Abstract
To inform regional conservation planning, we assessed mammalian and avian biodiversity in the Djéké Triangle, which is an intact forest with long-term research and tourism focused on western lowland gorillas (Gorilla gorilla gorilla). This critical region serves as a conservation conduit between the Nouabalé-Ndoki National Park (NNNP) in the Republic of Congo and the Dzanga-Ndoki National Park in Central African Republic. Wildlife inventories were conducted to determine if biodiversity in the Djéké Triangle (initially part of a logging concession) was equivalent to the NNNP. Camera traps (CTs) were deployed to estimate species richness, relative abundance, naïve occupancy, and activity patterns of medium-to-large species in mixed species and monodominant Gilbertiodendron forests that comprise the majority of regional terra firma. Species inventories were collected from CTs positioned on a grid and at termite nests throughout the Djéké Triangle and compared to CTs placed in the Goualougo Triangle located within the NNNP. From 10,534 camera days at 65 locations, we identified 34 mammal and 16 bird species. Allaying concerns of wildlife depletion, metrics of species richness in the Djéké Triangle surpassed those of the Goualougo Triangle. Many species were observed to occur across habitats, while others showed habitat specificity, with termite mounds indicated as an important microhabitat feature. Our comparisons of animal activity budgets in different habitat types provide important reference information for other populations and contexts. In conclusion, this study provided empirical evidence of the high conservation value of this region that contributed to increasing the protected status of the Djéké Triangle.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The unfolding crisis of tropical forest clearance and resource extraction has led to drastic declines in biodiversity (Gibson et al. 2011; Newbold et al. 2013; Phillips et al. 2017; Leclere et al. 2020; WWF/LZS 2020). As a result, safeguarding species-rich hotspots and a diverse range of tropical forest habitats that comprise these ecosystems remains an urgent priority (Watson et al. 2018). One mechanism that is often relied upon to conserv biodiversity and associated landscapes is the identification and use of charismatic megafauna as “flagship” species to raise awareness and facilitate protection efforts of broader biodiversity (Simberloff 1998; Western 1987; Leader-Williams and Dublin 2000). Given the high degree of spatial overlap of primates with a variety of taxa facing similar risks to survival it has been suggested that protection measures targeting the wellbeing of primates can also be of benefit to co-occurring species (Macdonald et al. 2013). Other studies have claimed, however, promoting research and protection of one species over others poses risks, including falling short on conservation expectations (Sun et al. 2022). In this study, we show how the conservation impact of long-term study sites focused on great apes was extended to facilitate biodiversity assessments and land preservation.
Primates, including great apes, have long played primary roles as ambassadors for conservation including land preservation initiatives (Mittermeier 1986, 1988; Wrangham et al. 2008). Three decades ago, this strategy was adopted by a team of local and international primatologists conducting research and surveys to lobby for the eventual creation of the Nouabalé-Ndoki National Park (NNNP) in northern Republic of Congo. These scientists envisioned gorillas and chimpanzees as the centerpieces of the protected area conservation strategy, and so supported the initiation of several long-term research sites with an emphasis on particular species. These included the Mbeli Bai Project in the NNNP, the Goualougo Triangle Ape Project in the forests south of the NNNP, and the Mondika Gorilla Project in the Djéké Triangle west of the NNNP. While the initial focus of these projects was to increase knowledge about gorillas and chimpanzees, long-term aspirations included studying broader primate community and ecosystem dynamics. These research sites are also uniquely positioned to conduct biodiversity monitoring more broadly for mammalian and avian taxa, which not only provides essential ecological information to contextualize studies of primates but also contributes to larger scale conservation efforts. Biodiversity assessments and subsequent monitoring are both time consuming and resource intensive, but essential in providing empirical means to assess the efficacy of conservation policies and practices. In this case, we show how such information contributed to elevating the protected status for the Djéké Triangle, a strategic and environmentally important forest enclave with the potential for long-term sustainable sources of funding for the NNNP.
In addition to surveying biodiversity, gaining a better understanding of habitat use is also crucial to successful wildlife management. Improved understanding on primate ecologies and activity patterns shaping resource partitioning and realized niches could also inform proposed linkages between guilds (Bourlière 1985), which is of particular relevance with regard to the semi- terrestrial primates found across the Ndoki landscape. Parity between habitat type and floral heterogeneity can also be a diversifying force in habitat use. This is particularly relevant in the Sanhga Tri National (TNS) landscape, where two main terra firma habitat types are widespread. These include Mixed Species Forest (MSF) that is characterized by high floral diversity (Harris 2002) and Gilbertiodendron dewevrei forest (GDF), a monodominant habitat with a single tree species comprising 75–88% of the basal area (Hart 1990). In contrast to MSF, GDF thrives in predominantly low-lying moist and infertile soil environments and constitutes an estimated 20% of the closed canopy terra firma habitat in the region (Hall et al. 2019). Assessments of GDF imply a low productivity habitat with scarce resources for wildlife (Hart 2001). Apart from periods of mast seed production that coincide with dramatic increases in the presence of primates and other wildlife in GDF (Blake and Fay 1997), the contribution of these monodominant forests to species co-existence warrants further investigation. Based on the lower abundance of GDF and distribution along waterways, we predict that larger and more terrestrial taxa will be more frequetly associated with both MSF and GDF than smaller or more arboreal taxa who tend to have smaller ranges.
While often overshadowed by the differences in habitat, the microhabitat features within these forest types are known to influence forest dynamics and harbor important resources for some species. For example, epigeal mounds constructed by termites have profound impacts on the composition, structure, and chemical makeup within the immediate location of the earthen structures (McComie and Dhanarajan 1993). The secondary consequences of alterations of the soil extend to vegetation heterogeneity and forest structure in the vicinity of the mounds. In several African savanna ecosystems, termite mounds were shown to positively impact floral diversity and the establishment of particular plant types on varying spatial scales (Crooks 2002; Boogert et al. 2006; Traore et al. 2008; Moe et al. 2009). In addition, soil macro-invertebrate abundances may also be elevated (Choosai, et al. 2009) altering the availability of resources and ultimately the distribution of mammalian and avian taxa that forage on these invertebrates. This could be particularly interesting in northern Congo where chimpanzees frequent termite nests, but the importance of this resource to other species is unknown.
While transect methodology has proven effective in monitoring large forest vertebrates such as elephants and great apes (Morgan et al. 2006; Stokes et al. 2010; Maisels et al. 2013), it tends to under-represent the presence of other forest dwelling species such as small- to medium- sized fauna (Bowkett et al. 2006; van Vliet and Nasi 2008; Fragoso et al. 2016). The NNNP, which is within the Sangha Tri National Landscape (TNS) landscape, is estimated to harbor more than 50 species of mammals (including 12 primates) and 429 species of birds (Maisels 2001). Most of these species are challenging to study during landscape-scale assessments, so it is no surprise that knowledge gaps still exist in fundamental aspects of their distribution, habitat use, and activity patterns. Fortunately, the widespread adoption of remote observations via camera traps (CTs) has addressed some of these challenges and expanded the taxonomic scope of species monitoring in cost-effective ways (Nichols et al. 2011; Howe et al. 2017; Agha et al. 2018). Systematic survey data from CTs yield traditional biodiversity monitoring metrics (e.g., species richness, relative abundance, and occupancy estimates: O'Brien et al. 2010; Fonteyn 2022) while also creating opportunities to document behavior relevant to accurately assessing the abundance of elusive species (Bessone et al. 2020).
Accurate and precise biodiversity monitoring is an essential aspect of conservation management, particularly in areas impacted by humans. The southern section of the Djéké Triangle is aligned with the hunting zone of nearby villages. Extending the research focus from flagship species to a broader range of taxa can provide indicators of ecosystem health and information for land use planning. This is increasingly relevant as overexploitation is recognized as a widespread driver of primate abundance and overall vertebrate diversity (Bennett et al. 2007; Fa and Brown 2009; Nasi et al. 2012; Abernethy et al. 2013; Cawthorn and Hoffman 2015; Ripple et al. 2017; Benítez-López et al. 2019). In Africa, roughly 35.5% of the biodiversity loss documented from 1970 to 2016 on the continent was attributed to overharvesting (WWF/LZS 2020). The impacts of hunting offtake are not spatially homogeneous across this region, with some remote, less-disturbed forests maintaining intact species assemblages while less distant and heavily exploited forests show shifts in species presence (Dupain et al. 2012; Fa et al. 2015; Ziegler et al. 2016). For some game species, such as duiker, overexploitation is argued to lead to predictable changes in the faunal community (Yasuoka et al. 2015). There are concerning indications that overharvesting of small, medium, and large duiker is already underway within the Kabo concession (Mockrin 2008; Brncic et al. 2018). Further, preliminary evidence from the Kabo concession (west sector including the Djéké Triangle) has also indicated extremely low encounter rates of guenons and mangabeys (Brncic et al. 2018). Poulsen et al. (2009) surmised the high diversity of species documented in bushmeat surveys recorded in the Kabo forestry management unit and neighboring concessions potentially signaled a tipping point towards depletion of the wildlife. If measures are not taken to stem the unsustainable rates of defaunation, it is projected that many species will vanish by 2050 (Fa et al. 2003) and overall biodiversity will spiral downward (Schipper et al. 2020).
Similar forecasts of decline have been made for pristine or 'Intact Forest Landscapes' (IFL) across Africa (Potapov et al. 2017). Such loss has far-reaching consequences for not only human livelihoods and food security, but floral diversity, regeneration, and structure of forests (Vanthomme et al. 2010; Effiom et al. 2013; Hempson et al. 2015). While habitat degradation and loss constitute the largest threats to biodiversity (WWF/LZS 2020), the Congo Basin still contains significant stands of forest with high environmental structural integrity (Hansen et al. 2020). Tracts of intact forest estate outside of protected areas in the Republic of Congo, including the Djéké Triangle, have previously been identified as targets for enhanced conservation planning. However, up to date information is needed to validate indices of intactness, condition, human pressure, and biodiversity that are used to identify such areas (Grantham et al. 2020).
Due to the interest in elevating the conservation status of the Djéké Triangle, we expanded the historic focus of research on great apes to conduct a broader assessment of biodiversity with a particular emphasis on game species and those at highest risk of extinction. More specifically, we compared measures of species richness, relative abundance indices (RAI), and naïve occupancy (NO) from camera trapping in the Djéké Triangle with surveys conducted in the Goualougo Triangle (located inside the boundaries of NNNP). Since both of these forests are intact, we expected to find similar species assemblages. However, there has been more human pressure on the wildlife of the Djéké Triangle which could result in decreased representation of game species. We also expected to find lower species diversity in monodominant Gilbertiodendron forest than Mixed Species Forest. Within these habitats, we compared observation of CTs placed in grid locations to those at termite nests. We anticipated that epigeal termite mounds would attract a wider variety of birds and mammals that consume insects than would otherwise be observed on CTs. Further, we examined activity patterns of particular species (e.g., duiker species) to fill knowledge gaps that could facilitate future surveys and identify meaningful changes over time. The findings of these biodiversity assessments were used to lobby for elevating the protection status of the Djéké Triangle by including it in the NNNP.
Methods
Study site location
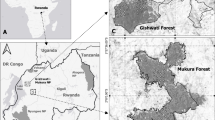
This inventory was conducted within the forests adjacent to and within the NNNP, an integral partner in the TNS. Camera trap surveys were conducted at two sites: the Djéké Triangle and the Goualougo Triangle (Fig. 1). The Djéké Triangle (100 km2) is an unlogged “conservation set-aside” forest in the Kabo Forest Management Unit (FMU) (2°15′–2°24′ N; 16°16′–16°21′E). Conservation set-aside status does not allow by law for industrial extraction such as timber exploitation. However, this designation is not by presidential decree meaning this land use status and protection can be downgraded. The southern border of the Djéké Triangle aligned with an adjacent village hunting zone that is part of a community development area. The Djéké Triangle is also home to the Mondika Gorilla Project (MDK) that has been ongoing since the mid-1990s investigating western lowland gorillas (Gorilla gorilla gorilla). At a distance of 30 km from the Djéké Triangle, the Goualougo Triangle (310 km2) lies within the NNNP. A comparable biodiversity inventory was conducted in the study area of the Goualougo Triangle Ape Project (GTAP), which is the site of long-term applied conservation research on the behavioral ecology of sympatric central chimpanzees (Pan troglodytes troglodytes) and western lowland gorillas.
The Sangha Trinational Conservation Complex includes national parks in Cameroon, the Central African Republic, and the Republic of Congo. The inset map shows camera placement in the study areas including the Djéké Triangle within the Kabo forestry concession and hunting zone. Zone B1 of the Goualougo Triangle is located within the Nouabalé-Ndoki National Park. Mixed and G. dewevrei forests types are also highlighted
The two most widespread terra firma habitat types were recognized in this study and based on species compositions described by Harris (2002): MSF and Monodominant GDF. MSF has a diverse flora and a canopy that is not always continuous. Families typical of this habitat type include Meliaceae, Leguminoseae, Irvingiaceae, Sterculiaceae, and Ebenaceae. It is semi-deciduous and occurs on terra firma areas. GDF is a single-species formation of G. dewevrei that features a continuous herbaceous understory from sparse to dense patches. It occurs along watercourses as well as on interfluvial plateaus.
Camera trapping surveys
Camera trap data were collected using Bushnell Trophy Cam units in the Djéké from 2017 to 2020, and at Goualougo from 2019 through 2020. Video recordings were collected using camera traps placed at a specific grid location (hereafter referred to as “grid cameras”) or at termite mounds (hereafter referred to as “termite cameras”). Comparable recording settings were used when deploying the Browning Dark Ops, BTC-6PXD units and Bushnell Trophy Cam HD units in the field. In total 19 grid cameras stationed for 3057 days and 15 termite mound-based units for 4332 days were employed in the Djéké Triangle. At Goualougo, a total of 29 camera units recorded data for 3145 days.
Termite cameras
In both the Djéké and Goualougo Triangle study areas, we placed CTs at above-ground epigeal mounds of Macrotermes muelleri or subterranean termite mounds constructed by Macrotermes lilljeborgi. CTs were oriented towards the base of a termite mound typically associated with a wildlife trail. Macrotermes are known to be sources of protein for chimpanzees and other wildlife in this region.
Grid cameras
Camera units were systematically placed at the midpoint of 1 km × 1 km spatial grids in the Djéké Triangle. To facilitate standardization of data collection with efforts underway across the region, we adopted the Pan African Programme: The Cultured Chimpanzee (PanAf) biodiversity monitoring protocols in placing grid cameras along animal trails and near forest features known to attract wildlife such as particular species of tree and sources water that support particular aquatic vegetation (http://panafrican.eva.mpg.de/). Cameras were placed 45 cm above the ground and oriented towards animal trails in an effort to survey medium- to large-sized mammals and birds. There is a recognized bias in species sampling when using ground-based camera trap surveys against more arboreal taxa but this height placement was selected to be comparable to large-scale monitoring programs in the region.
Species identification
All videos recorded on CTs were retrieved monthly and screened for species identification. We used the Kingdon Field Guide to African Mammals (Kingdon 2015), the Birds of the World database (Billerman et al. 2022), and the IUCN Red List of Threatened Species (IUCN 2022) to classify species and obtain their conservation status. In cases where individuals were not identifiable to the species level, effort was made to label them at the level of the taxonomic group. For example, individuals of the taxonomic group Viverridae, where species level identification was not possible, were identified as “genet”. When neither species nor taxonomic group identification were possible, the event was labeled “unidentified”. In cases where species identification was questionable, experts were consulted. An experienced observer conducted training on species identification. Considering the challenges of identifying wildlife from camera trap footage, we compiled a Visual Reference Guide for Identifying Bird and Mammals in Camera Traps (Supplementary Material S1) and Reference Videos for Identifying Bird and Mammals in Camera Traps (Supplemental Material S2) which highlight various species in different views and lighting. As multiple observers were involved in data extraction from camera trap footage, we conducted tests of interobserver reliability to ensure accuracy in species identification (Zett et al. 2022). Interobserver reliability was conducted using 100 video clips that contained a diverse and representative group of taxa, with > 80% agreement being the threshold for data collection. Techniques to reliably identify particularly rare species were discussed during training sessions and research meetings for this project.
Statistical analysis
To facilitate comparison of species richness across sites and habitat types, we constructed species-area curves (sensu Gotelli and Colwell 2001; Hedwig et al. 2018) using the specaccum function in the R package ‘vegan’ version 2.5–7 (Oksanen et al. 2015). We constructed species richness curves for each site and habitat type using random permutations of recordings (method random with 100 permutations in specaccum) as recommended by Gotelli and Colwell (Gotelli and Colwell 2001). Species richness was calculated for each site and camera placement option. Only observations that were resolved to the species level were used for construction of the species accumulation curves.
We calculated the relative abundance index (RAI) and naïve occupancy (NO) using the same methods as Hedwig and colleagues (Hedwig et al. 2018). The RAI was calculated as the number of camera events per 100 camera-trapping days (Rovero et al. 2014). Resampling of the same individual at the same location within a particular time frame was avoided (e.g., Tobler et al. 2008; Mugerwa et al. 2013), and following the procedure of Hedwig et al. (Hedwig et al. 2018), we considered all recordings of a species within 5 min at the same location to be the same animal and therefore the same recording. We calculated naïve occupancy as the proportion of camera trap locations where each species was found. Relative abundance index and naïve occupancy were calculated for all data resolved to each taxonomic level (class, order, family, genus, and species), by camera (grid or termite) and habitat type (MSF or GDF). We used Wilcoxon signed rank tests to test for differences between habitat types (sensu Hedwig et al. 2018).
We calculated a modified duiker index following Yasuoka et al. (2015) for camera trap locations to assess the percentage of duikers from a region outside the NNNP with a faunal community subject to hunting. For each camera unit, a detection rate was obtained and corresponds to the mean number of independent events over a 24-h period. The duiker index is referred to as the duiker percentage following Marrocoli et al. (2019), as this measure uses the percentage rather than ratio of duiker. For each camera unit and associated detection rate and number of independent events, we calculated a duiker index. In this study, we applied this to blue duiker Philantomba monticola and four red duiker species including Cephalophus callipygus, Cephalophus dorsalis, Cephalophus leucogaster and Cephalophus nigrifrons.
Finally, we used the hours of detection from camera trap triggers in order to determine activity patterns for select animals (sensu Barlow et al. 2016; Cappelle et al. 2021). Using the R package ‘activity’, we converted the recording times to radian time-of-day data, and then fit that data with a Von Mises kernel density distribution using the function ‘fitact’ in activity, which fits a smoothed circular distribution and confidence limits to the (now circular) radian time-of-day data in order to estimate the proportion of the time during a full 24-h camera-day where an animal was active (Rowcliffe et al. 2014). This analysis was performed for species with adequate observations to fit the curves, for both duikers (Cephalophus callipygus, Cephalophus dorsalis, Cephalophus leucogaster, Cephalophus silvicultor and Philantomba monticola) and threatened mammals (Gorilla gorilla, Pan troglodytes, Loxodonta cyclotis and Phataginus tricuspis). All analyses were done in R version 4.2.1 (R Core Team 2022).
Results
From December 2016 to March 2020, a total of 10,534 camera trap days at 65 locations were accumulated over the two sites (Table 1). The grid camera inventory at Djéké registered 3057 camera trap days while the termite camera sampling accumulated 4332 days of effort. Termite camera sampling effort at Goualougo was comparable, with 3145 camera trap days registered. Sampling effort across habitat types is reported in Table 2. A total of 16 species representing 16 genera of birds and 34 species representing 28 genera of mammals were recorded across the two sites (see Table 3 for complete listing of birds and Table 4 for complete listing mammals). Ungulates were the most frequently detected mammalian group, with blue duiker (Philantomba monticola) and Peters’s duiker (Cephalophus callipygus) being the most commonly documented species. Rodents were the second most documented group, with African brush-tailed porcupine (Atherurus africanus) being the most recorded species on CTs. CTs positioned at termite mounds detected 34 species in the Goualougo Triangle and 41 species in the Djéké Triangle, particularly species that consume insects including the endangered chimpanzee. In the Djéké forests, termite mound-based cameras recorded a similar number of species as the grid camera method in neighboring forests.
Impact of site, survey method and habitat on species richness
Overall species richness was slightly higher in the Djéké Triangle than Goualougo (Fig. 2). Cameras stationed at termite mounds in the Goualougo Triangle recorded marginally lower species richness than those stationed in the forests of Djéké in slightly fewer camera trap days, though differences fell within 95% confidence intervals (Fig. 2).
There were no discernible differences in species richness between MSF and neighboring GDF in the Djéké and Goualougo Triangles.
Relative abundance index and naïve occupancy
At both Djéké and Goualougo sites, we obtained observations for 50 species, from which we calculated RAIs and naïve occupancies (See Table 3 for birds and Table 4 for mammals). These included species identified as important to either sustainable hunting or conservation: Philantomba monticola, Cephalophus callipygus, Atherurus africanus, Loxodonta cyclotis and Gorilla gorilla gorilla. Of the avian fauna, there were six species of Passeriformes, comprising the largest representation among the birds.
Naïve occupancy estimates obtained at grid and termite cameras in the Djéké and Goualougo sites for threatened species such as forest elephants, great apes, African golden cats, and pangolins varied depending on species and camera placement option (Fig. 3). Grid-based cameras at Djéké recorded the highest occupancy estimates for elephants, African golden cats, and both species of great ape, whereas camera traps stationed at Djéké termite mounds recorded elevated levels of both species of pangolin. Several species of Cetartiodactyla also registered high naïve occupancy levels, including Philantomba monticola, Cephalophus callipygus, Cephalophus dorsalis, Cephalophus leucogaster, Cephalophus silvicultor and Potamochoerus porcus.
Comparing RAIs and naïve occupancy across forest types
We did not detect differences in relative abundance between forest types in either the Djéké Triangle (Wilcoxon test; V = 617, p value = 0.5783) or Goualougo Triangle (Wilcoxon test; V = 191, p = 0.0698). However, differences in RAIs were detected for specific species between the two forest types surveyed. Across both Djéké and Goualougo, higher RAI values for many species were associated with MSF than neighboring GDF. Cephalophus callipygus and Philantomba monticola were found to have the highest RAI values of all species in GDF as well as MSF. The African brush-tailed porcupine (Atherurus africanus), which is commonly hunted in the region, was detected more often in MSF in Djéké. Among threatened species, the grey-cheeked mangabey (Lophocebus albigena) was only recorded in MSF and the central chimpanzee (Pan troglodytes) was detected more frequently in MSF. Habitat selectivity for MSF was also documented in the avian fauna, as the spot-breasted ibis (Bostrychia rara) and the African wood owl (Strix woodfordii) were only recorded in MSF in the Djéké Triangle as well.
Naïve occupancy of many species in Djéké and Goualougo indicated overall similar widespread presence across locations in MSF and neighboring GDF habitats (Tables 3 and 4). However, several threatened species were found more frequently, or entirely, in one forest type or the other (Fig. 4).
Naïve occupancy for selected species (Left to right. Top row: gorilla, chimpanzee, grey-cheeked mangabey. Bottom row: African brush-tailed porcupine, spot-breasted ibis, and African wood owl) by study site (Djéké (DT) or Goualougo (GT) and forest type (Mixed Species Forest (MSF) or Monodominant Gilbertiodendron Forest (GDF)
Duiker percentages and activity patterns by forest type
As indicated by the above RAI measures, there were far more frequent recordings of blue duikers than red duikers in the un-hunted and unlogged Djéké Triangle. Calculated duiker percentages for each of the 19 camera traps stationed in the Djéké forests revealed similar values (Table 5). Duiker percentages were also comparable between MSF and GDF.
As expected, activity patterns varied across duiker species (Fig. 5). Philantomba monticola and Cephalophus callipygus appear to be largely diurnal, with peaks in activity around sunrise and sunset. Cephalophus dorsalis and Cephalophus silvicultor were nocturnal and showed only periodic activity during the day, while Cephalophus leucogaster was active during both day and night.
Tests comparing Philantomba monticola, Cephalophus callipygus, Cephalophus dorsalis and Cephalophus leucogaster activity patterns between contexts and sites indicated few significant differences between the estimates. For some of the species, this could be a product of sample size. However, species such as Philantomba monticola and Cephalophus dorsalis were among the most commonly observed mammals and the site-specific insight into their activity times makes biological sense. Both species showed similarly active times at grid-based cameras as well as those stationed at termite mounds.
Conservation status and activity patterns of threatened mammals
The IUCN Red List Index is a comprehensive and objective tool to assess the relative risk extinction poses for species (IUCN 2022). Critically Endangered taxa included African forest elephants (Loxodonta cyclotis) and western lowland gorillas (Gorilla gorilla gorilla). Endangered species included central chimpanzees (Pan troglodytes troglodytes), tree pangolins (Phataginus tricuspis) and giant ground pangolins (Smutsia gigantea). Vulnerable species included leopard (Panthera pardus), golden cat (Caracal aurata) and grey-cheeked mangabeys (Lophocebus albigena). We also detected nine other species classified as Near Threatened: two species of otter (Aonyx capensis congicus, Hydrictis maculicollis), forest buffalo (Syncerus caffer nanus), bongo (Tragelaphus eurycerus), yellow-backed duiker (Cephalophus silvicultor), bay duiker (Cephalophus dorsalis), and white-bellied duiker (Cephalophus leucogaster). A notable difference was also documented between the Djéké and Goualougo study sites in numbers of Least Concern species, with Djéké cameras capturing a greater number of species than in Goualougo (Table 6).
Activity patterns for each species indicate patterns of temporal availability for detection by CTs, which are relatively consistent across mixed species and monodominant Gilbertiodendron forests (Fig. 6). Findings also indicate the great apes (Gorilla gorilla and Pan troglodytes) are predominately diurnal, though multiple observations of Pan troglodytes occurred at night. As expected, Loxodonta cyclotis were recorded on camera traps during all hours. While there were insufficient recordings to estimate activity patterns, most recordings for Panthera pardus, as well as both species of pangolin (Phataginus tricuspis and Smutsia gigantea) were recorded during the night. Captures of Caracal aurata occurred both during the day and night. Only two captures of Lophocebus albigena were recorded; both occurred during the day.
Discussion
In this study, we compared species richness and relative abundances of medium to large mammals and birds in the Djéké Triangle to an area within the NNNP to inform conservation planning. The representation of species in the Djéké Triangle exceeded that of the Goualougo Triangle and demonstrated the rich biodiversity of this intact forest. Contrary to previous findings that large mammal visitation is largely restricted to the mast-fruiting in GDF, we documented comparable species richness in both GDF and MSF across this region. However, some species showed a stronger preference for one of these habitat types. CTs placed at termite nests complemented grid-based surveys by providing a different perspective on species distribution and activity patterns within the forest ecosystem. Several species that were rarely observed on grid-based cameras frequented termite mounds. Based on the taxonomic diversity and regular visitation, we argue that termite colonies and their nest structures should be considered high conservation attributes. While the flagship species approach was integral in advancing this conservation agenda, the systematic assessments to confirm the broader species richness across mammalian and avian taxonomic groups in the Djéké Triangle was important in lobbying for inclusion of this area into the NNNP. Further, the long-term investment in research and biodiversity monitoring that led to this change in the land-use status has created conditions that support the development of a sustainable gorilla-based tourism program in the Djéké Triangle.
Global initiatives established to monitor trends in populations of species around the planet such as the Living Planet Index (LPI) (Loh et al. 2005) and Biodiversity Intactness Index (BII) (Scholes and Biggs 2005) rely on monitoring data to provide indicators of overall trends in ecosystem and biodiversity health over large spatial scales. Simultaneously, there is a scientific imperative to increase our understanding of the factors shaping species abundances at local levels to inform local decision-making processes and species-specific action plans (e.g., Strier et al. 2021). Our combined approach of implementing CTs to assess biodiversity at long-term flagship sites provides a low-cost method for monitoring elusive and understudied species (Barlow et al. 2016). Over the last decade, the number and variety of CT studies across tropical Africa has substantially increased. Many of these have focused on documenting the behavior of a single species and/or targeted density estimation of particular mammals. For example, the PanAf Programme camera trapped at different study locations across the Congo Basin to document chimpanzee behavioral variation and estimate biodiversity across sites (http://panafrican.eva.mpg.de/). In this study, we show that long-term sites focused on great apes can facilitate broader biodiversity assessments and monitoring. Further, CTs deployed to study the behavior and cognition of chimpanzees produced comparable results to those deployed in a systematic grid and also new insights into the activity of other species (such as pangolins) who regularly visit these locations. Both environmental contexts presented here yielded important information about activity patterns of selected species, supporting previous investigations in the potential utility of drawing upon different camera applications to support larger more informed biodiversity monitoring programs (O'Brien 2008; Tobler et al. 2008; Fonteyn et al. 2021). The elevated species richness documented in the Djéké compared to the Goualougo may be related to higher incidences of rarer species being recorded on grid-based cameras at the site. We did not achieve complete surveys for all mammals or birds at the respective sites nor consider the undetected species in our species richness calculations (see e.g., Kéry and Royle 2008). Our CT survey did detect the presence of primates such as Lophocebus albigena, Cercocebus agilis, Cercopithecus cephus and Cercopithecus neglectus which were not recorded during the most recent transect census of the Kabo concession’s west sector (Brncic et al. 2018). Several rare observations of other species such as golden cat (Caracal aurata), giant pangolin (Smutsia gigantea), and the spot-breasted ibis (Bostrychia rara) were also obtained in this study which prompts further CT efforts to assess their occurrence across the region.
Studies of single species employed as surrogates or indicators for coexisting wildlife have been faulted for largely focusing on general overlap with other species, rather than other facets of community interaction such as commonalities of habitat use (Roberge and Angelstam 2004). Greater diversity of habitat types can elevate opportunities for species and support higher species diversity (e.g., Lindenmayer et al. 2000). However, findings from this study on GDF and MSF showed similar mammal species estimates as surveys of sites featuring a greater variety of habitat and microhabitat types. Similar census results on mammal were tallied in CT studies of mammals in forest-savannah mosaic landscapes of neighboring eastern Gabon including a national park (Hedwig et al. 2018) and a timber production concession (Fonteyn 2022). We detected differences in naïve occupancies of several species between forest types. Lophocebus albigena, and three species of Guenon (Cercopithecus cephus, C. neglectus and C. nictitans) were only detected in MSF. While all these species access multiple forest strata, our results imply that guenons in the Ndoki forests may prefer Mixed Species Forests and low forest strata levels. By contrast and as would be expected from ground stationed CTs, we did not document large-bodied, arboreal folivores like Colobus angolensis, Piliocolobus oustaleti which specialize on new leaves found in the middle to emergent canopies. The transition zones from MSF to monodominant Gilbertiodendron Forests are also often abrupt, and their close spatial proximity may support species capitalizing on resources found within both forest types. Larger bodied animals with larger home ranges would presumably have access to both habitat types. As expected, the larger and semi-terrestrial primates (Gorilla gorilla, Pan troglodytes) were observed in both habitats. Cercocebus agilis is the most terrestrial monkey species in the Ndoki primate community, and also spend time in both GDF and MSF. Their semi-terrestrial nature affords the possibility of expanded ecological niches accessing food, travel routes, shelter, or nesting materials (for apes) on or near the ground layer in both of these forest types. Data could also be interpreted to indicate these species may transition between Gilbertiodendron terra firma and accessing neighboring riparian swamp habitat not surveyed in this investigation but critical habitat for multiple primate species (Gautier-Hion and Brugière 2005). Based on our findings, future survey efforts in the Ndoki region should include sufficient monitoring effort in both GDF and MSF to generate accurate and precise density estimates as well as habitat suitability assessments.
In addition to habitat types, we also found that smaller scale structures such as Macrotermes mounds may be important features to mammalian and avian communities. These earthen mounds may function as “hotspots” for biodiversity (e.g., Choosai, et al. 2009) or potentially meaningful “structure-based indicators” that signal additional biodiversity features or forest health (e.g., (Tews et al. 2004)). Observations from Congo support previous conclusions that these engineered “patches” (Crooks 2002; Boogert et al. 2006; Traore et al. 2008) have the capacity to influence the patterning and abundance of biodiversity. Adding Macrotermes termite mounds to the list of features like trees and animal paths for improved monitoring efficiency of pangolins (e.g., Bruce et al. 2018; Khwaja et al. 2019) should be considered given the likelihood of increasing threat that pangolins face (Mambeya et al. 2018) and the uncertainty of Macrotermes persistence in modified and increasingly open canopy forests. Forest certification and international conservation conventions could also be expanded to consider the crucial role that such particular resources play in the maintenance of cultural processes (sensu Brakes et al. 2021; Carvalho et al. 2022), like the tool-using repertoires of chimpanzees known to be in decline across the African continent (Kuehl et al. 2019).
Few empirical data exist on the distribution, population sizes, demography, and behavior of most of the mammal species in northern Republic of Congo. One species that has received marginal scientific attention is the bongo (Tragelaphus eurycerus), which is the largest antelope in the region, and was detected in the Djéké Triangle at low relative abundance and naïve occupancy indices. These results support previous camera trap surveys indicating the presence of bongo tends to be lower in intact forests than modified forests (Elkan 2003). While bongo survival prospects appear to benefit from some low level of environmental disturbance, recent findings from the surrounding forestry concessions indicate this species is vulnerable to local extinction if other forms of extraction such as international trophy hunting are not intensively managed (Koopmans et al. 2021). Monitoring the impact that hunting poses to game species and the livelihoods and well-being of local communities are of critical conservation concern (Wilkie et al. 2001; van Vliet 2018).
Other species are also exploited in the region as sources of protein and/or revenue (Rowcliffe et al. 2005; Poulsen et al. 2009). In fact, the potential spatial impact of hunting on vertebrate species across the region is predicted to be quite extensive (Ziegler et al. 2016). A west and central African survey of bushmeat by Taylor and colleagues (Taylor et al. 2015) reported at 177 species including primates, rodents and duiker were among the most common taxonomic groups included in the capture and trade of wildlife (Fa et al. 2004; Fa and Brown 2009; Abernethy et al. 2013; Fontyen 2022). High mobility and larger body size are traits that hunters select for in mammals until over-harvesting forces extractors to shift their focus to less desirable targets (Wright 2003; Fa et al. 2015; Benítez-López et al. 2019). In a long-term hunting zone in the Sangha Trinational region, Noss (2000) tallied 18 species that were captured via species-specific hunting techniques, indicating that selectivity was at play. While selectivity of hunted species has been argued to lead to predictable changes in the duiker fauna composition (Yasuoka et al. 2015; Fontyen 2022), the high RAI values obtained for blue duiker and the duiker percentages calculated from the Djéké forests do not indicate duiker offtake by hunting. If hunting was having an impact on the mammal assemblages, we would expect considerably lower detections of artiodactyl and primates based on monitoring along gradients of human disturbance in Cameroonian forests (Yasuoka et al. 2015; Funteyn 2022). This assertion is also based on the lack of anthropogenic disturbance documented by research teams that maintain a year-round presence in the Djéké Triangle and the encounter rates of commonly targeted species. It has also been speculated that disease could contribute to declining duiker populations inside the Nouabalé-Ndoki National Park, but daily surveys have not detected any abnormal or elevated numbers of duiker carcasses which could signify such an event. Instead, our results provide empirical evidence that unlogged and less disturbed forests can maintain relatively high abundances of rapidly reproducing species like blue duiker as well as the medium to large and more slowly reproducing duiker taxa. Similarly, CT studies in other parts of the Congo Basin have found high abundances of blue duiker in relatively less disturbed forests (Houngbégnon et al. 2020), which should serve to caution against generalizations about the composition of duiker and other taxa across different sites and environments with varying levels of anthropogenic impacts.
Technological advancements and accessibility are also increasing the potential for local communities to develop site- and scale-specific management prescriptions around particular species and environmental indicators (Slough et al. 2021a, b). With the advent of more precise methods to estimate animal densities via camera traps (e.g., Howe et al. 2017), it is now possible to repeatedly assess indicator species over shorter time intervals, which is urgently needed if game species are to be managed at sustainable levels in human modified landscapes (e.g., Wilkie et al. 2019). Other technologically enhanced survey techniques such as DNA shed into the environment (eDNA: Bohmann et al. 2014) and monitoring tools such as the Spatial Monitoring and Reporting Tool (SMART) are evolving in their use and functions to facilitate the collection and timely reporting of wildlife inventories across multiple-use zones. Technology and knowledge sharing among scientists, law enforcement teams, conservation practitioners and local community members could bridge a longstanding gap in promoting effective management and monitoring practices in forests outside of national parks. It is at the community level that the most knowledgeable and inspired local actors (Getz et al. 1999; Gardner et al. 2010; Oldekop et al. 2019) can act upon resource management issues in expedient ways (Danielsen et al. 2010). Because biodiversity data are situation-dependent, more informed, and standardized monitoring is needed across what DeFries and colleagues (DeFries et al. 2010) refer to as “a zone of interaction” that includes a continuum of land-use options from highly disturbed to protected.
The modern land-use history of the Djéké Triangle since the inception of the Mondika Gorilla Project demonstrates how investing in a “flagship” species and commitment to the protection of a particular area can evolve to include broader biodiversity research and land use planning. Originally classified for timber production, it was then designated as a conservation set-aside which provided some protection of this intact forest. In 2023, the Djéké Triangle was included in the NNNP and is part of the Sangha Trinational World Heritage site. The southern portion of the Djéké Triangle aligns with the hunting zones of the local villages and remains accessible for hunting and gathering. Further, the IFL provides a variety of vital ecosystem services including watershed retention, climate regulation and natural source for maintaining depleted game species for an expanding village population that relies on locally sourced protein. Continued long-term monitoring of forests in and around the NNNP can inform park management about the potential existence of a “defaunation gradient” in the mammalian and avian communities of the Ndoki region and the broader Sangha Trinational Conservation Complex. It is therefore imperative to detect any such declines as quickly as possible so that concerns can be brought to the forefront of management decisions before irrevocable loss occurs.
Conclusions
The Djéké Triangle has long factored into the agenda of landscape protection in the Ndoki landscape for a variety of reasons, including its strategic location along the international borders of two national parks. We found that species diversity within Djéké is comparable to the remote forests of the Goualougo Triangle, despite increasing accessibility (Morgan et al. 2019) and hunting pressure in the landscape (Maisels et al. 2013). As we have demonstrated, it is also of high conservation value, with rich biodiversity and the presence of multiple integrally protected species residing in a mosaic of habitats. The continued integrity of this faunal assemblage and forest are credit to a steadfast commitment of local communities, government, industry partners, NGOs, and independent scientists to work collaboratively in implementing effective long-term protection and monitoring throughout the region. Regular ground patrols by armed ecoguards are bolstered by the year-round field presence of long-term ape research sites, which act as force multipliers in detecting illegal activities through their daily monitoring in these remote forests. Further, enhanced protection is provided by the Wildlife Crime Unit’s efforts to follow up on investigations of criminality and facilitate associated judicial processes. Amidst the expansion of industrial timber extraction in the early 2000s, Forest Stewardship Certification measures led to this area being set aside as a conservation zone which has fostered conditions for continued research and biodiversity monitoring. This investigation shows that prioritization of flagship species (in this case, gorillas and chimpanzees) did not act as a determinant or fail to include appreciable abundances of other species as some studies have surmised (Caro et. al. 2004; Ozaki et al. 2006; Sun et al. 2022). Conversely, discussions about these great apes have served as platforms for promoting the elevated protected status of this area and prompted expanded biodiversity monitoring. The result of which have included comparative estimates of species richness, identification of shared habitat use among a wide variety of species, new information about the activity patterns of rarely observed species, and empirical evidence of the high conservation value of the Djéké Triangle to support its inclusion into the NNNP. The synthesis of these actions is currently being met by further economic and conservation opportunities aimed at developing international and domestic tourism featuring habituated gorilla groups in the Djéké Triangle while aspiring to benefit local human populations. While challenges to the biodiversity of this region will persist, they can be managed if monitoring and mitigation efforts include social and economic values that continue to extend beyond the Djéké Triangle and into neighboring communities.
Data availability
A Visual Reference Guide for Identifying Birds and Mammals in Camara Traps is provided in the Supplemental Material S1.
References
Abernethy KA, Coad L, Taylor G, Lee ME, Maisels F (2013) Extent and ecological consequences of hunting in Central African rainforests in the twenty-first century. Philos Trans R Soc B 368(1625):20120303.
Agha M, Batter T, Bolas EC, Collins AC, Gomes da Rocha D, Monteza-Moreno CM, Preckler-Quisquater S, Sollmann R (2018) A review of wildlife camera trapping trends across Africa. Afr J Ecol 56(4):694–701
Barlow J, Lennox GD, Ferreira J, Berenguer E, Lees AC, Mac Nally R, Thomson JR, Ferraz SFD, Louzada J, Oliveira VHF, Parry L, Solar RRD, Vieira ICG, Aragao L, Begotti RA, Braga RF, Cardoso TM, de Oliveira RC, Souza CM, Moura NG, Nunes SS, Siqueira JV, Pardini R, Silveira JM, Vaz-de-Mello FZ, Veiga RCS, Venturieri A, Gardner TA (2016) Anthropogenic disturbance in tropical forests can double biodiversity loss from deforestation. Nature 535(7610):144-+
Benítez-López A, Santini L, Schipper AM, Busana M, Huijbregts MAJ (2019) Intact but empty forests? Patterns of hunting-induced mammal defaunation in the tropics. PLoS Biol 17(5):e3000247
Bennett EL, Blencowe E, Brandon K, Brown D, Burn RW, Cowlishaw G, Davies G, Dublin H, Fa JE, Milner-Gulland EJ, Robinson JG, Rowcliffe JM, Underwood FM, Wilkie DS (2007) Hunting for consensus: Reconciling bushmeat harvest, conservation, and development policy in west and central Africa. Conserv Biol 21(3):884–887
Bermejo M, Rodriguez-Teijeiro JD, Illera G, Barroso A, Vila C, Walsh PD (2006) Ebola outbreak killed 5000 gorillas. Science 314(5805):1564–1564
Bessone M, Kühl HS, Hohmann G, Herbinger I, N’Goran KP, Asanzi P, Da Costa PB, Dérozier V, Fotsing EDB, Beka BI, Iyomi MD, Iyatshi IB, Kafando P, Kambere MA, Moundzoho DB, Wanzalire MLK, Fruth B (2020) Drawn out of the shadows: surveying secretive forest species with camera trap distance sampling. J Appl Ecol 57(5):963–974
Billerman SM, Keeney BK, Rodewald PG, Schulenberg TS (2022) Birds of the World. Cornell Laboratory of Ornithology, Ithaca
Blake S, Fay JM (1997) Seed production by Gilbertiodendron dewevrei in the Nouabalé-Ndoki National Park, Congo, and its implications for large mammals. J Trop Ecol 14:885–891
Bohmann K, Evans A, Gilbert MT, Carvalho GR, Creer S, Knapp M, Yu DW, de Bruyn M (2014) Environmental DNA for wildlife biology and biodiversity monitoring. Trends Ecol Evol 29(6):358–367
Boogert NJ, Paterson DM, Laland KN (2006) The implications of niche construction and ecosystem engineering for conservation biology. Bioscience 56:570
Bourlière F (1985) Primate communities - their structure and role in the tropical ecosystems. Int J Primatol 6(1):1–26
Bowkett A, Pegg N, Rovero F, Plowman A (2006) How do you monitor rare and elusive mammals? Counting duikers in Kenya, Tanzania and Zimbabwe. In: Zgrabczyñska ĆPE, Ziomek J (eds) Animals, zoos and conservation. Zoological Garden in Poznan, Poland, pp 21–28
Brakes P, Carroll EL, Dall SRX, Keith SA, McGregor PK, Mesnick SL, Noad MJ, Rendell L, Robbins MM, Rutz C, Thornton A, Whiten A, Whiting MJ, Aplin LM, Bearhop S, Ciucci P, Fishlock V, Ford JKB, Notarbartolo di Sciara G, Simmonds MP, Spina F, Wade PR, Whitehead H, Williams J, Garland EC (2021) A deepening understanding of animal culture suggests lessons for conservation. Proc R Society B 288(1949):20202718
Brncic T, Maisels F, Strindberg S (2018) Results of the 2016–2017 large mammal survey of the Ndoki-Likouala landscape. New York Wildlife Conservation Society, New York
Bruce T, Kamta R, Tabue Mbobda RB, Talla Kanto S, Djibrilla D, Moses I, Deblauwe V, Njabo K, LeBreton M, Ndjassi C, Barichievy C, Olson D (2018) Locating giant ground pangolins (Smutsia gigantea) using camera traps on burrows in the Dja Biosphere reserve, Cameroon. Trop Conserv Sci 11:1940082917749224
Cappelle N, Howe EJ, Boesch C, Kühl HS (2021) Estimating animal abundance and effort–precision relationship with camera trap distance sampling. Ecosphere 12(1):e03299
Caro T, Engilis A, Fitzherbert E, Gardner T (2004) Preliminary assessment of the flagship species concept at a small scale. Anim Conserv 7(1):63–70
Carvalho S, Wessling EG, Abwe EE, Almeida-Warren K, Arandjelovic M, Boesch C, Danquah E, Diallo MS, Hobaiter C, Hockings K, Humle T, Ikemeh RA, Kalan AK, Luncz L, Ohashi G, Pascual-Garrido A, Piel A, Samuni L, Soiret S, Sanz C, Koops K (2022) Using nonhuman culture in conservation requires careful and concerted action. Conserv Lett 15(2):e12860
Cawthorn D-M, Hoffman LC (2015) The bushmeat and food security nexus: a global account of the contributions, conundrums and ethical collisions. Food Res Int 76:906–925
Choosai C, Mathieu J, Hanboonsong Y, Jouquet P (2009) Termite mounds and dykes are biodiversity refuges in paddy fields in North-Eastern Thailand. Environ Conserv 36(1):71–79
Crooks JA (2002) Characterizing ecosystem-level consequences of biological invasions: the role of ecosystem engineers. Oikos 97:153–166
Danielsen F, Burgess ND, Jensen PM, Pirhofer-Walzl K (2010) Environmental monitoring: the scale and speed of implementation varies according to the degree of peoples involvement. J Appl Ecol 47(6):1166–1168
DeFries R, Karanth KK, Pareeth S (2010) Interactions between protected areas and their surroundings in human-dominated tropical landscapes. Biol Cons 143(12):2870–2880
Dupain J, Nackoney J, Mario Vargas J, Johnson PJ, Farfan MA, Bofaso M, Fa JE (2012) Bushmeat characteristics vary with catchment conditions in a Congo market. Biol Cons 146(1):32–40
Effiom EO, Nuñez-Iturri G, Smith HG, Ottosson U, Olsson O (2013) Bushmeat hunting changes regeneration of African rainforests. Proc R Soc B 280(1759):20130246
Elkan P (2003) Ecology and conservation of bongo antelope (Tragelaphus eurycerus) in Lowland Forest, northern Republic of Congo. (Unpublished Ph.D. thesis), University of Minnesota
Elkan P, Parnell R, Smith D (2009) A die-off of large ungulates following a Stomoxys biting fly out-break in lowland forest, northern Republic of Congo. Afr J Ecol 47(4):528–536
Fa JE, Brown D (2009) Impacts of hunting on mammals in African tropical moist forests: a review and synthesis. Mammal Rev 39(4):231–264
Fa JE, Currie D, Meeuwig J (2003) Bushmeat and food security in the Congo Basin: linkages between wildlife and people’s future. Environ Conserv 30(1):71–78
Fa JE, Johnson PJ, Dupain J, Lapuente J, Koster P, Macdonald DW (2004) Sampling effort and dynamics of bushmeat markets. Anim Conserv 7:409–416
Fa JE, Olivero J, Real R, Farfan MA, Marquez AL, Vargas JM, Ziegler S, Wegmann M, Brown D, Margetts B, Nasi R (2015) Disentangling the relative effects of bushmeat availability on human nutrition in central Africa. Sci Rep. https://doi.org/10.1038/srep08168
Fontyen D (2022) Distribution and determinants of mammal assemblages across central African forests PhD Thesis, Liege University
Fonteyn D, Vermeulen C, Deflandre N, Cornelis D, Lhoest S, Houngbégnon FGA, Doucet JL, Fayolle A (2021) Wildlife trail or systematic? Camera trap placement has little effect on estimates of mammal diversity in a tropical forest in Gabon. Remote Sens Ecol Conserv 7(2):321–336
Fragoso JMV, Levi T, Oliveira LFB, Luzar JB, Overman H, Read JM, Silvius KM (2016) Line transect surveys underdetect terrestrial mammals: Implications for the sustainability of subsistence hunting. PLOS ONE 11(4):e0152659
Gardner TA, Barlow J, Sodhi NS, Peres CA (2010) A multi-region assessment of tropical forest biodiversity in a human-modified world. Biol Cons 143(10):2293–2300
Gautier-Hion A, Brugiere D (2005) Significance of riparian forests for the conservation of Central African primates. Int J Primatol 26(3):515–523
Getz WM, Fortmann L, Cumming D, du Toit J, Hilty J, Martin R, Murphree M, Owen-Smith N, Starfield AM, Westphal MI (1999) Sustaining natural and human capital: villagers and scientists. Science 283(5409):1855–1856
Gibson L, Lee TM, Koh LP, Brook BW, Gardner TA, Barlow J, Peres CA, Bradshaw CJA, Laurance WF, Lovejoy TE, Sodhi NS (2011) Primary forests are irreplaceable for sustaining tropical biodiversity. Nature 478(7369):378
Gotelli NJ, Colwell RK (2001) Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecol Lett 4(4):379–391
Grantham HS, Shapiro A, Bonfils D, Gond V, Goldman E, Maisels F, Plumptre AJ, Rayden T, Robinson JG, Strindberg S, Stokes E, Tulloch A, Watson JEM, Williams L, Rickenbach O (2020) Spatial priorities for conserving the most intact biodiverse forests within Central Africa. Environ Res Lett 15(9). https://doi.org/10.1088/1748-9326/ab9fae
Hall J, Harris D, Saltonstall K, Medjibe V, Ashton M, Turner B (2019) Resource acquisition strategies facilitate Gilbertiodendron dewevrei monodominance in African lowland forests. J Ecol 108:433–448
Hansen AJ, Burns P, Ervin J, Goetz SJ, Hansen M, Venter O, Watson JEM, Jantz PA, Virnig ALS, Barnett K, Pillay R, Atkinson S, Supples C, Rodríguez-Buritica S, Armenteras D (2020) A policy-driven framework for conserving the best of earth’s remaining moist tropical forests. Nat Ecol Evolut 4(10):1377–1384
Harris DJ (2002) The vascular plants of the Dzanga-Sangha reserve. National Botanic Garden, Belgium
Hart TB (1990) Monospecific dominance in tropical rain forests. Trends Ecol Evol 5(1):6–11
Hart TB (2001) Forest dynamics in the Ituri Basin (DR Congo). In: White LJT, Weber W, Vedder A, Naughton-Treves I (eds) African rain forest ecology and conservation. Yale University Press, New Heaven, pp 154–164
Hedwig D, Kienast I, Bonnet M, Curran BK, Courage A, Boesch C, Kuehl HS, King T (2018) A camera trap assessment of the forest mammal community within the transitional savannah-forest mosaic of the Bateke Plateau National park, Gabon. Afr J Ecol 56(4):777–790
Hempson GP, Archibald S, Bond WJ (2015) A continent-wide assessment of the form and intensity of large mammal herbivory in Africa. Science 350(6264):1056–1061
Houngbégnon FGA, Cornelis D, Vermeulen C, Sonké B, Ntie S, Fayolle A (2020) Daily Activity Patterns and Co-Occurrence of Duikers Revealed by an Intensive Camera Trap Survey across Central African Rainforests. Animals 10(12)
Howe EJ, Buckland ST, Despres-Einspenner ML, Kuhl HS (2017) Distance sampling with camera traps. Methods Ecol Evol 8(11):1558–1565
IUCN (2022) The IUCN Red List of Threatened Species. Version 2022–1. Retrieved September 19, 2022
Kéry M, Royle JA (2008) Hierarchical Bayes estimation of species richness and occupancy in spatially replicated surveys. J Appl Ecol 45(2):589–598
Khwaja H, Buchan C, Wearn OR, Bahaa-el-din L, Bantlin D, Bernard H, Bitariho R, Bohm T, Borah J, Brodie J, Chutipong W, du Preez B, Ebang-Mbele A, Edwards S, Fairet E, Frechette JL, Garside A, Gibson L, Giordano A, Gopi GV, Granados A, Gubbi S, Harich F, Haurez B, Havmoller RW, Helmy O, Isbell LA, Jenks K, Kalle R, Kamjing A, Khamcha D, Kiebou-Opepa C, Kinnaird M, Kruger C, Laudisoit A, Lynam A, Macdonald SE, Mathai J, Sienne JM, Meier A, Mills D, Mohd-Azlan J, Nakashima Y, Nash HC, Ngoprasert D, Nguyen A, O'Brien T, Olson D, Orbell C, Poulsen J, Ramesh T, Reeder D, Reyna R, Rich LN, Rode-Margono J, Rovero F, Sheil D, Shirley MH, Stratford K, Sukumal N, Suwanrat S, Tantipisanuh N, Tilker A, Van Berkel T, Van der Weyde LK, Varney M, Weise F, Wiesel I, Wilting A, Wong ST, Waterman C, Challender DWS (2019) Pangolins in global camera trap data: Implications for ecological monitoring. Global Ecology and Conservation 20:e00769
Kingdon J (2015) The Kingdon field guide to African mammals, 2nd edn. Princeton University Press, Princeton
Koopmans M, Stokes EJ, Opepa CK, Mouele AM, Abea G, Strindberg S (2021) Wild bongo density estimation and population viability analysis improves conservation management. Global Ecol Conserv 28:e01661
Kuehl HS, Boesch C, Kulik L, Haas F, Arandjelovic M, Dieguez P, Bocksberger G, McElreath MB, Agbor A, Angedakin S, Ayimisin EA, Bailey E, Barubiyo D, Bessone M, Brazzola G, Chancellor R, Cohen H, Coupland C, Danquah E, Deschner T, Dowd D, Dunn A, Egbe VE, Eshuis H, Goedmakers A, Granjon AC, Head J, Hedwig D, Hermans V, Imong I, Jeffery KJ, Jones S, Junker J, Kadam P, Kambere M, Kambi M, Kienast I, Kujirakwinja D, Langergraber KE, Lapuente J, Larson B, Lee K, Leinert V, Llana M, Maretti G, Marrocoli S, Martin R, Mbi TJ, Meier AC, Morgan B, Morgan D, Mulindahabi F, Murai M, Neil E, Niyigaba P, Ormsby LJ, Orume R, Pacheco L, Piel A, Preece J, Regnaut S, Rundus A, Sanz C, van Schijndel J, Sommer V, Stewart F, Tagg N, Vendras E, Vergnes V, Welsh A, Wessling EG, Willie J, Wittig RM, Yuh YG, Yurkiw K, Zuberbuhler K, Kalan AK (2019) Human impact erodes chimpanzee behavioral diversity. Science 363(6434):1453–1458
Leader-Williams N, Dublin HT (2000) Charismatic megafauna as ‘flagship species.’ In: Entwistle A, Dunstone N (eds) Priorities for the conservation of mammalian diversity. Has the panda had its day? Cambridge University Press, Cambridge, pp 53–81
Leclere D, Obersteiner M, Barrett M, Butchart SHM, Chaudhary A, De Palma A, DeClerck FAJ, Di Marco M, Doelman JC, Durauer M, Freeman R, Harfoot M, Hasegawa T, Hellweg S, Hilbers JP, Hill SLL, Humpenoder F, Jennings N, Krisztin T, Mace GM, Ohashi H, Popp A, Purvis A, Schipper AM, Tabeau A, Valin H, van Meijl H, Van Zeist WJ, Visconti P, Alkemade R, Almond R, Bunting G, Burgess ND, Cornell SE, Di Fulvio F, Ferrier S, Fritz S, Fujimori S, Grooten M, Harwood T, Havlik P, Herrero M, Hoskins AJ, Jung M, Kram T, Lotze-Campen H, Matsui T, Meyer C, Nel D, Newbold T, Schmidt-Traub G, Stehfest E, Strassburg BBN, van Vuuren DP, Ware C, Watson JEM, Wu WC, Young L (2020) Bending the curve of terrestrial biodiversity needs an integrated strategy. Nature 585(7826):551
Lindenmayer DB, Margules CR, Botkin DB (2000) Indicators of biodiversity for ecologically sustainable forest management. Conserv Biol 14(4):941–950
Loh J, Green RE, Ricketts T, Lamoreux J, Jenkins M, Kapos V, Randers J (2005) The Living Planet Index: using species population time series to track trends in biodiversity. Philos Trans R Soc B 360(1454):289–295
Macdonald DW, Burnham D, Hinks AE, Wrangham R (2013) A problem shared is a problem reduced: Seeking efficiency in the conservation of felids and primates. Folia Primatol 83(3–6):171–215
Maisels F (2001). Nouabale-Ndoki National Park, Republic du Congo, Management Plan. 2001–2006. C. d. l. P. e. d. R. H. Ministere de l'Economie Forestiere, Republique du Congo, Wildlife Conservation Society
Maisels F, Strindberg S, Blake S, Wittemyer G, Hart J, Williamson EA, Aba’a R, Abitsi G, Ambahe RD, Amsini F, Bakabana PC, Hicks TC, Bayogo RE, Bechem M, Beyers RL, Bezangoye AN, Boundja P, Bout N, Akou ME, Bene LB, Fosso B, Greengrass E, Grossmann F, Ikamba-Nkulu C, Ilambu O, Inogwabini B-I, Iyenguet F, Kiminou F, Kokangoye M, Kujirakwinja D, Latour S, Liengola I, Mackaya Q, Madidi J, Madzoke B, Makoumbou C, Malanda G-A, Malonga R, Mbani O, Mbendzo VA, Ambassa E, Ekinde A, Mihindou Y, Morgan BJ, Motsaba P, Moukala G, Mounguengui A, Mowawa BS, Ndzai C, Nixon S, Nkumu P, Nzolani F, Pintea L, Plumptre A, Rainey H, de Semboli BB, Serckx A, Stokes E, Turkalo A, Vanleeuwe H, Vosper A, Warren Y (2013) Devastating decline of forest elephants in central Africa. PLOS ONE 8(3):e59469
Mambeya MM, Baker F, Momboua BR, Koumba Pambo AF, Hega M, Okouyi Okouyi VJ, Onanga M, Challender DWS, Ingram DJ, Wang H, Abernethy K (2018) The emergence of a commercial trade in pangolins from Gabon. Afr J Ecol 56(3):601–609
Marrocoli S, Nielsen MR, Morgan D, van Loon T, Kulik L, Kühl H (2019) Using wildlife indicators to facilitate wildlife monitoring in hunter-self monitoring schemes. Ecol Ind 105:254–263
McComie LD, Dhanarajan G (1993) The physical and chemical composition of mounds of Macrotermes carbonarius (Hagen) (Termitidae, Macrotermitinae), in Penang. Malaysia J Soil Sci 44(3):427–433
Mittermeier RA (1986) Primate conservation priorities in the Neotropical region. In: Benirschke E (ed) Primates—the road to self-sustaining populations. Springer, New York, pp 221–240
Mittermeier RA (1988) Primate diversity and the tropical forest. In: Wilson EO (ed) Biodiversity. National Academy Press, Washington DC, pp 145–154
Mockrin MH (2008) The spatial structure and sustainability of subsistence wildlife harvesting in Kabo, Congo. PhD thesis. Columbia University
Mockrin MH (2010) Duiker demography and dispersal under hunting in Northern Congo. Afr J Ecol 48(1):239–247
Moe SR, Mobaek R, Narmo AK (2009) Mound building termites contribute to savanna vegetation heterogeneity. Plant Ecol 202:31–40
Morgan D, Sanz C, Onononga JR, Strindberg S (2006) Ape abundance and habitat use in the Goualougo Triangle, Republic of Congo. Int J Primatol 27(1):147–179
Morgan D, Strindberg S, Winston W, Stephens CR, Traub C, Ayina CE, Ndolo Ebika ST, Mayoukou W, Koni D, Iyenguet F, Sanz CM (2019) Impacts of selective logging and associated anthropogenic disturbance on intact forest landscapes and apes of northern Congo. Front Forest Glob Change 2(28). https://doi.org/10.3389/ffgc.2019.00028
Mugerwa B, Sheil D, Ssekiranda P, van Heist M, Ezuma P (2013) A camera trap assessment of terrestrial vertebrates in Bwindi Impenetrable National Park. Uganda Afr J Ecol 51(1):21–31
Nasi R, Billand A, van Vliet N (2012) Managing for timber and biodiversity in the Congo Basin. For Ecol Manage 268:103–111
Newbold T, Scharlemann JPW, Butchart SHM, Sekercioglu CH, Alkemade R, Booth H, Purves DW (2013) Ecological traits affect the response of tropical forest bird species to land-use intensity. Proc R Soc B 280(1750). https://doi.org/10.1098/rspb.2012.2131
Nichols JD, Karanth KU, O’Connell AF (2011) Science, conservation, and camera traps. In: O’Connell AF, Nichols JD, Karanth KU (eds) Camera traps in animal ecology. Springer, Tokyo
Noss A (2000) Cable snares and nets in the Central African Republic. In: Robinson J, Bennette (eds) Hunting for sustainability in tropical forests. Columbia University Press, New York, pp 282–304
O’Brien TG (2008) On the use of automated cameras to estimate species richness for large- and medium-sized rainforest mammals. Anim Conserv 11(3):179–181
O’Brien TG, Baillie JEM, Krueger L, Cuke M (2010) The wildlife picture index: monitoring top trophic levels. Anim Conserv 13(4):335–343
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara R, Simpson GL, Solymos P, Stevens MHH, Wagner H (2015) Vegan: community ecology package. 2019. R package version 2(10). https://github.com/vegandeus/vegan
Oldekop JA, Sims KRE, Karna BK, Whittingham MJ, Agrawal A (2019) Reductions in deforestation and poverty from decentralized forest management in Nepal. Nat Sustain 2(5):421–428
Ozaki K, Isono SM, Kawahara T, Iilda S, Kudo T, Fukuyama K (2006) A mechanistic approach to evaluation of umbrella species as conservation surrogates. Conserv Biol 20(5):1507–1515
Phillips HRP, Newbold T, Purvis A (2017) Land-use effects on local biodiversity in tropical forests vary between continents. Biodivers Conserv 26(9):2251–2270
Potapov P, Hansen MC, Laestadius L, Turubanova S, Yaroshenko A, Thies C, Smith W, Zhuravleva I, Komarova A, Minnemeyer S, Esipova E (2017) The last frontiers of wilderness: tracking loss of intact forest landscapes from 2000 to 2013. Sci Adv. https://doi.org/10.1126/sciadv.1600821
Poulsen JR, Clark CJ, Mavah G, Elkan PW (2009) Bushmeat supply and consumption in a tropical logging concession in Northern Congo. Conserv Biol 23(6):1597–1608
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Ripple WJ, Wolf C, Newsome TM, Hoffmann M, Wirsing AJ, McCauley DJ (2017) Extinction risk is most acute for the world’s largest and smallest vertebrates. Proc Natl Acad Sci USA 114(40):10678–10683
Roberge JM, Angelstam P (2004) Usefulness of the umbrella species concept as a conservation tool. Conserv Biol 18(1):76–85
Rovero F, Martin E, Rosa M, Ahumada JA, Spitale D (2014) Estimating species richness and modelling habitat preferences of tropical forest mammals from camera trap data. PLOS ONE 9(7):1–2. https://doi.org/10.1371/journal.pone.0103300
Rowcliffe JM, Milner-Gulland EJ, Cowlishaw G (2005) Do bushmeat consumers have other fish to fry? Trends Ecol Evol 20(6):274–276
Rowcliffe JM, Kays R, Kranstauber B, Carbone C, Jansen PA (2014) Quantifying levels of animal activity using camera trap data. Methods Ecol Evol 5(11):1170–1179
Schipper AM, Hilbers JP, Meijer JR, Antao LH, Benitez-Lopez A, de Jonge MMJ, Leemans LH, Scheper E, Alkemade R, Doelman JC, Mylius S, Stehfest E, van Vuuren DP, van Zeist WJ, Huijbregts MAJ (2020) Projecting terrestrial biodiversity intactness with GLOBIO 4. Glob Change Biol 26(2):760–771
Scholes RJ, Biggs R (2005) A biodiversity intactness index. Nature 434(7029):45–49
Simberloff D (1998) Flagships, umbrellas and keystones: is single-species management passe´ in the landscape era? Biol Cons 83:247–257
Slough T, Kopas J, Urpelainen J (2021a) Satellite-based deforestation alerts with training and incentives for patrolling facilitate community monitoring in the Peruvian Amazon. Proc Natl Acad Sci 118(29):e2015171118
Slough T, Rubenson D, Levy R, Alpizar Rodriguez F, Bernedo del Carpio M, Buntaine MT, Christensen D, Cooperman A, Eisenbarth S, Ferraro PJ, Graham L, Hartman AC, Kopas J, McLarty S, Rigterink AS, Samii C, Seim B, Urpelainen J, Zhang B (2021b) Adoption of community monitoring improves common pool resource management across contexts. Proc Natl Acad Sci 118(29):e2015367118
Stokes EJ, Strindberg S, Bakabana PC, Elkan PW, Iyenguet FC, Madzoke B, Malanda GAF, Mowawa BS, Moukoumbou C, Ouakabadio FK, Rainey HJ (2010) Monitoring great ape and elephant abundance at large spatial scales: Measuring effectiveness of a conservation landscape. PLOS ONE 5(4). https://doi.org/10.1371/journal.pone.0010294
Strier KB, Melo FR, Mendes SL, Valença-Montenegro MM, Rylands AB, Mittermeier RA, Jerusalinsky L (2021) Science, policy, and conservation management for a critically endangered primate in the Atlantic Forest of Brazil. Front Conserv Sci. https://doi.org/10.3389/fcosc.2021.734183
Sun P, Umuntunundi P, Wronski T (2022) Species richness, relative abundance and occupancy of ground-dwelling mammals denote the ineffectiveness of chimpanzee as flagship species.. Mamm Biol 102(5):1835–1850
Taylor G, Scharlemann JPW, Rowcliffe M, Kumpel N, Harfoot MBJ, Fa JE, Melisch R, Milner-Gulland EJ, Bhagwat S, Abernethy KA, Ajonina AS, Albrechtsen L, Allebone-Webb S, Brown E, Brugiere D, Clark C, Colell M, Cowlishaw G, Crookes D, De Merode E, Dupain J, East T, Edderai D, Elkan P, Gill D, Greengrass E, Hodgkinson C, Ilambu O, Jeanmart P, Juste J, Linder JM, Macdonald DW, Noss AJ, Okorie PU, Okouyi VJJ, Pallier S, Poulsen JR, Riddell M, Schleicher J, Schulte-Herbruutiggen B, Starkey M, van Vliet N, Whitham C, Willcox AS, Wilkie DS, Wright JH, Coad LM (2015) Synthesising bushmeat research effort in West and Central Africa: a new regional database. Biol Cons 181:199–205
Tews J, Brose U, Grimm V, Tielbörger K, Wichmann MC, Schwager M, Jeltsch F (2004) Animal species diversity driven by habitat heterogeneity/diversity: the importance of keystone structures. J Biogeogr 31(1):79–92
Tobler MW, Carrillo-Percastegui SE, Leite Pitman R, Mares R, Powell G (2008) An evaluation of camera traps for inventorying large- and medium-sized terrestrial rainforest mammals. Anim Conserv 11(3):169–178
Traore S, Tigabu M, Ouedraogo SJ, Boussim JI, Guinko S et al (2008) Macrotermes mounds as sites for tree regeneration in a Sudanian woodland (Burkina Faso). Plant Ecol 198:285–295
van Vliet N (2018) Bushmeat crisis and cultural imperialism in wildlife management? Taking value orientations into account for a more sustainable and culturally acceptable wildmeat sector. Front Ecol Evol 6(112):1–6. https://doi.org/10.3389/fevo.2018.00112
van Vliet N, Nasi R (2008) Why do models fail to assess properly the sustainability of duiker ( Cephalophus spp.) hunting in Central Africa? Oryx 42(3):392–399
Vanthomme H, Bellé B, Forget PM (2010) Bushmeat hunting alters recruitment of large-seeded plant species in central Africa. Biotropica 42(6):672–679
Watson JEM, Evans T, Venter O, Williams B, Tulloch A, Stewart C, Thompson I, Ray JC, Murray K, Salazar A, McAlpine C, Potapov P, Walston J, Robinson JG, Painter M, Wilkie D, Filardi C, Laurance WF, Houghton RA, Maxwell S, Grantham H, Samper C, Wang S, Laestadius L, Runting RK, Silva-Chavez GA, Ervin J, Lindenmayer D (2018) The exceptional value of intact forest ecosystems. Nat Ecol Evolut 2(4):599–610
Western D (1987) Africa’s elephants and rhinos: flagships in crisis. Trends in Ecol Evol 2:343–346
Wilkie DS, Sidle JG, Boundzanga GC, Auzel P, Blake S (2001) Defaunation, not deforestation: Commercial logging and market hunting in northern Congo. In: Fimbel RA, Grajal A, Robinson JG (eds) The cutting edge conserving wildlife in logged tropical forest. Columbia University Press, New York, pp 375–399
Wilkie DS, Wieland M, Poulsen JR (2019) Unsustainable vs. sustainable hunting for food in Gabon: modeling short- and long-term gains and losses. Front Ecol Evolut. https://doi.org/10.3389/fevo.2019.00357
Wrangham RW, Hagel G, Leighton M, Marshall AJ, Waldau P, Nishida T (2008) The great ape world heritage species project. In: Stoinski TS, Steklis HD, Mehlman PT (eds) Conservation in the 21st century Gorillas as a case study. Springer, Boston, pp 282–295
Wright SJ (2003) The myriad consequences of hunting for vertebrates and plants in tropical forests. Perspect Plant Ecol Evolut Syst 6(1):73–86
WWF/LZS (2020). WWF Living Planet Report 2020-Bending the curve of biodiversity loss. W. R. E. A. Almond, G. M. and T. Petersen. Gland, Switzerland, WWF
Yasuoka H, Hirai M, Kamgaing TOW, Dzefack Z, Kamdoum EC, Bobo KS (2015) Changes in the composition of hunting catches in southeastern Cameroon: a promising approach for collaborative wildlife management between ecologists and local hunters. Ecol Soc. https://doi.org/10.5751/ES-08041-200425
Zett T, Stratford KJ, Weise FJ (2022) Inter-observer variance and agreement of wildlife information extracted from camera trap images. Biodivers Conserv 31:3019–3037
Ziegler S, Fa JE, Wohlfart C, Streit B, Jacob S, Wegmann M (2016) Mapping bushmeat hunting pressure in Central Africa. Biotropica 48(3):405–412
Acknowledgements
We are deeply appreciative of the opportunity to work in the Djéké Triangle and the Nouabalé-Ndoki National Park. This research would not have been possible without the continued support of the Ministère de l’Economie Forestière du gouvernement de la République du Congo and the Agence Congolaise de la Faune et des Aires Protégées (ACFAP). The Wildlife Conservation Society’s Congo Program and the Nouabalé-Ndoki Foundation are integral partners in this continuing research. Special thanks are due to R. Malonga, M. Gately, E. Stokes, T. Breuer, P. Ngouembe, D. Dos Santos, E. Arnhem, B. Evans and M. Ngangoue. We would also like to recognize the tireless dedication of J. R. Onononga, C. Eyana-Ayina, S. Ndolo Ebika, W. Mayoukou, S. Kialiema, J. Wawa, D. Koni, I. Singono, M. Meguessa, and the Mondika and Goualougo tracking teams. We also thank A. Mitchell and E. Claisse for their assistance in reviewing and compiling materials for the species identification guide. This work was greatly improved by input from J. Ray, F. Maisels, and J. Kingdon who generously provided expert review of particular taxa. We are also grateful to N. Radford for feedback on this manuscript. Acknowledgment of funding is due to the Arcus Foundation; the Conservation, Food and Health Foundation; Lincoln Park Zoo, Indianapolis Zoo; the Saint Louis Zoo, Woodland Park Zoo, Zoo Atlanta and the Cincinnati Zoo and Botanical Garden. In addition, we are immensely grateful to S. Ross who provided enthusiastic encouragement and support for these field conservation efforts over many years. This research would not have been possible without him.
Author information
Authors and Affiliations
Contributions
Conceptualization, DM, SS and CS; Methodology, DM, SS and CS; Investigation, DM, AZ, IS, SH, SM, CEA, PJ, RC and CS; Data Curation, DM, SH, SM, PM, JF, HH and CS; Writing—Original Draft Preparation, DM and CS; Writing—Review and Editing, DM, SS, PM, AZ, IS, SH, SM, CEA, JF and CS; Visualization, DM, PM and JF; Supervision, DM, AZ, CEA and CS; Project Administration, DM, AZ, CEA, HH and CS; Funding Acquisition, DM, SM, HH and CS.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Our study adhered to the legal requirements of the Republic of Congo where the research was conducted. The research was approved by the Nouabalé-Ndoki Foundation and the Wildlife Conservation Society’s Congo Program. We also complied with ethics guidelines of the Association for the Study of Animal Behaviour (ASAB) and Animal Behavior Society. After reviewing the study’s camera trapping methods, Washington University issued an IACUC exemption.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Morgan, D., Strindberg, S., McElmurray, P. et al. Extending the conservation impact of great ape research: Flagship species sites facilitate biodiversity assessments and land preservation. Primates (2023). https://doi.org/10.1007/s10329-023-01080-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10329-023-01080-x