Abstract
The plant innate immune system deploys receptor proteins on the cell surface or inside the cell to sense microbial invasions. Perception of microbe associated molecular patterns (MAMPs), which are typically derived from conserved molecules in a class of microbes such as bacterial flagellin, triggers pattern-triggered immunity (PTI). While pathogens possess a repertoire of effectors that dampen PTI, plants detect the presence or actions of effectors and induce effector-triggered immunity (ETI) to prevent pathogen infection. PTI and ETI utilize a number of common signaling components such as reactive oxygen species, mitogen-activated protein kinases (MAPKs), and phytohormones to reprogram the cell state toward defense responses. It has become increasingly clear that these signaling components do not act in isolation but interact with each other to form an intricate network. Here, I discuss structure and dynamics of the plant immune signaling network that operate behind the interaction between the model plant Arabidopsis thaliana and the bacterial pathogen Pseudomonas syringae pv. tomato DC3000 (Pto).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The plant innate immune system deploys receptor proteins on the cell surface or inside the cell to sense microbial invasions (Jones and Dangl 2006). Perception of microbe-associated molecular patterns (MAMPs), which are typically derived from molecules such as bacterial flagellin that are conserved among a particular class of microbes, triggers pattern-triggered immunity (PTI). While pathogens possess a repertoire of effectors that dampen PTI, plants detect the presence or actions of effectors and induce effector-triggered immunity (ETI) to prevent pathogen infection. PTI and ETI utilize a number of common signaling components such as reactive oxygen species, mitogen-activated protein kinases (MAPKs), and phytohormones to reprogram the cell state toward defense responses. It has become increasingly clear that these signaling components do not act in isolation but interact with each other to form an intricate network (Mine et al. 2014). Here, I discuss structure and dynamics of the plant immune signaling network that operate behind the interaction between the model plant Arabidopsis thaliana and the bacterial pathogen Pseudomonas syringae pv. tomato DC3000 (Pto).
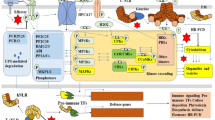
A signaling network that confers tunability and robustness to PTI
Plant immunity confers resistance against pathogens but often comes with a growth penalty (Karasov et al. 2017). Therefore, plant immunity needs to be tunable to avoid triggering unnecessary costly defense responses, especially against nonpathogenic microbes. Another property that should be intrinsic to plant immunity is a robustness that enables plants to withstand perturbations of defense responses by pathogen effectors or environmental factors. These properties, tunability and robustness, are particularly relevant to PTI, as it is induced by MAMPs, which do not distinguish pathogens from nonpathogenic microbes. However, how these properties emerge during PTI was poorly understood.
The phytohormones jasmonic acid (JA) and salicylic acid (SA) mediate immunity against necrotrophic and biotrophic pathogens, respectively (Glazebrook 2005). These phytohormones accumulate concomitantly during PTI (Tsuda and Katagiri 2010), suggesting the importance of JA–SA interplay for orchestrating PTI. Nevertheless, our understanding of the biological significance of JA–SA interplay was mostly limited to their mutual antagonism, which explains prioritization of resistance against necrotrophic or biotrophic pathogens over the other (Glazebrook 2005). I discovered that JA not only inhibits but also supports SA accumulation in a context-dependent manner during PTI triggered by the flagellin-derived MAMP, flg22 (Mine et al. 2017b). JA exerts a repressive effect on expression of PHYTOALEXIN DEFICIENT 4 (PAD4) that positively contributes to expression of ENHANCED DISEASE SUSCEPTIBILITY 5 (EDS5), a gene essential for SA accumulation. Paradoxically, JA also activates EDS5. The inhibitory effect of JA on SA is functionally dominant and mitigates SA accumulation to minimize the negative impact of SA on plant growth. However, the positive effect of JA on SA supports robust SA accumulation and bacterial resistance when PAD4 is perturbed by pathogen effectors or high temperature. Thus, the network structure discovered in this study simultaneously provides PTI with tunability and robustness.
Pathogen manipulation of the JA signaling network to inactivate MAPKs
Various strains of P. syringae including Pto produce the phytotoxin coronatine (COR) to promote virulence (Xin et al. 2018). COR as a structural mimic of the biologically active form of JA, ( +)-7-iso-jasmonoyl-l-Ile, is perceived by the JA receptor CORONATINE-INSENSITIVE 1 (COI1), triggering proteasome-dependent degradation of JASMONATE ZIM DOMAIN (JAZ) proteins, which liberates JAZ-mediated repression of the master transcription factor MYC2. Activation of MYC2 by COR transcriptionally activates the NAC transcription factors ANAC019, ANAC055, and ANAC072, which repress the SA biosynthesis gene SALYCYLIC ACID INDUCTION DEFICIENT 2 (SID2) and induce the SA catabolism gene SALICYLATE/BENZOATE CARBOXYL METHYLTRANSFERASE 1 (BSMT1), thereby suppressing SA-mediated immunity (Zheng et al. 2012). However, immune suppression by COR appeared to involve another target(s) in addition to SA (Zheng et al. 2012).
The activation status of the immune-related MAPKs, MPK3 and MPK6, changes dynamically during pathogen infection (Tsuda and Katagiri 2010). In an attempt to identify a regulator of MPK3/MPK6 activation during A. thaliana-Pto interactions, I discovered that COR inactivates these MAPKs (Mine et al. 2017a). COR-activated MYC2 directly regulates expression of HIGHLY ABA-INDUCED 1 (HAI1) encoding a serine/threonine phosphatase. HAI1 acts as a phosphatase on MPK3 and MPK6. Importantly, COR requires HAI1 to promote Pto growth in plants. These findings add a new aspect of how COR suppresses plant immunity: COR manipulates the JA signaling network to reduce SA accumulation on one hand and to inactivate MAPKs on the other hand.
ETI overcomes virulence actions of pathogen effectors and reestablishes bacterial resistance
PTI and ETI share immune signaling components such as SA and MAPK. Nevertheless, ETI is effective against pathogens that overcome PTI. For instance, Pto is highly virulent in A. thaliana accession Col-0 due to an arsenal of PTI-suppressing effectors but become avirulent when additionally carrying ETI-inducing effectors such as AvrRpt2 and AvrRpm1 (Jones and Dangl 2006). I found that ETI activation upon recognition of AvrRpt2 or AvrRpm1 counteracts induction of HAI1 and ANAC019/055/072 by COR (Mine et al. 2017a). This finding is analogous to the recent report that AvrRpt2-triggered ETI causes S-nitrosylation of and disarms the P. syringae effector HopAI1 (Ling et al. 2017). Moreover, ETI protects AtMIN7, an immune component involved in both PTI and ETI, from degradation by the P. syringae effector HopM1 (Nomura et al. 2011). Collectively, these findings support an emerging idea that plants can take back control of the immune signaling network over pathogens by counteracting effector actions, thereby reestablishing bacterial resistance during ETI.
The signaling network comprising JA, ethylene (ET), PAD4, and SA is responsible for the robust bacterial resistance in AvrRpt2-triggered ETI (Tsuda et al. 2009). To address how these signaling components contribute together to AvrRpt2-triggered ETI, I profiled the transcriptome of A. thaliana Col-0 and combinatorial mutants deficient in the network components over time after challenge with Pto carrying AvrRpt2 effector, and showed that susceptible plants, including a quadruple mutant lacking all four network components, had gene expression patterns that were almost identical to those in the resistant Col-0 plants but delayed by several hours (Mine et al. 2018). This delay in gene expression could hinder coexpression of functionally related genes. However, coexpression network analysis revealed that modules of coexpressed genes identified in the wild type are highly preserved in the quadruple mutant. Thus, the major role of the JA/ET/PAD4/SA signaling network is to establish transcriptional reprogramming within the early time window of ETI, which is critical for resistance against this bacterial pathogen.
Conclusion and perspectives
My research achievements have shed light on the molecular mechanisms underlying the dynamics of plant immune signaling networks of import in the outcome of plant–bacteria interactions. For instance, the effects of JA change from negative to positive to achieve tunable and robust SA accumulation depending on the status of the network component PAD4 during PTI. Such sophisticated interactions between network components are beneficial for plants to optimize their immune responses in a changing environment, but can be exploited by pathogens. Indeed, COR produced by Pto exploits JA-mediated suppression of SA and MAPK. However, plants can counteract these virulence actions of COR and reestablish disease resistance during ETI. I anticipate that these studies provide a foundation for further understanding of plant–pathogen interactions from a network perspective, which will eventually allow us to manipulate plant immune signaling networks for designing disease resistance according to pathogen virulence mechanisms and environmental conditions in agricultural fields.
References
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Jones JD, Dangl JL (2006) The plant immune system. Nature 444(7117):323–329
Karasov TL, Chae E, Herman JJ, Bergelson J (2017) Mechanisms to mitigate the trade-off between growth and defense. Plant Cell 29(4):666–680
Ling T, Bellin D, Vandelle E, Imanifard Z, Delledonne M (2017) Host-mediated S-nitrosylation disarms the bacterial effector HopAI1 to reestablish immunity. Plant Cell 29(11):2871–2881
Mine A, Sato M, Tsuda K (2014) Toward a systems understanding of plant–microbe interactions. Front Plant Sci 5:423
Mine A, Berens ML, Nobori T, Anver S, Fukumoto K, Winkelmuller TM, Takeda A, Becker D, Tsuda K (2017a) Pathogen exploitation of an abscisic acid- and jasmonate-inducible MAPK phosphatase and its interception by Arabidopsis immunity. Proc Natl Acad Sci USA 114(28):7456–7461
Mine A, Nobori T, Salazar-Rondon MC, Winkelmuller TM, Anver S, Becker D, Tsuda K (2017b) An incoherent feed-forward loop mediates robustness and tunability in a plant immune network. EMBO Rep 18(3):464–476
Mine A, Seyfferth C, Kracher B, Berens ML, Becker D, Tsuda K (2018) The defense phytohormone signaling network enables rapid, high-amplitude transcriptional reprogramming during effector-triggered immunity. Plant Cell 30(6):1199–1219
Nomura K, Mecey C, Lee YN, Imboden LA, Chang JH, He SY (2011) Effector-triggered immunity blocks pathogen degradation of an immunity-associated vesicle traffic regulator in Arabidopsis. Proc Natl Acad Sci USA 108(26):10774–10779
Tsuda K, Katagiri F (2010) Comparing signaling mechanisms engaged in pattern-triggered and effector-triggered immunity. Curr Opin Plant Biol 13(4):459–465
Tsuda K, Sato M, Stoddard T, Glazebrook J, Katagiri F (2009) Network properties of robust immunity in plants. PLoS Genet 5(12):e1000772
Xin XF, Kvitko B, He SY (2018) Pseudomonas syringae: what it takes to be a pathogen. Nat Rev Microbiol 16(5):316–328
Zheng XY, Spivey NW, Zeng W, Liu PP, Fu ZQ, Klessig DF, He SY, Dong X (2012) Coronatine promotes Pseudomonas syringae virulence in plants by activating a signaling cascade that inhibits salicylic acid accumulation. Cell Host Microbe 11(6):587–596
Acknowledgements
I express my sincere gratitude to Drs. Tetsuro Okuno, Yoshitaka Takano, Kazuyuki Mise and Masanori Kaido at Kyoto University, Dr. Kenichi Tsuda at Max Planck Institute for Plant Breeding Research, Dr. Yasuomi Tada at Nagoya University and Dr. Atsushi Takeda at Ritsumeikan University for their valuable suggestions, continuous support, and warm encouragement. I am also grateful to my colleagues and collaborators for their help and encouragement. My research was supported by a Postdoctoral Fellowship from Max Planck Society, a Postdoctoral Fellowship for Research Abroad from the Japanese Society for the Promotion of Science (JSPS), a Grant-in-Aid for JSPS Fellows (15J09701), a Grant-in-Aid for Young Scientists (B) (17K17802), a Grant-in-Aid for Scientific Research (B) (19H02960), Ritsumeikan Global Innovation Research Organization, and JST PRESTO (JPMJPR17Q6).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author has no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is an abstract of the paper which was going to be presented by a winner of the Young Scientist Award at the 2020 Annual Meeting of the Phytopathological Society of Japan in Kagoshima.
Rights and permissions
About this article
Cite this article
Mine, A. Structure and dynamics of the plant immune signaling network in plant–bacteria interactions. J Gen Plant Pathol 86, 528–530 (2020). https://doi.org/10.1007/s10327-020-00954-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-020-00954-y