Abstract
Introduction
Patients with vasovagal syncope (VVS) and positive tilt table test (TTT) were not found to benefit from pacing in the ISSUE-3 trial despite the presence of spontaneous asystole during monitoring. “Hypotensive susceptibility” unmasked by TTT was reported as a possible explanation. The purpose of this study was to assess the pathophysiologic mechanisms associated with hypotensive susceptibility.
Methods
366 consecutive patients with the diagnosis of VVS who also had TTT were identified. Baroreflex gain (BRG) in addition to blood pressure (BP) and heart rate (HR) responses during the first 20 min of TTT were analyzed and compared between patients with positive TTT (n = 275, 75 %) and negative TTT (n = 91, 25 %).
Results
The mean BRG was similar between the groups (12.5 ± 6.3 versus 12.4 ± 6.3 ms/mmHg, p = 0.72); however, an age-dependent decrease was noted (17.6 ± 4.8, 15.0 ± 6.0, 10.6 ± 4.2, 10.3 ± 6.4 and 9.9 ± 8.5 ms/mmHg for patients <21, 21–40, 41–60, 61–80 and >80 years old, respectively; p < 0.001). In addition, we saw a main effect of age on the type of response with a greater prevalence of a vasodepressor response in older subjects (p < 0.001). During the first 20 min of TTT, BP was similar in patients with tilt-positive VVS when compared with patients with tilt-negative VVS; however, HR was significantly lower.
Conclusion
BRG is similar in tilt-positive VVS patients when compared with tilt-negative VVS patients. An age-dependent decrease in BRG was noted with a higher prevalence of a vasodepressor response seen in older patients. The clinical significance of the blunted HR response in tilt-positive VVS remains to be determined.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Vasovagal syncope (VVS) is the most common type of syncope encountered in clinical practice [1]. While the underlying mechanisms remain poorly defined, the baroreflex has long been implicated in its pathogenesis, with tilt table testing (TTT) remaining the diagnostic test of choice in patients with suspected VVS [2]. The incidence of positive TTT depends on the clinical presentation with a higher positivity rate noted in patients with typical symptoms when compared to patients with atypical symptoms or unexplained syncope [3–6].
Management of patients with VVS remains a challenge. The Third International Study on Syncope of Uncertain Etiology (ISSUE-3) trial assessed the role of cardiac pacing in patients above the age of 40 years with neurally mediated spontaneous asystole [7]. The authors found a 57 % reduction in syncope recurrence with pacing therapy (57 versus 25 %, p = 0.04). In a subsequent study assessing the utility of TTT in predicting response to pacing, the benefit was found to be limited to patients with negative TTT [8]. Patients with VVS and positive TTT were found to derive no benefit from cardiac pacing despite the presence of spontaneous asystole during monitoring. “Hypotensive susceptibility” unmasked by TTT was reported as a possible explanation [9]. The authors hypothesized that patients with positive TTT have a significant vasodepressor response when subjected to orthostatic stress or other challenges such as cardiac arrhythmias rendering cardiac pacing ineffective.
The purpose of this study was to assess the baroreflex and hemodynamic response during TTT in patients with tilt-positive and tilt-negative VVS.
Methods
Subjects
We examined retrospectively the medical records of 482 consecutive patients referred to the Faint and Fall Clinic at the University of Wisconsin Hospital and Clinics between March 2013 and February 2015 for TTT. Inclusion criteria included certain or likely diagnosis of VVS as defined by the European Society of Cardiology and the Heart Rhythm Society [1, 10] and the completion of a TTT. Exclusion criteria included the presence of cardiac abnormalities suggestive of cardiac syncope or findings on examination consistent with orthostatic hypotension as the underlying mechanism of syncope. Additional exclusion criteria included the presence of cardiac pacing or atrial arrhythmias at the time of TTT or final diagnosis other than VVS following TTT including delayed orthostatic hypotension or psychogenic syncope. A total of 366 patients met the inclusion criteria and formed the cohort of this study. The study was approved by the institutional review board at the University of Wisconsin.
Tilt table test
Patients underwent TTT according to the University of Wisconsin protocol, a modification of the Italian protocol [6]. Briefly, after allowing the patient to rest in the supine position for 10 min, the table was tilted to 70°. At 20 min, patients who did not experience near syncope or syncope were given 0.3 mg of sublingual nitroglycerin. After nitroglycerin administration, TTT was continued for another 10 min. The modified Vasovagal Syncope International Study (VASIS) classification, which consists of Type 1 (mixed), Type 2A and 2B (cardio-inhibitory without and with asystole, respectively), or Type 3 (vasodepressor) was used to identify the type of response [11].
Measurements
Continuous blood pressure (BP) and heart rate (HR) monitoring were obtained non-invasively using the Task Force Monitor (CN Systems, Graz, Austria). Baroreflex gain (BRG) was measured using the sequence method [12] while the patient was in the supine position. Briefly, during the baseline monitoring period, spontaneously occurring increases or decreases in SBP of a minimum of 1 mmHg/beat for at least three consecutive heartbeats followed by progressive lengthening or shortening in RR intervals of at least 4 ms/beat, respectively, were identified. A regression line between SBP and RR changes for all identified sequences was plotted and the slopes were averaged and used to represent the sensitivity of arterial baroreflex modulation of HR. HR decreases and increases following spontaneous increases and decreases in BP have been shown to be the result of vagal activation and withdrawal, respectively, thus making BRG a reliable measure of vagal control of HR.
BP and HR measurements during TTT were recorded during the entire test; however, data analysis was limited to the first 20 min before the administration of sublingual nitroglycerin. We limited the data analysis to the first 20 min to assess patients’ hemodynamic responses to orthostatic stress independent of drug effects.
Statistical analysis
Statistical tests were performed using SigmaPlot v13 statistical software (Systat Software, Inc, San Jose, CA). One-way ANOVA was used to detect the effect of age on BRG and assess for significant differences in BRG, BP and HR responses between patients with a cardio-inhibitory response (VASIS Type 2A and 2B) and those with a mixed (VASIS Type 1) or vasodepressor (VASIS Type 3) responses. Post hoc multiple comparisons (Dunn’s method) were used where appropriate. Two-way repeated measures ANOVA was used to assess for statistically significant differences in BRG, BP and HR responses during the first 20 min of TTT between tilt-positive and tilt-negative VVS patients. Chi-square tests were used to determine if proportions in the various categories were different. P values less than 0.05 were considered statistically significant. All study data were expressed as mean ± SD.
Results
Patients’ characteristics
The mean age was 48 ± 20 years. Two hundred and seventy-five patients (75 %) had a positive TTT (tilt-positive VVS group; mean age 50 ± 21) and 91 patients had a negative TTT (tilt-negative VVS group; mean age 42 ± 18). The mean age and prevalence of hypertension were significantly higher in the tilt-positive VVS group when compared with the tilt-negative VVS group (p = 0.002 and p = 0.05, respectively). The mean BRG was similar in the tilt-positive and tilt-negative VVS groups (12.5 ± 6.3 versus 12.4 ± 6.3 ms/mmHg, p = 0.72). A summary of the clinical characteristics and BRG measurements is provided in Table 1.
In the tilt-positive VVS group, 33 (12 %), 161 (59 %) and 81 (29 %) patients had a cardio-inhibitory, mixed and vasodepressor responses, respectively. The mean ages were 40 ± 21, 48 ± 19 and 59 ± 22 years, respectively. Patients with a vasodepressor response were significantly older than those with a cardio-inhibitory or mixed response (p < 0.001). The mean BRG values in the three groups were 14.5 ± 6.2, 12.6 ± 6.2 and 11.5 ± 6.4 ms/mmHg, respectively. Patients with a vasodepressor response had a significantly lower mean BRG when compared with patients with a cardio-inhibitory response (p = 0.03). A summary of the clinical characteristics and BRG measurements in the various subgroups of patients with tilt-positive test is provided in Table 2.
Effects of age on BRG and response to TTT
Age had a significant effect on BRG measurements and response to TTT. BRG decreased with increasing age with mean BRG values of 17.6 ± 4.8, 15.0 ± 6.0, 10.6 ± 4.2, 10.3 ± 6.4 and 9.9 ± 8.5 ms/mmHg in patients <21, 21–40, 41–60, 61–80 and >80 years old, respectively; p < 0.001 (Fig. 1). The same trend was noted in tilt-positive and tilt-negative VVS patients (p < 0.001 and p = 0.002, respectively). Concomitant with the decrease in BRG, the prevalence of a vasodepressor response increased in older subjects (p < 0.001) (Fig. 2). Overall, the positivity rate for TTT was not different among the various age groups; however, the proportion of patients who had a positive TTT without NTG decreased with age (p = 0.008) (Fig. 3).
Mean baroreflex gain (BRG) measurements are shown as a function of age in all patients (open triangle), tilt-positive patients (filled circle) and tilt-negative patients (open circle). One-way ANOVA showed that age had a significant effect on BRG measurements. Mean BRG decreased with increasing age. This relationship was seen in all patients (p < 0.001) and in tilt-positive and tilt-negative patients (p < 0.001 and p = 0.002, respectively)
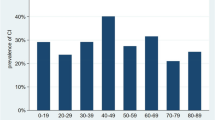
Tilt response according to the VASIS classification as a function of age. Each bar represents the overall percentage of patients who had a positive tilt table test (TTT) in the corresponding age group. Black, angled lines and white bars represent the percentages with a vasodepressor, mixed and cardio-inhibitory responses, respectively. We observed a significant relationship between age and type of tilt response with a greater prevalence of a vasodepressor response in older subjects (Chi-square test, p < 0.001)
Positivity rates (%) of tilt table testing (TTT) in the various age brackets. Each bar represents the overall percentage of patients who had a positive TTT in the corresponding age group. Black bars represent the percentage of patients who had a positive TTT without NTG and white bars represent the percentage of patients who had a positive TTT after NTG administration. We observed a significant relationship between age and test positivity rate with and without NTG. As age increased, the positivity rate without NTG administration decreased (Chi-square test, p = 0.008)
Hemodynamic response during the first 20 min of tilt table testing in patients with tilt-positive and tilt-negative VVS
When including all patients with positive TTT, BP during the first 20 min of TTT was similar in patients with tilt-positive VVS when compared to patients with tilt-negative VVS except for SBP at 10 min (p = 0.04). There were no significant differences in diastolic BP (p = 0.17) or mean BP (p = 0.08) at all measured time points (Fig. 4a). The HR response however, was significantly lower in tilt-positive VVS patients when compared with tilt-negative VVS patients at all measured time points (Fig. 4b).
a Systolic (SBP, open circle), diastolic (DBP, open triangle) and mean (MBP, open square) blood pressure responses during the first 20 min of tilt table testing (TTT) in tilt-positive (filled circle) and tilt-negative (open circle) VVS patients. Except for SBP at 10 min, there were no differences in BP between the groups. Asterisk indicates a significant difference in blood pressure between tilt-positive and tilt-negative groups. b Heart rate (HR) responses during the first 20 min of tilt table testing (TTT) in tilt-positive (filled circle) and tilt-negative (open circle) VVS patients. The HR was significantly lower in tilt-positive VVS patients when compared with tilt-negative VVS patients at all measured time points. Asterisk indicates a significant difference in HR between tilt-positive and tilt-negative groups
In the tilt-positive VVS group, 54 patients had a positive test during the first 20 min of TTT, i.e., did not require the administration of NTG. Theoretically, the decrease in HR associated with mixed or cardio-inhibitory responses in these patients might explain the above findings of decreased mean HR response in the tilt-positive VVS group. To investigate that possibility, we repeated the analysis after the exclusion of these patients. The above findings related to BP and HR responses remained unchanged.
Discussion
In the present study, we assessed the pathophysiologic mechanisms associated with hypotensive susceptibility in patients with the clinical diagnosis of VVS. Unlike previous studies but similar to the ISSUE-3 trial, we included only patients with the final diagnosis of VVS. We found that (1) patients with tilt-positive VVS have a BRG similar to patients with tilt-negative VVS; (2) aging was associated with a decrease in BRG and an increase in the prevalence of a vasodepressor response; and (3) the HR response during the first 20 min of tilt before NTG administration was lower in patients with tilt-positive VVS when compared with patients with tilt-negative VVS, with no significant changes in BP. Our results suggest that hypotensive susceptibility is not due to impairment of the baroreflex and that HR response during TTT might play a role in determining the outcome.
Pathophysiology of TTT-induced syncope
The normal physiologic response to orthostatic stress consists of a decrease in venous return and a reflex increase in HR and BP [13–15]. In patients who go on to develop near syncope or syncope, acute systemic vasodilatation triggered by forceful cardiac contractions is believed to be the underlying mechanism. This hypothesis has been supported by several studies showing reduction in cardiac volume, increase in fractional shortening [13–16], forearm vasodilatation and loss of sympathetic tone during syncope [17–22]. Increased vagal tone resulting in bradycardia and asystole plays a role, but is not always present during reflex syncope [23].
Recently, the above hypothesis has been challenged by studies showing that sympathetic withdrawal was not a pre-requisite for syncope [24–26]. Indeed, more than two decades ago, VVS was reported in heart transplant recipients and more recently several investigators made the observation that decreases in cardiac output contributed to the hypotension independent of reflex-mediated changes [25, 27, 28]. Fu et al. showed that up to one-third of subjects with tilt-induced presyncope had a severe drop in cardiac output with no change in total peripheral resistance. The decrease in cardiac output was thought to be due to a reduction in preload and stroke volume with or without associated bradycardia. The decrease in cardiac output hypothesis helps explain the significant incidence of positive TTT in patients with no history of reflex syncope, such as control subjects and in patients with clear arrhythmic etiology.
In patients with the clinical diagnosis of VVS, TTT is positive in the majority of patients but not all of them. Brignole and Sutton introduced the concept of “hypotensive susceptibility” as a possible explanation for the differences between patients with tilt-positive and tilt-negative VVS [9]. The authors suggested that patients with positive TTT have a greater vasodepressor response during spontaneous events when compared to patients with negative TTT. Therefore, even in the presence of spontaneous reflex-mediated asystole, these patients are less likely to benefit from pacing [8, 29]. This hypothesis was generated after learning from the ISSUE-3 trial that patients with positive TTT derived no benefit from cardiac pacing. The underlying mechanisms for the different responses between tilt-positive and tilt-negative VVS patients, however, remain poorly defined.
Present study
In the present study, the incidence of positive TTT was 75 %, consistent with previously published studies in patients with established or likely VVS (65–73 %) [3–5], but higher than that reported in the ISSUE-3 trial (56 %) [8]. When comparing BRG among the groups, we found no difference between the tilt-positive and tilt-negative VVS patients. Our results are in contrast with those from Shinohara et al. who found that baroreflex sensitivity indices were significantly lower in tilt-positive patients when compared with tilt-negative patients (6.1 ± 5.5 versus 9.8 ± 7.6 mmHg/s, p = 0.02) [30], but are aligned with the study by Klemenc et al. where baroreflex sensitivity was found to be similar in patients with tilt-positive and tilt-negative VVS [31]. The different results are best explained by the different patients’ characteristics including age, clinical presentation and time of BRG assessment. We have also found a relationship between age, BRG measurements and response to TTT. Previously, our group made a similar observation in all patients referred for tilt table test. The results of this study, which included only patients with VVS, reinforce our previous observation. It is generally accepted that the sympathetic component of the baroreflex remains intact with aging [32, 33], while the cardiovagal response might decrease [34, 35]. Therefore, our results of a decrease in BRG with age could be explained by either the presence of a blunted efferent cardiovagal response of the reflex arc or intrinsic sinus node disease [36]. Our findings of the relationship between age and type of tilt response are aligned with the work of Kurbaan et al. who showed that age was associated with a greater prevalence of a vasodepressor response in older patients (>65 years; OR 29.5, p < 0.0001) [37].
Our findings of no difference in BRG between tilt-positive and tilt-negative VVS support a lesser role of the baroreflex in determining the outcome during tilt in patients with VVS and favor a greater contribution of a drop in cardiac output in the development of hypotension. Indeed, the presence of a blunted HR response in subjects with positive TTT supports the hypothesis that tilt-induced hypotension is primarily due to a drop in cardiac output with the HR playing a role. It is important to note that a blunted HR response was still present after excluding patients who had a positive TTT during the first 20 min of TTT. Therefore, our findings are not due to the bradycardia present in patients with mixed or cardio-inhibitory responses during TTT.
There are several possible explanations for the blunted HR response. One possible mechanism is greater HR variability during TTT in patients with tilt-positive VVS when compared to patients with tilt-negative VVS, with progressive increases in variability until the occurrence of syncope. Theodorakis et al. had previously shown that HR variability defined as standard deviation (SD) of the mean RR interval and %SD was higher at the 10th minute of tilt testing in patients with VVS when compared with a control group [38]. Hausenloy et al. demonstrated that the presence of oscillations in BP during tilt table testing was a predictor for VVS with a sensitivity and specificity of 88 and 40 %, respectively [39]. Another possible explanation for the decreased mean HR is sinus node dysfunction. The mean age of tilt-positive VVS patients was significantly higher than the mean age of tilt-negative VVS supporting this hypothesis (50 ± 21 versus 42 ± 18, p < 0.01). Folino et al. evaluated the effect of age on tilt results in patients undergoing TTT for unexplained syncope [40]. The authors found that patients with ages 31–50 and >50 years had a lower increase in HR during the first 20 min of TTT when compared with patients younger than 30 years. In another study by Kurbaan et al., the authors made a similar observation that age was associated with blunted HR response (OR 1.04, p < 0.0002) [37]. Therefore, the observed blunted HR response might very well be related to age, although its role in determining the outcome remains unclear.
Limitations
This was a retrospective study with all the limitations associated with such analysis. The BRG measurement is an assessment of the HR response to fluctuations in BP, which is vagally mediated and may not correlate with the sympathetic response to orthostatic stress. Lastly, the tilt-positive VVS patients were significantly older than the tilt-negative VVS patients. While age might help explain the TTT results, mean BRG measurements were similar between the groups and all patients carried the diagnosis of VVS.
Conclusion
Our findings suggest that patients with VVS and positive TTT, i.e., hypotensive susceptibility, have no evidence of impairment of the baroreflex. An age-dependent decrease in BRG with a higher prevalence of a vasodepressor response was noted in older patients. While BP response was similar during the first 20 min of tilt testing, the HR response was blunted in tilt-positive VVS patients when compared with tilt-negative VVS patients. The clinical significance of the blunted HR response in tilt-positive VVS remains to be determined.
Abbreviations
- BP:
-
Blood pressure
- BRG:
-
Baroreflex gain
- DBP:
-
Diastolic blood pressure
- EF:
-
Ejection fraction
- HR:
-
Heart rate
- MBP:
-
Mean blood pressure
- SBP:
-
Systolic blood pressure
- TTT:
-
Tilt table test
- VVS:
-
Vasovagal syncope
References
Moya A, Sutton R, Ammirati F, Blanc JJ, Brignole M, Dahm JB, Deharo JC, Gajek J, Gjesdal K, Krahn A, Massin M, Pepi M, Pezawas T, Ruiz Granell R, Sarasin F, Ungar A, van Dijk JG, Walma EP, Wieling W, Task Force for the D, Management of S, European Society of C, European Heart Rhythm A, Heart Failure A, Heart Rhythm S (2009) Guidelines for the diagnosis and management of syncope (version 2009). Eur Heart J 30(21):2631–2671. doi:10.1093/eurheartj/ehp298
Kenny RA, Ingram A, Bayliss J, Sutton R (1986) Head-up tilt: a useful test for investigating unexplained syncope. Lancet 1(8494):1352–1355
Flevari P, Leftheriotis D, Komborozos C, Fountoulaki K, Dagres N, Theodorakis G, Kremastinos D (2009) Recurrent vasovagal syncope: comparison between clomipramine and nitroglycerin as drug challenges during head-up tilt testing. Eur Heart J 30(18):2249–2253. doi:10.1093/eurheartj/ehp255
Furukawa T, Maggi R, Solano A, Croci F, Brignole M (2011) Effect of clinical triggers on positive responses to tilt-table testing potentiated with nitroglycerin or clomipramine. Am J Cardiol 107(11):1693–1697. doi:10.1016/j.amjcard.2011.01.057
Petix NR, Del Rosso A, Furlan R, Guarnaccia V, Zipoli A (2014) Nitrate-potentiated head-up tilt testing (HUT) has a low diagnostic yield in patients with likely vasovagal syncope. Pacing Clin Electrophysiol 37(2):164–172. doi:10.1111/pace.12235
Raviele A, Menozzi C, Brignole M, Gasparini G, Alboni P, Musso G, Lolli G, Oddone D, Dinelli M, Mureddu R (1995) Value of head-up tilt testing potentiated with sublingual nitroglycerin to assess the origin of unexplained syncope. Am J Cardiol 76(4):267–272
Brignole M, Menozzi C, Moya A, Andresen D, Blanc JJ, Krahn AD, Wieling W, Beiras X, Deharo JC, Russo V, Tomaino M, Sutton R, International Study on Syncope of Uncertain Etiology I (2012) Pacemaker therapy in patients with neurally mediated syncope and documented asystole: Third International Study on Syncope of Uncertain Etiology (ISSUE-3): a randomized trial. Circulation 125(21):2566–2571. doi:10.1161/CIRCULATIONAHA.111.082313
Brignole M, Donateo P, Tomaino M, Massa R, Iori M, Beiras X, Moya A, Kus T, Deharo JC, Giuli S, Gentili A, Sutton R, International Study on Syncope of Uncertain Etiology I (2014) Benefit of pacemaker therapy in patients with presumed neurally mediated syncope and documented asystole is greater when tilt test is negative: an analysis from the third International Study on Syncope of Uncertain Etiology (ISSUE-3). Circ Arrhythm Electrophysiol 7(1):10–16. doi:10.1161/CIRCEP.113.001103
Sutton R, Brignole M (2014) Twenty-eight years of research permit reinterpretation of tilt-testing: hypotensive susceptibility rather than diagnosis. Eur Heart J 35(33):2211–2212. doi:10.1093/eurheartj/ehu255
Sheldon RS, Grubb BP 2nd, Olshansky B, Shen WK, Calkins H, Brignole M, Raj SR, Krahn AD, Morillo CA, Stewart JM, Sutton R, Sandroni P, Friday KJ, Hachul DT, Cohen MI, Lau DH, Mayuga KA, Moak JP, Sandhu RK, Kanjwal K (2015) 2015 heart rhythm society expert consensus statement on the diagnosis and treatment of postural tachycardia syndrome, inappropriate sinus tachycardia, and vasovagal syncope. Heart Rhythm 12(6):e41–e63. doi:10.1016/j.hrthm.2015.03.029
Brignole M, Menozzi C, Del Rosso A, Costa S, Gaggioli G, Bottoni N, Bartoli P, Sutton R (2000) New classification of haemodynamics of vasovagal syncope: beyond the VASIS classification. Analysis of the pre-syncopal phase of the tilt test without and with nitroglycerin challenge. Vasovagal Syncope International Study. Europace 2(1):66–76
Hughson RL, Quintin L, Annat G, Yamamoto Y, Gharib C (1993) Spontaneous baroreflex by sequence and power spectral methods in humans. Clin Physiol 13(6):663–676
Borst C, Wieling W, van Brederode JF, Hond A, de Rijk LG, Dunning AJ (1982) Mechanisms of initial heart rate response to postural change. Am J Physiol 243(5):H676–H681
Dambrink JH, Wieling W (1987) Circulatory response to postural change in healthy male subjects in relation to age. Clin Sci (Lond) 72(3):335–341
Petersen ME, Williams TR, Gordon C, Chamberlain-Webber R, Sutton R (2000) The normal response to prolonged passive head up tilt testing. Heart 84(5):509–514
Borst C, Meijler FL (1984) Baroreflex modulation of ventricular rhythm in atrial fibrillation. Eur Heart J 5(11):870–875
Barcroft H, Edholm OG (1945) On the vasodilatation in human skeletal muscle during post-haemorrhagic fainting. J Physiol 104(2):161–175
Epstein SE, Stampfer M, Beiser GD (1968) Role of the capacitance and resistance vessels in vasovagal syncope. Circulation 37(4):524–533
Morillo CA, Eckberg DL, Ellenbogen KA, Beightol LA, Hoag JB, Tahvanainen KU, Kuusela TA, Diedrich AM (1997) Vagal and sympathetic mechanisms in patients with orthostatic vasovagal syncope. Circulation 96(8):2509–2513
Mosqueda-Garcia R, Furlan R, Fernandez-Violante R, Desai T, Snell M, Jarai Z, Ananthram V, Robertson RM, Robertson D (1997) Sympathetic and baroreceptor reflex function in neurally mediated syncope evoked by tilt. J Clin Invest 99(11):2736–2744
Sharpey-Schafer EP (1956) Syncope. Br Med J 1(4965):506–509
Wallin BG, Sundlof G (1982) Sympathetic outflow to muscles during vasovagal syncope. J Auton Nerv Syst 6(3):287–291
Moya A, Brignole M, Menozzi C, Garcia-Civera R, Tognarini S, Mont L, Botto G, Giada F, Cornacchia D, International Study on Syncope of Uncertain Etiology I (2001) Mechanism of syncope in patients with isolated syncope and in patients with tilt-positive syncope. Circulation 104(11):1261–1267
Cooke WH, Rickards CA, Ryan KL, Kuusela TA, Convertino VA (2009) Muscle sympathetic nerve activity during intense lower body negative pressure to presyncope in humans. J Physiol 587(Pt 20):4987–4999. doi:10.1113/jphysiol.2009.177352
Fu Q, Verheyden B, Wieling W, Levine BD (2012) Cardiac output and sympathetic vasoconstrictor responses during upright tilt to presyncope in healthy humans. J Physiol 590(Pt 8):1839–1848. doi:10.1113/jphysiol.2011.224998
Vaddadi G, Esler MD, Dawood T, Lambert E (2010) Persistence of muscle sympathetic nerve activity during vasovagal syncope. Eur Heart J 31(16):2027–2033. doi:10.1093/eurheartj/ehq071
Jardine DL, Melton IC, Crozier IG, English S, Bennett SI, Frampton CM, Ikram H (2002) Decrease in cardiac output and muscle sympathetic activity during vasovagal syncope. Am J Physiol Heart Circ Physiol 282(5):H1804–H1809. doi:10.1152/ajpheart.00640.2001
Verheyden B, Liu J, van Dijk N, Westerhof BE, Reybrouck T, Aubert AE, Wieling W (2008) Steep fall in cardiac output is main determinant of hypotension during drug-free and nitroglycerine-induced orthostatic vasovagal syncope. Heart Rhythm 5(12):1695–1701. doi:10.1016/j.hrthm.2008.09.003
Brignole M, Ammirati F, Arabia F, Quartieri F, Tomaino M, Ungar A, Lunati M, Russo V, Del Rosso A, Gaggioli G, Syncope Unit Project Two I (2015) Assessment of a standardized algorithm for cardiac pacing in older patients affected by severe unpredictable reflex syncopes. Eur Heart J 36(24):1529–1535. doi:10.1093/eurheartj/ehv069
Shinohara T, Ebata Y, Ayabe R, Fukui A, Okada N, Yufu K, Nakagawa M, Takahashi N (2014) Cardiac autonomic dysfunction in patients with head-up tilt test-induced vasovagal syncope. Pacing Clin Electrophysiol 37(12):1694–1701. doi:10.1111/pace.12484
Klemenc M, Strumbelj E (2015) Predicting the outcome of head-up tilt test using heart rate variability and baroreflex sensitivity parameters in patients with vasovagal syncope. Clin Auton Res 25(6):391–398. doi:10.1007/s10286-015-0318-6
Ebert TJ, Morgan BJ, Barney JA, Denahan T, Smith JJ (1992) Effects of aging on baroreflex regulation of sympathetic activity in humans. Am J Physiol 263(3 Pt 2):H798–H803
Monahan KD (2007) Effect of aging on baroreflex function in humans. Am J Physiol Regul Integr Comp Physiol 293(1):R3–R12. doi:10.1152/ajpregu.00031.2007
Greene NM, Bachand RG (1971) Vagal component of the chronotropic response to baroreceptor stimulation in man. Am Heart J 82(1):22–27
Gribbin B, Pickering TG, Sleight P, Peto R (1971) Effect of age and high blood pressure on baroreflex sensitivity in man. Circ Res 29(4):424–431
Kardos A, Watterich G, de Menezes R, Csanady M, Casadei B, Rudas L (2001) Determinants of spontaneous baroreflex sensitivity in a healthy working population. Hypertension 37(3):911–916
Kurbaan AS, Bowker TJ, Wijesekera N, Franzen AC, Heaven D, Itty S, Sutton R (2003) Age and hemodynamic responses to tilt testing in those with syncope of unknown origin. J Am Coll Cardiol 41(6):1004–1007
Theodorakis GN, Kremastinos DT, Avrambos GT, Stefanakis GS, Karavolias GK, Toutouzas PK (1992) Heart rate variability in patients with vasovagal syndrome. Pacing Clin Electrophysiol 15(11 Pt 2):2221–2225
Hausenloy DJ, Arhi C, Chandra N, Franzen-McManus AC, Meyer A, Sutton R (2009) Blood pressure oscillations during tilt testing as a predictive marker of vasovagal syncope. Europace 11(12):1696–1701. doi:10.1093/europace/eup338
Folino AF, Migliore F, Marinelli A, Iliceto S, Buja G (2010) Age-related hemodynamic changes during vasovagal syncope. Auton Neurosci 156(1–2):131–137. doi:10.1016/j.autneu.2010.04.009
Acknowledgments
This work was supported in part by funds provided from the Mildred and Marv Conney Chair in Cardiology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Chaddha, A., Rafanelli, M., Brignole, M. et al. The pathophysiologic mechanisms associated with hypotensive susceptibility. Clin Auton Res 26, 261–268 (2016). https://doi.org/10.1007/s10286-016-0362-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10286-016-0362-x