Abstract
Background
To determine the renal and cardiovascular prognosis and all-cause mortality of Japanese patients with type 2 diabetes showing a reduced estimated glomerular filtration rate (eGFR) without albuminuria.
Methods
A population of 675 patients with type 2 diabetes was prospectively observed for 4 years to determine the renal and cardiovascular outcomes and mortality. The subjects were divided into the four groups: those with a preserved eGFR and no albuminuria (n = 306), a preserved eGFR and albuminuria (n = 151), a reduced eGFR and no albuminuria (n = 96), and a reduced eGFR and albuminuria (n = 122). The Cox proportional hazard model and Fine and Gray method were used to assess between-group differences in the risk of mortality and cardiovascular events.
Results
In the group with a reduced eGFR, the eGFR value did not significantly change in the subjects without albuminuria (0 ± 8 mL/min/1.73 m2), whereas it decreased continuously in those with albuminuria (-6 ± 12 mL/min/1.73 m2). The incidence of cardiovascular events was significantly (P = 0.03) higher in the subjects with albuminuria (17%) than those without albuminuria (7%) in the group with a reduced eGFR. Cardiovascular events were significantly (P < 0.01) more frequent in the group with a reduced eGFR than in those with a preserved eGFR in both subjects with and without albuminuria.
Conclusions
The risk of end-stage kidney disease in non-albuminuric subjects with a reduced eGFR is considered to be low. We should focus on cardiovascular prognosis, because these patients are still at high risk of cardiovascular events, even though the prognosis is better in comparison to albuminuric patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetic nephropathy has been staged based on the degree of urinary albumin excretion [1], and it is well known that the cardiovascular mortality increases with the progression of albuminuria in patients with type 2 diabetes [2]. However, subjects who showed a decrease in the glomerular filtration rate (GFR) without albuminuria were often identified among type 2 diabetic patients, including in Japanese population [3,4,5]. Vascular and/or tubulointerstitial lesions in addition to glomerular injury are pathologically observed in such normoalbuminuric patients with a reduced GFR [6, 7]. Despite decreases in the number of diabetic subjects showing typical nephropathy graded by albuminuria, the number of patients with renal dysfunction without albuminuria have been increasing with the aging of subjects and advances in diabetes treatment [3, 8, 9].
It was reported that approximately 20% of the Japanese adult population have an estimated GFR (eGFR) of ≤ 30 mL/min/1.73 m2, and this prevalence was estimated to be more than double that in the general population of the United States [10]. The eGFR was directly correlated with the number of glomeruli without sclerosis based on a study of autopsied kidneys, and the number of nephrons was found to be lower in Japanese patients, regardless of the presence of hypertension or chronic kidney disease (CKD), than in Caucasian subjects [11]. Therefore, it seems that there will be more opportunities in the future to treat type 2 diabetic patients showing a reduced eGFR in Japan, where the proportion of diabetes is increasing among the elderly [12].
We previously reported that diabetic macroangiopathies were more frequently found, independently of albuminuria, in Japanese patients with a reduced eGFR than in those with a normal eGFR according to a cross-sectional study [13]. Furthermore, the intima-media thickness of the carotid artery, a surrogate marker of atherosclerosis, was increased [14], and the flow-mediated dilatation, a clinical parameter for detecting the initial stage of atherosclerosis, was decreased [15] in normoalbuminuric patients with a reduced eGFR compared to those with a normal eGFR.
In summary, it is considered necessary to prevent the progression of atherosclerosis as well as renal dysfunction in the treatment of type 2 diabetic patients showing a reduced eGFR without albuminuria. Although diabetic kidney disease (DKD), the definition of which was recently proposed [3], is a concept including atypical renal impairment as described above, clinical studies investigating type 2 diabetic patients with a reduced GFR without albuminuria are still insufficient, especially in Asian population.
We, therefore, conducted a prospective observational study to determine the renal and cardiovascular prognosis in Japanese patients with type 2 diabetes showing a reduced eGFR without albuminuria. We believe that the accumulation of results from such studies will contributes to the development of a new strategy for treating patients with the DKD.
Methods
Study population and outcomes
Initially, 788 patients with type 2 diabetes who were registered for a previous cross-sectional study [16] were prospectively observed. All patients were evaluated for the eGFR, urinary albumin-to-creatinine ratio (uACR) in random spot urine samples, and diabetic macroangiopathies in the Department of Diabetes, Metabolism and Kidney Disease of Edogawa Hospital between August and November 2014. After excluding the subjects receiving the administration of sodium-glucose cotransporter 2 (SGLT2) inhibitors (n = 10) and glucagon-like peptide-1 (GLP-1) receptor agonists (n = 1), that are considered to improve renal and cardiovascular prognosis [17,18,19,20], we analyzed 675 patients who had been observed for more than 1 year as the study subjects for the present cohort.
The study subjects were divided into the following 4 groups: those with (1) a preserved eGFR (≥ 60 mL/min/1.73 m2) and no albuminuria (uACR < 30 mg/gCr), (2) a preserved eGFR and albuminuria (uACR ≥ 30 mg/gCr), (3) a reduced eGFR (< 60 mL/min/1.73 m2) and no albuminuria, and (4) a reduced eGFR and albuminuria.
We continuously treated the study subjects for 4 years until the end of 2018 or death. The outcomes for the present study were the change in the eGFR, incidence of end-stage kidney disease (ESKD), cardiovascular events (cardiovascular death, nonfatal myocardial infarction, coronary interventions, or nonfatal stroke), and all-cause mortality.
Confounding factors
Diabetes mellitus was diagnosed when the subject met one of the following criteria at the past hospital visit: fasting blood glucose ≥ 7.0 mmol/L (126 mg/dL), random blood glucose ≥ 11.1 mmol/L (200 mg/dL), HbA1c ≥ 6.5% (48 mmol/mol), or the use of antidiabetic agents. Obese individuals were defined as those with a body mass index (BMI) of ≥ 25 kg/m2 according to the criteria set by the Japan Society for the Study of Obesity [21]. A current drinker was defined as a person consuming > 20 g ethanol equivalent/day. Hypertension was defined as a systolic blood pressure ≥ 140 mmHg and/or a diastolic blood pressure ≥ 90 mmHg. The participants currently using antihypertensive medications were also classified as positive for hypertension. Hyper-LDL-cholesterolemia was defined as either a serum concentration of LDL-cholesterol ≥ 3.62 mmol/L (140 mg/dL) or the current use of statins or ezetimibe. Hypo-HDL-cholesterolemia was defined as a serum concentration of HDL-cholesterol < 1.03 mmol/L (40 mg/dL). The eGFR was calculated using the formula which is recommended by the Japanese Society of Nephrology [22]. Albuminuria was defined as a uACR ≥ 30 mg/gCr. Hyperuricemia was defined by a serum uric acid level > 416 μmol/L (7 mg/dL) or as patients using urate-lowering agents. Diabetic retinopathy, peripheral neuropathy, cerebrovascular disease, coronary heart disease and peripheral artery disease were diagnosed as previously described [5, 13,14,15].
Statistical analyses
All data are presented as the mean ± standard deviations. The χ2 test was used for between-group comparisons of categorical variables. None of the continuous variables (age, duration of diabetes, BMI, blood pressure, serum lipid concentrations, HbA1c, eGFR, or uric acid) showed a normal distribution in the Shapiro–Wilk tests. Wilcoxon’s rank-sum test was, therefore, used to assess the significance of differences in the continuous variables. Wilcoxon’s signed-rank test was used to assess the significance of differences in the eGFR during the observation period in comparison to baseline values. A least squares model was used to evaluate the associations between the clinical background factors of the patients and the changes in the eGFR at the final evaluation during the observation period (ΔeGFR). The odds ratio (OR), and respective 95% confidence intervals (95% CIs) were determined to examine the strength of the relationship between the clinical characteristics of the patients and the incidence of ESKD, cardiovascular events, and all-cause mortality by a logistic regression analysis. Independent variables in the logistic regression analysis were determined according to forward stepwise selection. Factors that showed a significant association with the outcomes in a univariate analysis were included in a multivariable analysis. The Cox proportional hazard model was used to assess between-group differences in the risk of all-cause mortality. The risk of cardiovascular events was assessed by the Fine and Gray method for analyzing competing risks data [23]. P values of < 0.05 (two-tailed) were considered to indicate statistical significance. Data analyses were performed using the statistical software package JMP version 12.2.0 (SAS Institute, Cary, NC, USA), and analyses including competing risk were performed with EZR version 1.42 (Saitama Medical Center, Jichi Medical University, Saitama, Japan), which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics and clinical features associated with a reduced eGFR without albuminuria
The clinical characteristics of the subjects at the baseline are shown in Table 1. Subjects showing a reduced eGFR and those without albuminuria were found in 32 and 59%, respectively. In the group showing a reduced eGFR without albuminuria, the patients were older and had a longer duration of diabetes than in the group showing a preserved eGFR. Hypertension, hyperuricemia, cerebrovascular disease, and coronary heart disease were also more frequent in this group than in the group showing a preserved eGFR. Diabetic retinopathy and peripheral neuropathy were less frequent in this group than in the group showing a reduced eGFR with albuminuria.
Outcomes
The eGFR value continuously decreased during the observation period in whole subjects (Fig. 1a) and the group showing a preserved eGFR (Fig. 1b). In the group showing a preserved eGFR, the ΔeGFR was significantly (P = 0.01) greater in subjects with albuminuria (− 10 ± 14 mL/min/1.73 m2) than in those without albuminuria (− 6 ± 11 mL/min/1.73 m2). In the group showing a reduced eGFR, the eGFR did not significantly change in the subjects without albuminuria, whereas it decreased continuously in those with albuminuria (Fig. 1c). The ΔeGFR was significantly (P < 0.01) greater in the subjects with albuminuria (− 6 ± 12 mL/min/1.73 m2) than in those without albuminuria (0 ± 8 mL/min/1.73 m2).
In whole subjects, the cumulative incidence of ESKD, cardiovascular events, and all-cause mortality during the observation period was 2% (n = 14), 5% (n = 35), and 5% (n = 35), respectively. All patients who developed ESKD were in the group showing a reduced eGFR with albuminuria. Among the group showing a reduced eGFR, the incidence of ESKD was significantly (P < 0.01) more frequent in the subjects with albuminuria than in those without albuminuria.
The level of uACR continuously increased during the observation period in whole subjects (Fig. 2a) and the group showing a reduced eGFR (Fig. 2c). In the group showing a preserved eGFR, the uACR did not significantly change in the subjects with albuminuria, whereas it increased continuously in those without albuminuria (Fig. 2b).
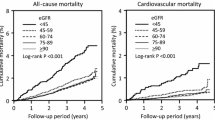
Although the incidence of cardiovascular events was not significantly different between the subjects with (n = 4, 3%) and without albuminuria (n = 3, 1%) in the group showing a preserved eGFR (Fig. 3a), it was significantly (P = 0.03) higher in the subjects with albuminuria (n = 21, 17%) than in those without albuminuria (n = 7, 7%) in the group showing a reduced eGFR (Fig. 3b). Cardiovascular events were significantly more frequent in the group showing a reduced eGFR than in those showing a preserved eGFR in both subjects with and without albuminuria (P < 0.01 and P < 0.01, respectively).
A Kaplan–Meier analysis of cardiovascular events and all-cause mortality in the group showing a preserved eGFR (a) and the group showing a reduced eGFR (b). Solid and dashed lines indicate the subjects with and without albuminuria, respectively. *P = 0.06 and **P < 0.01 vs. corresponding subjects of eGFR ≥ 60 mL/min/1.73 m2 group
The all-cause mortality was not significantly different between the subjects with (n = 3, 2%) and without albuminuria (n = 9, 3%) in the group showing a preserved eGFR (Fig. 3a). Similarly, it was not significantly different between the subjects with (n = 16, 13%) and without albuminuria (n = 7, 7%) in the group showing a reduced eGFR (Fig. 3b). The all-cause mortality was significantly (P < 0.01) higher in the group showing a reduced eGFR than in those showing a preserved eGFR in the subjects with albuminuria. It also showed a tendency to be higher (P = 0.06) in the group showing a reduced eGFR than in those with a preserved eGFR in the subjects without albuminuria.
Risk factors for outcomes
In single regression models, the smoking history, HbA1c level, eGFR, albuminuria, serum LDL-cholesterol concentration, and presence of diabetic retinopathy at the baseline were significantly associated with the ΔeGFR (Table 2). The smoking history, eGFR, albuminuria, serum LDL-cholesterol concentration, and presence of diabetic retinopathy were significantly independent variables associated with the ΔeGFR according to a multiple regression analysis. Statistical analyses stratified by albuminuria showed that the eGFR and LDL-cholesterol concentration were independently associated with the ΔeGFR in subjects without albuminuria. The eGFR was the only value that was significantly associated with the ΔeGFR in subjects with albuminuria (Table S1 in the Supplementary Appendix). HbA1c, albuminuria and peripheral artery disease were significantly associated with the ΔeGFR in the group showing a preserved eGFR. Patient age, albuminuria and diabetic retinopathy were significantly associated with the ΔeGFR in the group showing a reduced eGFR (Table S2).
In univariate logistic regression analyses, the patient’s age, drinking habit, biguanides use, renin–angiotensin system (RAS) inhibitors use, eGFR, albuminuria, hypo-HDL-cholesterolemia, hyperuricemia, and presence of diabetic retinopathy at the baseline were significantly associated with the incidence of cardiovascular events (Table 3). Only the eGFR was significantly associated with the incidence of cardiovascular events according to a multivariable logistic regression analysis. Statistical analyses stratified by albuminuria showed that hypo-HDL-cholesterolemia was significantly associated with the incidence of cardiovascular events in subjects without albuminuria. The eGFR and hyperuricemia were significantly associated with the incidence of cardiovascular events in subjects with albuminuria (Table S3). Only the serum HDL-cholesterol concentration was significantly associated with the incidence of cardiovascular events in the group showing a preserved eGFR. Albuminuria was independently associated with the incidence of cardiovascular events in subjects showing a reduced eGFR (Table S4). Furthermore, male, age and use of biguanides were significantly associated with the incidence of all-cause mortality (Table 3). Statistical analyses stratified by albuminuria showed that the BMI was significantly associated with mortality in the subjects without albuminuria. Patient age, use of biguanides, HbA1c, and hypo-HDL-cholesterolemia were significantly associated with mortality in subjects with albuminuria (Table S5). The BMI and use of RAS inhibitors were significantly associated with mortality in the group showing a preserved eGFR. Patient age was the only variable that was significantly associated with mortality in the group showing a reduced eGFR (Table S6).
Discussion
In the present prospective observation study for 4 years, the group showing a reduced eGFR without albuminuria demonstrated a significantly better prognosis for the renal function and cardiovascular events than in those showing a reduced eGFR with albuminuria. However, the incidence of cardiovascular events was higher in the group showing a reduced eGFR without albuminuria than in those showing a preserved eGFR, and the all-cause mortality was not significantly different between the group showing a reduced eGFR without and with albuminuria.
Because the prevalence of diabetic retinopathy and neuropathy were significantly lower in the group showing a reduced eGFR without albuminuria than in those with albuminuria, renal dysfunction in the former group is considered to have mainly been caused by factors other than those specific to classical diabetic nephropathy [24]. Ekinci et al. reported that structural changes caused by aging, hypertension, and arteriosclerosis were dominant in type 2 diabetic patients with normoalbuminuria and renal impairment (eGFR < 60 mL/min/1.73 m2) based on renal biopsy specimens [25]. Moriya et al. also reported that 29, 62, and 20 specimens obtained by a renal biopsy showed a normal pattern, typical diabetic glomerulopathy, and tubulointerstitial/vascular injury with no/mild glomerulopathy, respectively, among 111 Japanese patients (59 normoalbuminuria and 52 microalbuminuria) with type 2 diabetes [26]. Shimizu and Wada et al. retrospectively investigated the association of renal pathology with the long-term (approximately 8 years) outcomes in Japanese patients with type 2 diabetes. Nodular and/or exudative lesions were frequently observed in the kidneys obtained from the patients with typical diabetic nephropathy. During the follow-up period, macroalbuminuria was closely related to a high incidence of renal events (requirement of dialysis or 50% decline in the eGFR from baseline) and all-cause mortality. In contrast, vascular and/or tubulointerstitial lesions were observed in normoalbuminuric patient with both a preserved (n = 24) and reduced eGFR (n = 14). The incidence of renal events and mortality were not significantly different between the two groups [6]. Although the present study included no information on renal histopathology, the outcomes do not conflict with that report. The strength of our study is that the prognosis was examined in a large number of patients with the detailed clinical characteristics.
The GFR is reportedly an independent risk factor for cardiovascular events and mortality in patients with type 2 diabetes. Rigalleau et al. reported that the risks for the progress of renal dysfunction and death were lower in type 2 diabetic patients showing a reduced GFR without albuminuria than in those with albuminuria [27]. It was also demonstrated that the incidence of cardiovascular events and mortality was significantly lower in the subjects showing a reduced eGFR without albuminuria than in those with albuminuria according to the multicenter trials conducted in Caucasians [28,29,30]. Similar results were obtained from the studies conducted in Chinese type 2 diabetic patients whose BMI values were markedly lower than those in Caucasians [31, 32]. Bouchi and Babazono et al. reported that a reduced eGFR was independently associated with the incidence of coronary heart disease in Japanese patients with type 2 diabetes [33]. According to the registration prospective multicenter cohort with an average follow-up of 7 years for 4328 patients with type 2 diabetes in Japan, the renal and cardiovascular events and all-cause mortality increased in albuminuric patients, and the risk of mortality increased in normoalbuminuric patients when the eGFR was less than 30 mL/min/1.73 m2 [34]. A meta-analysis of 31 cohort studies describing the risk of albuminuria and the eGFR for cardiovascular mortality, all-cause mortality, and renal events by Toyama et al. [35] showed that the risk of cardiovascular mortality tended to be 1.70-fold higher in diabetic subjects showing a reduced eGFR without albuminuria than in those showing a preserved eGFR. Although the all-cause mortality was significantly higher by 1.42-fold in diabetic subjects showing a reduced eGFR without albuminuria than in those showing a preserved eGFR, the incidence of renal events was not significantly different between the two groups. Taking the above into consideration, medical intervention to prevent the development of cardiovascular events is considered to be more important than the prognosis of renal function in type 2 diabetic patients showing a reduced eGFR without albuminuria.
Recently, Yamanouchi and Wada et al. reported the renal prognosis and mortality in Japanese patients with type 2 diabetes and a reduced eGFR (< 60 mL/min/1.73 m2) without proteinuria (uACR < 300 mg/gCr) according to a nationwide, biopsy-based cohort study [7]. The baseline characteristics were evaluated at the time of renal biopsy for patients managed from 1985 to 2016. The mortality rate in the proteinuric group was significantly higher than that in the non-proteinuric group, although this incidence showed a similar trend without reaching statistical significance in our study. Patient age, BMI and the rate of statin usage were obviously higher in our study in comparison to previous studies performed in Japan [6, 7, 33, 34]. Aging and weight gain are recent clinical characteristics of Japanese patients with type 2 diabetes [12, 36]. These different baseline characteristics might have been responsible for the difference in the result of the current study.
In the treatment of the type 2 diabetic patients showing a reduced eGFR without albuminuria, the management of the risk factors is considered to be similarly necessary to typical diabetes care. The efficacy of intensified multifactorial intervention for preventing cardiovascular events and all-cause mortality was shown in previous trials, such as the Steno-2 study [37] and the J-DOIT study [38]. In particular, intervention to manage a smoking habit, albuminuria, and serum LDL-C concentration, all of which were significantly associated with the ΔeGFR in the present study, might be important for improving the renal prognosis. Based on our results, maintaining the eGFR is expected to prevent cardiovascular events. It is established that RAS inhibitors protect the renal function in patients with diabetes. Furthermore, it has been recently reported that SGLT2 inhibitors and GLP-1 receptor agonists exert renoprotective effects in addition to preventing cardiovascular events in patients with type 2 diabetes [17,18,19], even among those with renal impairment [19, 20]. Because SGLT2 inhibitors and GLP-1 receptor antagonists were not used in the present study, the effects of these drugs should be examined in the type 2 diabetic patients showing a reduced eGFR without albuminuria in another study.
Several limitations associated with the present study warrant mention. First, the present study was unable to investigate the effects of changing treatment methods, including antidiabetic, antihypertensive, lipid-lowering agents, during the observation period. The changes in the clinical parameters such as the body weight, uACR, HbA1c value, blood pressure, and serum lipid concentration were also not evaluated. It should be noted that the current study demonstrated only the prognosis of the groups defined by the eGFR and albuminuria at the baseline, although the clinical course differed among individuals depending on the treatment method and treatment response. Second, type 2 diabetic patients showing a reduced eGFR without albuminuria, who were the main targets of the present study, are heterogeneous. For example, this population includes subjects with nephrosclerosis caused by aging, hypertension, and arteriosclerosis; subjects with tubulointerstitial injury; and subjects with remission of albuminuria thanks to successful treatment [39]. Because more than half of the subjects received RAS inhibitors or calcium channel blockers administration at the baseline in the present study, there might have been many patients whose albuminuria caused by diabetic glomerulopathy disappeared thanks to treatment modification. If this is indeed the case, our study results thereby also confirm the findings of previous trials showing that RAS inhibitors obtained good renal and cardiovascular prognoses in patients with type 2 diabetes by reducing proteinuria [40,41,42]. Third, the measurement of uACR was based on a single spot urine sample obtained when the patients visited our department. Variability should be considered when confirming the DKD group with staging by uACR because it has been recommended that evaluations should be performed in two or more samples. Fourth, the eGFR was calculated by the formula that is recommended by the Japanese Society of Nephrology [22] but was not measured using inulin clearance, which is the gold standard for determining the GFR [43]. Because the eGFR was calculated using the serum creatinine concentration, it should be noted that the kidney function may be overestimated in elderly individuals with a reduced muscle mass. Despite these limitations, however, we believe that this cohort study is useful for managing patients presenting with atypical clinical feature in DKD.
Conclusion
Preventing the development of cardiovascular events is considered to be more important than the prognosis of the renal function in type 2 diabetic patients showing a reduced eGFR without albuminuria.
References
Mogensen CE. Microalbuminuria, blood pressure and diabetic renal disease: origin and development of ideas. Diabetologia. 1999;42:263–85.
Adler AI, Stevens RJ, Manley SE, Bilous RW, Cull CA, Holman RR, UKPDS GROUP. Development and progression of nephropathy in type 2 diabetes: the United Kingdom Prospective Diabetes Study (UKPDS 64). Kidney Int. 2003;63:225–32.
de Boer IH, Rue TC, Hall YN, Heagerty PJ, Weiss NS, Himmelfarb J. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA. 2011;305:2532–9.
Yokoyama H, Sone H, Oishi M, Kawai K, Fukumoto Y, Kobayashi M. Prevalence of albuminuria and renal insufficiency and associated clinical factors in type 2 diabetes: the Japan Diabetes Clinical Data Management study (JDDM15). Nephrol Dial Transplant. 2009;24:1212–9.
Ito H, Oshikiri K, Mifune M, Abe M, Antoku S, Takeuchi Y, Togane M, Yukawa C. The usefulness of the revised classification for chronic kidney disease by the KDIGO for determining the frequency of diabetic micro- and macroangiopathies in Japanese patients with type 2 diabetes mellitus. J Diabetes Complications. 2012;24:286–90.
Shimizu M, Furuichi K, Toyama T, Kitajima S, Hara A, Kitagawa K, Iwata Y, Sakai N, Takamura T, Yoshimura M, Yokoyama H, Kaneko S, Wada T, Kanazawa Study Group for Renal Diseases, and Hypertension. Long-term outcomes of Japanese type 2 diabetic patients with biopsy-proven diabetic nephropathy. Diabetes Care. 2013;36:3655–62.
Yamanouchi M, Furuichi K, Hoshino J, Toyama T, Hara A, Shimizu M, Kinowaki K, Fujii T, Ohashi K, Yuzawa Y, Kitamura H, Suzuki Y, Sato H, Uesugi N, Hisano S, Ueda Y, Nishi S, Yokoyama H, Nishino T, Samejima K, Kohagura K, Shibagaki Y, Mise K, Makino H, Matsuo S, Ubara Y, Wada T, Research Group of Diabetic Nephropathy, the Ministry of Health, Labour, and Welfare, and the Japan Agency for Medical Research, and Development. Nonproteinuric versus proteinuric phenotypes in diabetic kidney disease: a propensity score-matched analysis of a nationwide, biopsy-based cohort study. Diabetes Care. 2019;42:891–902.
Afkarian M, Zelnick LR, Hall YN, Heagerty PJ, Tuttle K, Weiss NS, de Boer IH. Clinical manifestations of kidney disease among US adults with diabetes, 1988–2014. JAMA. 2016;316:602–10.
Kume S, Araki SI, Ugi S, Morino K, Koya D, Nishio Y, Haneda M, Kashiwagi A, Maegawa H. Secular changes in clinical manifestations of kidney disease among Japanese adults with type 2 diabetes from 1996 to 2014. J Diabetes Investig. 2019;10:1032–40.
Imai E, Horio M, Iseki K, Yamagata K, Watanabe T, Hara S, Ura N, Kiyohara Y, Hirakata H, Moriyama T, Ando Y, Nitta K, Inaguma D, Narita I, Iso H, Wakai K, Yasuda Y, Tsukamoto Y, Ito S, Makino H, Hishida A, Matsuo S. Prevalence of chronic kidney disease (CKD) in the Japanese general population predicted by the MDRD equation modified by a Japanese coefficient. Clin Exp Nephrol. 2007;11:156–63.
Kanzaki G, Puelles VG, Cullen-McEwen LA, Hoy WE, Okabayashi Y, Tsuboi N, Shimizu A, Denton KM, Hughson MD, Yokoo T, Bertram JF. New insights on glomerular hyperfiltration: a Japanese autopsy study. JCI Insight. 2017;2:e94334.
Ministry of Health, Labor and Welfare. In: The National Health and Nutrition Survey in Japan, 2017. 2018. https://www.mhlw.go.jp/content/000451755.pdf, Accessed 1 July 2020.
Ito H, Takeuchi Y, Ishida H, Antoku S, Abe M, Mifune M, Togane M. High frequencies of diabetic micro- and macroangiopathies in patients with type 2 diabetes mellitus with decreased estimated glomerular filtration rate and normoalbuminuria. Nephrol Dial Transplant. 2010;25:1161–7.
Ito H, Komatsu Y, Mifune M, Antoku S, Ishida H, Takeuchi Y, Togane M. The estimated GFR, but not the stage of diabetic nephropathy graded by the urinary albumin excretion, is associated with the carotid intima-media thickness in patients with type 2 diabetes mellitus: a cross-sectional study. Cardiovasc Diabetol. 2010;9:18.
Ito H, Nakashima M, Meguro K, Furukawa H, Yamashita H, Takaki A, Yukawa C, Omoto T, Shinozaki M, Nishio S, Abe M, Antoku S, Mifune M, Togane M. Flow mediated dilatation is reduced with the progressive stages of glomerular filtration rate and albuminuria in type 2 diabetic patients without coronary heart disease. J Diabetes Res. 2015;2015:728127.
Ito H, Yamashita H, Nakashima M, Takaki A, Yukawa C, Matsumoto S, Omoto T, Shinozaki M, Nishio S, Abe M, Antoku S, Mifune M, Togane M. Current metabolic status affects urinary liver-type fatty-acid binding protein in normoalbuminuric patients with type 2 diabetes. J Clin Med Res. 2017;9:366–73.
American Diabetes Association. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes-2019. Diabetes Care. 2019;42(Suppl 1):S90–102.
Giugliano D, De Nicola L, Maiorino MI, Bellastella G, Esposito K. Type 2 diabetes and the kidney: insights from cardiovascular outcome trials. Diabetes Obes Metab. 2019;21:1790–800.
Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Furtado RHM, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Sabatine MS. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31–9.
Ito H, Matsumoto S, Izutsu T, Kusano E, Nishio S, Antoku S, Yamasaki T, Mori T, Togane M, Ando S, Tsugami E. Comparison of the changes in the factors associated with the renal prognosis of non-elderly and elderly subjects treated with empagliflozin- a retrospective observation study in Japanese patients with type 2 diabetes. Diabetes Metab Syndr Obes. 2019;12:1783–94.
Examination Committee of Criteria for ‘Obesity Disease’ in Japan; Japan Society for the Study of Obesity. New criteria for ‘obesity disease’ in Japan. Circ J. 2002;66:987–92.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, Yamagata K, Tomino Y, Yokoyama H, Hishida A, Collaborators developing the Japanese equation for estimated GFR. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
Luis-Rodríguez D, Martínez-Castelao A, Górriz JL, De-Álvaro F, Navarro-González JF. Pathophysiological role and therapeutic implications of inflammation in diabetic nephropathy. World J Diabetes. 2012;3:7–18.
Ekinci EI, Jerums G, Skene A, Crammer P, Power D, Cheong KY, Panagiotopoulos S, McNeil K, Baker ST, Fioretto P, Macisaac RJ. Renal structure in normoalbuminuric and albuminuric patients with type 2 diabetes and impaired renal function. Diabetes Care. 2013;36:3620–6.
Moriya T, Suzuki Y, Inomata S, Iwano M, Kanauchi M, Haneda M. Renal histological heterogeneity and functional progress in normoalbuminuric and microalbuminuric Japanese patients with type 2 diabetes. BMJ Open Diabetes Res Care. 2014;2:e000029.
Rigalleau V, Lasseur C, Raffaitin C, Beauvieux MC, Barthe N, Chauveau P, Combe C, Gin H. Normoalbuminuric renal-insufficient diabetic patients: a lower-risk group. Diabetes Care. 2007;30:2034–9.
Bruno G, Merletti F, Bargero G, Novelli G, Melis D, Soddu A, Perotto M, Pagano G, Cavallo-Perin P. Estimated glomerular filtration rate, albuminuria and mortality in type 2 diabetes: the Casale Monferrato study. Diabetologia. 2007;50:941–8.
Penno G, Solini A, Orsi E, Bonora E, Fondelli C, Trevisan R, Vedovato M, Cavalot F, Lamacchia O, Scardapane M, Nicolucci A, Pugliese G, Renal Insufficiency And Cardiovascular Events (RIACE) Study Group. Non-albuminuric renal impairment is a strong predictor of mortality in individuals with type 2 diabetes: the Renal Insufficiency And Cardiovascular Events (RIACE) Italian multicentre study. Diabetologia. 2018;61:2277–89.
Drury PL, Ting R, Zannino D, Ehnholm C, Flack J, Whiting M, Fassett R, Ansquer JC, Dixon P, Davis TM, Pardy C, Colman P, Keech A. Estimated glomerular filtration rate and albuminuria are independent predictors of cardiovascular events and death in type 2 diabetes mellitus: the Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study. Diabetologia. 2011;54:32–433.
So WY, Kong AP, Ma RC, Ozaki R, Szeto CC, Chan NN, Ng V, Ho CS, Lam CW, Chow CC, Cockram CS, Chan JC, Tong PC. Glomerular filtration rate, cardiorenal end points, and all-cause mortality in type 2 diabetic patients. Diabetes Care. 2006;29:2046–52.
Fung CS, Wan EY, Chan AK, Lam CL. Association of estimated glomerular filtration rate and urine albumin-to-creatinine ratio with incidence of cardiovascular diseases and mortality in Chinese patients with type 2 diabetes mellitus - a population-based retrospective cohort study. BMC Nephrol. 2017;18:47.
Bouchi R, Babazono T, Yoshida N, Nyumura I, Toya K, Hayashi T, Hanai K, Tanaka N, Ishii A, Iwamoto Y. Association of albuminuria and reduced estimated glomerular filtration rate with incident stroke and coronary artery disease in patients with type 2 diabetes. Hypertens Res. 2010;33:1298–304.
Wada T, Haneda M, Furuichi K, Babazono T, Yokoyama H, Iseki K, Araki S, Ninomiya T, Hara S, Suzuki Y, Iwano M, Kusano E, Moriya T, Satoh H, Nakamura H, Shimizu M, Toyama T, Hara A, Makino H, Research Group of Diabetic Nephropathy, Ministry of Health, Labour, and Welfare of Japan. Clinical impact of albuminuria and glomerular filtration rate on renal and cardiovascular events, and all-cause mortality in Japanese patients with type 2 diabetes. Clin Exp Nephrol. 2014;18:613–20.
Toyama T, Furuichi K, Ninomiya T, Shimizu M, Hara A, Iwata Y, Kaneko S, Wada T. The impacts of albuminuria and low eGFR on the risk of cardiovascular death, all-cause mortality, and renal events in diabetic patients: meta-analysis. PLoS ONE. 2013;8:e71810.
Japan Diabetes Clinical Data Management Study Group. https://jddm.jp/data/index-2018. Accessed 1 July, 2020.
Gaede P, Lund-Andersen H, Parving HH, Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N Engl J Med. 2008;358:580–91.
Ueki K, Sasako T, Okazaki Y, Kato M, Okahata S, Katsuyama H, Haraguchi M, Morita A, Ohashi K, Hara K, Morise A, Izumi K, Ishizuka N, Ohashi Y, Noda M, Kadowaki T, J-DOIT3 Study Group. Effect of an intensified multifactorial intervention on cardiovascular outcomes and mortality in type 2 diabetes (J-DOIT3): an open-label, randomised controlled trial. Lancet Diabetes Endocrinol. 2017;5:951–64.
Yokoyama H, Araki S, Honjo J, Okizaki S, Yamada D, Shudo R, Shimizu H, Sone H, Moriya T, Haneda M. Association between remission of macroalbuminuria and preservation of renal function in patients with type 2 diabetes with overt proteinuria. Diabetes Care. 2013;36:3227–333.
Parving HH, Lehnert H, Bröchner-Mortensen J, Gomis R, Andersen S, Arner P, Irbesartan in Patients with Type 2 Diabetes, and Microalbuminuria Study Group. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med. 2001;345:870–8.
Brenner BM, Cooper ME, de Zeeuw D, Keane WF, Mitch WE, Parving HH, Remuzzi G, Snapinn SM, Zhang Z, Shahinfar S, RENAAL Study Investigators. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med. 2001;345:861–9.
Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet. 2000;355:253–9.
Soveri I, Berg UB, Björk J, Elinder CG, Grubb A, Mejare I, Sterner G, Bäck SE, SBU GFR Review Group. Measuring GFR: a systematic review. Am J Kidney Dis. 2014;64:411–24.
Acknowledgements
The authors thank Tomoko Koyanagi in the secretarial section of Edogawa Hospital for her valuable help with data collection.
Funding
No funds were received in support of this work.
Author information
Authors and Affiliations
Contributions
HI contributed to conception, design, analysis, interpretation, writing first draft, editing, and final approval. SA contributed to reviewing drafts, editing, and final approval. TI, EK, SM, TY, TM and MT contributed to data collection and final approval.
Corresponding author
Ethics declarations
Conflict of interest
Hiroyuki Ito has received lecture fees from Eli Lilly Japan KK and Nippon Boehringer Ingelheim Co., Ltd.
Human and animal rights
The study was conducted in accordance with the principles expressed in the 2008 Declaration of Helsinki. The Ethics Committee of Edogawa Hospital approved the study protocol (approved number: 2015-18). The trial is registered on UMIN-CTR, identifier UMIN000038835.
Informed consent
The Ethics Committee of Edogawa Hospital waived the need for written informed consent because the data were analyzed anonymously for this analysis based on information stored in the hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Ito, H., Antoku, S., Izutsu, T. et al. The prognosis of subjects showing a reduced estimated glomerular filtration rate without albuminuria in Japanese patients with type 2 diabetes: a cohort study for diabetic kidney disease. Clin Exp Nephrol 24, 1033–1043 (2020). https://doi.org/10.1007/s10157-020-01935-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-020-01935-3