Abstract
Even without diseases that cause dysphagia, physiological swallowing function declines with age, increasing the risk of aspiration. This study analyzed age-related changes in laryngeal movement in older adults. The study population consisted of 10 volunteers in their 80s and six in their 20s. A videofluoroscopic study of 3 and 10 mL barium swallows was performed laterally using a digital fluorographic. The recorded images were retrieved to a personal computer and analyzed frame-by-frame using video analysis software. The movement of the larynx during swallowing, barium's pharyngeal transit time (PTT), and laryngeal elevation delay time (LEDT) were analyzed. Results were compared between the 20s and 80s age groups using statistical analyses. The PTT was shorter in the 20s than in the 80s age group. The PTT was significantly longer in the 80s group than in the 20s for both 3 and 10 mL barium swallows. LEDT in the 80s was statistically significantly longer than that in the 20s for the 10 ml barium. No statistically significant differences were found; however, there was a tendency for the 80s group to have more types of laryngeal movement velocity peaks. In this study, LEDT was prolonged in the 80s with 10 ml barium swallowing than in the 20s. Two peak patterns of laryngeal elevation during swallowing were observed. The velocity peaks showed a two-peak pattern when the patients were in their 80s and when the barium volume was tested at 10 mL. Our results suggest that aging’s effect on swallowing relates to laryngeal elevation.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Dysphagia can be caused by various diseases, such as cerebrovascular disease, head and neck cancer, head trauma and neuromuscular disease [1]. Aging is also a factor in the deterioration of swallowing functions [2], and age-related changes in swallowing function have also been reported [3,4,5,6,7,8,9]. They include a delay in initiation of maximal hyolaryngeal excursion and prolongation of the total duration of oropharyngeal swallowing [3], insufficient pharyngeal contraction [4], the reduction of maximum vertical and anterior hyoid movements [5], high threshold amount of induction of pharyngeal swallowing [6], prolongation of apnea time during swallowing [7], increase in the frequency of multiple swallowing in single swallowing, and increase in the frequency of coughing during or after swallowing [8] or lower larynx [9].
The pharyngeal stage of swallowing begins when the food bolus reaches the level of the epiglottis valley and ends when the esophageal inlet closes [10]. In the pharyngeal phase, the larynx is pulled anteriorly and upward in accordance with the hyoid bone movement. This movement of the larynx from the penetration by the bolus expands the hypopharyngeal chamber, causing a decrease in pressure in the pharyngoesophageal segment [1, 11]. The larynx plays an important role in the defense of the trachea and the delivery of a bolus into the esophagus. It has been reported that laryngeal penetration increases in patients over 50 years of age, possibly due to the aging of the swallowing function [12]. Laryngeal proptosis is a physiological age-related change that increases laryngeal penetration and aspiration.
Therefore, the present study focused on the movement of the larynx. By observing the movements of the larynx in detail, it is possible to assess the decrease in swallowing function in older adults. The videofluoroscopic study of swallowing (VFSS) is considered the most appropriate examination for laryngeal movement. The results are typically reported qualitatively and quantitatively [13]. In this study, to investigate age-related changes in the larynx during swallowing, we compared the position and movement of the larynx during swallowing in younger and older adults using VFSS images.
Materials and methods
This study included six participants in their 20s (aged 24–27 years) and 10 in their 80s (aged 80–89 years) with no swallowing problems. There were four men and two women in their 20s and five men and five women in their 80s.
VFSS of swallowing 3 and 10 mL 10% barium liquid were examined using a digital fluorographic system (DIGTEX2400UX; Shimadzu, Kyoto, Japan) in the sitting upright position at 30 frames/s. The 3 and 10 ml volumes of barium to be swallowed were determined with reference to the method of Logemann [14]. Although the subjects had previously confirmed that they had no symptoms of dysphagia, VFSS was first performed with 3 ml of barium liquid to confirm that there was no aspiration. Then VFSS was performed with 10 ml of barium liquid. The reason for using 3 ml for a small amount and 10 ml for a large amount is that the small amount was set at 3 ml in case of aspiration, while the large amount of 10 ml was set as the amount that could be swallowed up in one swallow to avoid more than one swallow. The recorded images were retrieved on a personal computer and analyzed frame-by-frame using the video analysis software Videopoint (Lenox Softworks, Lee, MA, USA). Videopoint is a video analysis software package for Windows-based computers that allows the collection of position and time data from digital video as "Video Points." Data are collected by clicking on the location of the items of interest for each frame of a QuickTime movie. These points can be combined to form other calculations, such as center of mass locations and distances between points. The elevation of the assigned point of the larynx was traced, and the velocity was analyzed using the software.
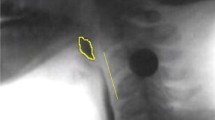
To investigate the movement of the larynx, the present study used the lower posterior point of the glottis as the larynx (Fig. 1). The position of the larynx at the start and end of pharyngeal transit of barium liquid is shown in Fig. 2. The start of pharyngeal transit is considered as the start of glottis closure, and the end of pharyngeal transit is considered as the end of laryngeal closure. Regarding the position of the larynx at the start of glottis closure and the end of laryngeal closure, the entire larynx was divided into three sections in the vertical direction, represented by the upper 1/3, middle 1/3, and lower 1/3.
Start (A) and end (B) of pharyngeal transit. A The point at which the bolus head passes through the lower edge of the mandible. B The point at which the bolus tail enters the esophagus. Green dotted circle: the glottis. Red area: bolus. Orange arrow: lower posterior point of the glottis. Blue line: lower edge of the mandible
Additionally, the peak velocity of laryngeal elevation during swallowing (PV, pixels/s) and pharyngeal transit time of barium bolus (PTT; s) [14], and laryngeal elevation delay time (LEDT; s) [15] were examined. LEDT was defined as the time from the arrival of the bolus head at the bottom of either side of the pyriform sinus until the first highest position of laryngeal elevation. The velocity of elevation of the larynx in the y-axis direction of the image was measured (Fig. 1), and the maximum value of the velocity was considered the peak velocity. All examined swallows were commanded.
Statistical analysis
The participants were divided into two groups: younger (20s, four men and two women) and older (80s, six men and four women) adults. The results were compared between the 20s and 80s groups using the Wilcoxon rank-sum test for numerical data and the Chi-square test, and Fisher’s exact test for categorical data. Statistical significance was set at P < 0.05. JMP Pro version 16.0 (SAS Institute, Cary, NC, USA) was used for all the statistical analyses.
Ethics approval and informed consent
All procedures were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and the Helsinki Declaration of 1975. Consideration was given to the ethical aspects of the participants, and a full explanation was given in advance, including possible disadvantages such as radiation exposure in VFSS, and written informed consent was obtained from all participants.
Results
The relationships between the laryngeal position and the start of the glottis or end of laryngeal closure are shown in Table 1. The position of the larynx at the start of the glottis closure was in the middle 1/3 for both 3 and 10 mL in the 20s group. In contrast, the position of the larynx at the start of the glottis closure was in the lower 1/3 for one participant in the 80s group at 3 mL and for two participants at 10 mL. No statistical difference in the level of the larynx at the start of the glottis closure was found between patients in their 20s and 80s. The position of the larynx at the end of laryngeal closure was the upper 1/3 in all participants in their 20s at 3 mL swallow, the upper 1/3 in nine participants in their 80s, and the middle 1/3 in one participant in their 80s. No statistically significant difference was found between those in their 20s and 80s. Although the position of the larynx at the end of laryngeal closure increased the number of middle 1/3 in both the 20s and 80s groups at the 10 mL swallow compared to the 3 ml swallow, there was no statistical difference between the 20s and 80s groups.
Regarding the velocity of laryngeal movement, either one or two peaks of peak velocity were observed (Fig. 3). In the 20s group, the two-peak type was observed only in the 10 mL swallow (3/6). In the 80s group, the two-peak swallow was observed at 3 (2/10) and 10 mL (4/10) (Table 2). Table 3 presents the samples' peak velocity, PTT values, and LEDT. The peak velocity was not statistically different between the 20s and 80s groups for both the 3 and 10 mL barium swallows. The PTT was significantly longer in the 80s group than that in the 20s group for both the 3 and 10 mL swallows. Regarding the LEDT, no statistically significant difference was found between the 20s and 80s groups for the 3 ml barium swallow. In contrast, the LEDT in the 80s group was statistically significantly longer than that in the 20s for the 10 ml barium (P = 0.0040).
Discussion
In this study, the pharyngeal stages of swallowing were compared between patients in their 20s and 80s using videofluoroscopic examinations. The PTT was longer in the 80s than that in the 20s group. The PTT is longer in older adults because muscular contractions of the tongue and pharynx become weaker with age, resulting in a reduced ability to squeeze in the bolus. In our study, the peak velocity of laryngeal movement tended to decrease in the 80s age group, suggesting oropharyngeal muscle weakness. Bolus transit time has been reported to examine oral transit time [16,17,18], pharyngeal transit time [16,17,18,19,20,21,22], and esophageal transit time [23, 24], whereas the definition of those terms depends on each study. PTT was used as bolus transit time in this study. Of the bolus pharyngeal transit times examined, PTT was reported to be prolonged in older adults [16,17,18, 20,21,22]. These results support our study. However, PTT has been considered susceptible to the oral phase, and some studies have examined transit times only in the hypopharynx [25]. The definition is critical with respect to the bolus transit time.
In the 10 ml barium swallow, LEDT in the 80s group was significantly longer than that in the 20s. The prolongation of the LEDT means a decrease in laryngeal elevation velocity or a delay in the timing of laryngeal elevation, suggesting a decrease in the contraction velocity of the laryngeal elevator muscles and decreased sensation of the pharynx [26]. Prolongation of LEDT has also been reported to increase the incidence of aspiration [26], and the relationship between LEDT and laryngeal movement will need further investigation.
Many studies have reported the relationship between the arrival of the bolus to the pharynx and the onset of the swallowing action [8, 16,17,18, 27,28,29,30]. Pharyngeal delay time (PDT) has been used to measure the delay of the pharyngeal phase. PDT is generally defined as the time from when the bolus head crosses the inferior margin of the mandible until the larynx begins to elevate. All studies report that older adults have prolonged PDT, although some have slightly different definitions of PDT. In our study, LEDT was used instead of PDT as a measure of the delay in swallow onset. Since the position of the larynx can easily change, the PDT is not defined to account for the rapid laryngeal elevation phase directly related to the swallow reflex, and the PDT is not appropriate for assessing the delayed pharyngeal phase [15]. As LEDT was defined to assess the delay of rapid laryngeal elevation relative to bolus inflow into the pharyngeal cavity, LEDT was used in this study to investigate the delay time of the pharyngeal phase. Since the present study aimed to investigate age-related changes in laryngeal elevation, the LEDT was chosen because it also considers laryngeal elevation.
Velocity peaks tended to show two-peak patterns in the 80s group and when the volume of barium was 10 mL, although no statistically significant differences were found. This is probably due to the longer distance of laryngeal elevation due to age-related laryngeal lowering; the 80s group may have performed a slight laryngeal movement to prepare for the elevation of the larynx before the swallowing reflex. In addition, when the bolus volume increased, the two-peak type appeared even in the 20s group, suggesting that they may be performing preparatory movements for laryngeal elevation depending on the bolus volume.
Leonard et al. reported that the pharynx is a larger, longer space that constricts to a lesser extent in older adults, which may mean that these individuals have to do more work than younger adults to achieve the same effects in terms of the displacement, constriction, and approximation gestures of swallowing [10]. The two-peak type of swallowing observed in our study could represent one of the various preparations made by older adults before swallowing.
With the global transition to an aging society, the age at which people are recognized as older adults is increasing, and research publications are increasingly focusing on people over the age of 85 [31]. Swallowing research will continue to be an important part of the study of swallowing in older adults in their 80s and older.
Limitations
This study had several limitations. First, this study has a small sample size. This makes it difficult to perform a comprehensive statistical analysis. And, because gender also affects swallowing, we also examined differences by gender; however, the number of cases was too small to obtain statistically reliable values for differences in swallowing by gender, so we could not include analyses by gender classification in this study. The number of participants should be increased; however, it is difficult to include a large number of participants because of the problem of radiation exposure in healthy participants and the risk of barium aspiration. Second, the movement of the posterior-inferior edge of the glottis was investigated to observe the movement of the larynx. Since the posterior-inferior edge of the glottis can be best visualized in the larynx, this position was set as the observation point; however, some images were not easily visible in some frames. This is also a limitation of the image quality of the VFSS. In this study, the image quality was limited because the detector of the digital fluoroscopic system uses an image intensifier. However, the detector design is now being shifted to a flat panel for enhanced image quality and decreased radiation dose [32]; as flat panel detectors provide better resolution, the quality of analysis of VF images is expected to improve in the future.
Conclusion
In this study, the PTT was longer in the 80s group than in the 20s group. The LEDT was prolonged in the 80s with 10 ml barium swallow than in the 20s. In addition, there are two types of laryngeal elevation during swallowing. Velocity peaks tended to show two-peak patterns in the 80s group and when the volume of barium was 10 mL. These interesting findings are expected to be useful for further research into age-related changes in swallowing.
Data availability
Not applicable.
References
Groher ME, Crary MA. Dysphagia: clinical management in adults and children. 1st ed. Missouri: Mosby; 2010.
Gleeson DC. Oropharyngeal swallowing and aging: a review. J Commun Disord. 1999;32:373–95 (quiz 395-6).
Robbins J, Hamilton JW, Lof GL, Kempster GB. Oropharyngeal swallowing in normal adults of different ages. Gastroenterology. 1992;103:823–9.
Nilsson H, Ekberg O, Olsson R, Hindfelt B. Quantitative aspects of swallowing in an elderly nondysphagic population. Dysphagia. 1996;11:180–4.
Leonard RJ, Kendall KA, McKenzie S, Gonçalves MI, Walker A. Structural displacements in normal swallowing: a videofluoroscopic study. Dysphagia. 2000;15:146–52.
Shaker R, Ren J, Zamir Z, Sarna A, Liu J, Sui Z. Effect of aging, position, and temperature on the threshold volume triggering pharyngeal swallows. Gastroenterology. 1994;107:396–402.
Hiss SG, Treole K, Stuart A. Effects of age, gender, bolus volume, and trial on swallowing apnea duration and swallow/respiratory phase relationships of normal adults. Dysphagia. 2001;16:128–35.
Logemann JA, Pauloski BR, Rademaker AW, Colangelo LA, Kahrilas PJ, Smith CH. Temporal and biomechanical characteristics of oropharyngeal swallow in younger and older men. J Speech Lang Hear Res. 2000;43:1264–74.
Furukawa K. Cineradiographic analysis of laryngeal movement during deglutition–with special reference to aging. Nihon Jibiinkoka Gakkai Kaiho. 1984;87:169–81.
Leonard R, Kendall KA, McKenzie S. Structural displacements affecting pharyngeal constriction in nondysphagic elderly and nonelderly adults. Dysphagia. 2004;19:133–41.
Leonard R, Kendall K. Dysphagia assessment and treatment planning: a team approach. 2nd ed. San Diego: Plural Publishing, Inc.; 2007. pp. 12–7.
Daggett A, Logemann J, Rademaker A, Pauloski B. Laryngeal penetration during deglutition in normal subjects of various ages. Dysphagia. 2006;21:270–4.
Konishi M, Yasuhara Y, Nagasaki T, Hossain A, Tanimoto K, Rohlin M. Differences of aspiration between liquid and solid foods in videofluoroscopic swallowing study: a review of literature. Int J Phys Med Rehabil. 2018;6:1–9.
Logemann JA. Evaluation and treatment of swallowing disorders. 2nd ed. Texas: PRO-ED Inc; 1998.
Miyaji H, Umezaki T, Adachi K, Sawatsubashi M, Kiyohara H, Inoguchi T, To S, Komune S. Videofluoroscopic assessment of pharyngeal stage delay reflects pathophysiology after brain infarction. Laryngoscope. 2012;122:2793–9.
Rademaker AW, Pauloski BR, Colangelo LA, Logemann JA. Age and volume effects on liquid swallowing function in normal women. J Speech Lang Hear Res. 1998;41:275–84.
Yoshikawa M, Yoshida M, Nagasaki T, Tanimoto K, Tsuga K, Akagawa Y, Komatsu T. Aspects of swallowing in healthy dentate elderly persons older than 80 years. J Gerontol A Biol Sci Med Sci. 2005;60:506–9.
McCullough GH, Rosenbek JC, Wertz RT, Suiter D, McCoy SC. Defining swallowing function by age: promises and pitfalls of pigeonholing. Top Geriatr Rehabil. 2007;23:290–307.
Dejaeger E, Pelemans W, Bibau G, Ponette E. Manofluorographic analysis of swallowing in the elderly. Dysphagia. 1994;9:156–61.
Yokoyama M, Mitomi N, Tetsuka K, Tayama N, Niimi S. Role of laryngeal movement and effect of aging on swallowing pressure in the pharynx and upper esophageal sphincter. Laryngoscope. 2000;110:434–9.
Im I, Kim Y, Oommen E, Kim H, Ko MH. The effects of bolus consistency in pharyngeal transit duration during normal swallowing. Ann Rehabil Med. 2012;36:220–5.
Nishikubo K, Mise K, Ameya M, Hirose K, Kobayashi T, Hyodo M. Quantitative evaluation of age-related alteration of swallowing function: Videofluoroscopic and manometric studies. Auris Nasus Larynx. 2015;42:134–8.
Miles A, Clark S, Jardine M, Allen J. Esophageal swallowing timing measures in healthy adults during videofluoroscopy. Ann Otol Rhinol Laryngol. 2016;125:764–9.
Jardine M, Miles A, Allen J. Dysphagia onset in older adults during unrelated hospital admission: quantitative videofluoroscopic measures. Geriatrics (Basel). 2018;3:66.
Takeda S, Saitoh E, Matsuo K, Baba M, Fujii W, Palmer JB. Influence of chewing on food transport and swallowing. Jpn J Rehabil Med. 2002;39:322–30.
Ameya M, Nishikubo K, Mise K, Motoyoshi K, Hyodo M. Dysphagia associated with advancing age. Jibi to Rinsho. 2006;52:S249–55.
Logemann JA, Pauloski BR, Rademaker AW, Kahrilas PJ. Oropharyngeal swallow in younger and older women: videofluoroscopic analysis. J Speech Lang Hear Res. 2002;45:434–45.
Kim Y, McCullough GH, Asp CW. Temporal measurements of pharyngeal swallowing in normal populations. Dysphagia. 2005;20:290–6.
Martin-Harris B, Brodsky MB, Michel Y, Lee FS, Walters B. Delayed initiation of the pharyngeal swallow: normal variability in adult swallows. J Speech Lang Hear Res. 2007;50:585–94.
Ayala KJ, Logemann JA. Effects of altered sensory bolus characteristics and repeated swallows in healthy young and elderly subjects. J Med Speech Lang Pathol. 2010;18:34–58.
Jardine M, Miles A, Allen J. A systematic review of physiological changes in swallowing in the oldest old. Dysphagia. 2020;35:509–32.
Weis M, Hagelstein C, Diehm T, Schoenberg SO, Neff KW. Comparison of image quality and radiation dose between an image-intensifier system and a newer-generation flat-panel detector system—technical phantom measurements and evaluation of clinical imaging in children. Pediatr Radiol. 2016;46:286–92.
Acknowledgements
The authors would like to express our gratitude to all the staff members in the Department of Oral and Maxillofacial Radiology, Hiroshima University Hospital.
Funding
None.
Author information
Authors and Affiliations
Contributions
MK contributed to the conceptualization, methodology, software, data curation, writing—original draft preparation, and reviewing and editing; TN contributed to the data curation and writing—reviewing and editing; NK contributed to the writing—reviewing and editing.
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts of interest to disclose.
Ethics approval and informed consent
All procedures were performed in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and the Helsinki Declaration of 1975. Written informed consent was obtained from all participants.
Clinical trial registration
Not applicable.
Animal statements
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Konishi, M., Nagasaki, T. & Kakimoto, N. Videofluoroscopic analysis of the laryngeal movement of older adults in swallowing. Odontology 112, 624–629 (2024). https://doi.org/10.1007/s10266-023-00852-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-023-00852-7