, other factors must also play a role. Duncan et al. (2003) distinguished between three broad categories of determinants of success: location-, species-, and event-level factors (the last category being effects that can differ between 2 introductions of a given species to a given location, such as propagule pressure). It makes sense that whether a population succeeds or fails will depend on the characteristics of the species concerned and on the location to which it is introduced. Indeed, these effects were the primary scientific focus of the SCOPE programme (questions 1 and 2) and resulted in substantial literature on these issues (Simberloff 2011).
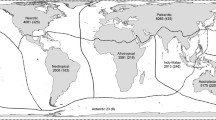
Focusing on location-level effects, Shea and Chesson (2002) distinguished three broad features of the area of introduction that are likely to influence establishment, hypothesizing that fewer enemies, more resources and benign environmental conditions should all promote success. These predictions make sense, and are easy to state, but in practice there are so many axes along which their effects might act that studies rarely address them directly. Instead, most studies of location-level effects consider surrogate variables, such as the latitude at which a location lies, whether it is on an island or continental mainland or whether it represents “pristine” or disturbed environments (Blackburn et al. 2009a). Spatial structure in the distribution of the bird species listed in Long (1981) identifies rich assemblages of aliens at mid-latitudes, as well as on archipelagoes such as the Hawaiian Islands, New Zealand and the Greater Antilles (Fig. 3). These patterns, together with formal analyses of alien species richness, have been used to conclude that areas with simpler biotic communities (i.e. islands, high latitudes) are easier to invade than species-rich areas (i.e. the continental tropics), as long as those simpler communities are not in environmentally harsh (i.e. polar or desert) regions.
Unfortunately, however, there is an analytical problem that has often been overlooked by studies of location-level effects on establishment success, and which may invalidate the conclusions drawn from them. The number of alien species established at a location, E is necessarily some fraction of the total number of species introduced there (or close to there), I such that
where S is the establishment probability (Lonsdale 1999). Lockwood et al. (2009) term I “colonization pressure”. Thus, the hotspots of alien species richness in Fig. 3 may be hotspots (high E) because they are easy to invade (high S) or because they are not easier (or even harder) to invade, but have higher colonization pressure (high I). For example, the Hawaiian Island of Maui has E = 27 alien bird species established, whereas Mauritius has E = 19. It would be easy to conclude that Maui is easier for birds to invade than Mauritius. However, the colonization pressure for Maui was I = 47 species, while that for Mauritius was I = 33 species. Thus, S is more or less identical for these two islands (0.57 vs. 0.58, respectively); Maui only appears to be easier to invade because of its higher colonization pressure (Lockwood et al. 2009).
The concept of colonization pressure makes life difficult for anyone wishing to understand the causes of spatial or temporal variation in establishment success or as the determinants of alien species richness. E and I in Eq. 1 are not independent because E is a subset of I and, hence, is always constrained to be <I. The null hypothesis for the relationship between E and I is therefore not zero, but a positive relationship (see Brett 2004). This means that one cannot analyse causes of variation in E directly, but must instead model the number of established species out of the total number of introduced species as a binomial response variable (Lonsdale 1999; Lockwood et al. 2009). This is straightforward enough for some questions, such as causes of variation in establishment success in different acclimatization regions (Blackburn et al. 2011b; see above), or in temporal variation in establishment success at a location (Lockwood et al. 2009). However, it creates substantial problems for attempts to answer other questions.
For example, consider tests of hypotheses for the spatial variation in alien species richness in Fig. 3. Any such test would need to account for colonization pressure. However, most locations where invasive species occur are not locations where the species were introduced, because the species have subsequently spread. Richness variation across regions may be attributable to colonization pressure (i.e. Maui vs. Mauritius), whereas richness variation within regions may or may not (e.g. E can and does vary across Maui, but I = 47 for the whole island). However, models are likely to need to address the causes of richness at both levels.
Failing to account for colonization pressure has led to some erroneous conclusions in the body of literature on invasion. For example, Brown (1989) showed a negative relationship between the number of native and number of alien bird species in four regions (Hawaii, New Zealand, Australia and North America) as evidence that historically isolated areas with low native species diversity are relatively easily invaded. However, this is not a valid conclusion in the absence of data on colonization pressure. Sol (2000) subsequently showed that the percentage success rates for bird introductions did not differ between Australia and New Zealand, although they did differ between Hawaii and North America. Sol (2000) nevertheless further showed that the latter difference could be attributed to the characteristics of the different species introduced at the different locations: there was no location-level difference in the success rate for those species introduced to both Hawaii and North America. The notion that simpler communities, and especially oceanic islands, are easier to invade is a seductive one, but robust evidence in support of such statements that also accounts for colonization pressure is surprisingly elusive.
There is no evidence that escape from parasites promotes alien (bird) invasions
While colonization pressure undoubtedly needs to be factored into any consideration of location-level effects on establishment success, there is still evidence that success varies across locations (Duncan et al. 2014). One or all of Shea and Chesson’s (2002) broad categories of determinants of success (fewer enemies, more resources or benign environmental conditions) are therefore likely to be the cause of the variation in establishment success.
There is abundant evidence that biotic interactions regulate natural populations of animals and plants, and so it comes as little surprise that these processes are frequently invoked to explain the success or otherwise of human-mediated biological invasions (e.g. Elton 1958). One mechanism by which this success might be mediated is now generally termed the “enemy release hypothesis” (ERH) (Keane and Crawley 2002). This hypothesis proposes that alien species escape the negative impacts on reproduction and survival they experience from their natural enemies, including predators/herbivores and parasites/pathogens, left behind in their native geographic ranges. In consequence, they experience lower levels of enemy impact than native species in their new ranges. If the reduction in enemy regulation translates into increased population growth, then alien species may be able to increase rapidly in numbers, escaping the stochastic effects that afflict small populations (Allendorf et al. 2012) and, thereby, increasing the probability of establishment and invasive spread.
The last few years have seen an upsurge of interest in the role of parasites (including pathogens) in the context of enemy release, following the publication of an influential paper by Torchin et al. (2003; cited more than 760 times by December 2014 according to Google Scholar). Torchin et al. (2003) showed that parasite species richness and prevalence were generally lower in the alien than the native range for a variety of introduced species, including molluscs, crustaceans, fishes, amphibians, reptiles, mammals and birds. For example, 44 parasite species have been recorded on the European starling Sturnus vulgaris in its native range, but only nine have been recorded on this bird species in its North American alien range. Torchin et al. (2003) do not explicitly conclude that the observed escape from parasites causes invasion success, but they do note the implications of their findings for potential application in the biocontrol of invasive species.
A range of studies has assessed evidence of release from parasites in biological invasions (reviewed by Colautti et al. 2004; Mitchell et al. 2006), with outcomes that depend on the type of study undertaken (Colautti et al. 2004). Biogeographical studies compare rates of parasitism (richness and/or prevalence) in native and introduced populations of the same species (e.g. Torchin et al. 2003) and tend to support the ERH. Thus, introduced plant populations are exposed to fewer species of pathogens, are less impacted by pathogens and herbivores and are less negatively impacted by soil organisms than are native conspecific populations (Callaway et al. 2004; Mitchell et al. 2006; Inderjit and van der Putten 2010). In contrast, community studies compare rates of parasitism for native and introduced populations of different species within the same community and are much more equivocal in their support for the ERH (Colautti et al. 2004).
Differences in the general outcomes of biogeographical versus community studies has resulted in the validity of these tests being questioned in the literature. On the one hand, Colautti et al. (2004) argued that biogeographical tests are flawed because they compare the parasite richness of introduced populations with that of entire native species, rather than with the specific source population(s) for the introduction. However, this bias should not affect some measures of prevalence, or experimental tests of pathogen impact (Inderjit and van der Putten 2010). In contrast, Mitchell et al. (2006) argued that many within-community comparisons of plants, at least, are poor tests of the ERH because they rely on estimates of visible damage from natural enemies, rather than on measures of performance or demographics. They also tend to compare congeneric species of native and aliens and will thus be conservative tests of ERH if invaders with close relatives in the native community are more likely to acquire new enemies from them. Whether biogeographical or community studies best represent the true situation with respect to both escape from parasites and the ERH therefore remains an open question.
There is, however, a much more fundamental problem with evidence from both biogeographical and community studies for the ERH in terms of escape from parasitism: none of it actually provides information on the causes of invasion success. A species must pass through a succession of stages—transport, introduction, establishment and spread—in order to negotiate the pathway from native to alien invasive, and it may fail to become an invader if it falls at barriers imposed at any of these stages (Blackburn et al. 2011a). The ERH is likely to operate primarily at the establishment and spread stages. In terms of establishment, increased population growth arising from a reduction in enemy regulation may increase the likelihood that an introduction can escape the perils of small population size (and so establish). In terms of spread, release from enemies may increase the likelihood that an established population can produce enough colonists to enable dispersal and establishment at further new locations. For the ERH to influence establishment, however, it is necessary for populations that successfully establish to have benefitted more from release from the impacts of parasites than those that fail to establish. As most tests are framed in terms of parasite species richness, this would equate to successful species having fewer parasites (or to have escaped more) than those that fail to establish. Similarly, if the ERH influences the ability of an established population to spread, we would expect a positive relationship between the extent of spread and the degree of release from parasitism. However, neither biogeographical nor community studies provide information on these comparisons: they say nothing about the level of parasitism in those populations that failed to establish relative to those that succeeded, nor do they tell us whether level of parasitism influences tendency to, or extent of, spread.
To be informative about the influence of the ERH on invasion success, therefore, studies need to compare the extent of escape from parasitism in populations that are introduced and become established versus those that are introduced but do not, or in populations that establish and spread to varying extent. [In terms of the framework for comparisons in invasion studies proposed by van Kleunen et al. (2010), this is a comparison of type B, rather than A (community studies) or F1 (biogeographic studies).] We are aware of only two studies that have explored the ERH in this fashion for parasites (Mitchell and Power 2003; van Kleunen and Fischer 2009), and in alien plant species rather than birds. Mitchell and Power (2003) showed that, among plant species listed as natural area invaders, species that experienced more complete pathogen release were more widely invasive. Here, however, invasive refers to the number of states and non-governmental organizations that list the species as an invasive problem in natural areas; consequently, it is not a direct measure of the extent of spread. Moreover, the relationship was only found when non-noxious and non-invasive alien species were excluded from the data set, and even then the analysis is not robust to the inclusion of an outlying data point. Van Kleunen and Fischer (2009) showed that the geographic spread of alien plants introduced from North America to Europe was negatively associated with their release from fungal pathogens, in direct opposition to the ERH. Additionally, Klironomos (2002) showed that interactions between plants and soil microbes were more likely to be positive for invasive alien species than for rare native plant species, and that the strength and sign of such interactions were positively related to abundance for plants in an old field meadow community in Canada. Callaway et al. (2004) showed that plant–soil interactions were more likely to be positive in the alien range than the native range of the plant Centaurea maculosa. Taken together, these studies suggest that the extent of plant invasions may be related to the degree of escape from negative interactions with soil biota, but they stop short of demonstrating such a relationship. To date, therefore, there is arguably no study that can be considered to support the ERH in terms of the influence of escape from parasitism on invasion success, not only for birds, but for any taxon.
References
Allendorf FW, Luikart GH, Aitken SN (2012) Conservation and the genetics of populations. Wiley, New York
Blackburn TM, Duncan RP (2001) Establishment patterns of exotic birds are constrained by non-random patterns in introduction. J Biogeogr 28:927–939
Blackburn TM, Lockwood JL, Cassey P (2009a) Avian invasions. The ecology and evolution of exotic birds. Oxford University Press, Oxford
Blackburn TM, Lockwood JL, Cassey P (2009b) Following birds along the pathway to invasion. The Biologist 56:80–85
Blackburn TM, Prowse TAA, Lockwood JL, Cassey P (2011a) Passerine introductions to New Zealand support a positive effect of propagule pressure on establishment success. Biodivers Conserv 20:2189–2199
Blackburn TM, Pyšek P, Bacher S, Carlton JT, Duncan RP, Jarošík V, Wilson JRU, Richardson DM (2011b) A proposed unified framework for biological invasions. Trends Ecol Evol 26:333–339
Blackburn TM, Prowse TAA, Lockwood JL, Cassey P (2013) Propagule pressure as a driver of establishment success in deliberately introduced exotic species: fact or artefact? Biol Invasions 15:1459–1469
Brett MT (2004) When is correlation between non-independent variables “spurious”? Oikos 105:647–656
Brown JH (1989) Patterns, modes and extents of invasions by vertebrates. In: Drake JA, di Castri F, Groves RH, Kruger FJ, Rejmánek M, Williamson M (eds) Biological invasions, a global perspective. Wiley, Chichester, pp 85–109
Callaway RM, Thelen GC, Rodriguez A, Holben WE (2004) Soil biota and exotic plant invasion. Nature 427:731–733
Cassey P (2002) Life history and ecology influences establishment success of introduced land birds. Biol J Linn Soc 76:465–480
Cassey P, Blackburn TM, Sol D, Duncan RP, Lockwood JL (2004) Introduction effort and establishment success in birds. Proc R Soc Lond B [Suppl] 271:S405–S408
Cassey P, Blackburn TM, Duncan RP, Lockwood JL (2005) Lessons from the establishment of exotic species: a meta-analytical case study using birds. J Anim Ecol 74:250–258
Caswell H (2001) Matrix population models. Sinauer Assoc, Sunderland
Cohen AN, Carlton JT (1998) Accelerating invasion rate in a highly invaded estuary. Science 279:555–558
Colautti RI, Ricciardi A, Grigorovich IA, MacIsaac HJ (2004) Is invasion success explained by the enemy release hypothesis? Ecol Lett 7:721–733
Colautti RI, Grigorovich IA, MacIsaac HJ (2006) Propagule pressure: a null model for biological invasions. Biol Invasions 8:1023–1037
Cooney R, Jepson P (2006) The international wild bird trade: what’s wrong with blanket bans? Oryx 40:18–23
Crosby AW (1993) Ecological imperialism: the biological expansion of Europe, 900–1900. Cambridge University Press, Cambridge
Dawson JC (1984) A statistical analysis of species characteristics affecting the success of bird introductions. BSc Thesis, University of York, York
Drake JA, di Castri F, Groves RH, Kruger FJ, Rejmánek M, Williamson M (eds) (1989) Biological invasions, a global perspective. Wiley, Chichester
Duncan RP, Blackburn TM, Sol D (2003) The ecology of bird introductions. Annu Rev Ecol Evol Syst 34:71–98
Duncan RP, Blackburn TM, Rossinelli S, Bacher S (2014) Quantifying invasion risk: the relationship between establishment probability and founding population size. Methods Ecol Evol 5:1255–1263
Elton C (1958) The ecology of invasions by animals and plants. Methuen, London
Environment and Animal Society of Taiwan (EAST) (2009) The investigation of prayer animal releases in Taiwan. Environment and Animal Society of Taiwan, Taiwan. Available at http://www.east.org.tw/that_content.php?s_id=14&m_id=0&id=89
Food and Agricultural Organization (FAO) (2011) International trade in wild birds, and related bird movements, in Latin America and the Caribbean. Animal Production and Health Paper No. 166, FAO, Rome
Fox GA (2005) Extinction risk of heterogeneous populations. Ecology 86:1191–1198
Genovesi P, Bacher S, Kobelt M, Pascal M, Scalera R (2009) Alien mammals of Europe. In: DAISIE (ed) Handbook of alien species in Europe. Springer, Dordrecht, pp 119–128
Hayes KR, Barry SC (2008) Are there any consistent predictors of invasion success? Biol Invasions 10:483–506
Inderjit, van der Putten WH (2010) Impacts of soil microbial communities on exotic plant invasions. Trends Ecol Evol 25:512–519
International Union for the Conservation of Nature (IUCN) (2014) IUCN Red list of threatened species. Available at http://www.iucnredlist.org
Jepson P, Ladle RJ (2005) Bird-keeping in Indonesia: conservation impacts and the potential for substitution-based conservation responses. Oryx 39:1–6
Jerde CL, Lewis MA (2007) Waiting for invasions: a framework for the arrival of nonindigenous species. Am Nat 170:1–9
Kark S, Solarz W, Chiron F, Clergeau P, Shirley S (2009) Alien birds, amphibians and reptiles of Europe. In: DAISIE (ed) Handbook of alien species in Europe. Springer, Dordrecht, pp 105–118
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17:164–170
Klironomos JN (2002) Feedback with soil biota contributes to plant rarity and invasiveness in communities. Nature 417:67–70
Leader-Williams N, Tibanyenda RK (eds) (1996) The live bird trade in Tanzania. International Union for the Conservation of Nature, Gland
Leung B, Drake JM, Lodge DM (2004) Predicting invasions: propagule pressure and the gravity of Allee effects. Ecology 85:1651–1660
Lever C (2005) Naturalised birds of the world. T & AD Poyser, London
Li L, Jiang Z (2014) International trade of CITES listed bird species in China. PLoS ONE 9:1–8
Li S-H, Yeung CK-L, Han L, Le MH, Wang C et al (2010) Genetic introgression between an introduced babbler, the Chinese hwamei Leucodioptron c. canorum, and the endemic Taiwan hwamei L. taewanus: a multiple marker systems analysis. J Avian Biol 41:64–73
Lockwood JL (1999) Using taxonomy to predict success among introduced avifauna: relative importance of transport and establishment. Conserv Biol 13:560–567
Lockwood JL, Cassey P, Blackburn T (2005) The role of propagule pressure in explaining species invasions. Trends Ecol Evol 20:223–228
Lockwood JL, Cassey P, Blackburn TM (2009) The more you introduce the more you get: the role of colonization pressure and propagule pressure in invasion ecology. Divers Distrib 15:904–910
Long JL (1981) Introduced birds of the world. The worldwide history, distribution and influence of birds introduced to new environments. David & Charles, London
Lonsdale WM (1999) Global patterns of plant invasions and the concept of invisibility. Ecology 80:1522–1536
Mawson J (2003) Prologue to the introduction. In: Long JL (ed) Introduced mammals of the world. CSIRO Publishing, Collingwood
McDowall RM (1994) On size and growth in freshwater fish. Ecol Freshw Fish 3:67–79
Mitchell CE, Power AG (2003) Release of invasive plants from fungal and viral pathogens. Nature 421:625–627
Mitchell CE, Agrawal AA, Bever JD, Gilbert GS, Hufbauer RA, Klironomos JN, Maron JL, Morris WF, Parker IM, Power AG et al (2006) Biotic interactions and plant invasions. Ecol Lett 9:726–740
Moulton MP (1993) The all-or-none pattern in introduced Hawaiian passeriforms: the role of competition sustained. Am Nat 141:105–119
Moulton MP, Cropper WP Jr (2014) Establishment success in introduced passeriforms of New Zealand: evidence for a Franklin Delano Roosevelt effect. Biol Invasions 16:233–237
Moulton MP, Pimm SL (1983) The introduced Hawaiian avifauna: biogeographic evidence for competition. Am Nat 121:669–690
Moulton MP, Cropper WP Jr, Avery ML, Moulton LE (2010) The earliest house sparrow introductions to North America. Biol Invasions 12:2955–2958
Moulton MP, Cropper WP Jr, Avery ML (2011) A reassessment of the role of propagule pressure in influencing fates of passerine introductions to New Zealand. Biodivers Conserv 20:607–623
Moulton M, Cropper WP Jr, Moulton LE, Avery ML, Peacock D (2012a) A reassessment of historical records of avian introductions to Australia: no case for propagule pressure. Biodivers Conserv 21:155–174
Moulton MP, Cropper WP Jr, Avery ML (2012b) Historical records of passerine introductions to New Zealand fail to support the propagule pressure hypothesis. Biodivers Conserv 21:297–307
Moulton MP, Cropper WP Jr, Avery ML (2013) Is propagule size the critical factor in predicting introduction outcomes in passeriform birds? Biol Invasions 15:1449–1458
Pipek P, Pyšek P, Blackburn TM (2014) How the Yellowhammer became a Kiwi: the history of an alien invasion revealed. NeoBiota 24:1–31
Regueira RFS, Bernard E (2012) Wildlife sinks: quantifying the impact of illegal bird trade in street markets in Brazil. Biol Conserv 149:16–22
Richardson DM, Pyšek P, Rejmanek M, Barbour MG, Panetta FD, West CJ (2000) Naturalization and invasion of alien plants: concepts and definitions. Divers Distrib 6:93–107
Roques A, Rabitsch W, Rasplus J-Y, Lopez-Vaamonde C, Nentwig W, Kenis M (2009) Alien terrestrial invertebrates of Europe. In: DAISIE (ed) Handbook of alien species in Europe. Springer, Dordrecht, pp 63–80
Roy HE, Peyton J, Aldridge DC, Bantock T, Blackburn TM, Bishop J, Britton R, Clark P, Cook E, Dehnen-Schmutz K, Dines T, Dobson M, Edwards F, Harrower C, Harvey MC, Minchin D, Noble DG, Parrott D, Pocock MJO, Preston CD, Roy S, Salisbury A, Schönrogge K, Sewell J, Shaw RH, Stebbing P, Stewart AJA, Walker KJ (2014) Horizon-scanning for invasive alien species with the potential to threaten biodiversity in Great Britain. Glob Change Biol 20:3859–3871
Severinghaus LL (2007) The breeding ecology and dispersal of Taiwan and light-vented bulbuls in Kenting National Park. J Natl Park 17:27–41
Shea K, Chesson P (2002) Community ecology as a framework for biological invasions. Trends Ecol Evol 17:170–176
Shieh B-S, Lin Y-H, Lee T-W, Chang C-C, Cheng K-T (2006) Pet trade as sources of introduced bird species in Taiwan. Taiwania 51:81–86
Simberloff D (2009) The role of propagule pressure in biological invasions. Annu Rev Ecol Evol Syst 40:81–102
Simberloff D (2011) Charles Elton: neither founder nor siren, but prophet. In: Richardson DM (ed) Fifty years of invasion ecology. The legacy of Charles Elton. Wiley, Oxford, pp 11–24
Sol D (2000) Are islands more susceptible to be invaded than continents? Birds say no. Ecography 23:687–692
Sol D, Duncan RP, Blackburn TM, Cassey P, Lefebvre L (2005) Big brains, enhanced cognition, and response of birds to novel environments. Proc Natl Acad Sci USA 102:5460–5465
Sol D, Maspons J, Vall-llosera M, Bartomeus I, García-Peña GE, Piñol J, Freckleton RP (2012) Unraveling the life history of successful invaders. Science 337:580–583
Su S, Cassey P, Blackburn TM (2014) Patterns of non-randomness in the composition and characteristics of the Taiwanese bird trade. Biol Invasions 16. doi:10.1007/s10530-014-0686-1
Thomson GM (1922) The naturalisation of plants and animals in New Zealand. Cambridge University Press, Cambridge
Torchin ME, Lafferty KD, Dobson AP, McKenzie VJ, Kuris AM (2003) Introduced species and their missing parasites. Nature 421:628–630
van Kleunen M, Fischer M (2009) Release from foliar and floral fungal pathogen species does not explain the geographic spread of naturalized North American plants in Europe. J Ecol 97:385–392
van Kleunen M, Dawson W, Schlaepfer D, Jeschke JM, Fischer M (2010) Are invaders different? A conceptual framework of comparative approaches for assessing determinants of invasiveness. Ecol Lett 13:947–958
Westphal MI, Browne M, MacKinnon K, Noble I (2008) The link between international trade and the global distribution of invasive alien species. Biol Invasions 10:391–398
Williamson M (1996) Biological invasions. Chapman and Hall, London
Williamson MH, Kornberg H, Holdgate MW, Gray AJ, Conway GR (1986) Preface: the British contribution to the SCOPE program on the ecology of biological invasions. Philos Trans R Soc Lond B 314:1–2
Yao C-T (2007) The elimination and caged raise of exotic Red-billed blue magpie Urocissa erythrorhyncha in Wu-ling National Forest Recreation Area. Endemic Species Research Institute, Council of Agriculture, Executive Yuan, Taiwan/Wu-ling National Forest Recreation Area, Taiwan
Acknowledgements
We thank the organizing committee of the 26th International Ornithological Congress for the opportunity to present our work, and two anonymous referees for their helpful comments on an earlier version of the paper. This study was supported by the King Saud University Distinguished Scientist Fellowship Program (TMB), and by an ARC Discovery Grant (DP140102319) and Future Fellowship (FT0991420) to PC.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Matthysen.
Rights and permissions
About this article
Cite this article
Blackburn, T.M., Dyer, E., Su, S. et al. Long after the event, or four things we (should) know about bird invasions. J Ornithol 156 (Suppl 1), 15–25 (2015). https://doi.org/10.1007/s10336-015-1155-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-015-1155-z