Abstract
Grouping species may provide some degree of simplification to understand the ecological function of plants on key ecosystem processes. We asked whether groups of plant species based on morpho-chemical traits associated with plant persistence and stress/disturbance resistance reflect dominant plant growth forms in arid ecosystems. We selected twelve sites across an aridity gradient in northern Patagonia. At each site, we identified modal size plants of each dominant species and assessed specific leaf area (SLA), plant height, seed mass, N and soluble phenol concentration in green and senesced leaves at each plant. Plant species were grouped according with plant growth forms (perennial grasses, evergreen shrubs and deciduous shrubs) and plant morphological and/or chemical traits using cluster analysis. We calculated mean values of each plant trait for each species group and plant growth form. Plant growth forms significantly differed among them in most of the morpho-chemical traits. Evergreen shrubs were tall plants with the highest seed mass and soluble phenols in leaves, deciduous shrubs were also tall plants with high SLA and the highest N in leaves, and perennial grasses were short plants with high SLA and low concentration of N and soluble phenols in leaves. Grouping species by the combination of morpho-chemical traits yielded 4 groups in which species from one growth form prevailed. These species groups differed in soluble phenol concentration in senesced leaves and plant height. These traits were highly correlated. We concluded that (1) plant height is a relevant synthetic variable, (2) growth forms adequately summarize ecological strategies of species in arid ecosystems, and (3) the inclusion of plant morphological and chemical traits related to defenses against environmental stresses and herbivory enhanced the potential of species grouping, particularly within shrubby growth forms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The main biomes of the world show a remarkable degree of convergence in physiognomy, despite of the great differences in the taxonomic composition of their floras (Crawley 1998; Moncrieff et al. 2015). This convergence of plant physiognomy reflects the importance of climatic factors as agents of natural selection (Crawley 1998) and facilitates the simplification of the diversity of plant species to a few, easily-defined categories based on attributes with meaningful ecological significance (Golodets et al. 2009). In the last decades, there has been an ongoing effort to identify plant functional groups based on attributes related to tolerance and avoidance to environmental stresses (e.g. Bertiller et al. 2006; de Bello et al. 2010; Díaz and Cabido 1997; Díaz et al. 2004; Körner 1994; Pérez- Harguindeguy et al. 2013; Vesk and Westoby 2001; Westoby 1998). Grouping species sharing similar structure and functioning would increase our ability to predict vegetation dynamics and ecosystem functioning under natural or man-induced environmental changes at a global scale (e.g. Díaz et al. 2004; Körner 1994; Rusch et al. 2003; Woodward and Cramer 1996). Several plant traits summarize the major dimensions of variations in plant ecological strategies and provide insights on the main opportunities and selective forces that shaped life history, architecture, resource allocation, and physiology of plants (Lambers et al. 2000; Navas et al. 2009; Westoby 1998; Westoby et al. 2002). Some of these traits converge across a wide range of environments and were successfully used to identify plant functional groups across broad environmental gradients (Cornwell et al. 2008; Díaz et al. 2004; Fortunel et al. 2009; Reich 2014). However, variation of meaningful functional traits potentially useful to group plants in narrow environmental gradients such as those characteristic of arid ecosystems were scarcely described.
In arid ecosystems, drought exerts a strong selective pressure on morpho-chemical traits and plant life histories (Aerts and Chapin 2000; Bertiller and Bisigato 1998; Díaz et al. 2004; Lambers et al. 2000; Niu et al. 2010; Noy Meir 1973; Vesk and Westoby 2001; Violle et al. 2007). In these environments, vegetation is dominated by shrubs and perennial grasses (Bertiller et al. 1991; Bertiller and Bisigato 1998; Sala et al. 1989; West 1983). These plant growth forms display strong morphological and functional differences (Bertiller et al. 1991; Carrera et al. 2003; Díaz et al. 2004; Lambers et al. 2000; Noy Meir 1973). Trait variation across aridity gradients may be related to species replacement and/or plant phenotypic plasticity having different effects on the patterns of relevant ecosystem processes such as primary production, litter decomposition, and nutrient release and cycling (Carrera et al. 2009; Golodets et al. 2009; Lambers et al. 2000; Moreno et al. 2010; Moreno and Bertiller 2015; Pazos et al. 2010; Weiher et al. 1999; Wright et al. 2004). Accordingly, the selection of a meaningful set of plant traits reflecting differential ecological functions of plants may increase our ability to predict vegetation functional responses to environmental changes across arid ecosystems (de Bello et al. 2010; Díaz and Cabido 1997; Díaz et al. 2004; Rusch et al. 2003; Woodward and Cramer 1996).
Plant grouping by growth form has been widely used in eco-physiological studies encompassing a wide range of environmental variation (e.g. Cornwell et al. 2008; Pérez- Harguindeguy et al. 2013, Shaver and Chapin 1980; Zhao et al. 2014). However, other studies showed a large overlapping in attributes related to C fixation and N conservation among plant growth forms (Aerts and Chapin 2000; Cornelissen 1999) mostly in narrow gradients of aridity (Bertiller et al. 2006; Moreno et al. 2010). Alternatively, classifications based on leading dimensions of variation of easily measurable traits summarizing fundamental plant trade-offs were proposed to assess plant functional groups (e.g. Westoby 1998). In this context, we explored whether plant grouping based on morpho-chemical traits related to the persistence, stress/disturbance resistance, and functioning (e.g. specific leaf area, plant height, seed mass, concentrations of N and soluble phenols in green and senesced leaves) is consistent with plant grouping by dominant growth forms (perennial grasses, evergreen shrubs, and deciduous shrubs) in constrained gradients of arid ecosystems.
Materials and methods
Study area
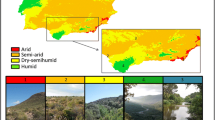
We selected twelve study sites (Table 1a) evenly separated across an area of 300 km lenght and 165 km width in northern Patagonia (Argentina), including the southern portion of the Monte Phytogeographical Province and the northern portion of the Central District of the Patagonian Phytogeographical Province (León et al. 1998). Vegetation corresponds to xeromorphic tall shrublands in the Patagonian Monte and to shrub or shrub-grass steppes dominated by medium shrubs in the Central District (Ares et al. 1990). Mean annual temperature ranges from 9.8 to 13.5 °C, the mean annual precipitation varies from 125 to 150 mm (Barros and Rivero 1982; Mitchell and Jones 2005), and the aridity index (AI) ranges from 4.5 to 7.3 (Table 1a) (Le Houérou 1990; Moreno et al. 2010). The study area has been grazed by sheep since the early 1900s (Ares et al. 1990) with a mean historical stocking rate (period 1960–1991) of 0.20 ± 0.02 sheep ha−1 (Chubut Province 1960–1970–1978–1991).
Sampling
Sampling was carried out in summer 2011–2012 and 2012–2013, corresponding to the late vegetative-early reproductive growth and seed dispersal periods of shrubs and perennial grasses (Bertiller et al. 1991; Campanella and Bertiller 2008). Vegetation sampling was done at physiognomically and floristically homogeneous stands representative of each site in an area of about 3 ha (minimal area sensu Mueller-Dombois and Ellenberg 1974). We recorded species richness and plant species cover (Mueller-Dombois and Ellenberg 1974) at each site. Cover was visually estimated using 1 % cover intervals (Bär Lamas et al. 2013; Bertiller and Ares 2008). At each site, we randomly selected between 9 and 14 adult plants of modal size (most frequent crown diameter and plant height) of each dominant perennial plant species (those species accounted for more than 75 % of the total cover of the site). Annual plant species were not included in the analysis since the abundance of this plant group is very low in all Patagonian ecosystems contributing with 0.1–3 % of the total plant cover (Bertiller and Carrera 2015; Golluscio and Sala 1993; Larreguy et al. 2014).
Selection of plant traits
We selected 7 plant traits (specific leaf area: SLA, plant height, seed mass, N concentration in green and senesced leaves, and soluble phenol concentration in green and senesced leaves) which are associated with the persistence and stress/disturbance resistance. These traits may influence the rates of ecosystem processes (Bertiller et al. 2006; Chapin et al. 2000; Díaz and Cabido 1997; Moreno et al. 2010; Pérez-Harguindeguy et al. 2013; Westoby 1998). The SLA represents a fundamental trade-off between resource acquisition and conservation (Moog et al. 2005; Westoby 1998). Moreover, this trait is related to the patterns of plant growth, carbon and nutrient investment in tissues, development of structural and chemical defenses, leaf life span, nutrient resorption rates, and litter chemistry (Aerts and Chapin 2000; Bertiller et al. 2006; Cornelissen 1999; Mediavilla and Escudero 2003; Moreno et al. 2010; Saraví Cisneros et al. 2013). The SLA could also be used as an indicator of structural chemical components such as lignin (Pérez-Harguindeguy et al. 2013). Plant height at maturity indicates a trade-off between the competitive ability for light and the construction cost of stem biomass, and it is associated with the ability to tolerate or avoid environmental stress (Pérez-Harguindeguy et al. 2013; Westoby 1998). The seed mass represents a trade-off between seed dispersal and seedling establishment ability (Murray et al. 2002). Nitrogen is one of the most limiting nutrients for plant growth (Lambers et al. 2000). The concentration of N in green leaves is associated with photosynthetic proteins (Lambers et al. 2000) while N in senesced leaves is strongly related to patterns of N conservation in plants (del Arco et al. 1991; Niinemets and Tamm 2005). In arid and semiarid ecosystems, plant species exhibit different mechanisms of N conservation (Aerts 1996; Aerts and Chapin 2000; Carrera et al. 2000; Lambers et al. 2000). In the Patagonian Monte, evergreen non-leguminous woody species show long-lived leaves with low N concentration and high concentration of secondary compounds in green leaves. These latter induce low N resorption from senescing leaves and consequently N concentration in senesced leaves is higher than that in non-woody species such as perennial grasses (Mazzarino et al. 1998; Mazzarino and Bertiller 1999; Carrera et al. 2000). Soluble phenols are the secondary metabolites most widely distributed in plants (Hättenschwiler and Vitousek 2000). The concentration of soluble phenols in green leaves is involved in the absorption of UV radiation, tolerance to water shortage, thermal protection, allelopathy, and defenses against herbivores or pathogens (Hättenschwiler and Vitousek 2000). The concentration of soluble phenols in senesced leaves may negatively affect soil microbial activity, and soil organic matter decomposition and mineralization (Aerts and Chapin 2000; Hartley and Jones 1998; Lambers et al. 2000). Moreover, Saraví Cisneros et al. (2013) provided evidence that the diversity and amount of phenolic compounds in green leaves of coexisting shrubs of an arid ecosystem of Patagonia was related to plant traits such as rooting depth, leaf turnover, and leaf N.
Plant sampling
We measured the height of each selected plant excluding the reproductive structures in the case to be present (Westoby 1998), and harvested three branches of the external canopy crown (with sunny and partially sunny leaves) from each dominant woody plant, and three tillers of each dominant perennial grass plant. The harvested material was air dried. Then, we randomly selected five totally expanded green leaves per plant, rehydrated them up to full expansion, placed each of them on the flat bed of an HP ScanJet ADF scanner and scanned them. Leaf area was assessed by analyzing the images with the AxioVision 4 software. After that, leaves were oven dried at 60 °C for 48 h and weighed to assess leaf dry mass (Bertiller et al. 2006). We further calculated the mean SLA in green leaves of each species at each site as the leaf area divided by the leaf mass. The rest of the totally green leaves without signs of deterioration (uniformity in coloration and absence of damage by herbivores or pathogens) was oven dried at 60 °C during 48 h. We also harvested senesced standing leaves (without deterioration signs) from 20 to 40 modal size plants of each dominant species at each site. Senesced leaves were subsequently pooled in a single sample per species and site. We assessed N concentration in green and senesced leaves by semi-micro Kjeldahl, and soluble phenol concentration by the Folin-Ciocalteu method using 50 % methanol as extract solution and tannic acid as standard (Waterman and Mole 1994). Additionally, we collected 10–20 seeds of at least three reproductive branches or tillers of each plant and site at the end of the reproductive season (January–February 2012 and 2013). Seeds were air dried to constant weight and weighed (Pérez-Harguindeguy et al. 2013; Westoby 1998).
Statistical analysis
Plant species were grouped according to (1) growth forms, (2) plant morphological traits (plant height, SLA, seed mass), (3) SLA (as indicator of structural chemical components such as lignin) and chemical traits (N and soluble phenol concentrations in green and senesced leaves), (4) morphological traits (plant height, SLA, seed mass) and chemical traits of green leaves (N and soluble phenol concentrations), and (5) morphological traits (plant height, SLA, seed mass) and chemical traits of senesced leaves (N and soluble phenol concentrations). Plant grouping (4) was aimed to identify plant species groups based on traits related to different key ecosystem processes such as plant growth and light capture while plant grouping (5) was attempted to identify species groups sharing attributes closely related to decomposition processes. Consistency among the three groupings would indicate the feasibility to expedite assess groups of plant species sharing traits related to main key processes. Moreover, consistency between groupings (4) and (5) would indicate that plant groups sharing traits related to decomposition processes could be addressed by grouping species accordingly to green leaf traits. Green leaf traits are easier to assess than those of senesced leaves without deterioration signs due to green leaves being more abundant than senesced ones in arid ecosystems. By grouping according to growth forms (1), we assigned each species to one of the following plant growth forms (Pérez-Harguindeguy et al. 2013): shrubs with green leaves during summer (evergreen shrubs during drought: ES), including evergreen tall and dwarf shrubs and a winter deciduous shrub; summer deciduous shrubs (deciduous shrubs during drought: DS); and perennial grasses (PG) (Table 1b). To perform the other groups (2–5), we used cluster analysis with Euclidean square distances and the un-weighted paired group method with arithmetic average (UPGMA) (Sneath and Sokal 1973). We selected this statistical analysis since it is widely used in ecological studies to identify plant functional groups based on single plant traits, independently of growth forms (e.g. Belbin and McDonald 1993; Casanoves et al. 2011; Leishman and Westoby 1992). Before the analyses, plant traits were standardized. We used the level of 10 % of the maximum distance to identify plant species groups at each dendrogram (2–5). After grouping, the mean values of the 7 plant traits of each group were calculated. The significance of differences in mean values of plant traits among plant groups within each grouping procedure (growth forms, and 4 groups of species resulting from cluster analyses) was assessed by one way ANOVA. Relationships between all traits were assessed by Spearman correlation analysis to identify those traits that summarize fundamental trade-offs related to ecological strategies independently of plant groupings. In those cases in which assumptions of ANOVA were not met, variables were logarithmic transformed (Sokal and Rohlf 1981). Least significant difference test (in the cases of homogeneity of variances) or Dunnett’s T3 test (in the case of non-homogeneous variances) were used for multiple comparisons (Norusis 1997).
Results
Plant groups based on plant growth forms
Growth forms differed in plant height (F 2,47 = 14.48, P < 0.01, Fig. 1a), specific leaf area (SLA) (F 2,47 = 8.25, P < 0.01, Fig. 1b), seed mass (F 2,47 = 3.96, P = 0.03, Fig. 1c), concentration of N and soluble phenols in green (F 2,47 = 11.86, P < 0.01, Fig. 1d; and F 2,47 = 19.74, P < 0.01, Fig. 1f, respectively) and senesced (F 2,47 = 16.92, P < 0.01, Fig. 1e; and F 2,47 = 17.07, P < 0.01, Fig. 1g, respectively) leaves. We found differences among the three growth forms in most of the traits, except for plant height and SLA, in which mean values were overlapped between evergreen shrubs (ES) and deciduous shrubs (DS), and DS and perennial grasses (PG), respectively (Fig. 1a, b). Despite this, each growth form was characterized by a particular combination of traits. ES were tall plants with the lowest SLA, the highest seed mass, the highest concentration of soluble phenols in green and senesced leaves and intermediate concentrations of N in leaves. DS were also tall plants with high SLA, the highest concentration of N in both green and senesced leaves and intermediate values of soluble phenols in leaves. PG were short plants with high SLA and low concentration of N and soluble phenols in green and senesced leaves.
Mean values of a plant height (cm), b specific leaf area (SLA, mm2 mg−1), c seed mass (mg seed−1), d N concentration in green leaves (mg g−1), e N concentration in senesced leaves (mg g−1), f soluble phenol concentration in green leaves (mg g−1), and g soluble phenol concentration in green leaves (mg g−1) by plant growth form. DS deciduous shrubs, ES evergreen shrubs, PG perennial grasses. Lowercase letters indicate significant differences among plant growth forms. Vertical lines indicate one standard error
Plant groups based on morphological traits
We identified three groups of species based on morphological traits (plant height, SLA and seed mass). The species Colliguaja integerrima (Ci), with the highest seed mass (216 mg seed−1), was not grouped. All groups clustered species from different plant growth forms (Fig. 2a) and they differed in plant height (F 2,46 = 52.09, P < 0.01, Fig. 2b), SLA (F 2,46 = 33.77, P < 0.01, Fig. 2c), seed mass (F 2,46 = 3.95, P = 0.03, Fig. 2d), concentrations of N in senesced leaves (F 2,46 = 4.66, P < 0.02, Fig. 2f), and soluble phenols in green and senesced leaves (F 2,46 = 11.36, P < 0.01, Fig. 2g; and F 2,46 = 14.99, P < 0.01, Fig. 2h, respectively). We found differences among the three groups only in the SLA, while mean values of plant height, seed mass, N concentration in senesced leaves and soluble phenol concentration in green and senesced leaves were overlapped between two groups. Moreover, we did not find significant differences in N concentration in green leaves among groups (F 2,46 = 2.52, P = 0.09, Fig. 2e). Despite this, each group was characterized by a particular combination of traits. Group 1 consisted of species of all growth forms, having low values of all traits except for seed mass (Fig. 2d). Group 2 consisted of evergreen and deciduous shrub species (relative contribution of 60 and 40 %, respectively) with high mean values of most traits, and intermediate values of SLA (Fig. 2c). Group 3 clustered one evergreen shrub, deciduous shrub and perennial grass species (relative contribution of 9, 18 and 73 %, respectively) with the highest value of SLA (Fig. 2c), and low values in the rest of the traits.
Plants groups based on morphological traits: plant height, specific leaf area (SLA), and seed mass. a Dendrogram, groups of plant species: 1, 2, 3. Acronyms of plant growth forms as in Fig. 1, aridity index (AI) in parentheses, and acronyms of plant species as in Table 1. Mean values of b plant height (cm), c SLA (mm2 mg−1), d seed mass (mg seed−1), e N concentration in green leaves (mg g−1), f N concentration in senesced leaves (mg g−1), g soluble phenol concentration in green leaves (mg g−1), h soluble phenol concentration in senesced leaves (mg g−1) by plant group 1, 2 and 3. Lowercase letters indicate significant differences among plant groups. Vertical lines indicate one standard error
Plant groups based on SLA and chemical traits in green and senesced leaves (N, soluble phenols)
We recognized four groups of species (Fig. 3a) and the species Prosopis alpataco (Pa) that had the highest mean values of N concentration in green and senesced leaves (36.29 and 27.06 mg g−1, respectively) was not clustered. Species groups showed significant differences in plant height (F 3,46 = 10.58, P < 0.01, Fig. 3a), SLA (F 3,46 = 12.85, P < 0.01, Fig. 3b), seed mass (F 3,46 = 5.42, P < 0.01, Fig. 3c), N concentration in green and senesced leaves (F 3,46 = 13.59, P < 0.01; F 3,46 = 24.14, P < 0.01; Fig. 3e and f), and soluble phenol concentration in green and senesced leaves (F 3,46 = 30.70, P < 0.01, F 3,46 = 68.39, P < 0.01; Fig. 3g, h). We found significant differences among three groups in the SLA, while one group exhibited intermediated values in this trait. Mean values of soluble phenols in senesced leaves were overlapped between two groups and mean values of the rest of the plant traits were overlapped in more than two groups. Despite this overlapping, species groups showed particular combinations of traits. Group 1 consisted of species of all growth forms (Fig. 3a) having low values in all traits (Fig. 3b–h). Group 2 clustered tall deciduous shrub species (Fig. 3b) having high N concentration in green and senesced leaves (Fig. 3e, f), intermediate values of SLA (Fig. 3c), low seed mass and low concentration of soluble phenols in green leaves (Fig. 3d, g). Group 3 gathered deciduous shrub and perennial grass species (relative contribution of 25 and 75 %, respectively, Fig. 3a) showing the highest SLA (Fig. 3c), intermediate concentration of N in green leaves and low values in the other traits. Group 4 clustered evergreen shrub species (Fig. 3a) and had the highest values of seed mass (Fig. 3d) and soluble phenol concentration in green and senesced leaves (Fig. 3g, h).
Plants groups based on chemical traits: concentrations of N and soluble phenols in green and senesced leaves, and specific leaf area (SLA). a Dendrogram, groups of plant species: 1, 2, 3, 4. Acronyms of plant growth forms as in Fig. 1, aridity index (AI) in parentheses, and acronyms of plant species as in Table 1. Mean values of b plant height (cm), c SLA (mm2 mg−1), d seed mass (mg seed−1), e N concentration in green leaves (mg g−1), f N concentration in senesced leaves (mg g−1), g soluble phenol concentration in green leaves (mg g−1), h soluble phenol concentration in senesced leaves (mg g−1) by group of species 1, 2, 3, and 4. Lowercase letters indicate significant differences among plant groups. Vertical lines indicate one standard error
Plant groups based on morphological traits and chemical traits of green leaves
We identified four groups of species based on morphological traits and chemical traits of green leaves (Fig. 4a), and three species were not grouped (Colliguaja integerrima: Ci, Prosopis alpataco: Pa, and Schinus johnstonii: Sj from the site with aridity index of 6.44 that had low height). Species groups differed in plant height (F 3,44 = 40.83, P < 0.01, Fig. 4b), SLA (F 3,44 = 16.49, P < 0.01, Fig. 4c), seed mass (F 3,44 = 4.36, P = 0.01, Fig. 4d), N concentration in green (F 3,44 = 10.82, P < 0.01, Fig. 4e) and senesced (F 3,44 = 24.19, P < 0.01, Fig. 4f) leaves, and soluble phenol concentration in green (F 3,44 = 48.03, P < 0.01, Fig. 4g) and senesced (F 3,44 = 77.65, P < 0.01, Fig. 4h) leaves. We found differences among the four groups in plant height and soluble phenols in senesced leaves traits. Mean values of SLA, N in senesced leaves, and soluble phenols in green leaves traits were overlapped between two groups while mean values of seed mass and N in green leaves were overlapped in more than two groups. Regardless of this overlapping, species groups differed among them in the combination of traits. Group 1 clustered evergreen shrub and grass species (relative contribution of 78 and 22 %, respectively, Fig. 4a) with low SLA and N concentration in green leaves (Fig. 4b, e). Group 2 gathered deciduous shrubs and one evergreen shrub (relative contribution of 89 and 11 %, respectively, Fig. 4a) with high N concentration in green and senesced leaves (Fig. 4e, f). Group 3 had only perennial grasses with the highest SLA (Fig. 4c), and low values in the other traits. Group 4 clustered evergreen shrubs and one deciduous shrub (relative contribution of 90 and 10 %, respectively, Fig. 4a) and exhibited high values in all traits.
Plants groups based on morphological and chemical traits in green leaves: plant height, specific leaf area (SLA), seed mass, concentrations of N and soluble phenols in green leaves. a Dendrogram, groups of plant species: 1, 2, 3, 4. Acronyms of plant growth forms as in Fig. 1, aridity index (AI) in parentheses, and acronyms of plant species as in Table 1. Mean values of b plant height (cm), c SLA (mm2 mg−1), d seed mass (mg seed−1), e N concentration in green leaves (mg g−1), f N concentration in senesced leaves (mg g−1), g soluble phenol concentration in green leaves (mg g−1), h soluble phenols concentration in senesced leaves (mg g−1) by group of species 1, 2, 3, and 4. Lowercase letters indicate significant differences among plant groups. Vertical lines indicate one standard error
Plant groups based on morphological traits and chemical traits of senesced leaves
We recognized four groups of species (Fig. 5a) and three species were not grouped (Colliguaja integerrima: Ci, Prosopis alpataco: Pa, and Schinus johnstonii: Sj from the site with aridity index of 6.44). Plant groups differed in plant height (F 3,44 = 46.89, P < 0.01, Fig. 5a), SLA (F 3,44 = 15.23, P < 0.01, Fig. 5b), seed mass (F 3,44 = 3.43, P = 0.03, Fig. 5c), N concentration in green and senesced leaves (F 3,44 = 8.96, P < 0.01; F 3,44 = 25.72, P < 0.01; Fig. 5e, f) and soluble phenol concentration in green and senesced leaves (F 3,44 = 22.38, P < 0.01, F 3,44 = 50.45, P < 0.01; Fig. 5g, h). We found differences among the four groups in plant height and soluble phenols in senesced leaves traits. The mean values of SLA, N in senesced leaves, and soluble phenols in green leaves traits were overlapped between two groups while mean values of seed mass and N in green leaves traits were overlapped in more than two groups. Regardless of this overlapping, each species group was characterized by a particular combination of traits. Group 1 clustered evergreen shrub and perennial grass species (relative contribution of 67 and 33 %, respectively, Fig. 5a) with low SLA (Fig. 5c) and N concentration in green leaves (Fig. 5e). Group 2 gathered evergreen and deciduous shrub species (relative contribution of 18 and 82 %, respectively, Fig. 5a) and had high N concentration in green and senesced leaves (Fig. 5e, f). Group 3 included perennial grass species (Fig. 5a) with the highest SLA (Fig. 5c) and the lowest values in the rest of the traits. Group 4 clustered tall evergreen shrub species (Fig. 5b) with the highest soluble phenol concentration in green and senesced leaves (Fig. 5g, h).
Plants groups based on morphological and chemical trait in senesced leaves chemical traits: plant height, specific leaf area (SLA), seed mass, concentrations of N and soluble phenols in senesced leaves. a Dendrogram, groups of plant species: 1, 2, 3, 4. Acronyms of plant growth forms as in Fig. 1, aridity index (AI) in parentheses, and acronyms of plant species as in Table 1. Mean values of b plant height (cm), c SLA (mm2 mg−1), d seed mass (mg seed−1), e N concentration in green leaves (mg g−1), f N concentration in senesced leaves (mg g−1), g soluble phenol concentration in green leaves (mg g−1), h soluble phenol concentration in senesced leaves (mg g−1) by group of species 1, 2, 3, and 4. Lowercase letters indicate significant differences among plant groups. Vertical lines indicate one standard error
Relationships among morpho-chemical traits and the aridity index
Plant height was positively related to all morpho-chemical traits except for SLA. Seed mass was negatively related to SLA and positively associated with N concentration in senesced leaves and soluble phenols in both green and senesced leaves. Both N and soluble phenols in green and senesced leaves were positively correlated between them (Table 2). All morpho-chemical traits did not significantly vary with the aridity index (Table 2).
Discussion
Grouping species might provide some degree of simplification to scaling up from species to vegetation, to understand the ecological function of plants on key ecosystem processes, and to predict how vegetation and ecosystems respond to natural or man-induced disturbances (Cornwell et al. 2008; de Bello et al. 2010; Díaz and Cabido 1997; Körner 1994; Pietsch et al. 2014). In doing this, the selection of traits reflecting ecological strategies closely linked to ecosystem functioning (i.e. soil organic matter turnover and nutrient cycling) is quite relevant (Fry et al. 2013; Hillebrand and Matthiessen 2009).
Our results showed that grouping plants by growth forms (perennial grasses, evergreen shrubs and deciduous shrubs) yielded homogeneous species groups in ecological meaningful traits related to persistence, stress/disturbance resistance and ecosystem functioning. Plant growth forms differed in seed mass and in all chemical traits showing convergence in the pattern of traits associated with plant lifespan, opportunistic behavior, defenses against herbivory, and tolerance or avoidance to environmental stress in constrained environmental gradients in arid ecosystems (Briske 1996; Díaz et al. 2004; Grassein et al. 2010; Moreno and Bertiller 2015; Pazos et al. 2010; Westoby 1998; Wright et al. 2004). Thus, high SLA and N concentration in green leaves of deciduous shrubs could be associated with short-lived leaves which are produced during a relative short period in the wet season (Campanella and Bertiller 2008; Chesson et al. 2004). In contrast, evergreen shrubs had long lasting leaves with high concentration and diversity of secondary compounds protecting them against environmental stress and herbivory (Campanella and Bertiller 2008; Díaz et al. 2004; Hartley and Jones 1998; Moreno et al. 2010; Saraví Cisneros et al. 2013; Zunzunegui et al. 2005). In the case of perennial grasses, high SLA and low soluble phenol concentration in leaves could be associated with opportunistic behavior and high phenological plasticity (Couso and Fernández 2012; Tieszen and Archer 1990). Furthermore, differences in seed mass could be related to higher reproductive allocation (as numerous small seeds) in short-lived perennial grasses compared to long-lived shrubs (Crawley 1998; Westoby et al. 1999; Wright et al. 2004).
However, groups resulting from those groupings including either morphological or chemical traits showed scarce differences among them mainly in those plant traits not included in the respective cluster analysis. In contrast, plant groupings including both morphological and chemical traits yielded species groups internally consistent in relation to relevant functional traits such as soluble phenols in senesced leaves and plant height (Pérez-Harguindeguy et al. 2013; Saraví Cisneros et al. 2013; Westoby 1998). Moreover, these groups were similar to those identified by growth forms and further discriminated subgroups of evergreen shrubs and perennial grasses. Within evergreen shrubs, we identified two subgroups showing different combinations of plant traits (a subgroup represented by short plants with low SLA and low or intermediate concentrations of N and soluble phenols in leaves and other subgroup consisting of tall plants with intermediate values of SLA, and high concentrations of N and soluble phenols in leaves). Both subgroups could indicate different adaptive advantages to natural and anthropic disturbances and environmental stresses (Wright and Westoby 2003; Moreno et al. 2010; Saraví Cisneros et al. 2013). Plant height at maturity indicates a trade-off between the competitive ability for light and the construction cost of stem biomass and is usually associated with the ability to avoid or tolerate environmental stress (Pérez-Harguindeguy et al. 2013; Westoby 1998). On the other hand, long lasting leaves with low SLA and large accumulations of secondary metabolites (e.g. soluble phenols) not only increase N-residence time in the plant but also enhance the tolerance to water shortage, and resistance to solar radiation and herbivores (Grace 1998; Hartley and Jones 1998; Lambers et al. 2000). This reinforces the evidence that evergreen shrubs are a structurally and chemically heterogeneous plant group as previously reported in relation to the amount and diversity of chemical and structural defenses against drought and herbivory (Moreno et al. 2010; Saraví Cisneros et al. 2013). Moreover, plant height was highly correlated to almost all the other morpho-chemical traits considered in this study. This finding highlighted that plant height is a trait that summarizes fundamental trade-offs associated with the ability to tolerate or avoid environmental stress and grazing disturbance (Briske 1996; Jardim and Batalha 2008; Moles et al. 2009; Westoby 1998). Unlike shrubs, perennial grass species only showed heterogeneity in relation to plant height. However, plant height is a plastic trait in several perennial grass species of arid ecosystem (Couso and Fernández 2012) mostly in relation to the proximity to shrub canopies (i.e. plants growing underlying or in the periphery of shrub canopies vs. those occupying open areas) (Moreno and Bertiller 2015). These findings emphasize that perennial grasses are mostly morphologically and functionally more homogeneous than shrubs (Carrera et al. 2003; 2009; Díaz et al. 2004; Lambers et al. 2000; Moreno et al. 2010).
Additionally, grouping plants by morpho-chemical traits failed to include some species (Colliguaja integerrima, Prosopis alpataco, and Schinus johnstonii) with extreme values in particular traits. Moreover, in some cases, groupings based on morphological and/or chemical traits clustered the same species in different groups, depending on the growing site (i.e. the deciduous shrub Lycium chilense, the evergreen shrubs Atriplex lampa, Schinus johnstonii and Larrea divaricata, and the perennial grasses Jarava neaei, Poa ligularis, Pappostipa humilis and Nassella tenuis). These results could be related to adaptations or plastic responses to differences in water shortage among sites. We discarded differences due to domestic grazing pressure among sites based on the similar grazing pressure across the aridity gradient and studies reporting no changes in mean values of some plant traits due to herbivory in arid ecosystems (Adler et al. 2005; Bär Lamas et al. 2013; Lázaro-Nogal et al. 2013; Milchunas et al. 1998; Moreno et al. 2010).
We concluded that growth forms adequately summarize ecological strategies of species across the aridity gradient and that the inclusion of plant morphological and chemical traits related to defenses against water shortage and herbivory may clearly enhance the power of species grouping particularly within shrubby growth forms. This result highlighted the importance of using additional sets of traits to those used to identify growth forms when the objective is grouping species with different ecological function in relation to relevant ecosystem processes in narrow aridity gradients. Furthermore, our findings indicated that plant height is a key trait that could be used as an expedite indicator of plant ecological function in arid ecosystems.
References
Adler PB, Milchunas DG, Sala OE, Burke IC, Lauenroth WK (2005) Plant traits and ecosystem grazing effects: comparison of US Sagebrush steppe and Patagonian steppe. Ecol Appl 15:774–792
Aerts R (1996) Nutrient resorption from senescing leaves of perennials: are there general patterns? J Ecol 84:597–608
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67
Ares JO, Beeskow AM, Bertiller MB et al (1990) Structural and dynamic characteristics of overgrazed lands of northern Patagonia, Argentina. In: Breymeyer A (ed) Managed grasslands. Elsevier, The Netherlands, pp 149–175
Bär Lamas MI, Larreguy C, Carrera AL, Bertiller MB (2013) Changes in plant cover and functional traits induced by grazing in the arid Patagonian Monte. Acta Oecol 51:66–73
Barros V, Rivero M (1982) Mapas de probabilidad de precipitación de la Provincia del Chubut. Monografía 54. Centro Nacional Patagónico. Puerto Madryn, Chubut, AR
Belbin L, McDonald C (1993) Comparing three classification strategies for use in ecology. J Veg Sci 4:341–348
Bertiller MB, Ares JO (2008) Sheep spatial grazing strategies at the arid Patagonian Monte, Argentina. Range Ecol Manage 61:38–47
Bertiller MB, Bisigato A (1998) Vegetation dynamics under grazing disturbance. The state-and-transition model for the Patagonian steppes. Ecol Aust 8:191–199
Bertiller MB, Carrera AL (2015) Aboveground vegetation and perennial grass seed bank in arid rangelands disturbed by grazing. Range Ecol Manage 68:71–78
Bertiller MB, Beeskow AM, Coronato FR (1991) Seasonal environmental variation and plant phenology in arid Patagonia (Argentina). J Arid Environ 21:1–11
Bertiller MB, Mazzarino MJ, Carrera AL et al (2006) Leaf strategies and soil N across a regional humidity gradient in Patagonia. Oecologia 148:612–624
Briske DD (1996) Strategies of plant survival in grazed systems: a functional interpretation. In: Hodgson J, Illius AW (eds) The ecology and management of grazed systems. CAB International, Wallingford, UK
Campanella MV, Bertiller MB (2008) Plant phenology, leaf traits, and leaf litterfall of contrasting life forms in arid Patagonian Monte, Argentina. J Veg Sci 19:75–85
Carrera AL, Sain CL, Bertiller MB (2000) Patterns of nitrogen conservation in shrubs and grasses in the Patagonian Monte, Argentina. Plant Soil 224:185–193
Carrera AL, Bertiller MB, Sain CL, Mazzarino MJ (2003) Relationship between plant nitrogen conservation strategies and the dynamics of soil nitrogen in the arid Patagonian Monte, Argentina. Plant Soil 255:595–604
Carrera AL, Mazzarino MJ, Bertiller MB, del Valle HF, Carretero EM (2009) Plant impacts on nitrogen and carbon cycling in the Monte Phytogeographical Province, Argentina. J Arid Environ 73:192–201
Casanoves F, Pla L, Di Rienzo JA (eds) (2011) Valoración y análisis de la diversidad funcional y su relación con los servicios ecosistémicos. Centro Agronómico Tropical de Investigación y Enseñanza, CATIE Turrialba, Costa Rica
Chapin FS, Zavaleta ES, Eviner VT et al (2000) Consequences of changing biodiversity. Nature 405:234–242
Chesson P, Gebauer RL, Schwinning S et al (2004) Resource pulses, species interactions, and diversity maintenance in arid and semi-arid environments. Oecologia 141:236–253
Chubut Province (1960–1970–1978–1991) Statistics of the Chubut Province. http://www.chubut.gov.ar
Cornelissen JHC (1999) A triangular relationship between leaf size and seed size among woody species: allometry, ontogeny, ecology and taxonomy. Oecologia 118:248–255
Cornwell WK, Cornelissen JH, Amatangelo K et al (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071
Couso LL, Fernández RJ (2012) Phenotypic plasticity as an index of drought tolerance in three Patagonian steppe grasses. Ann Bot 110:849–857
Crawley MJ (ed) (1998) Plant ecology. Blackwell, Cambridge
de Bello F, Lavergne S, Meynard CN, Lepš J, Thuiller W (2010) The partitioning of diversity: showing Theseus a way out of the labyrinth. J Veg Sci 21:92–1000
del Arco JM, Escudero A, Garrido MV (1991) Effects of site characteristics on nitrogen retranslocation from senescing leaves. Ecol 72:701–708
Díaz S, Cabido M (1997) Plant functional types and ecosystem function in relation to global change. J Veg Sci 8:463–474
Díaz S, Hodgson JG, Thompson K et al (2004) The plant traits that drive ecosystems: evidence from three continents. J Veg Sci 15:295–304
Fortunel C, Garnier E, Joffre R et al (2009) Leaf traits capture the effects of land use changes and climate on litter decomposability of grasslands across Europe. Ecol 90:598–611
Fry EL, Power SA, Manning P (2013) Trait-based classification and manipulation of plant functional groups for biodiversity–ecosystem function experiments. J Veg Sci 25:248–261
Golluscio RA, Sala OE (1993) Plant functional types and ecological strategies in Patagonian forbs. J Veg Sci 4:839–846
Golodets C, Sternberg M, Kigel J (2009) A community-level test of the leaf-height-seed ecology strategy scheme in relation to grazing conditions. J Veg Sci 20:392–402
Grace J (1998) Plant water relation. In: Crawley MJ (ed) Plant ecology. Blackwell Science, Oxford, pp 284–324
Grassein F, Till-Bottraud I, Lavorel S (2010) Plant resource-use strategies: the importance of phenotypic plasticity in response to a productivity gradient for two subalpine species. Ann Bot 106:637–645
Hartley SE, Jones CG (1998) Plant chemistry and herbivory, or why the world is green. In: Crawley MJ (ed) Plant ecology. Blackwell Science, Oxford, pp 284–324
Hättenschwiler S, Vitousek PM (2000) The role of polyphenols in terrestrial ecosystem nutrient cycling. TREE 15:238–243
Hillebrand H, Matthiessen B (2009) Biodiversity in a complex world: consolidation and progress in functional biodiversity research. Ecol Lett 12:1405–1419
Jardim AVF, Batalha MA (2008) Can we predict dispersal guilds based on the leaf-height-seed scheme in a disjunct cerrado woodland? Braz J Biol 68:553–559
Körner C (1994) Scaling from species to vegetation: the usefulness of functional groups. In: Schulze ED, Mooney HA (eds) Biodiversity and ecosystem function. Springer, Berlin, pp 117–139
Lambers H, Chapin FS, Pons T (eds) (2000) Plant physiological ecology. Springer-Verlag, New York
Larreguy C, Carrera AL, Bertiller MB (2014) Effects of long-term grazing disturbance on the belowground storage of organic carbon in the Patagonian Monte, Argentina. J Environ Manage 134:47–55
Lázaro-Nogal A, Forner A, Traveset A, Valladares F (2013) Contrasting water strategies of two Mediterranean shrubs of limited distribution: uncertain future under a drier climate. Tree Physiol 33:1284–1295
Le Houérou H (1990) Bioclimatologie comparative des Zones Arides s.l. de l’Afrique et l’Amerique Latine. Terra Arida 7:26–55
Leishman MR, Westoby M (1992) Classifying plants into groups on the basis of associations of individual traits-Evidence from Australian semi-arid woodlands. J Ecol 80:417–424
León RJC, Bran D, Collantes M, Paruelo JM, Soriano A (1998) Grandes unidades de vegetación de la Patagonia extra andina. Ecol Aust 8:125–144
Mazzarino MJ, Bertiller MB (1999) Soil N pools and processes as indicators of desertification in semi-arid woodlands and semi-arid steppes of Argentina. In: Proceedings of the VV International Rangeland Congress. Townsville, Australia, pp 101–105
Mazzarino MJ, Bertiller M, Schlichter T, Gobbi M (1998) Nutrient cycling in Patagonian ecosystems. Ecol Aust 8:167–181
Mediavilla S, Escudero A (2003) Relative growth rate of leaf biomass and leaf nitrogen content in several Mediterranean woody species. Plant Ecol 168:321–332
Milchunas DG, Lauenroth WK, Burke IC (1998) Livestock grazing: animal and plant biodiversity of shortgrass steppe and the relationship to ecosystem function. Oikos 83:65–74
Mitchell TD, Jones PD (2005) An improved method of constructing a database of monthly climate observations and associated high-resolution grids. Int J Climatol 25:693–712
Moles AT, Warton DI, Warman L et al (2009) Global patterns in plant height. J Ecol 97:923–932
Moncrieff GR, Hickler T, Higgins SI (2015) Intercontinental divergence in the climate envelope of major plant biomes. Global Ecol Biogeogr 24:324–334
Moog D, Kahmen S, Poschlod P (2005) Application of CSR and LHS strategies for the distinction of differently managed grasslands. Basic Appl Ecol 6:133–143
Moreno L, Bertiller MB (2015) Phenotypic plasticity of morpho-chemical traits of perennial grasses from contrasting environments of arid Patagonia. J Arid Environ 116:96–102
Moreno L, Bertiller MB, Carrera AL (2010) Changes in traits of shrub canopies across an aridity gradient in northern Patagonia, Argentina. Basic Appl Ecol 11:693–701
Mueller-Dombois D, Ellenberg H (1974) Aims and Methods of Vegetation Ecology. Wiley, New York
Murray BR, Thrall PH, Gill AM, Nicotra AB (2002) How plant life-history and ecological traits relate to species rarity and commonness at varying spatial scales. Aust Ecol 27:291–310
Navas ML, Roumet C, Bellmann A, Laurent G, Garnier E (2009) Suites of plant traits in species from different stages of a Mediterranean secondary succession. Plant Biol 12:183–196
Niinemets Ü, Tamm Ü (2005) Species differences in timing of leaf fall and foliage chemistry modify nutrient resorption efficiency in deciduous temperate forest stands. Tree Physiol 25:1001–1014
Niu K, Zhang S, Zhao B, Du G (2010) Linking grazing response of species abundance to functional traits in the Tibetan alpine meadow. Plant Soil 330:215–223
Norusis MJ (1997) SPSS advanced statistics 7.5. SPSS. Inc., Chicago
Noy Meir I (1973) Desert ecosystems: environment and producers. Ann Rev Ecol Sys 4:25–52
Pazos GE, Ares JO, Bertiller MB (2010) Quantitative assessment of shrub–grass mosaic development in grazed shrublands: an example in the Patagonian Monte (Argentina). J Arid Environ 74:998–1002
Pérez- Harguindeguy N, Díaz S, Garnier E et al (2013) New handbook for standardised measurement of plant functional traits worldwide. Aust J Bot 61:167–234
Pietsch KA, Ogle K, Cornelissen JH et al (2014) Global relationship of wood and leaf litter decomposability: the role of functional traits within and across plant organs. Glob Ecol Biogeogr 23:1046–1057
Reich PB (2014) The worldwide ‘fast–slow’ plant economics spectrum: a traits manifesto. J Ecol 102:275–301
Rusch GM, Pausas JG, Lepš J (2003) Plant functional types in relation to disturbance and land use: introduction. J Veg Sci 14:307–310
Sala OE, Golluscio RA, Lauenroth WK, Soriano A (1989) Resource partitioning between shrubs and grasses in the Patagonian steppe. Oecologia 81:501–505
Saraví Cisneros H, Bertiller MB, Carrera AL, Larreguy C (2013) Diversity of phenolic compounds and plant traits in coexisting Patagonian desert shrub species of Argentina. Plant Ecol 11:1335–1343
Shaver GR, Chapin FS (1980) Response to fertilization by various plant growth forms in an Alaskan tundra: nutrient accumulation and growth. Ecol 61:662–675
Sneath P, Sokal R (1973) Numerical taxonomy: the principles and practice of numerical classification. SW Freeman, San Francisco
Sokal RR, Rohlf FJ (1981) Biometry. Freeman, San Francisco
Tieszen LL, Archer S (1990) Isotopic assessment of vegetation changes in grassland and woodland systems. In: Osmond CB, Pitelka LF, Hidy GM (eds) Plant biology of the basin and range. Springer-Verlag, New York, pp 293–321
Vesk PA, Westoby M (2001) Predicting plant species’ responses to grazing. J Appl Ecol 38:897–909
Violle C, Navas ML, Vile D et al (2007) Let the concept of trait be functional. Oikos 116:882–892
Waterman PG, Mole S (1994) Analysis of phenolic plant metabolites. Blackwell Scientific, Oxford
Weiher E, van der Werf A, Thompson K et al (1999) Challenging Theophrastus: a common core list of plant traits for functional ecology. J Veg Sci 10:609–620
West NE (ed) (1983) Temperate deserts and semi-deserts. Ecosystems of the world 5. Amsterdam: Elsevier
Westoby M (1998) A leaf-height-seed (LHS) plant ecology strategy scheme. Plant Soil 199:213–227
Westoby M, Eldridge D, Freudenberger D (1999) The LHS strategy scheme in relation to grazing and fire. Proc Int Range Cong 2:893–896
Westoby M, Falster DS, Moles AT, Vesk PA, Wright IJ (2002) Plant ecological strategies: some leading dimensions of variation between species. Ann Rev Ecol Sys 33:125–159
Woodward FI, Cramer W (1996) Plant functional types and climatic changes: introduction. J Veg Sci 7:306–308
Wright IJ, Westoby M (2003) Nutrient concentration, resorption and lifespan: leaf traits of Australian sclerophyll species. Funct Ecol 17:10–19
Wright IJ, Reich PB, Cornelissen JHC et al (2004) Assessing the generality of global leaf trait relationships. New Phytol 166:485–496
Zhao N, He N, Wang Q et al (2014) The altitudinal patterns of leaf C:N: P stoichiometry are regulated by plant growth form, climate and soil on Changbai mountain, China. PLoS One 9:e95196. doi:10.1371/journal.pone.0095196
Zunzunegui M, Barradas MD, Ain-Lhout F, Clavijo A, Novo FG (2005) To live or to survive in Doñana dunes: adaptive responses of woody species under a Mediterranean climate. Plant Soil 273:77–89
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding information
This work was supported by the National Agency for Scientific, Technological Promotion (PICTs 1349, 1368) and the National Research Council of Argentina (CONICET PIPs-112-200801-01664 and 112-201301-00449). M. I. Bär Lamas fellowship is supported by CONICET.
Rights and permissions
About this article
Cite this article
Bär Lamas, M.I., Carrera, A.L. & Bertiller, M.B. Meaningful traits for grouping plant species across arid ecosystems. J Plant Res 129, 449–461 (2016). https://doi.org/10.1007/s10265-016-0803-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-016-0803-6