Abstract
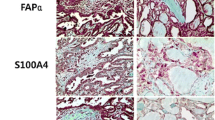
Papillary thyroid carcinoma (PTC) has a relatively good prognosis, yet there are some invasive PTC cases with worse clinicopathological features and poor outcome. Cancer-associated fibroblasts (CAFs) play an important role in cancer invasion and metastasis. This study aimed to investigate the expression of marker proteins of CAFs in PTC and their correlations with clinicopathological features through immunohistochemistry. The medical records of 125 PTC patients were reviewed in this study, whose specimens were retrieved for immunohistochemistry. Four CAFs marker proteins, FAP fibroblast activated protein (FAP), α-smooth muscle actin (α-SMA), Vimentin and platelet-derived growth factor receptor-α(PDGFR-α), were stained and scored. Then, statistical analyses were performed. The immunoreactivity scores of FAP and α-SMA correlated with tumor size, BRAF mutation, extrathyroidal, invasion, pathological subtype, lymph node metastasis and ATA risk stratification. Moreover, binary logistic regression analysis and receiver operating characteristic curves showed that high FAP and α-SMA immunoreactivity scores were risk factors for extrathyroidal invasion, BRAF mutation, multi-focality and lymph node metastasis (especially N1b) with good sensitivity and accuracy in prediction. A better performance was found in FAP than α-SMA. Strong expressions of CAFs were risk factors for worse thyroid cancer clinicopathological features. FAP was the better CAFs marker for PTC.



Similar content being viewed by others
References
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16(9):582–98.
Calvo F, Ranftl R, Hooper S, et al. Cdc42EP3/BORG2 and septin network enables mechano-transduction and the emergence of cancer-associated fibroblasts. Cell Rep. 2015;13(12):2699–714.
Ren Y, Zhou X, Liu X, et al. Reprogramming carcinoma associated fibroblasts by AC1MMYR2 impedes tumor metastasis and improves chemotherapy efficacy. Cancer Lett. 2016;374(1):96–106.
Kharaishvili G, Simkova D, Bouchalova K, Gachechiladze M, Narsia N, Bouchal J. The role of cancer-associated fibroblasts, solid stress and other microenvironmental factors in tumor progression and therapy resistance. Cancer Cell Int. 2014;14:41.
Kobayashi H, Enomoto A, Woods SL, Burt AD, Takahashi M, Worthley DL. Cancer-associated fibroblasts in gastrointestinal cancer. Nat Rev Gastroenterol Hepatol. 2019;16(5):282–95.
Gu J, Wang C, Xu X, et al. Immunohistochemical detection of cancer-associated fibroblasts in gastrointestinal cancer as a potential prognostic biomarker of survival: meta-analysis. Transl Cancer Res. 2020;9(11):6629–38.
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424.
Cho JG, Byeon HK, Oh KH, et al. Clinicopathological significance of cancer-associated fibroblasts in papillary thyroid carcinoma: a predictive marker of cervical lymph node metastasis. Eur Arch Otorhinolaryngol. 2018;275(9):2355–61.
Wen S, Qu N, Ma B, et al. Cancer-associated fibroblasts positively correlate with dedifferentiation and aggressiveness of thyroid cancer. Onco Targets Ther. 2021;14:1205–17.
Sun WY, Jung WH, Koo JS. Expression of cancer-associated fibroblast-related proteins in thyroid papillary carcinoma. Tumour Biol. 2016;37(6):8197–207.
Haugen BR, Alexander EK, Bible KC, et al. 2015 American thyroid association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American thyroid association guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid. 2016;26(1):1–133.
Perrier ND, Brierley JD, Tuttle RM (2018) Differentiated and anaplastic thyroid carcinoma: major changes in the American joint committee on cancer eighth edition cancer staging manual. Ca-a Cancer J Clin, 68(1):56–63.
Zhang Y, Meng Z, Zhang M, et al. Immunohistochemical evaluation of midkine and nuclear factor-kappa B as diagnostic biomarkers for papillary thyroid cancer and synchronous metastasis. Life Sci. 2014;118(1):39–45.
Zhang J, Qi J, Wei H, et al. TGFbeta1 in cancer-associated fibroblasts is associated with progression and radiosensitivity in small-cell lung cancer. Front Cell Dev Biol. 2021;9: 667645.
Wu F, Yang J, Liu J, et al. Signaling pathways in cancer-associated fibroblasts and targeted therapy for cancer. Signal Transduct Target Ther. 2021;6(1):218.
Wang Z, Liu J, Huang H, et al. Metastasis-associated fibroblasts: an emerging target for metastatic cancer. Biomark Res. 2021;9(1):47.
Toti A, Santi A, Pardella E, et al. Activated fibroblasts enhance cancer cell migration by microvesicles-mediated transfer of Galectin-1. J Cell Commun Signal. 2021;15(3):405–19.
Desbois M, Wang Y. Cancer-associated fibroblasts: Key players in shaping the tumor immune microenvironment. Immunol Rev. 2021;302(1):241–58.
Vokurka M, Lacina L, Brabek J, Kolar M, Ng YZ, Smetana K Jr. Cancer-associated fibroblasts influence the biological properties of malignant tumours via paracrine secretion and exosome production. Int J Mol Sci. 2022;23(2):964.
McAndrews KM, Chen Y, Darpolor JK et al: Identification of functional heterogeneity of carcinoma-associated fibroblasts with distinct il-6 mediated therapy resistance in pancreatic cancer. Cancer Discov 2022.
Henry LR, Lee HO, Lee JS, et al. Clinical implications of fibroblast activation protein in patients with colon cancer. Clin Cancer Res. 2007;13(6):1736–41.
Zadka L, Chabowski M, Grybowski D, Piotrowska A, Dziegiel P. Interplay of stromal tumor-infiltrating lymphocytes, normal colonic mucosa, cancer-associated fibroblasts, clinicopathological data and the immunoregulatory molecules of patients diagnosed with colorectal cancer. Cancer Immunol Immunother. 2021;70(9):2681–700.
Wang L, Cao L, Wang H, et al. Cancer-associated fibroblasts enhance metastatic potential of lung cancer cells through IL-6/STAT3 signaling pathway. Oncotarget. 2017;8(44):76116–28.
Zhang J, Wang Y, Li D, Jing S. Notch and TGF-beta/Smad3 pathways are involved in the interaction between cancer cells and cancer-associated fibroblasts in papillary thyroid carcinoma. Tumour Biol. 2014;35(1):379–85.
Geng X, Chen H, Zhao L, et al. Cancer-associated fibroblast (CAF) heterogeneity and targeting therapy of CAFs in pancreatic cancer. Front Cell Dev Biol. 2021;9: 655152.
Bota-Rabassedas N, Banerjee P, Niu Y, et al. Contextual cues from cancer cells govern cancer-associated fibroblast heterogeneity. Cell Rep. 2021;35(3): 109009.
LeBleu VS, Neilson EG. Origin and functional heterogeneity of fibroblasts. Faseb J. 2020;34(3):3519–36.
Yan T, Qiu W, Weng H, Fan Y, Zhou G, Yang Z. Single-cell transcriptomic analysis of ecosystems in papillary thyroid carcinoma progression. Front Endocrinol (Lausanne). 2021;12: 729565.
Li H, Courtois ET, Sengupta D, et al. Reference component analysis of single-cell transcriptomes elucidates cellular heterogeneity in human colorectal tumors. Nat Genet. 2017;49(5):708–18.
Herrera M, Islam AB, Herrera A, et al. Functional heterogeneity of cancer-associated fibroblasts from human colon tumors shows specific prognostic gene expression signature. Clin Cancer Res. 2013;19(21):5914–26.
Kilvaer TK, Khanehkenari MR, Hellevik T, et al. Cancer associated fibroblasts in stage I-IIIA NSCLC: prognostic impact and their correlations with tumor molecular markers. PLoS ONE. 2015;10(8): e0134965.
Imlimthan S, Moon ES, Rathke H, et al. New frontiers in cancer imaging and therapy based on radiolabeled fibroblast activation protein inhibitors: a rational review and current progress. Pharmaceuticals (Basel). 2021;14(10):1023.
Ulisse S, Baldini E, Lauro A, et al. Papillary thyroid cancer prognosis: an evolving field. Cancers (Basel). 2021;13(21):5567.
Harries V, Wang LY, McGill M, et al. Should multifocality be an indication for completion thyroidectomy in papillary thyroid carcinoma? Surgery. 2020;167(1):10–7.
Kim H, Kwon H, Moon BI. Association of multifocality with prognosis of papillary thyroid carcinoma: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg. 2021;147(10):847–54.
Ohshima A, Yamashita H, Noguchi S, et al. Indications for bilateral modified radical neck dissection in patients with papillary carcinoma of the thyroid. Arch Surg. 2000;135(10):1194–8 (discussion 1199).
Lee IA, Moon G, Kang S, et al. Predictive factors indicative of hemithyroidectomy and close follow-up versus bilateral total thyroidectomy for aggressive variants of papillary thyroid cancer. Cancers (Basel). 2022;14(11):2757.
Rashid FA, Munkhdelger J, Fukuoka J, Bychkov A. Prevalence of BRAF(V600E) mutation in Asian series of papillary thyroid carcinoma-a contemporary systematic review. Gland Surg. 2020;9(5):1878–900.
Ye Z, Xia X, Xu P, et al. The prognostic implication of the BRAF V600E mutation in papillary thyroid cancer in a Chinese population. Int J Endocrinol. 2022;2022:6562149.
Meng Z, Matsuse M, Saenko V, et al. TERT promoter mutation in primary papillary thyroid carcinoma lesions predicts absent or lower (131) I uptake in metastases. IUBMB Life. 2019;71(7):1030–40.
Yu XM, Schneider DF, Leverson G, Chen H, Sippel RS. Follicular variant of papillary thyroid carcinoma is a unique clinical entity: a population-based study of 10,740 cases. Thyroid. 2013;23(10):1263–8.
Baloch ZW, LiVolsi VA. Encapsulated follicular variant of papillary thyroid carcinoma with bone metastases. Mod Pathol. 2000;13(8):861–5.
Giani C, Torregrossa L, Piaggi P, et al. Outcome of classical (CVPTC) and follicular (FVPTC) variants of papillary thyroid cancer: 15 years of follow-up. Endocrine. 2020;68(3):607–16.
Maletta F, Falco EC, Gambella A, Metovic J, Papotti M. Noninvasive follicular thyroid neoplasm with papillary-like nuclear features: from echography to genetic profile. Tohoku J Exp Med. 2020;252(3):209–18.
Funding
This work was supported by Tianjin Science and Technology Committee Foundation Grant 21JCYBJC01820. This study was also funded by the National Natural Science Foundation of China grants (#81571709 and #81971650).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study implementation (patient treatment, specimen management, immunohistochemistry, etc.). Material preparation, data collection and analysis were performed by LZ, XZ, SZ, ZM. ZM,GZ and KX contributed in conception and design of the study. The first draft was written by LZ, and all authors commented and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests. The authors have no relevant financial or non-financial interests to disclose.
Ethical Statement
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Tianjin Medical University General Hospital.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, L., Zhang, X., Zhang, S. et al. Cancer-associated fibroblasts in papillary thyroid carcinoma. Clin Exp Med 23, 2209–2220 (2023). https://doi.org/10.1007/s10238-023-00998-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-023-00998-2