Abstract
Background
Surgical strategies for acute perforated diverticulitis with generalised peritonitis remain controversial. This study aimed to meta-analyse trials comparing primary resection and anastomosis (PRA) to Hartmann’s procedure (HP) for Hinchey III/IV diverticulitis.
Methods
A systematic literature search was conducted to identify observational studies and randomised control trials (RCTs) of patients with Hinchey III/IV diverticulitis undergoing sigmoidectomy that compared PRA to HP. The methodological quality of the included studies was assessed systematically (Newcastle–Ottawa, Jadad and Cochrane risk of bias scores) and a meta-analysis was performed.
Results
After removal of duplicates, 12 studies including 4 RCTs were identified. The analysis included 918 patients, of whom 367 (39.98%) underwent PRA. Both the initial stoma rate (risk ratio [RR] persistent stoma 0.43, 95% confidence interval [CI] 0.26, 0.71, p = 0.001; I2 = 99%, p < 0.0001) and the rate of permanent stoma after combining the first (emergency surgery) and second (stoma reversal) procedures were lower in the PRA group. There was no difference in in 30-day mortality; however, PRA resulted in a reduction in overall mortality as well as major complications after the initial operation (RR 0.67, 95% CI 0.46, 0.97, p = 0.03; I2 = 22%, p = 0.26), stoma reversal (RR 0.48, 95% CI 0.26, 0.92, p = 0.03; I2 = 0%, p = 0.58) and when combining both procedures (RR 0.67, 95% CI 0.51, 0.88, p = 0.005; I2 = 0%, heterogeneity p = 0.58). A subgroup analysis of stoma reversal rates using data from only RCTs were consistent (RR permanent stoma, 0.33, 95% CI 0.13, 0.85, p = 0.02; I2 = 77%, p = 0.004) with the findings of the overall analysis.
Conclusions
This meta-analysis demonstrates that PRA used in the management of haemodynamically stable patients with Hinchey grade III/IV diverticulitis leads to a lower overall persistent stoma rate, with reduced morbidity compared with the traditional management.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Colonic diverticula are a common gastrointestinal pathology in Western and industrialised societies [1, 2]. They occur in up to 70% of the population over 80 years of age [3] and are associated with a spectrum of clinical issues ranging from uncomplicated diverticulosis to complex diverticular disease with abscess, peritonitis, stricture/obstruction, fistula or haemorrhage [4,5,6,7,8,9,10]. The prevalence of diverticulitis was historically reported as > 20%. More recent data suggest that only 4% of patients with diverticulosis will develop acute complicated diverticulitis [11]. Acute diverticulitis with colonic perforation is associated with high morbidity and mortality [12,13,14,15]. The severity and subsequent management of acute perforated diverticulitis is most commonly classified according to the modified Hinchey staging system [16, 17].
There has been considerable change in and debate regarding the management of perforated diverticulitis with generalised peritonitis (Hinchey Stage III and IV). Historically, a Hartmann’s procedure (HP) was the gold standard approach for all such cases. However, this has been associated with considerable morbidity and mortality, and over half of the patients never have their stoma reversed [18, 19]. As a consequence, primary resection and anastomosis (PRA) of the colon with or without diverting loop ileostomy has been advocated as an alternative to HP for the management of Hinchey III/IV peritonitis [15, 20,21,22]. Alternatively, laparoscopic peritoneal lavage (LPL) has been proposed as a viable strategy to treat “select” patients with perforated colonic diverticulitis and purulent peritonitis (Hinchey III). A number of studies have suggested it to be associated with low morbidity, stoma, and mortality rates [23,24,25].
In recent years, there have been several observational studies comparing PRA to HP [26,27,28,29,30,31,32,33] and LPL to HP [34, 35] in terms of mortality and postoperative morbidity. However, there have been limited randomised data until now [36,37,38]. Contemporary meta-analyses have examined outcomes from well-conducted, modern randomised controlled trials (RCTs) of LPL and HP [39,40,41], but there has been little evidence of this quality comparing PRA to HP until recently [42,43,44,45]. In synthesis, LPL is associated with increased risk of reintervention compared to HP for Hinchey III diverticulitis. The lower rate of stoma reversal and higher rate of complications after HP suggest PRA may provide the optimal management of perforated diverticulitis.
Since these pooled analyses, a further RCT [46] and the largest observational study [47] comparing outcomes between PRA and HP have been published. Therefore, our aim was to compare the outcome of PRA versus HP in patients with perforated diverticulitis and generalised peritonitis (Hinchey III/IV) to further inform the debate regarding the optimal management of this challenging surgical emergency.
Materials and methods
This systematic review (CRD42019139160) was performed according to the guidelines of the preferred reporting items for systematic reviews and meta-analyses (PRISMA) [48] and meta-analyses and systematic reviews of observational studies (MOOSE) checklists [49]. Institutional review board approval was not required.
Search strategy
An electronic search for relevant publications was performed using the PubMed, Embase, Scopus, and the Cochrane Collaboration databases. Each of the relevant publication reference section and Google Scholar was also screened for other applicable publications. The function “related article” in PubMed was also used to identify articles. In addition, clinicaltrials.gov was searched for proposed or ongoing trials. The search of the databases was performed by combining the following search terms using the Boolean AND/OR operators: "Primary Anastomosis", “Resection”, "Hartmann's Procedure", "Hartmann Procedure", "Diverticulitis". Publications were limited to those published in the English language. All titles were initially screened, and appropriate abstracts were reviewed. The last date of search was June 31, 2019.
Inclusion criteria and exclusion criteria
To be included in the analysis, studies had to (1) have an RCT or a prospective or retrospective observational cohort study design 92) report the operative results on the “colon” or use the words “splenic flexure”, “descending colon” or “sigmoid colon’’ to describe the study group; (3) compare PRA and HP operations; (4) have at least ten patients in each treatment arm; (5) reported on at least one of the outcome measures mentioned below; and (6) contain a previously unreported group (where two studies were reported by the same institution, the analysis included the highest quality publication). Studies were excluded from the analysis if they: (1) reported on diseases other than diverticulitis of the colon; (2) did not report on the outcome measures mentioned below; or (3) contained a previously reported group. The historic three-step approach (diversion by colostomy, followed by colonic resection and closure of the colostomy) was excluded due to strong evidence for superiority of primary resection [21].
Outcomes of interest
The primary end points of this review were stoma formation, reversal and persistent stoma. Secondary outcomes were 30-day and overall mortality, and major morbidity. Specific morbidities including abdominal sepsis, anastomotic leak, wound infection and stoma complications were examined independently. Continuous outcomes including operating time, length of stay (LOS) in the intensive care unit (ICU) and length of inpatient stay were also recorded. Complications were assessed according to the widely used Clavien–Dindo classification [50]. Complications grades IIIa–IVb were defined as major complications.
Study selection and data extraction
The bibliographical details of all retrieved articles were stored in an Endnote file and duplicate records were removed. Two reviewers (ÉJR, OKR) independently assessed all identified citations and recorded details of selected studies including bibliography, study design, interventions, patient and outcome measures in a predefined form. In the case of disagreement, resolution was by a third reviewer (MK).
Risk of bias and quality assessment
An independent reviewer (BC) assessed the risk of bias in the included studies as either high, low or unclear, using the Cochrane’ Risk of bias’ tool [51]. With regard to differences between surgical strategies, the quality of the evidence was assessed according to the Grading of Recommendations Assessment, Development and Evaluation (GRADE) criteria [52]. The quality of RCTs and observational studies was also assessed using the Jadad criteria and the Newcastle–Ottawa scale (NOS), respectively. The quality score rating was determined for each publication, with a Jadad scale of 3 out of 5 and a NOS of 7 or more stars representing studies deemed to be of higher quality.
Statistical analysis
Statistical analysis was performed using Review Manager (RevMan) [Computer program] Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014. Outcome measures (mean and standard deviation and median and interquartile range) were recorded. Binary outcome data were reported as risk ratios (RR) and 95% confidence interval (95% CI) were estimated using the Mantel–Haenszel method. OR of < 1 favoured PRA. Weighted mean differences (WMD) were calculated for the effect size of PRA on continuous variables. Where means and standard deviations were not available, estimates were derived from study data by the methods described by Hozo et al. [53], as well as Luo et al. [54] and Wan et al. [55] for studies reporting interquartile range [55]. Pooled estimates of differences were calculated, using random effects models if necessary to account for potential interstudy heterogeneity. Heterogeneity was assessed by I-squared statistics (I2), with > 50% being considered as considerable heterogeneity. Sensitivity analyses were carried out where appropriate. p values < 0.050 were considered significant.
Results
Search overview
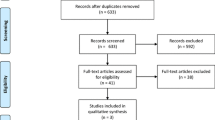
Figure 1 shows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flowchart of our search strategy. A total of 251 potential studies remained after the removal of duplicates. One hundred and thirty-two studies were removed after title and abstract screen. We identified 119 full-text articles of potential relevance, 107 of which were excluded for the following reasons: non-English language (n = 83); did not differentiate based on Hinchey stage (n = 14); did not pertain to perforated diverticulitis (n = 6); study other than RCT or observational study (n = 2); inadequate numbers (n = 2). Twelve studies [26,27,28,29,30,31,32, 46, 56,57,58,59], including 4 RCTs met the predefined inclusion criteria [46, 56,57,58].
Characteristics of included studies and patient population
Characteristics of included studies
The included studies were conducted in multiple centres throughout Europe and the USA, over a time period that ranged from 1990 to 2019. The number of participants in the included trials ranged from 40 (PRA n = 21 [52.5%] and n = 19 [47.5%]) to 130 (PRA n = 64 [49.2%] and HP n = 66 [50.8%]) patients. All the studies had an observational, retrospective [26, 29, 59] or prospective [27, 28, 30,31,32] cohort study design, except the aforementioned four RCTs [46, 56,57,58]. The RCTs had a Jadad score of 3 of 5, while in the non-randomised studies, NOS rating ranged from 6 to 9, and all but one of the studies were considered to have a high rating (NOS ≥ 7). The characteristics and findings of the included studies are shown in Tables 1, 2 and 3.
Characteristics of included patients
A total of 918 patients were included in the qualitative and quantitative analysis (Table 2). There were 367 patients (39.98%) in the PRA group as compared to 551 participants in the HP group (60%). The mean age of the groups was 63.8 ± 11.6 SD and 67.3 ± 11.6 SD in the PRA and HP groups, respectively, while the distribution of the sexes between the groups was broadly similar {sex, female (PRA n = 159 [43.32%] and HP n = 279 [50.64%])}. Of the studies, a significant proportion (44%) of the patients who underwent both procedures had an American Society of Anesthesiologists (ASA) score of III/IV, with a higher proportion of such patients in the HP group (PRA n = 65 [40%] and HP n = 212 [67.1%]). The rate of Hinchey IV patients within the included studies varied from 3.7 to 37%, with the HP group having a higher preponderance of Hinchey IV patients (PRA n = 48 [28.7%] and HP n = 123 [71.9%]). With regard to the degree of peritoneal contamination, the Manheim peritoneal index (MPI) was also higher in the HP group (PRA 20.5 ± 5.7 SD versus HP 21.7 ± 6.7 SD).
Characteristics of RCTs and included randomised patients
The four RCTs yielded a total of 387 patients for inclusion in the analysis (Table 4). The PRA group contained 181 patients (46.77%) compared to 206 HP patients (53.23%). The mean ages of the groups were 64.2 ± 11 SD and 64.3 ± 9.3 SD, respectively. Lambrichts et al. [46] was the only study to report the number of patients with an ASA score of III/IV, with a higher proportion of patients in the HP group (PRA n = 14 [24%] and HP n = 22 [37%]), although in general the HP group had higher ASA grades compared to the PRA group [46, 56,57,58]. The proportion of Hinchey IV patients within the RCTs varied from 16.7 to 30.1%, with the HP group having a higher preponderance of Hinchey IV patients (PRA n = 38 [20.99%] and HP n = 50 [24.27%]). The MPI was also higher in the HP group (PRA 21.2 ± 5.4 SD versus HP 20.8 ± 5.5 SD).
Primary outcome
Stoma reversal rates
There was a reduced incidence of stoma formation after the first operation (RR 0.43, 95% CI 0.26, 0.71, p = 0.001, I2 = 99%, p < 0.0001) (Fig. 2). The lower rate of patients with a stoma remained after combining the first and second procedures, and the stoma non-reversal rate was 11.8% in the PRA group and 37.3% in the HP group. The overall analysis shows that the RR of having a persistent stoma was 0.34 in favour of PRA (95% CI 0.24, 0.49, p < 0.001, I2 = 65%, p = 0.009) (Fig. 3).
Subgroup analysis of RCT data only
In contrast to the main analysis, there was no difference in RR with regard to initial stoma formation rate between the two procedures (RR 0.89, 95% CI 0.75, 1.06, p = 0.19, I2 = 93%, p = < 0.0001) (Fig. 4). However, the reduced incidence of persistent stoma with PRA remained consistent in this subgroup analysis. The stoma non-reversal rate was 13.7% in the PRA group and 37.5% in the HP group (RR .0.33, 95% CI 0.13, 0.85, p = 0.02, I2 = 77%, p = 0.004) (Fig. 5).
Binary secondary outcomes
The forest plot for the major morbidity and overall mortality after both operations (i.e. including stoma reversal) are shown in Figs. 6, 7, 8 and 9. The results for the remaining binary outcomes of the included studies are shown in Table 4.
Perioperative morbidity, reoperation and perioperative mortality rates of the first operation only
There was no difference in the postoperative morbidity (RR 0.9, 95% CI 0.68, 1.21, p = 0.5, I2 = 69%, p = 0.002) and 30-day mortality (RR 0.65, 95% CI 0.40, 1.06, p = 0.09, I2 = 0%, p = 0.97) between the groups. However, PRA was associated with a reduced incidence of major morbidity (RR 0.67, 95% CI 0.46, 0.97, p = 0.03, I2 = 22%, p = 0.26 [Fig. 6]) despite similar unplanned reoperation rates (RR 0.68, 95% CI 0.35, 1.31, p = 0.25, I2 = 0%, p = 0.95). The results remained similar when only RCT data were included.
Specific complication rates of the first operation only
There was a significant reduction in anastomotic leak/abdominal sepsis rates with PRA after the first operation (RR 0.38, 95% CI 0.21, 0.68, p = 0.001, I2 = 0%, p = 0.82) as compared to HP, and this reduction in anastomotic leak/abdominal sepsis (RR 0.27, 95% CI 0.11, 0.62, p = 0.002, I2 = 0%, p = 0.79) remained when including only data from RCTs. There was no difference in wound complications such as wound dehiscence (RR 1.19, 95% CI 0.60, 2.35, p = 0.63, I2 = 0%, p = 0.50) and SSI (RR 0.77, 95% CI 0.54, 1.11, p = 0.16, I2 = 0%, p = 0.57).
Perioperative morbidity for the stoma reversal operation
There was no difference in surgical site infection (SSI) rates in both the main (RR 0.75, 95% CI 0.28, 2.04, p = 0.57, I2 = 47%, p = 0.09) and subgroup analyses (RR 0.37, 95% CI 0.08, 1.64, p = 0.19, I2 = 16%, p = 0.30) for the stoma reversal procedure. PRA was associated with decreased major morbidity for the stoma reversal operation in comparison to HP (RR 0.54, 95% CI 0.34, 0.86, p = 0.009, I2 = 0%, p = 0.67 [Fig. 9]); however, there was no difference in the incidence of anastomotic leak/abdominal sepsis (RR 0.39, 95% CI 0.11, 1.40, p = 0.15, I2 = 0%, p = 0.74) compared to HP for the second operation alone, with both these outcomes being supplied by the RCT subgroup only.
Perioperative morbidity, reoperation and perioperative mortality rates after both (emergency and soma reversal) operations
While there was no difference in the overall morbidity (RR 1.11, 95% CI 0.84, 1.48, p = 0.45, I2 = 73%, p = 0.01) between the two procedures, PRA was associated with a reduced incidence of severe complications (RR 0.67, 95% CI 0.51, 0.88, p = 0.005, I2 = 0%, p = 0.58) and overall unplanned reoperation rates (RR 0.55, 95% CI 0.31, 0.98, p = 0.04, I2 = 0%, p = 1.0) when compared to HP. This translated into reduced overall mortality (RR 0.64, 95% CI 0.43, 0.93, p = 0.02, I2 = 29%, p = 0.17). However, these differences were lost in subgroup analysis of RCT data (Fig. 10).
Continuous secondary outcomes
Intraoperative and postoperative continuous data for both the initial operation and the combined operations, including derived data, are shown in Table 5.
Operating time, ICU stay and initial LOS of the first operation only
The pooled, weighted data demonstrated that while there was no significant difference in operating time (WMD, 0.30, 95% CI − 0.21, 0.81 min) or initial LOS (WMD, − 4.09, 95% CI − 8.36, 0.17 min), after the emergency procedure PRA was associated with a shorter ICU stay (WMD, − 1.00, 95% CI − 1.30, − 0.69 days). Subgroup analysis of RCT data only again demonstrated a reduced ICU stay (WMD, − 0.98, 95% CI − 1.29, − 0.67 days), but also a reduced inpatient LOS (WMD, − 1.87, 95% CI − 3.39, − 0.35 days) in favour of PRA.
Operating time and LOS in the second (stoma reversal) operation only
While there was no difference in operating time for stoma reversal between the two procedures in the main (WMD, − 1.25, 95% CI − 2.6, 0.10 mins) and subgroup analyses (WMD − 1.13, 95% CI − 2.85, 0.60, p = 0.2), PRA was associated with a shorter LOS (WMD, − 3.38, 95% CI − 5.53, − 1.23 days) compared to HP when using only data from the included RCTs.
Operating time, ICU stay and initial LOS of the first operation and second (stoma reversal) operation combined
There was no difference in overall operating time between the two procedures in the main (WMD − 0.90, 95% CI − 3.85, 2.05, p = 0.55) and subgroup analyses (WMD − 0.90, 95% CI − 3.85, 2.05, p = 0.55). However, PRA was associated with a shorter overall ICU stay (WMD 0.99, 95% CI 0.67, 1.31 days) and overall LOS (WMD − 1.83, 95% CI − 3.48, − 0.17, p = 0.03) compared to HP using data from the included RCTs.
Risk of bias
The grade criteria for the prespecified main outcomes are shown in Table 6. Using these criteria for RCT data only, the pooled data demonstrating a reduced rate of persistent stoma with PRA represents a high level of evidence. A risk of bias summary and graph for the included studies are included in Fig. 11. There was a low risk of reporting, attrition and other bias. Graphical exploration of the results with funnel plots did not demonstrate any remarkable asymmetry or publication bias (Fig. 12).
Discussion
The results of the present meta-analysis demonstrate that PRA leads to a significantly reduced incidence of initial stoma formation, as well as permanent stoma rate, and consequently stoma complications when compared to HP. There was also a reduced incidence of abdominal sepsis, major morbidity, and overall mortality rates compared with the standard management. Subgroup analyses using data from only RCTs of stoma reversal rates, major morbidity for stoma reversal, stoma complications and abdominal sepsis remained consistent with the findings of the overall analysis, and PRA was also associated with a shorter overall ICU stay and inpatient LOS (all level 1a evidence).
Eliminating the source of contamination and controlling sepsis are the immediate management steps in acute perforated diverticulitis. LPL may avoid the formation of a stoma in select cases [36,37,38,39]; however, due to the degree of contamination and the clinical status of the patient, many are left with an end colostomy following an HP. The extent of reversing an end colostomy and the morbidity, mortality and quality of life (QOL) issues associated with this are significant [60, 61]. This ultimately impacts stoma reversal rates, with > 50% of patients not having their stoma reversed after HP [18, 19]. Improved stoma reversal rates are encountered when a PRA and defunctioning loop ileostomy (DLI) are performed, with reversal rates as high as 90% reported [62]. This is consistent with the results of the present meta-analysis. Furthermore, the associated morbidity of ileostomy reversal is known to be lower than that of an end colostomy [63] and this is reflected in the finding of reduced major morbidity after stoma reversal with PRA.
While permanent stoma is associated with considerable morbidity [60, 61], there is little patient-reported outcome (PROM) data on QOL outcomes after HP reversal or PRA for diverticulitis, and restoration of intestinal continuity may be associated with under-appreciated morbidity. A short rectal stump (< 7.5 cm) appears to be the main independent risk factor for long-term persistent stoma after attempted HP reversal, and low anterior resection syndrome (LARS) may affect up to one-third of reversed patients [64]. Similarly, PRA patients should be warned about the risk of low anterior resection syndrome (LARS) and bowel dysfunction preoperatively [65]. Interestingly, the formation of a DLI does not appear to be associated with the development of LARS, suggesting this should be performed if clinically indicated [66]. In the present study, the DLI reversal rate was almost 90%%, as compared to approximately 60% stoma reversal rate in the HP group, although no data on the functional status of reversed patients were available. Greater PROM data are required to help inform management decisions, especially in patients that are at high risk for LARS, and to allow an increased appreciation of the patient’s perspective and what drives their decision-making processes.
Apart from stoma formation, the morbidity and mortality associated with Hinchey III/IV diverticulitis is significant [67]. Minimally invasive surgery helped to minimise morbidity, but studies examining its role in perforated diverticulitis have yielded conflicting results. While the clinical effectiveness of LPL seems to be equivalent, higher reintervention rates have been observed with a lavage approach [39]. However, this appears to be primarily due to interventional radiological drainage, and mortality at both 30 and 90 days is similar to HP. Historically, HP was associated with considerable morbidity and mortality (70% and 28% respectively) [31, 56]. Initial PRA study results demonstrated better rates of morbidity and mortality; however, patients were highly selective [68]. Pooling of data from RCTs in the present meta-analysis showed a significant reduction in major morbidity for stoma reversal with PRA. Surprisingly, the operative time for stoma reversal in the two groups was similar, as in the authors’ experience it takes longer to re-establish bowel continuity after HP compared to loop-ileostomy closure. However, PRA was associated with a reduced incidence of initial and overall abdominal sepsis. The explanation for this is unclear, but may be related to rectal stump leak in the HP group, as well as a higher incidence of anastomotic leak after HP reversal.
Decision making and guidelines on the best approach to the treatment of perforated diverticulitis have previously been limited to single centre retrospective studies, ultimately leading to selection bias [57]. With recent publication of RCTs on the various surgical techniques (HP, PRA, LPL) for Hinchey III/IV diverticulitis management, opinions on the best approach appear to be changing. Although the latest American Society of Colon and Rectal Surgeons guidelines still recommend an HP for the management of perforated diverticulitis, the European Association of Endoscopic Surgery is now favouring resection with PRA in select cases [69]. Despite this, many surgeons have been hesitant to perform PRA in the setting of peritoneal contamination for fear of anastomotic leakage. A recent study using the National Surgical Quality Improvement Program database from 2012 to 2016 found that a minority of patients undergoing an emergency sigmoidectomy for perforated diverticulitis have PRA (7.6% versus 92.4% receiving HP) [47] Unfortunately data from this, the largest observational study on the topic, could not be included in the analysis as no breakdown of included Hinchey grades was provided [47]. It must also be noted that the present study observed differences in a number of adverse features, such as higher ASA grade, Hinchey IV and MPI between the groups in the overall analysis, highlighting the selection biases inherent in many of the studies favouring PRA. Consequently, sound clinical judgement should be used regarding the appropriate operative approach for patients who are haemodynamically unstable and severely septic, because such patients were not included in the trials.
Our study has a number of limitations. The inclusion of non-randomised studies increases the risk of selection bias. However, the groups are broadly similar and sensitivity analysis, including analysis of RCT data exclusively, was undertaken to account for this heterogeneity [18, 40]. Despite this, it must be noted that all four of the RCTs were prematurely terminated due to logistical reasons and only about a third (n = 387/1218) of the proposed sample size was reached. Moreover, recruitment of patients was hindered by decision making and enrolling patients into a research trial in life-threatening situations, reluctance of surgeons to change practice and the implementation of less invasive surgical techniques [46, 56, 57, 70]. No study reported on the number of patients who refused consent or were deemed unfit for enrolment in the study suggesting surgeon influence was a factor. One RCT alluded to the interest of LPL while patients were being recruited for the study, with surgeons reluctant to randomise their patients into alternative management regimes [57]. Furthermore, although three of the four RCTs claimed that participants were randomised preoperatively, there is no mention of patients that went on to have other diagnoses, such as a lower Hinchey grade or malignancy as there was in the LPL RCTs, raising concerns of attrition bias. One RCT, while claiming consecutive enrolment, enrolled 68 of 90 patients within 2 years while the remaining patients from 14 centres were recruited over seven more years [58]. Binda et al. even highlighted the limitations of blinding and sample size in RCTs of emergency surgical presentations, concluding that conducting an RCT in the acute setting for diverticulitis was ‘practically unfeasible’ [58].
The issues highlighted above are extremely pertinent to all studies investigating the management of surgical emergencies. While RCTs provide the best level of evidence, they are not without significant limitations especially in the acute setting. It is unclear whether premature termination of the RCTs influenced their results and all were conducted at academic and referral centres. Larger hospitals may have the capacity to have specialist colorectal expertise on call [71], but this may not be possible in smaller regional/rural hospitals. Future prospective cohort studies with short- and long-term follow-up will help improve the understanding of the role of PRA in Hinchey III/IV diverticulitis. Any possible cost-effectiveness analyses arising from the DIVA arm of the LADIES Trial [46] and the recent RCT by Bridoux et al. [57] are also eagerly awaited. However, the conclusion of three RCTs and this as well as previous meta-analyses are favourable for PRA with regard to stoma reversal [42, 43]. Ultimately, careful patient selection, with clear discussion and counselling may increase the rates of PRA in the acute setting. This is likely to have a positive impact on patients’ QOL, by reducing permanent end-colostomy rates with reduced associated morbidity.
References
Bharucha AE, Parthasarathy G, Ditah I et al (2015) Temporal trends in the incidence and natural history of diverticulitis: a population-based study. Am J Gastroenterol 110(11):1589–1596
Etzioni DA, Mack TM, Beart RW Jr, Kaiser AM (2009) Diverticulitis in the United States: 1998–2005: changing patterns of disease and treatment. Ann Surg 249(2):210–217
Feingold D, Steele SR, Lee S et al (2014) Practice parameters for the treatment of sigmoid diverticulitis. Dis Colon Rectum 57(3):284–294
Sheth AA, Longo W, Floch MH (2008) Diverticular disease and diverticulitis. Am J Gastroenterol 103(6):1550–1556
Tursi A, Brandimarte G, Giorgetti G, Elisei W, Maiorano M, Aiello F (2008) The clinical picture of uncomplicated versus complicated diverticulitis of the colon. Dig Dis Sci 53(9):2474–2479
Stollman NH, Raskin JB (1999) Diagnosis and management of diverticular disease of the colon in adults. Ad Hoc Practice Parameters. Committee of the American College of Gastroenterology. Am J Gastroenterol 94(11):3110–3121
Hernandez-Guerrero A, Abdo-Francis J, Brito-Lugo P, Dela Torre-Bravo A, Marin-Pineda R, Stoopen-Rometti M (2008) Gastroenterology diagnosis and treatment guidelines of diverticular disease of the colon. Clinical and diagnostic. Rev Gastroenterol Mex 73(4):258–260
Andersen JC, Bundgaard L, Elbrond H, Laurberg S, Walker LR, Stovring J (2012) Danish national guidelines for treatment of diverticular disease. Dan Med J 59(5):C4453
Kruis W, Germer CT, Leifeld L (2014) Diverticular disease: guidelines of the German society for gastroenterology, digestive and metabolic diseases and the German society for general and visceral surgery. Digestion 90(3):190–207
Cuomo R, Barbara G, Pace F et al (2014) Italian consensus conference for colonic diverticulosis and diverticular disease. United Eur Gastroenterol J 2(5):413–442
Shahedi K, Fuller G, Bolus R et al (2013) Long-term risk of acute diverticulitis among patients with incidental diverticulosis found during colonoscopy. Clin Gastroenterol Hepatol 11(12):1609–1613
Stollman N, Smalley W, Hirano I (2015) American gastroenterological association institute guideline on the management of acute diverticulitis. Gastroenterology 149(7):1944–1949
Zorcolo L, Covotta L, Carlomagno N, Bartolo DC (2003) Safety of primary anastomosis in emergency colo-rectal surgery. Colorectal Dis 5(3):262–269
Bielecki K, Kaminski P, Klukowski M (2002) Large bowel perforation: morbidity and mortality. Tech Coloproctol 6(3):177–182
Krukowski ZH, Matheson NA (1984) Emergency surgery for diverticular disease complicated by generalized and faecal peritonitis: a review. Br J Surg 71(12):921–927
Hinchey EJ, Schaal PG, Richards GK (1978) Treatment of perforated diverticular disease of the colon. Adv Surg 12:85–109
Sher ME, Agachan F, Bortul M, Nogueras JJ, Weiss EG, Wexner SD (1997) Laparoscopic surgery for diverticulitis. Surg Endosc 11(3):264–267
Salem L, Anaya DA, Roberts KE, Flum DR (2005) Hartmann's colectomy and reversal in diverticulitis: a population-level assessment. Dis Colon Rectum 48(5):988–995
Constantinides VA, Heriot A, Remzi F et al (2007) Operative strategies for diverticular peritonitis: a decision analysis between primary resection and anastomosis versus Hartmann's procedures. Ann Surg 245(1):94–103
Dandekar NV, McCann WJ (1969) Primary resection and anastomosis in the management of perforation of diverticulitis of the sigmoid flexure and diffuse peritonitis. Dis Colon Rectum 12(3):172–175
Zeitoun G, Laurent A, Rouffet F et al (2000) Multicentre, randomized clinical trial of primary versus secondary sigmoid resection in generalized peritonitis complicating sigmoid diverticulitis. Br J Surg 87(10):1366–1374
Greif JM, Fried G, McSherry CK (1980) Surgical treatment of perforated diverticulitis of the sigmoid colon. Dis Colon Rectum 23(7):483–487
O'Sullivan GC, Murphy D, O'Brien MG, Ireland A (1996) Laparoscopic management of generalized peritonitis due to perforated colonic diverticula. Am J Surg 171(4):432–434
Myers E, Hurley M, O'Sullivan GC, Kavanagh D, Wilson I, Winter DC (2008) Laparoscopic peritoneal lavage for generalized peritonitis due to perforated diverticulitis. Br J Surg 95(1):97–101
Taylor CJ, Layani L, Ghusn MA, White SI (2006) Perforated diverticulitis managed by laparoscopic lavage. ANZ J Surg 76(11):962–965
Hold M, Denck H, Bull P (1990) Surgical management of perforating diverticular disease in Austria. Int J Colorectal Dis 5(4):195–199
Thaler K, Neumann F, Gero A, Kreuzer W (2000) Utility of appropriate peritonitis grading in the surgical management of perforated sigmoid diverticulitis. Colorectal Dis 2(6):359–363
Schilling MK, Maurer CA, Kollmar O, Buchler MW (2001) Primary vs. secondary anastomosis after sigmoid colon resection for perforated diverticulitis (Hinchey Stage III and IV): a prospective outcome and cost analysis. Dis Colon Rectum 44(5):699–703 (discussion 703–695)
Gooszen AW, Gooszen HG, Veerman W et al (2001) Operative treatment of acute complications of diverticular disease: primary or secondary anastomosis after sigmoid resection. Eur J Surg 167(1):35–39
Regenet N, Pessaux P, Hennekinne S et al (2003) Primary anastomosis after intraoperative colonic lavage vs. Hartmann's procedure in generalized peritonitis complicating diverticular disease of the colon. Int J Colorectal Dis 18(6):503–507
Vermeulen J, Akkersdijk GP, Gosselink MP et al (2007) Outcome after emergency surgery for acute perforated diverticulitis in 200 cases. Digest Surg 24(5):361–366
Trenti L, Biondo S, Golda T et al (2011) Generalized peritonitis due to perforated diverticulitis: Hartmann's procedure or primary anastomosis? Int J Colorectal Dis 26(3):377–384
Alizai PH, Schulze-Hagen M, Klink CD et al (2013) Primary anastomosis with a defunctioning stoma versus Hartmann's procedure for perforated diverticulitis—a comparison of stoma reversal rates. Int J Colorectal Dis 28(12):1681–1688
Liang S, Russek K, Franklin ME Jr (2012) Damage control strategy for the management of perforated diverticulitis with generalized peritonitis: laparoscopic lavage and drainage vs. laparoscopic Hartmann's procedure. Surg Endosc 26(10):2835–2842
Karoui M, Champault A, Pautrat K, Valleur P, Cherqui D, Champault G (2009) Laparoscopic peritoneal lavage or primary anastomosis with defunctioning stoma for Hinchey 3 complicated diverticulitis: results of a comparative study. Dis Colon Rectum 52(4):609–615
Angenete E, Thornell A, Burcharth J et al (2016) Laparoscopic lavage is feasible and safe for the treatment of perforated diverticulitis with purulent peritonitis: the first results from the randomized controlled trial DILALA. Ann Surg 263(1):117–122
Schultz JK, Yaqub S, Wallon C et al (2015) Laparoscopic lavage vs primary resection for acute perforated diverticulitis: the SCANDIV randomized clinical trial. JAMA 314(13):1364–1375
Vennix S, Musters GD, Mulder IM et al (2015) Laparoscopic peritoneal lavage or sigmoidectomy for perforated diverticulitis with purulent peritonitis: a multicentre, parallel-group, randomised, open-label trial. Lancet 386(10000):1269–1277
Marshall JR, Buchwald PL, Gandhi J et al (2017) Laparoscopic lavage in the management of Hinchey grade III diverticulitis: a systematic review. Ann Surg 265(4):670–676
Cirocchi R, Di Saverio S, Weber DG et al (2017) Laparoscopic lavage versus surgical resection for acute diverticulitis with generalised peritonitis: a systematic review and meta-analysis. Tech Coloproctol 21(2):93–110
Shaikh FM, Stewart PM, Walsh SR, Davies RJ (2017) Laparoscopic peritoneal lavage or surgical resection for acute perforated sigmoid diverticulitis: a systematic review and meta-analysis. Int J Surg 38:130–137
Acuna SA, Wood T, Chesney TR et al (2018) Operative strategies for perforated diverticulitis: a systematic review and meta-analysis. Dis Colon Rectum 61(12):1442–1453
Cirocchi R, Afshar S, Shaban F et al (2018) Perforated sigmoid diverticulitis: Hartmann's procedure or resection with primary anastomosis—a systematic review and meta-analysis of randomised control trials. Tech Coloproctol 22(10):743–753
Shaban F, Carney K, McGarry K, Holtham S (2018) Perforated diverticulitis: To anastomose or not to anastomose? A systematic review and meta-analysis. Int J Surg 58:11–21
Gachabayov M, Oberkofler CE, Tuech JJ, Hahnloser D, Bergamaschi R (2018) Resection with primary anastomosis vs nonrestorative resection for perforated diverticulitis with peritonitis: a systematic review and meta-analysis. Colorectal Dis 20(9):753–770
Lambrichts DPV, Vennix S, Musters GD et al (2019) Hartmann's procedure versus sigmoidectomy with primary anastomosis for perforated diverticulitis with purulent or faecal peritonitis (LADIES): a multicentre, parallel-group, randomised, open-label, superiority trial. Lancet Gastroenterol Hepatol 20:20
Lee JM, Bai PCJ, El Hechi M et al (2019) Hartmann's procedure vs primary anastomosis with diverting loop ileostomy for acute diverticulitis: nationwide analysis of 2,729 emergency surgery patients. J Am Coll Surg 20:20
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 339:b2535
Stroup DF, Berlin JA, Morton SC et al (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis of observational studies in epidemiology (MOOSE) group. JAMA 283(15):2008–2012
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Higgins JP, Altman DG, Gotzsche PC et al (2011) The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Guyatt G, Oxman AD, Akl EA et al (2011) GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol 64(4):383–394
Hozo SP, Djulbegovic B, Hozo I (2005) Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 5:13
Luo D, Wan X, Liu J, Tong T (2018) Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Stat Methods Med Res 27(6):1785–1805
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14(1):135
Oberkofler CE, Rickenbacher A, Raptis DA et al (2012) A multicenter randomized clinical trial of primary anastomosis or Hartmann's procedure for perforated left colonic diverticulitis with purulent or fecal peritonitis. Ann Surg 256(5):819–826 (discussion 826–817)
Bridoux V, Regimbeau JM, Ouaissi M et al (2017) Hartmann's procedure or primary anastomosis for generalized peritonitis due to perforated diverticulitis: a prospective multicenter randomized trial (DIVERTI). J Am Coll Surg 225(6):798–805
Binda GA, Karas JR, Serventi A et al (2012) Primary anastomosis vs nonrestorative resection for perforated diverticulitis with peritonitis: a prematurely terminated randomized controlled trial. Colorect Dis 14(11):1403–1410
Richter S, Lindemann W, Kollmar O, Pistorius GA, Maurer CA, Schilling MK (2006) One-stage sigmoid colon resection for perforated sigmoid diverticulitis (Hinchey stages III and IV). World J Surg 30(6):1027–1032
Downing A, Morris EJ, Richards M et al (2015) Health-related quality of life after colorectal cancer in England: a patient-reported outcomes study of individuals 12 to 36 months after diagnosis. J Clin Oncol 33(6):616–624
Banerjee S, Leather AJ, Rennie JA, Samano N, Gonzalez JG, Papagrigoriadis S (2005) Feasibility and morbidity of reversal of Hartmann's. Colorectal Dis 7(5):454–459
Aziz O, Constantinides V, Tekkis PP et al (2006) Laparoscopic versus open surgery for rectal cancer: a meta-analysis. Ann Surg Oncol 13(3):413–424
Tilney HS, Sains PS, Lovegrove RE, Reese GE, Heriot AG, Tekkis PP (2007) Comparison of outcomes following ileostomy versus colostomy for defunctioning colorectal anastomoses. World J Surg 31(5):1142–1151
Caille C, Collard M, Moszkowicz D, AlDJ P, Maggiori L, Panis Y (2019) Reversal of Hartmann's procedure in patients following failed colorectal or coloanal anastomosis: an analysis of 45 consecutive cases. Colorectal Dis 20:20
Levack MM, Savitt LR, Berger DL et al (2012) Sigmoidectomy syndrome? Patients' perspectives on the functional outcomes following surgery for diverticulitis. Dis Colon Rectum 55(1):10–17
Jimenez-Rodriguez RM, Segura-Sampedro JJ, Rivero-Belenchon I et al (2017) Is the interval from surgery to ileostomy closure a risk factor for low anterior resection syndrome? Colorectal Dis 19(5):485–490
Stollman N, Raskin JB (2004) Diverticular disease of the colon. Lancet 363(9409):631–639
Breitenstein S, Kraus A, Hahnloser D, Decurtins M, Clavien PA, Demartines N (2007) Emergency left colon resection for acute perforation: primary anastomosis or Hartmann's procedure? A case-matched control study. World J Surg 31(11):2117–2124
Wong WD, Wexner SD, Lowry A et al (2000) Practice parameters for the treatment of sigmoid diverticulitis—supporting documentation. The Standards Task Force. The American Society of Colon and Rectal Surgeons. Dis Colon Rectum 43(3):290–297
Binda G, Karas J, Serventi A, Bergamaschi R, Amato A (2011) Primary anastomosis versus Hartmann's for perforated diverticulitis: a randomized controlled trial [Journal: Conference Abstract]. Colorectal Dis 13:5. https://doi.org/10.1111/j.1463-1318.2011.02704.x
Boyd-Carson H, Doleman B, Herrod PJJ et al (2019) Association between surgeon special interest and mortality after emergency laparotomy. Br J Surg 106(7):940–948
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no external funding or conflict of interests to declare.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors. Institutional review board approval was not required due to the studies meta-analytic and retrospective nature.
Informed consent
For this type of study formal consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ryan, O.K., Ryan, É.J., Creavin, B. et al. Systematic review and meta-analysis comparing primary resection and anastomosis versus Hartmann’s procedure for the management of acute perforated diverticulitis with generalised peritonitis. Tech Coloproctol 24, 527–543 (2020). https://doi.org/10.1007/s10151-020-02172-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10151-020-02172-2