Abstract
Phylogeography infers the demographic history of various species by resolving genetic relationships among populations across a geographic range. Comparison of phylogeographic patterns of multiple species with similar geographic distributions, i.e., comparative phylogeography, allows us to assess the roles of regional historical events and species-specific ecological factors in shaping intraspecific genetic diversity, providing model systems for studying the processes and genetic basis of adaptive evolution and speciation. In addition, these patterns can also be compared among species from different regions to infer the impact of global-scale historical and present events, such as Pleistocene glacial cycles, on current biodiversity. In this review, I summarize and integrate recent findings of phylogeographic studies for two geographic lineages, Pacific Ocean (PO) and Sea of Japan (SJ) lineages, of five coastal gobies distributed around the Japanese Archipelago. Using mitochondrial and nuclear DNA markers, these studies revealed genetic divergences of the PO and SJ lineages in the coastal gobies. However, the two types of markers showed discordant patterns for genetic divergences between the two lineages (mitonuclear discordance), suggesting different coalescent processes between these genetic markers. Ecological features that depend on shallow coastal resources may contribute to restricted gene flow and be responsible for the existence of the two lineages that are predominantly found in the coastal gobies. All coastal gobies shared sharp contrasts in genetic diversity between the two lineages, with the SJ lineages rapidly expanding during Pleistocene interglacial periods. Such rapid population expansions can provide alternative ways to obtain recent calibration points to estimate divergence times of the two lineages. A comparison of the geographic distributions between the two lineages indicated that the centers of their secondary contact zones vary, which may appear to represent the continuous transition process of the two lineage distributions. Compared with results of phylogeographic studies from other oceans, multiple divergence times, sharply contrasting demographic histories, and two independent secondary contact zones are unique characteristics of the divergence between the PO and SJ lineages. These characteristics will allow us to test the continuous processes of genomic divergence during geographical isolation with environmental fluctuations and the consequences of hybridization of divergent lineages in independent secondary contact zones with different environmental settings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Distributions of present-day species have been strongly affected by past climatic and topographical changes. As evidence, phylogeographic studies identified significant genetic divergences within species or between sister-species caused by past geographic barriers, mostly accompanied by environmental changes (Avise 2000). Comparative phylogeography, i.e., synthesis and comparisons of these phylogeographic findings from multiple species with similar distributions, is useful for assessing the relative roles of regional historical events and species-specific ecological factors, such as habitat and tolerance, in shaping intraspecific genetic diversity at the regional scale. Furthermore, the phylogeographic patterns of species from different regions can be compared to infer the impact of global-scale historical and present events on current global species diversity (Bernatchez and Wilson 1998; Bowen et al. 2016).
Phylogeographic knowledge also provides a key platform for evolutionary biology because geographic isolation is the most common mechanism behind the separation of gene pools, which can result in speciation (Coyne and Orr 2004). Specifically, genome-wide analyses of geographically separated and divergent populations can help to understand the genomic processes of speciation (Ravinet et al. 2018; Yamasaki et al. 2020). Moreover, if such genetic divergences occurred in concert with hybridization in secondary contact zones, such hybrid zones will provide useful research basis to investigate the genomic consequences of hybridization between divergent populations (April et al. 2013; Montanari et al. 2014; Hirase et al. 2020a). In addition, if multiple species displayed a wide range of genetic divergences owing to geographic events occurring at different times, we could address the relationships between genetic and phenotypic divergence, and the consequence of their hybridization, by considering them a continuous process of divergence (Hendry 2009).
Marine fishes often show low genetic differentiation throughout their geographic distribution areas, while phylogeographic studies of freshwater fishes have often demonstrated substantial genetic divergences within species (Beheregaray 2008). In general, extensive dispersal promoted by ocean currents in open environments is believed to counteract genetic differentiation in marine environments (Beheregaray 2008). Nonetheless, recent advances indicate that a significant number of marine, especially coastal, fish populations possess deep genetic divergences generated by sea-level fluctuations during Pleistocene glacial–interglacial cycles in multiple regions. Indeed, such genetic divergences were also found in marine geographic areas worldwide (Soltis 2006; Patarnello et al. 2007; Gaither and Rocha 2013; Ni et al. 2014). Accumulating knowledge about these phylogeographic patterns in coastal fishes would provide useful insights into how past global-scale environmental changes and subsequent geographic isolations have generated modern-day species diversity in local and global marine environments.
This review focused on recent phylogeographic studies of the five coastal gobies distributed in the Japanese Archipelago, which have distinct Pacific Ocean (PO) and Sea of Japan (SJ) lineages (Akihito et al. 2008; Hirase et al. 2012a; Kokita and Nohara 2011; Hirase and Ikeda 2014a). Sea of Japan (SJ) is a semi-enclosed sea located between the Japanese Archipelago and the Asian continent and connected to PO and other seas through narrow and shallow (≈ 130 m depth) straits. Currently, the warm Tsushima Current flows into SJ through the southern Tsushima Straits, but paleoenvironmental studies have suggested that this sea was largely isolated in Pleistocene glacial periods when the surrounding straits were closed by sea-level declines (Tada 1994) (Fig. 1). Previous phylogeographic studies based on samples collected extensively along the Japanese Archipelago coast suggested that these isolation events arose in the PO and SJ lineages of multiple coastal gobies (Akihito et al. 2008; Hirase et al. 2012a; Hirase and Ikeda 2014a; Kokita and Nohara 2011), and three-spined stickleback, Gasterosteus aculeatus (Higuchi and Goto 1996). Considering the accumulated paleoenvironmental data (Oba et al. 1991; Tada 1994; Crusius et al. 1999; Gorbarenko and Southon 2000; Kitamura et al. 2001; Kitamura and Kimoto 2006) and parallel genetic divergence, PO and SJ lineages provide a significant testbed to perform comparative phylogeographic analyses. The PO and SJ lineages of Gasterosteus aculeatus showed reproductive isolation (Kitano et al. 2009; Ishikawa et al. 2019; the SJ lineage was described as Gasterosteus nipponicus; Higuchi et al. 2014), while lineage pairs of coastal gobies are not reproductively isolated and generate later-generation hybrids in secondary contact zones (Hirase and Ikeda 2015; Hirase et al. 2020a, c; Kato et al. 2021). Therefore, such lineage pairs are presumably in the preliminary step of speciation and would provide significant opportunities to investigate the evolutionary processes in this phase. This review summarizes and integrates previous phylogeographic findings about the PO and SJ lineages of coastal gobies, demonstrating their potential as a model system for future evolutionary biology studies.
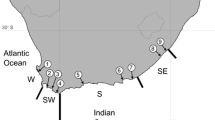
Submarine topography around the Japanese Archipelago illustrated by GMT 4.0 (Wessel et al. 2013). Gray represented the land area when sea level dropped 125 m during the last glacial period
Existence of the Pacific Ocean and Sea of Japan lineages and mitonuclear discordance
In Japanese coastal species, the PO and SJ lineages of mitochondrial DNA (mtDNA) were first reported in the late 1990s. To date, the presence of two lineages in animals has been reported clearly in two mollusk species [Turbo (Batillus) cornutus, Kojima et al. 1997; Batillaria cumingi, Kojima et al. 2004] and five coastal fishes (Pterogobius elapoides, Akihito et al. 2008; Pterogobius zonoleucus, Akihito et al. 2008; Leucopsarion petersii, Kokita and Nohara 2011; Chaenogobius annularis, Hirase et al. 2012a; Chaenogobius gulosus, Hirase and Ikeda 2014b), with divergences estimated to date back into the early to late Pleistocene. Among coastal fishes with two lineages, all fishes are gobiid (Fig. 2), suggesting that unique ecological features of gobiid fishes (see the next section) lead to genetic divergence. Although mtDNA is a useful genetic tool for phylogeographic studies owing to its rapid evolution and maternal transmission without intermolecular recombination (Avise 2000), this marker can represent only a minute fraction of the total historical record (Degnan 1993; Palumbi and Baker 1994), and the stochastic nature of coalescent processes in mtDNA could differ from that in the nuclear genome (Wilson and Veraguth 2010). In particular, mtDNA analyses could yield a misleading picture of population-level processes owing to hybridization and introgression (Toews and Brelsford 2012). Therefore, assessing congruence patterns among multi-locus nuclear DNA data will allow us to resolve reliable demography of the PO and SJ lineages.
Neighbor-joining tree of mitochondrial DNA sequences of the Pacific Ocean and Sea of Japan lineages for the five coastal gobies in the Japanese Archipelago. I used cytochrome b of mitochondrial DNA sequences of Chaenogobius annularis (Hirase et al. 2012a, AB684846–AB684973), C. gulosus (Hirase and Ikeda 2014a, AB775375–AB775405), Leucopsarion petersii (Kokita and Nohara 2011, AB562156–AB562280), Pterogobius elapoides (Akihito et al. 2008, AB440403–AB440472), and P. zonoleucus (Akihito et al. 2008, AB440363–AB440397). Acanthogobius flavimanus (AB021249) was used as an outgroup. Mitochondrial DNA sequences of the five species and an outgroup were aligned using mafft v7.402 (Katoh and Standley 2013), and a neighbor-joining trees of the aligned sequences was constructed with the Jukes-Cantor distance and complete deletion options using MEGA 7 (Tamura et al. 2011). Branch lengths were proportional to the estimated numbers of nucleotide substitutions per site. Values in nodes (closed circles) showed divergence times (million years ago: Ma) based on five calibration points (open circles). Pictures of the five coastal gobies are provided by Shuya Kato
Genetic analyses using nuclear DNA markers have been performed for four of the five coastal gobies using allozymes (C. annularis, Hirase et al. 2012c), sequences of several nuclear genes (P. elapoides and P. zonoleucus, Akihito et al. 2016), microsatellite DNA (L. petersii, Kokita et al. 2013; C. annularis, Hirase and Ikeda 2014a), and nuclear single nucleotide polymorphism (SNP) markers (L. petersii, Hirase et al. 2020a; C. annularis, Hirase et al. 2020c). In these studies, mtDNA and nuclear DNA markers discerned both concordance and discordance patterns for genetic divergences between the PO and SJ lineages. For example, C. annularis showed a concordant pattern between the genetic structures revealed by mtDNA and nuclear DNA markers and lack of gene flow between the two lineages (Hirase and Ikeda 2014a), except in two secondary contact zones (Hirase and Ikeda 2015; Kato et al. 2021). Contrastingly, L. petersii revealed discordant patterns in two secondary contact zones; PO mtDNA widely introgressed in the southern secondary contact zone, whereas SJ mtDNA extensively introgressed in the eastern secondary contact zone in comparison with nuclear genomic results (Hirase et al. 2020a). Extreme cases of discordance were observed in P. zonoleucus, with mtDNA sequences of its SJ lineage appearing closely related to P. elapoides but not to the PO lineage of P. zonoleucus (Akihito et al. 2008). In subsequent phylogenetic analyses of P. elapoides and P. zonoleucus based on three nuclear genes, two of the nuclear genes revealed monophyly for the two lineages of P. zonoleucus, although one nuclear gene showed similar patterns to those of mtDNA (Akihito et al. 2016). These conflicting patterns between mtDNA and nuclear DNA markers, i.e., mitonuclear discordance, have been observed in many phylogeographic studies (Toews and Brelsford 2012). These findings highlight the need for analyzing multiple nuclear DNA markers to investigate the genetic divergence of the two lineages, which will be possible by the application of high-throughput sequencing methods, such as RAD-seq (Peterson et al. 2012), MIG-seq (Suyama et al. 2015), and GRAS-Di (Hosoya et al. 2019).
Ecological features leading to genetic divergence
Low-dispersal species are often composed of genetically and geographically highly structured populations that reflect past geographical events (Pelc et al. 2009). The five coastal gobies in this study deposit sticky eggs on rocks or shells along coasts or rivers, and their parent fish defend the eggs until the larvae hatch (Dotsu and Tsutsumi 1959; Tsutsumi and Dotsu 1961; Sasaki and Hattori 1969; Matsui 1986). Therefore, dispersal presumably only occurs during the pelagic larval stages and limited dispersal of their larvae and adults should be associated with the genetic divergence between PO and SJ lineages. Indeed, a population genetic study of Chaenogobius annularis across a small geographic scale revealed genetic differentiation within 10 km, suggesting a remarkable low dispersal for this species owing to site fidelity of adults, larval retention, and presumably short (a few days or 1 week) pelagic larvae duration which may characterize tidepool fishes (Hirase et al. 2012b). As C. gulosus is a tidepool fish as well as C. annularis, one would expect its dispersal to be limited by similar ecological characteristics. Leucopsarion petersii has an anadromous life history, with adults ascending to the lower reaches of rivers for reproduction in spring and dying after reproduction. Then, newly-hatched larvae descend to the sea where they grow in coastal waters until the following spring (Matsui 1986). Therefore, larval retention in coastal areas near natal rivers is expected to prevent dispersal. Additionally, as Pterogobius elapoides and P. zonoleucus have free-floating life history throughout their lifetime (Suzuki et al. 2004), site fidelity of adults and larval retention in these coastal gobies may also contribute to limited dispersal.
In contrast to the five gobiid fishes, genetic divergences within the yellowfin goby Acanthogobius flavimanus have not been found neither by mtDNA nor nuclear DNA markers (Hirase et al. 2017, 2020b) despite A. flavimanus being distributed in both coastal areas of PO and SJ as the five gobiid fishes. A similar discordance phylogeographic pattern was also reported in gobiid fishes distributed along coastal areas in California, by showing a considerably shallower phylogeographic structure in Clevelandia ios compared to that of Eucyclogobius newberryi, which suggested that the high dispersal ability of C. ios owing to its open habitat, high abundance, and long pelagic larvae duration contributed to the structure (Dawson et al. 2002). Among these factors, Hirase et al. (2017) inferred that longer pelagic larvae duration of A. flavimanus contributed to its shallow phylogeographic structure. The deeper breeding site of A. flavimanus compared to those of the other five gobies may also promote the dispersal of their pelagic larvae. Additionally, Hirase et al. (2017) proposed another possible scenario by focusing on A. flavimanus breeding sites, i.e., the extinction of its SJ lineage and subsequent SJ colonization by its PO lineage. Juvenile of A. flavimanus settles mainly in shallow estuarine waters such as tidal flats and boulder areas, while adults are observed to inhabit and reproduce in deeper bay waters (Dotsu and Mito 1955; Katayama et al. 2000; Sakai et al. 2000; Kanou et al. 2005). In contrast, the five gobies that showed genetic divergences reproduce in shallow coastal areas or rivers (Dotsu and Tsutsumi 1959; Tsutsumi and Dotu 1961; Sasaki and Hattori 1969). If we assume environments in deep bay waters to be more severe than those in shallow coastal areas during Pleistocene glacial periods, when the SJ was isolated (Oba et al. 1991; Tada 1994; Gorbarenko and Southon 2000), the absence of genetic differentiation between the PO and SJ populations in A. flavimanus can be explained by the extinction of the ancestral SJ lineage owing to the disappearance of spawning sites. Conversely, the presence of SJ lineages in the five coastal gobies may suggest their survival during past severe environments during the glacial periods. Local extinctions were suggested as important in eliminating genetic divergence resulting from past geographic barriers (Cunningham and Collins 1998). Although it is generally difficult to determine the involvement of local extinctions (Provan and Bennett 2008), further accumulation of phylogeographic studies on other coastal gobies with various ecological characteristics will help us address this issue.
Contrasting demography between the Pacific Ocean and Sea of Japan lineages and its application to calibration of mitochondrial DNA evolutionary rates
Previous phylogeographic studies found sharp contrasts in genetic diversity and branching patterns of phylogenetic trees between the PO and SJ mtDNA lineages for coastal gobies (Akihito et al. 2008; Kokita and Nohara 2011; Hirase et al. 2012a). Notably, the PO mtDNA lineage showed a deep branching pattern, whereas the SJ mtDNA lineage had a shallow branching pattern with a “star-like” phylogeny (Table 1; Fig. 3). In general, the deep branching pattern indicates a comparatively long evolutionary history, while the shallow star-like branching pattern suggests a rapid increase in the survival probability of mtDNA lineages owing to rapid population expansion (Avise et al. 1984; Di Rienzo and Wilson 1991; Grant and Bowen 1998). Low nucleotide diversity and negative Tajima’s D value in the SJ mtDNA lineages of the five coastal gobies also suggested their rapid population expansion (Table 1). Sea of Japan (SJ) was expected to represent a severe environment during the glacial periods for marine warm-water species, such as coastal gobies, but a much less severe one during the interglacial interludes owing to the inflow of the warm Tsushima Current (Kitamura et al. 2001; Kitamura and Kimoto 2006). Hence, following severe bottlenecks during glacial periods, populations of the SJ lineages are believed to have rapidly expanded during the Pleistocene interglacial periods (Akihito et al. 2008; Kokita and Nohara 2011; Hirase et al. 2012a, 2016).
TCS networks of mitochondrial DNA sequences of the Pacific Ocean (red) and Sea of Japan (blue) lineages for five coastal gobies drown by PopArt (Leigh and Bryant 2015). I used cytochrome b of mitochondrial DNA sequences that used for the neighbor joining tree in Fig. 2. Dashes and small black circles represented mutational steps and branching points, respectively
Estimating when rapid population expansion occurs is an important issue (Provan and Bennett 2008). Rapid population expansion after the end of the last glacial period (LGM), called as postglacial expansion, is a common phenomenon in several warm-dwelling terrestrial species (Hewitt 2004). However, previous estimates suggested that populations of SJ lineages expanded before LGM (pre-LGM) because the mtDNA evolutionary rate would be too fast assuming it expanded after LGM (Kokita and Nohara 2011; Hirase et al. 2012a, 2016). Actually, pre-LGM population expansion has been suggested in several marine realms, including the northeastern Pacific (Marko et al. 2010), northeastern Atlantic (Hoarau et al. 2007; Derycke et al. 2008), northeast European seas (Laakkonen et al. 2013), and East China Sea (Ni et al. 2014).
Divergence and expansion time estimation in phylogeography is important and has mainly be conducted using mtDNA. Its maternal transmission, extensive intraspecific variation, and general absence of intermolecular recombination have facilitated evolutionary rate estimations (Galtier et al. 2009), with data accumulation of these rates providing a standard for evaluating newly calculated ones. mtDNA evolutionary rate estimation is of also a challenging task, with being calculated by calibrating nucleotide divergences with calibration points, such as divergence time or lineage expansion time, based on fossil or paleobiogeographic data (Tringali et al. 1999; Watanabe et al. 2003). In phylogeographic studies focusing on closely related lineages, recent calibration points are preferred because of the time dependency of molecular clocks (Ho et al. 2015). However, a general lack of recent calibration points for marine species often prevents using this estimation procedure for their evolutionary rates (Marko 2004; Lessios 2008). Therefore, several studies have used “universal” evolutionary rates that have been estimated for other related species. However, these universal rates may not be applicable to all species because evolutionary rates often vary among lineages (Zhang and Ryder 1995).
Based on the above problem, the evolutionary rate of Chaenogobius annularis was tried to be calibrated based on paleoenvironmental events (Hirase et al. 2016). In this previous study, based on the assumption that the SJ lineage of C. annularis expanded during any interglacial periods, the evolutionary rates were calculated using any interglacial periods as the calibration point and average pairwise sequence divergences within the SJ lineage (i.e., average pairwise sequence divergences were divided by the times of any interglacial periods), and reasonable rates were selected by comparing with previously reported mtDNA evolutionary rates of fishes. Consequently, Hirase et al. (2016) inferred that the SJ lineage of C. annularis expanded during the last interglacial period, 130 thousand years ago (ka). Such rapid population expansions during interglacial periods have provided alternative ways to obtain recent calibration points in other studies (Akihito et al. 2008; Crandall et al. 2012; Sakuma et al. 2019). I applied the above strategy to the other four gobies, and obtained their evolutionary rates of mitochondrial cytochrome b (cyt b), as shown in Table 2. Assuming rapid population expansion in the last interglacial period, the evolutionary rates of Leucopsarion petersii (4.2%/million years:Myr) and Pterogobius zonoleucus (3.1%/Myr) were faster than previously reported cyt b evolutionary rates of gobiid fishes [those of the Gymnogobius genus (2.2–2.7%/Myr, Harada et al. 2002; Sota et al. 2005)], whereas the rates of C. annularis (2.3%/Myr), C. gulosus (2.3%/Myr), and P. elapoides (2.5%/Myr) were similar to those. It is difficult to determine whether the faster rates were caused by artifacts (i.e., incorrect calibration) or differences in evolutionary rate. However, given that rapid population expansion during the last interglacial periods has been estimated for marine species in other areas (Provan et al. 2005; Hoarau et al. 2007; Ni et al. 2014), simultaneous rapid population expansion of SJ lineages and differences in the evolutionary rate may be a reasonable inference. Such pre-LGM expansions mean that the five coastal gobies survived despite the presumed severe environments in SJ during LGM (Oba et al. 1991). In contrast, SJ lineages of deep-sea fishes expanded after LGM (Sakuma et al. 2014), which suggests that environmental changes during LGM may be severe for deep-sea fishes if the evolutionary rates of these species were correctly calibrated. On the other hand, we could not eliminate the possibility that postglacial expansions of the SJ lineages of coastal gobies, which lead to too fast mtDNA evolutionary rates, when considering the acceleration of evolutionary rates in a recent period (< 1 million years ago: Ma) suggested by Ho et al. (2005). Therefore, the timing of expansion of the SJ lineages should also be examined carefully using the other strategies, such as Pairwise Sequentially Markovian Coalescent model based on nuclear SNP loci and their mutation rates (Li and Durbin 2011).
The SJ lineages of five coastal gobies show a star-like phylogeny lacking large genetic divergences, and the southern populations of C. annularis and L. petersii SJ lineages around Tsushima Strait have the highest genetic diversity (Kokita and Nohara 2011; Hirase et al. 2012a; Hirase and Ikeda 2014b). In general, such genetic signatures are believed to reflect the rapid postglacial expansion from southern refugia (Provan and Bennett 2008). Accordingly, SJ lineages likely rapidly expanded from southern refugia during Pleistocene interglacial periods (Kokita and Nohara 2011; Hirase and Ikeda 2014b). The star-like phylogeny, lacking large genetic divergences within SJ lineages, was key evidence for the southern refugia hypothesis. However, this finding was indicated by analyses of partial genes of the mitochondrial genome, and several signatures of genetic divergence within the lineage may have been missed owing to the lack of informative nucleotide sites. As it is expected that the mitochondrial genome as a whole (mitogenome) would provide greater resolution of possible genetic differentiation within the SJ lineage (Jacobsen et al. 2012), Hirase et al. (2016) investigated genetic differentiation within C. annularis SJ lineage using parallel mitogenome sequencing. They discovered that the northern sub-lineage is clearly distinct from southern sub-lineages, and likely experienced rapid population expansion (Fig. 4; Hirase et al. 2016); this finding was a result that the mitogenome data successfully rooted the SJ lineage by alleviating the “random rooting effect”, which hindered phylogenetic analysis based on specific gene sequences (Fig. 4). Consequently, Hirase et al. (2016) proposed the presence of glacial refugia in northern SJ coastlines, and that the entire SJ lineage did not expand from the southern refugia alone. Identification of cryptic refugia offers important insights for predicting biological responses to future global climate change (Provan and Bennett 2008). It will be a future work to clarify whether the northern sub-lineage is shared by other coastal gobies that possess PO and SJ lineages. Although the importance of using multiple nuclear DNA markers has been highlighted, further mitogenomic phylogeographic investigations will provide additional important information about the biological responses to past environmental changes in the SJ and other seas.
Phylogenetic tree of 75 Chaenogobius annularis mitogenomes [reproduced with from Hirase et al. (2016)]. Branch lengths were proportional to the estimated numbers of nucleotide substitutions per site. Closed circles and numbers indicated highly (> 80%) supported geographical clades and their bootstrap values, respectively. In the upper-left side, proportions of branches of the Sea of Japan lineage to which the outgroup branch was connected among the bootstrap trees of a single mitochondrial gene (ND1) were indicated. The outgroup branch was connected to diverse ingroup branches, thus failing to root the tree reliably, i.e., random rooting effect. Collectively, the use of mitogenome sequence solved the random rooting problem in the Sea of Japan lineage
Although mtDNA markers have shown lower genetic diversity in SJ than in PO mtDNA lineages in coastal gobies commonly, it should be noted that nuclear genetic markers do not necessarily show such contrasts. For example, whereas C. annularis showed lower genetic diversity of the SJ lineage also in microsatellite DNA loci (Hirase and Ikeda 2014b), L. petersii did not show such trends in neither microsatellite DNA (Kokita et al. 2013) nor SNP loci (Hirase et al. 2020a). Kokita et al. (2013) explained that similar levels of microsatellite DNA diversity between the PO and SJ lineages were caused because sufficient time has passed for microsatellite DNA, which is characterized by high mutation rates, to lose the molecular footprint of old demographic events and restore the genetic diversity. However, given that Hirase et al. (2020a) found similar levels of genetic diversity also in SNP loci, whose mutation rate is not high, between the two lineages, it is reasonable to assume that the diversity of the mitogenome, which has a small effective population size, decreased more severely in the SJ lineages (Ballard and Whitlock 2004). The difference in genetic diversity between PO and SJ lineages using nuclear DNA markers for other species should be more investigated to infer the demographic history of each lineage.
Re-estimation of divergence times between the Pacific Ocean and Sea of Japan lineages
Accumulation of paleoenvironmental data is important for estimating the historical processes of population divergence. In this regard, SJ has been a target field for several paleoenvironmental studies (Oba et al. 1991; Tada 1994; Crusius et al. 1999; Gorbarenko and Southon 2000; Kitamura et al. 2001; Kitamura and Kimoto 2006). The Tsushima Strait in the southwest of SJ was closed from 10.5 to 3.5 Ma (Tada 1994), and after 3.5 Ma, the Tsushima Current flowed into SJ during the interglacial periods. Kitamura et al. (2001) proposed that the Tsushima Strait was closed again from 2.5 to 1.71 Ma, and suggested that 1.7 Ma was an important turning point for SJ because its southern straits widely opened (Kitamura et al. 2001; Kitamura and Kimoto 2006). Finally, during each interglacial period after 1.7 Ma, SJ was connected to other seas, accompanied by inflow from the Tsushima Current (Kitamura et al. 2001; Kitamura and Kimoto 2006). Thus, we can expect several timings of geographical isolation for SJ lineages, and multiple events might have led to various divergence times between the SJ and PO lineages among the five coastal gobies.
As expected, my reanalysis based on a uniform phylogenetic method suggested variation of genetic distance between PO and SJ lineages among species, which could be attributed to multiple vicariance events (Fig. 2; Table 1), and allow us to statistically distinguish between “single-event” and “multiple-event” biogeographic hypotheses (Edwards and Beerli 2000). However, owing to potential differences in mtDNA evolutionary rates among species (Gissi et al. 2000), it is difficult to conclude whether a wide range of genetic distances reflects multiple vicariant events. In this regard, pairs of closely related species that possess similar ecological features may facilitate ensuring the robustness of differences in divergence time (Hills and Moriz 1994). Hirase and Ikeda (2014a) focused on closely related Chaenogobius gulosus and C. annularis as suitable models to compare levels of genetic divergence, and tested the hypothesis of multiple vicariance events. Both species have the same geographic distribution (Akihito et al. 2002), and mainly inhabit tide pools and possess similar ecological features (Sasaki and Hattori 1969). On the basis of the molecular clock and a hierarchical approximate Bayesian computation (ABC) model, they showed more recent divergence time in the two lineages of C. gulosus than those of C. annularis, suggesting multiple isolation events for SJ in the Pleistocene.
Accumulated paleoenvironmental data for SJ could provide suitable calibration points for estimating evolutionary rates and divergence times, as the time of the most recent common ancestor (TMRCA) of the SJ lineages possibly correspond to interglacial periods when warm water flows into SJ and the rapid expansion of the SJ lineages occurred (Akihito et al. 2008; Hirase et al. 2016). As mentioned in the previous section, Hirase et al. (2016) assumed rapid population expansion of the SJ lineage of C. annularis (i.e., the SJ northern sublineage suggested by the mitogenomic sequences) in the last interglacial period (130 ka), and estimated that the two lineages diverged 3.37 Ma. Here, I re-estimated divergence times between the two lineages for the other four gobies using Reltime of MEGA 7, by relaxing the assumption of a strict molecular clock in a phylogeny (Tamura et al. 2018). In this analysis, I assumed that the TMRCA of the SJ lineage for each of the four coastal gobies (Pterogobius elapoides, P. zonoleucus, Leucopsarion petersii, and C. gulosus) was 130 ka, implying simultaneous rapid population expansion of the SJ lineages in last interglacial periods, and that divergence between the SJ and PO lineages of C. annularis occurred 3.37 Ma (Hirase et al. 2016) (Fig. 2). [In C. annularis, the northern sub-lineage of the SJ lineage showed a rapid population expansion (Hirase et al. 2016) and was not used as a calibration point here because this sublineage was not shown in the cyt b tree (Fig. 2). Instead, the divergence time between the PO and SJ lineages (i.e., 3.37 Ma), which was estimated by the northern sublineage calibration point, was used as an older calibration point.] The two lineages of the four gobies were estimated to diverge from 340−460 ka (Fig. 2), suggesting that they diverged in a similar period. Contrastingly, the divergence time between the SJ lineage of P. zonoleucus and P. elapoides (3.09 Ma) was close to that between the two lineages of C. annularis (Fig. 2). The unexpected monophyletic relationship of mtDNA between the SJ lineage of P. zonoleucus and P. elapoides was inferred to be caused by introgression of mtDNA from P. elapoides to the SJ lineage of P. zonoleucus upon ancient hybridization (Akihito et al. 2016). Given that the opening of the Tsushima Strait 3.5 Ma was estimated to cause divergence between the two lineages of C. annularis (Hirase et al. 2016), this same geographic event may also be associated with ancient hybridization between P. elapoides and the SJ lineage of P. zonoleucus. In addition, this event has been reported to account for the divergence between the continental and Japanese Archipelago populations of a related group, the Gymnogobius castaneus and Gymnogobius taranetzi species complex (Sota et al. 2005). Therefore, this re-estimation of divergence times between the two lineages may suggest two geographic isolation event waves in the SJ.
Although mtDNA is a good molecular tool to estimate divergence time, we should note that it may not always share the same topology and branch lengths as other genomic regions (Toews and Brelsford 2012). Moreover, the timing of gene divergence predates the population divergence event, leading to overestimation of population divergence times (Edwards and Beerli 2000; Carstens and Knowles 2007). In this regard, multi-locus coalescent approach allows for much more accurate estimation of the divergence history (McCormack et al. 2011) and should be applied to divergence time estimation of the SJ and PO lineages for coastal marine species. For example, Akihito et al. (2016) estimated divergence times of the two lineages of P. elapoides and P. zonoleucus based on three nuclear genes (P. elapoides, 810 ka; P. zonoleucus, 560 ka). In addition, Ravinet et al. (2018) used coalescent analyses and ABC based on genome-wide SNP markers and estimated that Gasterosteus aculeatus and G. nipponicus diverged approximately 0.68–1 Ma. In the future, comparison of divergence times between the two lineages based on mtDNA and multi-locus coalescent analyses will be important to increase dating precision of genetic divergences.
Secondary contact zones between the Pacific Ocean and Sea of Japan lineages
There are two potential secondary contact zones between the PO and SJ lineages of coastal gobies, i.e., the western and eastern coastal areas of the Japanese Archipelago, where these seas are connected. Both secondary contact zones were identified in Leucopsarion petersii and Chaenogobius annularis based on mtDNA and nuclear DNA markers. The western and eastern secondary contact zones were the Seto Inland Sea and the Joban-Kashimanada or the Sanriku coast, respectively (Kokita and Nohara 2011; Hirase and Ikeda 2015; Hirase et al. 2020a; Kato et al. 2021) (Fig. 5). These two secondary contact zones have been suggested to be formed at different periods in both species (Hirase et al. 2020a; Kato et al. 2021). As the Seto Inland Sea was terrestrial during LGM (Fig. 1), it is probable that the secondary contact zones in this inland sea formed as a consequence of subsequent postglacial colonization (Kokita and Nohara 2011; Hirase et al. 2020c; Kato et al. 2021). In contrast, assuming that the SJ lineage expanded and formed geographic distribution into the PO side under the influence of the Tsugaru Current, which is part of the Tsushima Current, during the last interglacial period as discussed above, the secondary contact zones in Joban-Kashimanada or Sanriku Coast were inferred to establish before LGM (Fig. 1) (Hirase and Ikeda 2015; Hirase et al. 2020a).
Geographical distribution of the Pacific Ocean (red) and Sea of Japan (blue) lineages for five coastal gobies. Yellow denotes populations where the two mitochondrial lineages co-exist. Pie graphs showed ratios of both mitochondrial lineages, and bar plots show the individual genotype membership in populations where nuclear admixture between the two lineages has been detected. Bar plots of Chaenogobius annularis in the Sanriku Coast and Leucopsarion petersii were based on SNP markers (Hirase et al. 2020a, b), and that of C. annularis in Seto Inland Sea was based on microsatellite DNA markers (Kato et al. 2021). Dashed line in the map of C. annularis shows the region where the third lineage, the East China Sea lineage, is distributed (Kato et al. 2021)
Hybridization between the PO and SJ lineages occurred in the two secondary contact zones in both L. petersii and C. annularis; however, mtDNA and nuclear DNA markers demonstrated different genetic structures in these zones. For C. annularis, an admixture of both DNA markers was observed in the center of the secondary contact zone and showed their concordance pattern (Fig. 5). Contrastingly, in the secondary contact zones of L. petersii, the dominant mtDNA is inconsistent with the results of nuclear genomic clustering, suggesting that mating preference and/or local adaptive pressure may cause asymmetric introgression of mtDNA in both secondary contact zones (Fig. 5) (Hirase et al. 2020a). This mitonuclear discordance in the secondary contact zone has been commonly observed across a wide range of organisms (Toews and Brelsford 2012), and the secondary contact zone between the PO and SJ lineages are no exception to this phenomenon.
A comparison of the geographic locations of secondary contact zones may represent the continuous transition process of formation of the PO and SJ lineage distributions (Fig. 5). For L. petersii, the eastern secondary contact zone appears around the southern Joban-Kashimanada coast, whereas that of C. annularis is located on the Sanriku coast north of Joban-Kashimanada (Hirase and Ikeda 2015). Moreover, although secondary contact zones between lineages have not reported for the other three coastal gobies, C. gulosus, Pterogobius elapoides, considering the geographic distributions of the two lineages (Fig. 5), their secondary contact zones are expected to be formed in coastal areas around Joban-Kashimanada as those of L. petersii. Therefore, this means that only the SJ lineage of C. annularis has not expanded its distribution southward. Hirase and Ikeda (2015) hypothesized that the cold environments of the Sanriku coast, located in the cold Oyashio Current (Fig. 1), have restricted dispersion and lowered the abundance of the C. annularis population. Indeed, the cold Oyashio Current restricts the distribution of P. zonoleucus, which is believed to be less cold-tolerant (Akihito et al. 2008). Therefore, the southern expansion of the SJ lineages of L. petersii and the other two gobies (C. gulosus and P. elapoides) suggests that their dispersal abilities and/or cold tolerance are stronger than those of C. annularis, and they have colonized through the cold area. Enhanced expansion of the SJ lineage of L. petersii compared to that of C. annularis was also observed in western secondary contact zones in the Seto Inland Sea (Kokita and Nohara 2011; Hirase et al. 2020a; Kato et al. 2021), where the SJ lineage of L. petersii has been shown to be extend further eastward than that of C. annularis (Fig. 5). Given that the geographic isolation of SJ occurred due to the closing of the Tsugaru and Tsushima Straits (Fig. 1), both of western and eastern secondary contact zones of the two species in the PO side suggest unidirectional expansion of the SJ lineages and contraction of the PO lineages. This may indicate selective advantage of the SJ lineages and/or the contribution of ocean currents in these zones, including the dominant southward currents (Tsugaru Currents; Fig. 1) around the Sanriku coast and eastward currents around the Seto Inland Sea, which could promote expansion of the SJ lineages (Ishizu et al. 2017; Kurogi and Hasumi 2019). Intensive sampling is needed to determine the population genetic structures around these secondary contact zones.
Future perspectives: advantages of the Japanese Archipelago for evolutionary studies
Genetic divergences of coastal fishes caused by the decline in Pleistocene glacial sea levels have also been detected in other geographic regions, such as between the Gulf of Mexico (GM) and Atlantic Ocean (AO) (Soltis et al. 2006), Mediterranean Sea (MS) and AO (Patarnello et al. 2007), South China Sea (SCS) and East China Sea (ECS) (Ni et al. 2014), and Indian Ocean (IO) and PO (Gaither and Rocha 2013) (Fig. 6). I compare the genetic divergences with those between PO and SJ lineages, focusing on their divergence times, population demography, and hybridization, and discuss the advantages of SJ and PO lineages for evolutionary study.
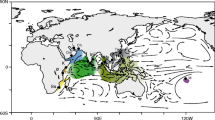
Four geographic regions where genetic divergences of coastal fishes have been caused by sea-level fall during glaciation: Gulf of Mexico versus the Atlantic Ocean (Soltis et al. 2006), the Mediterranean Sea versus the Atlantic Ocean (Patarnello et al. 2007), the South China Sea versus the East China Sea (Ni et al. 2014), and the Indian Ocean and the Pacific Ocean (Gaither and Rocha 2013). Range of divergence times in each geographic region are also shown. Submarine topography around each geographic region is illustrated by GMT 4.0 (Wessel et al. 2013). Gray represented the land area when sea level dropped 125 m during the Last Glacial Maximum. Scale bar corresponds to 500 km
The divergence times between GM and AO and between SCS and ECS were estimated to be in the middle Pleistocene (Centropristis striata, and the Brevoortia tyrannus and Brevoortia patronus pair: 0.35 Ma, Bowen and Avise 1990; Coilia mystus and Chelon haematocheilus: 0.4–0.7 Ma, Ni et al. 2014) (Fig. 6). Therefore, the genetic divergences detected in those regions were younger than those between the PO and SJ lineages (0.34–3.37 Ma; Fig. 2). Contrastingly, divergence times between the MS and AO lineages (0.7–3.26 Ma, Patarnello et al. 2007) and between the IO and PO lineages (0.3–6.6 Ma, Gaither and Rocha 2013) show a wide time range, including old divergence (Fig. 6). Therefore, the level of genetic divergence between these lineages is similar to that between the PO and SJ lineages. However, the nature of population demography and secondary contact zones of these pairs (MS and AO, and IO and PO) differs from that of the PO and SJ lineages. In the former two pairs, rapid population expansion was estimated for both lineages (Patarnello et al. 2007; Gaither and Rocha 2013). In addition, each of their hybrid zones was located in one contact region; hybridization between MS and AO lineages occurred in the Alboran Sea east of the Strait of Gibraltar that connects AO to MS (Lemaire et al. 2005), while that between IO and PO lineages was in the Christmas and Cocos (Keeling) Islands, where the IO and PO regional biotas meet (Hobbs et al. 2008) (Fig. 6). Thus, multiple divergence times, characterized by sharp contrasts of demographic histories, and two independent hybrid zones are unique characteristics of the divergence between the PO and SJ lineages.
The unique characteristics of the PO and SJ lineages can provide two major advantages for evolutionary studies. First, the PO and SJ lineages were under different environmental scenarios, as suggested by their demographic histories. Paleoenvironmental studies have demonstrated that SJ was nearly isolated from surrounding seas after the closing of the Tsushima Straits because of sea level falling during glacial periods, which resulted in salinity and temperature decrease owing to freshwater inputs from rivers and the loss of the warm Tsushima Current, respectively (Oba et al. 1991; Tada 1994; Gorbarenko and Southon 2000). Therefore, the PO and SJ lineages of Japanese coastal species were subjected to vastly differing selection regimes, offering a useful model system for studies of adaptive divergence (Kokita et al. 2013; Hirase et al. 2020a). Indeed, for Leucopsarion petersii, it was suggested that past and present low seawater temperatures in SJ promoted the evolution of fast growth and large body sizes in SJ populations (Kokita et al. 2013). Additionally, divergence in female mate choice patterns owing to body size changes was detected in L. petersii, with the SJ lineage females preferring larger and same-lineage males, while the PO lineage females showed no preference (Kokita et al. 2017). However, ecological and phenotypic differences between the two lineages have not yet been evaluated for most of the coastal gobies having both lineages. Further studies on these perspectives will allow us to test the continuous process of genomic divergence during geographical isolation and their evolutionary consequences in the speciation continuum (Hendry 2009) owing to their wide spectrum of genetic divergences.
Second, there are two independent hybrid zones between the PO and SJ lineages (Hirase and Ikeda 2015; Hirase et al. 2020a; Kato et al. 2021). In general, the genomic features in independent hybrid zones vary (Nolte et al. 2009; Aboim et al. 2010), suggesting that the evolutionary outcomes of hybridization are influenced by various exogenous factors due to local environments. Genomic and phenotypic features in such multiple independent hybrid zones will provide key information about the evolutionary consequences of hybridization (Morgan-Richards and Wallis 2003; Mandeville et al. 2017). As the environments vary spatially with oceanographic conditions, hybridization impact may play a substantial role in shaping biodiversity in marine environments. However, genomic studies for hybridization on species inhabiting marine ecosystems are rare. In this regard, the two potential hybrid zones between the PO and SJ lineages, i.e., western and eastern coastal areas of the Japanese Archipelago, constitute unique systems to study the genomic features of independent hybrid zones. For example, Hirase et al. (2020a) performed integrated analyses of genotypic and phenotypic data for two independent hybrid populations of both lineages of L. petersii, revealing that these populations have different genotypic and phenotypic features. Moreover, phenotypic features were not explained by their nuclear genomic ancestry, suggesting that adaptive introgression of genes regulates the respective phenotypes, and they concluded that hybridization could lead to variable genomic and phenotypic consequences under different environmental conditions in marine ecosystems (Hirase et al. 2020a).
Two independent hybrid zones have also been found in Chaenogobius annularis (Hirase and Ikeda 2015; Kato et al. 2021). Hirase et al. (2020c) analyzed the genomic status of one hybrid zone formed on the Sanriku coast by comprehensive genome sequencing, including RNA-sequencing, RAD-sequencing, whole genome sequencing, whole mitogenome sequencing, amplicon-sequencing, and small RNA-sequencing. Although all hybrids in this population have the genomes of both groups at a ratio of approximately 50%, they also showed excess ancestry of either group in several genomic regions, suggesting mosaic features of hybrid genomes. In addition, they showed accumulation of nonsynonymous substitutions in the PO lineage mitogenomes, including two convergent substitutions as well as signals of mitochondrial lineage-specific selection on mitochondria-related nuclear genes. These substitutions were likely caused by mismatch combinations between mitogenomes and nuclear mitochondria-related genes, i.e., mitonuclear incompatibility (Hill 2019). These results suggest that novel genomic combinations formed by hybridization promoted novel mitogenome mutations. Moreover, Hirase et al. (2020c) suggested that the novel substitutions in the PO lineage mitogenomes may not only resolve the mitonuclear incompatibility with SJ-lineage nuclear genes but also aid in the local adaptation to the Sanriku coast, which is characterized by cold seawater temperature due to the Oyashio Current (Fig. 1). Whether environmental effects play a direct and general role in driving mitonuclear incompatibility in natural hybrid populations remains unclear and requires further investigation (Arnqvist et al. 2010; Zhang et al. 2017; Hill 2019), and two independent hybrid zones with different environmental settings could provide a unique system for pursuing this question.
References
Aboim M, Mavárez J, Bernatchez L, Coelho M (2010) Introgressive hybridization between two Iberian endemic cyprinid fish: a comparison between two independent hybrid zones. J Evol Biol 23:817–828
Akihito, Sakamoto K, Ikeda Y, Sugiyama K (2002) Gobioidei. In: Nakabo T (ed) Fishes of Japan with pictorial keys to the species, English edn. Tokai University Press, Tokyo, pp1139–1310, 1596–1619
Akihito, Fumihito A, Ikeda Y, Aizawa M, Makino T, Umehara Y, Kai Y, Nishimoto Y, Hasegawa M, Nakabo T, Gojobori T (2008) Evolution of Pacific Ocean and the Sea of Japan populations of the gobiid species, Pterogobius elapoides and Pterogobius zonoleucus, based on molecular and morphological analyses. Gene 427:7–18
Akihito, Akishinonomiya F, Ikeda Y, Aizawa M, Nakagawa S, Umehara Y, Yonezawa T, Mano S, Hasegawa M, Nakabo T, Gojobori T (2016) Speciation of two gobioid species, Pterogobius elapoides and Pterogobius zonoleucus revealed by multi-locus nuclear and mitochondrial DNA analyses. Gene 576:593–602
April J, Hanner RH, Dion‐Côté AM, Bernatchez L (2013) Glacial cycles as an allopatric speciation pump in north‐eastern American freshwater fishes. Mol Ecol 22:409–422
Arnqvist G, Dowling DK, Eady P, Gay L, Tregenza T, Tuda M, Hosken DJ (2010) Genetic architecture of metabolic rate: environment specific epistasis between mitochondrial and nuclear genes in an insect. Evolution 64:3354–3363
Avise JC (2000) Phylogeography: the history and formation of species. Harvard University Press, Cambridge, MA
Avise JC, Neigel JE, Arnold J (1984) Demographic influences on mitochondrial DNA lineage survivorship in animal populations. J Mol Evol 20:99–105
Ballard JWO, Whitlock MC (2004) The incomplete natural history of mitochondria. Mol Ecol 13:729–744
Beheregaray LB (2008) Twenty years of phylogeography: the state of the field and the challenges for the Southern Hemisphere. Mol Ecol 17:3754–3774
Bernatchez L, Wilson CC (1998) Comparative phylogeography of Nearctic and Palearctic fishes. Mol Ecol 7:431–452
Bowen B, Avise J (1990) Genetic structure of Atlantic and Gulf of Mexico populations of sea bass, menhaden, and sturgeon: influence of zoogeographic factors and life-history patterns. Mar Biol 107:371–381
Bowen BW, Gaither MR, DiBattista JD, Iacchei M, Andrews KR, Grant WS, Toonen RJ, Briggs JC (2016) Comparative phylogeography of the ocean planet. Proc Natl Acad Sci U S A 113:7962–7969
Carstens BC, Knowles LL (2007) Shifting distributions and speciation: species divergence during rapid climate change. Mol Ecol 16:619–627
Coyne JA, Orr HA (2004) Speciation. Sinauer Associates, Sunderland, MA
Crandall ED, Sbrocco EJ, DeBoer TS, Barber PH, Carpenter KE (2012) Expansion dating: calibrating molecular clocks in marine species from expansions onto the Sunda Shelf following the Last Glacial Maximum. Mol Biol Evol 29:707–719
Crusius J, Pedersen TF, Calvert SE, Cowie GL, Oba T (1999) A 36 kyr geochemical record from the Sea of Japan of organic matter flux variations and changes in intermediate water oxygen concentrations. Paleoceanography 14:248–259
Cunningham CW, Collins TM (1998) Beyond area relationships: extinction and recolonization in molecular marine biogeography. In: Schierwater B, Streit B, Wagner G, DeSalle R (eds) Molecular ecology and evolution: approaches and applications. Birkhäuser, Basel, pp 297–321
Dawson M, Louie K, Barlow M, Jacobs D, Swift C (2002) Comparative phylogeography of sympatric sister species, Clevelandia ios and Eucyclogobius newberryi (Teleostei, Gobiidae), across the California Transition Zone. Mol Ecol 11:1065–1075
Degnan SM (1993) The perils of single gene trees—mitochondrial versus single-copy nuclear DNA variation in white-eyes (Aves: Zosteropidae). Mol Ecol 2:219–225
Derycke S, Remerie T, Backeljau T, Vierstraete A, Vanfleteren J, Vincx M, Moens T (2008) Phylogeography of the Rhabditis (Pellioditis) marina species complex: evidence for long‐distance dispersal, and for range expansions and restricted gene flow in the northeast Atlantic. Mol Ecol 17:3306–3322
Di Rienzo A, Wilson AC (1991) Branching pattern in the evolutionary tree for human mitochondrial DNA. Proc Natl Acad Sci U S A 88:1597–1601
Dotsu Y, Mito S (1955) On the breeding-habits, larvae and young of a goby, Acanthogobius flavimanus (Temminck et Schlegel). Jpn J Ichthyol 4:153–161 (in Japanese)
Dotsu Y, Tsutsumi T (1959) The reproductive behaviour in the gobiid fish, Pterogobius elapoides (Günther). Bull Fac Fish, Nagasaki Univ 8:186–190 (in Japanese)
Edwards SV, Beerli P (2000) Perspective: gene divergence, population divergence, and the variance in coalescence time in phylogeographic studies. Evolution 54:1839–1854
Gaither MR, Rocha LA (2013) Origins of species richness in the Indo‐Malay‐Philippine biodiversity hotspot: evidence for the centre of overlap hypothesis. J Biogeogr 40:1638–1648
Galtier N, Nabholz B, Glémin S, Hurst G (2009) Mitochondrial DNA as a marker of molecular diversity: a reappraisal. Mol Ecol 18:4541–4550
Gissi C, Reyes A, Pesole G, Saccone C (2000) Lineage-specific evolutionary rate in mammalian mtDNA. Mol Biol Evol 17:1022–1031
Gorbarenko S, Southon J (2000) Detailed Japan Sea paleoceanography during the last 25 kyr: constraints from AMS dating and \(\delta\)18O of planktonic foraminifera. Palaeogeogr Palaeoclimatol Palaeoecol 156:177–193
Grant W, Bowen B (1998) Shallow population histories in deep evolutionary lineages of marine fishes: insights from sardines and anchovies and lessons for conservation. J Hered 89:415–426
Harada S, Jeon S-R, Kinoshita I, Tanaka M, Nishida M (2002) Phylogenetic relationships of four species of floating gobies (Gymnogobius) as inferred from partial mitochondrial cytochrome b gene sequences. Ichthyol Res 49: 324-332
Hendry AP (2009) Ecological speciation! Or the lack thereof? Can J Fish Aquat Sci 66:1383–1398
Hewitt G (2004) Genetic consequences of climatic oscillations in the Quaternary. Philos Trans R Soc Lond B Biol Sci 359:183–195
Higuchi M, Goto A (1996) Genetic evidence supporting the existence of two distinct species in the genus Gasterosteus around Japan. Environ Biol Fishes 47:1–16
Higuchi M, Sakai H, Goto A (2014) A new threespine stickleback, Gasterosteus nipponicus sp. nov. (Teleostei: Gasterosteidae), from the Japan Sea region. Ichthyol Res 61:341–351
Hill GE (2019) Mitonuclear Ecology. Oxford University Press, Oxford
Hillis DM, Moritz C (1994) An overview of applications of molecular systematics. In: Hillis DM, Moritz C (eds) Molecular Systematics. Sinauer, Sunderland, pp 502–515
Hirase S, Ikeda M (2014a) Divergence of mitochondrial DNA lineage of the rocky intertidal goby Chaenogobius gulosus around the Japanese Archipelago: reference to multiple Pleistocene isolation events in the Sea of Japan. Mar Biol 161:565–574
Hirase S, Ikeda M (2014b) Long-term vicariance and post-glacial expansion in the Japanese rocky intertidal goby Chaenogobius annularis. Mar Ecol Prog Ser 499: 217–231
Hirase S, Ikeda M (2015) Hybrid population of highly divergent groups of the intertidal goby Chaenogobius annularis. J Exp Mar Biol Ecol 473:121–128
Hirase S, Ikeda M, Kanno M, Kijima A (2012a) Phylogeography of the intertidal goby Chaenogobius annularis associated with paleoenvironmental changes around the Japanese Archipelago. Mar Ecol Prog Ser 450:167–179
Hirase S, Kanno M, Ikeda M, Kijima A (2012b) Evidence of the restricted gene flow within a small spatial scale in the Japanese common intertidal goby Chaenogobius annularis. Mar Ecol 33:481–489
Hirase S, Ikeda M, Kanno M, Kijima A (2012c) Detection of regional allozyme divergence in the rocky intertidal goby Chaenogobius annularis. Ichthyol Res 59:264–267
Hirase S, Takeshima H, Nishida M, Iwasaki W (2016) Parallel mitogenome sequencing alleviates random rooting effect in phylogeography. Genome Biol Evol 8:1267–1278
Hirase S, Chambers S, Hassell K, Carew M, Pettigrove V, Soyano K, Nagae M, Iwasaki W (2017) Phylogeography of the yellowfin goby Acanthogobius flavimanus in native and non-native distributions. Mar Biol 164:106
Hirase S, Kokita T, Nagano AJ, Kikuchi K (2020a) Genomic and phenotypic consequences of two independent secondary contact zones between allopatric lineages of the anadromous ice goby Leucopsarion petersii. Heredity 124:223–235
Hirase S, Tezuka A, Nagano AJ, Kikuchi K, Iwasaki W (2020b) Genetic isolation by distance in the yellowfin goby populations revealed by RAD sequencing. Ichthyol Res 67:98–104
Hirase S, Tezuka A, Nagano AJ, Sato M, Hosoya S, Kikuchi K, Iwasaki W (2020c) Integrative genomic phylogeography reveals signs of mitonuclear incompatibility in a natural hybrid goby population. Evolution 75:176–194
Ho SY, Phillips MJ, Cooper A, Drummond AJ (2005) Time dependency of molecular rate estimates and systematic overestimation of recent divergence times. Mol Biol Evol 22:1561–1568
Hoarau G, Coyer J, Veldsink J, Stam W, Olsen J (2007) Glacial refugia and recolonization pathways in the brown seaweed Fucus serratus. Mol Ecol 16:3606–3616
Hobbs J-PA, Frisch AJ, Allen GR, Van Herwerden L (2008) Marine hybrid hotspot at Indo-Pacific biogeographic border. Biol Lett 5:258–261
Hosoya S, Hirase S, Kikuchi K, Nanjo K, Nakamura Y, Kohno H, Sano M (2019) Random PCR‐based genotyping by sequencing technology GRAS‐Di (genotyping by random amplicon sequencing, direct) reveals genetic structure of mangrove fishes. Mol Ecol Resour 19:1153–1163
Ishikawa A, Kabeya N, Ikeya K, Kakioka R, Cech JN, Osada N, Leal MC, Inoue J, Kume M, Toyoda A, Tezuka A, Nagano AJ, Yamasaki YY, Suzuki Y, Kokita T, Takahashi H, Lucek K, Marques D, Takehana Y, Naruse K, Mori S, Monroig O, Ladd N, Schubert CJ, Matthews B, Peichel CL, Seehausen O, Yoshizaki G, Kitano J (2019) A key metabolic gene for recurrent freshwater colonization and radiation in fishes. Science 364:886–889
Ishizu M, Itoh S, Tanaka K, Komatsu K (2017) Influence of the Oyashio Current and Tsugaru Warm Current on the circulation and water properties of Otsuchi Bay, Japan. J Oceanogr 73:115–131
Jacobsen MW, Hansen MM, Orlando L, Bekkevold D, Bernatchez L, Willerslev E, Gilbert MTP (2012) Mitogenome sequencing reveals shallow evolutionary histories and recent divergence time between morphologically and ecologically distinct European whitefish (Coregonus spp.). Mol Ecol 21:2727–2742
Kanou K, Sano M, Kohno H (2005) Ontogenetic diet shift, feeding rhythm, and daily ration of juvenile yellowfin goby Acanthogobius flavimanus on a tidal mudflat in the Tama River estuary, central Japan. Ichthyol Res 52:319–324
Katayama S, Sakai K, Iwata T, Honda H (2000) Life history of Japanese common goby Acanthogobius flavimanus in Hiroura Lagoon of Natori River mouth. Bull Miyagi Pref Fish Res Dev Center 16:93–97
Kato S, Arakaki S, Kikuchi K, Hirase S (2021) Complex phylogeographic patterns in the intertidal goby Chaenogobius annularis around Kyushu Island as a boundary zone of three different seas. Ichthyol Res:1–15
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780
Kitamura A, Kimoto K (2006) History of the inflow of the warm Tsushima Current into the Sea of Japan between 3.5 and 0.8 Ma. Palaeogeogr Palaeoclimatol Palaeoecol 236:355–366
Kitamura A, Takano O, Takata H, Omote H (2001) Late Pliocene-early Pleistocene paleoceanographic evolution of the Sea of Japan. Palaeogeogr Palaeoclimatol Palaeoecol 172:81–98
Kitano J, Ross JA, Mori S, Kume M, Jones FC, Chan YF, Absher DM, Grimwood J, Schmutz J, Myers RM (2009) A role for a neo-sex chromosome in stickleback speciation. Nature 461:1079–1083
Kojima S, Segawa R, Hayashi I (1997) Genetic differentiation among populations of the Japanese turban shell Turbo (Batillus) cornutus corresponding to warm currents. Mar Ecol Prog Ser 150:149–155
Kojima S, Hayashi I, Kim D, Iijima A, Furota T (2004) Phylogeography of an intertidal direct-developing gastropod Batillaria cumingi around the Japanese Islands. Mar Ecol Prog Ser 276:161–172
Kokita T, Nohara K (2011) Phylogeography and historical demography of the anadromous fish Leucopsarion petersii in relation to geological history and oceanography around the Japanese Archipelago. Mol Ecol 20:143–164
Kokita T, Takahashi S, Kumada H (2013) Molecular signatures of lineage‐specific adaptive evolution in a unique sea basin: the example of an anadromous goby Leucopsarion petersii. Mol Ecol 22:1341–1355
Kokita T, Takahashi S, Kinoshita M (2017) Evolution of gigantism and size-based female mate choice in ice goby (Leucopsarion petersii) populations in a semi-enclosed sea basin. Biol J Linn Soc 120:563–577
Kurogi M, Hasumi H (2019) Tidal control of the flow through long, narrow straits: a modeling study for the Seto Inland Sea. Sci Rep 9:1–9
Laakkonen HM, Lajus DL, Strelkov P, Väinölä R (2013) Phylogeography of amphi-boreal fish: tracing the history of the Pacific herring Clupea pallasii in North-East European seas. BMC Evol Biol 13:67
Leigh JW, Bryant D (2015) PopART: full-feature software for haplotype network construction. Methods Ecol Evol 6:1110–1116
Lemaire C, Versini JJ, Bonhomme F (2005) Maintenance of genetic differentiation across a transition zone in the sea: discordance between nuclear and cytoplasmic markers. J Evol Biol 18:70–80
Lessios HA (2008) The great American schism: divergence of marine organisms after the rise of the Central American Isthmus. Annu Rev Ecol Evol Syst 39:6–91
Li H, Durbin R (2011) Inference of human population history from individual whole-genome sequences. Nature 475:493–496
Mandeville EG, Parchman TL, Thompson KG, Compton RI, Gelwicks KR, Song SJ, Buerkle CA (2017) Inconsistent reproductive isolation revealed by interactions between Catostomus fish species. Evol Lett 1:255–268
Marko PB (2004) 'What's larvae got to do with it?' Disparate patterns of post-glacial population structure in two benthic marine gastropods with identical dispersal potential. Mol Ecol 13:597–611
Marko PB, Hoffman JM, Emme SA, McGovern TM, Keever CC, Nicole Cox L (2010) The ‘Expansion-Contraction’ model of Pleistocene biogeography: rocky shores suffer a sea change? Mol Ecol 19:146–169
Matsui S (1986) Studies on the ecology and the propagation of the ice goby, Leucopsarion petersi Hilgendorf. Sci Bull Fac Agr, Kyushu Univ 40:135–174 (in Japanese)
McCormack JE, Heled J, Delaney KS, Peterson AT, Knowles LL (2011) Calibrating divergence times on species trees versus gene trees: implications for speciation history of Aphelocoma jays. Evolution 65:184–202
Montanari SR, Hobbs JPA, Pratchett MS, Bay LK, Van Herwerden L (2014) Does genetic distance between parental species influence outcomes of hybridization among coral reef butterflyfishes? Mol Ecol 23:2757–2770
Morgan-Richards M, Wallis GP (2003) A comparison of five hybrid zones of the weta Hemideina thoracica (Orthoptera: Anostostomatidae): degree of cytogenetic differentiation fails to predict zone width. Evolution 57:849–861
Ni G, Li Q, Kong L, Yu H (2014) Comparative phylogeography in marginal seas of the northwestern Pacific. Mol Ecol 23:534–548
Nolte A, Gompert Z, Buerkle C (2009) Variable patterns of introgression in two sculpin hybrid zones suggest that genomic isolation differs among populations. Mol Ecol 18:2615–2627
Oba T, Kato M, Kitazato H, Koizumi I, Omura A, Sakai T, Takayama T (1991) Paleoenvironmental changes in the Japan Sea during the last 85,000 years. Paleoceanography 6:499–518
Palumbi SR, Baker CS (1994) Contrasting population structure from nuclear intron sequences and mtDNA of humpback whales. Mol Biol Evol 11:426–435
Patarnello T, Volckaert FA, Castilho R (2007) Pillars of Hercules: is the Atlantic–Mediterranean transition a phylogeographical break? Mol Ecol 16:4426–4444
Pelc R, Warner R, Gaines S (2009) Geographical patterns of genetic structure in marine species with contrasting life histories. J Biogeogr 36:1881–1890
Peterson BK, Weber JN, Kay EH, Fisher HS, Hoekstra HE (2012) Double digest RADseq: an inexpensive method for de novo SNP discovery and genotyping in model and non-model species. PLoS One 7:e37135
Provan J, Bennett K (2008) Phylogeographic insights into cryptic glacial refugia. Trends Ecol Evol 23:564–571
Provan J, Wattier RA, Maggs CA (2005) Phylogeographic analysis of the red seaweed Palmaria palmata reveals a Pleistocene marine glacial refugium in the English Channel. Mol Ecol 14:793–803
Ravinet M, Yoshida K, Shigenobu S, Toyoda A, Fujiyama A, Kitano J (2018) The genomic landscape at a late stage of stickleback speciation: high genomic divergence interspersed by small localized regions of introgression. PLoS Genet 14:e1007358
Sakai K, Katayama S, Iwata T (2000) Life history of the Japanese common goby, Acanthogobius flavimanus in the Matsushima Bay. Bull Miyagi Pref Fish Res Dev Center 16:85–92
Sakuma K, Ueda Y, Hamatsu T, Kojima S (2014) Contrasting population histories of the deep-sea demersal fish, Lycodes matsubarai, in the Sea of Japan and the Sea of Okhotsk. Zool Sci 31:375–383
Sakuma K, Ishida R, Kodama T, Takada Y (2019) Reconstructing the population history of the sandy beach amphipod Haustorioides japonicus using the calibration of demographic transition (CDT) approach. PLoS One 14:e0223624
Sasaki T, Hattori J (1969) Comparative ecology of two closely related sympatric gobiid fishes living in tide pools. Jpn J Ichthyol 15:143–155 (in Japanese)
Soltis DE, Morris AB, McLachlan JS, Manos PS, Soltis PS (2006) Comparative phylogeography of unglaciated eastern North America. Mol Ecol 15:4261–4293
Sota T, Mukai T, Shinozaki T, Sato H, Yodoe Ki (2005) Genetic differentiation of the gobies Gymnogobius castaneus and G. taranetzi in the region surrounding the Sea of Japan as inferred from a mitochondrial gene genealogy. Zool Sci 22:87–93
Suyama Y, Matsuki Y (2015) MIG-seq: an effective PCR-based method for genome-wide single-nucleotide polymorphism genotyping using the next-generation sequencing platform. Sci Repo 5:1–12
Suzuki T, Shibukawa K, Yano K (2004) Pterogobius elapoides, Pterogobius zonoleucus. In: Senou H (ed) A photographic guide to the gobioid fishes of Japan. Heibonsha, Tokyo, pp 231, 233 (in Japanese)
Tada R (1994) Paleoceanographic evolution of the Japan Sea. Palaeogeogr, Palaeoclimatol, Palaeoecol 108:487–508
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Tamura K, Tao Q, Kumar S (2018) Theoretical foundation of the RelTime method for estimating divergence times from variable evolutionary rates. Mol Biol Evol 35:1770–1782
Toews DP, Brelsford A (2012) The biogeography of mitochondrial and nuclear discordance in animals. Mol Ecol 21:3907–3930
Tringali MD, Bert TM, Seyoum S, Bermingham E, Bartolacci D (1999) Molecular phylogenetics and ecological diversification of the transisthmian fish genus Centropomus (Perciformes: Centropomidae). Mol Phylogenet Evol 13:193–207
Tsutsumi T, Dotsu Y (1961) The reproductive behavior in the gobiid fish, Pterogobius zonoleucus Jordan et Snyder. Bull Fac Fish, Nagasaki Univ 10:149–155 (in Japanese)
Watanabe K, Mori S, Nishida M (2003) Genetic relationships and origin of two geographic groups of the freshwater threespine stickleback, 'Hariyo'. Zool Sci 20:265–274
Wessel P, Smith WH, Scharroo R, Luis J, Wobbe F (2013) Generic mapping tools: improved version released. Eos Trans A G U 94:409–410
Wilson AB, Veraguth EI (2010) The impact of Pleistocene glaciation across the range of a widespread European coastal species. Mol Ecol 19:4535–4553
Yamasaki YY, Kakioka R, Takahashi H, Toyoda A, Nagano AJ, Machida Y, Møller PR, Kitano J (2020) Genome-wide patterns of divergence and introgression after secondary contact between Pungitius sticklebacks. Philos Trans R Soc Lond B Biol Sci 375:20190548
Zhang Y, Ryder O (1995) Different Rates of Mitochondrial DNA Sequence Evolution in Kirk's Dik-dik (Madoqua-kirkii) Populations. Mol Phylogenet Evol 4:291–297
Zhang C, Montooth KL, Calvi BR (2017) Incompatibility between mitochondrial and nuclear genomes during oogenesis results in ovarian failure and embryonic lethality. Development 144:2490–2503
Acknowledgements
I sincerely thank Seishi Kimura and other organizing committee members of the Commemoration of the 50th Anniversary of the Ichthyological Society of Japan for providing me with an opportunity for writing this review article. I am grateful to Carol A. Stepien and Kiyoshi Kikuchi for their constructive feedback and language improvements. I am grateful to Shuya Kato for providing me with the photos of the five gobies. I would like to thank all my collaborators. This work was supported by the Japan Society for the Promotion of Science (KAKENHI 18H02493). Publication of this article was also supported by the Commemoration of the 50th Anniversary of the Ichthyological Society of Japan Society. I would like to thank the editor and two anonymous reviewers for their helpful comments on earlier versions of this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Hirase, S. Comparative phylogeography of coastal gobies in the Japanese Archipelago: future perspectives for the study of adaptive divergence and speciation. Ichthyol Res 69, 1–16 (2022). https://doi.org/10.1007/s10228-021-00824-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10228-021-00824-3