Abstract
Background
Transanal total mesorectal excision (taTME) is an altogether different approach to rectal cancer surgery, and the effects of carbon dioxide (CO2) on this dissection remain poorly described.
Methods
This article critically examines the effect of carbon dioxide insufflation and the workspace it creates during the process of taTME. The unique aspects of insulation with this approach are governed by the laws of physics, especially the principles of fluid dynamics, an area that remains poorly described for laparoscopy and not at all described for taTME.
Results
A summary of established factors which affect the operative field of the taTME surgeon is delineated and further explored. In addition, new concepts regarding gas delivery, such as insufflation vectors, anatomic distortion, hyper-dissection, and workspace volume rate of change as a function of taTME dissection time, are addressed. Collectively, these factors pose important challenges which increase case complexity and are thus essential for taTME trainers and trainees alike to understand.
Conclusions
Although an invisible gas, CO2 insufflation with taTME produces markedly visible effects which are imposed upon the operative field. This can result in anatomic distortion and misperception of operative planes. Thus, practicing taTME surgeons should be cognizant of these effects.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transanal total mesorectal excision (taTME) is a new surgical technique which provides unprecedented access to the distal, horizontal rectum [1, 2]. With prerequisite skill, core study, ongoing training, and operative experience, it represents a quantum leap forward in our approach to the anatomically constrained rectum in selected patients [3, 4]. But mastery of this technique has proved to be arduous with the potential for serious morbidity [5].
It is often stated that taTME poses an unfamiliar vantage point for the operating surgeon and, because of this, the operation is more complex and thus more difficult to perform. However, this is an oversimplification of reality, as more than mere vantage point accounts for taTME’s complexity. Importantly, there are many aspects to taTME that are quite unique to this operation [6,7,8,9,10] including the delivery of gas insufflation for creation of a so-called pneumopelvis.
In this report, the specific and rather curious effects of carbon dioxide (CO2) insufflation within the subperitoneal pelvis are delineated. Collectively, they form a composite that differentiates taTME from conventional minimally invasive surgical (MIS) approaches to the rectum, and even differentiate it from the original down-to-up sphincter-preserving operation, transanal abdominal transanal (TATA), first performed in 1984 by Gerald Marks [11]. As described here, insufflation with taTME contributes to the complexity of the operation, primarily because it can render anatomic planes more difficult to interpret.
Unique effects of CO2 insufflation in taTME
Insufflation in taTME is fundamentally different from insufflation delivered via laparoscopy, as outlined in Table 1. CO2 insufflation in taTME imparts these procedure-specific effects: (a) anatomic distortion (with both global and local effects), (b) extra-mesenteric pneumatic dissection resultant in hyper-mesorectal excision, (c) cyclic billowing, (d) inadvertent luminal insufflation, (e) CO2 entrainment with potential air embolization, and (f) CO2 embolization of bacteria and tumor cells (theoretical). These effects are summarized (Table 2), and each is explored in further detail, with a glossary of key terminology provided (Table 3). The specific effects of gas flow, or fluid (gas) dynamics, within the taTME apparatus are also examined.
Newtonian fluid dynamics and gas kinetics
Gas kinetics is a branch of study in physics that examines the motion of gases, their fluid dynamics, and the effect they impose on physical systems. Remarkably, although gas insufflation has been used for decades and although the physiologic effects of insufflation have been exhaustively studied, there remains a paucity of information about the continuum mechanics of gas insufflation during laparoscopy, let alone taTME. In other words, precisely what are the direct effects of insufflation on the operative field?
Relying on known physical principles of continuum mechanics and fluid dynamics, concepts which may have a relevant impact on taTME can be formulated. CO2, a Newtonian fluid, exhibits properties in accordance with laws pertaining to fluid dynamics. There are two important principles in physics that govern CO2 delivery during taTME. First, the Hagen–Poiseuille law defines volume rate of flow dependent on location within a conduit (or cylinder, e.g., taTME access channel), such that flow of a liquid (or gas) has the highest velocity at the center and lowest at the periphery, thereby creating a velocity gradient; there also exists a pressure gradient along the length of a cylinder. This means that the pressure at the end of the taTME access channel (exiting to the operative field) will be less than what is was at the beginning of the channel, and that the flow velocity of CO2 gas at the periphery is less than it is at the center (Fig. 1). Furthermore, Bernoulli’s law states that velocity flow rates are conserved, and mathematically, the gas velocity will be decreased as it exits the taTME access channel, since the radius is differentially larger than the trocar radius (Fig. 1). The effect of Bernoulli’s law and the Hagen–Poiseuille law on CO2 flow through the taTME apparatus is important to understand. However, the effect on the work space created by CO2 within the taTME operative field is likely complex and remains poorly understood.
Principles of flow dynamics for CO2 through a cylindrical system, such as for taTME, are illustrated in a simple model. There are two physical laws that govern the laminar flow of CO2. Bernoulli’s law states that velocity flow rate is conserved as the diameter of a cylinder changes, so that as the CO2 flows from a trocar to the cylinder of the taTME access channel, velocity decreases since the cross-sectional area increases. The Hagen–Poiseuille law indicates that pressure diminishes in the direction of flow, thus creating a pressure gradient, ∆P (P 1 − P 2). Furthermore, the velocity of a fluid or gas within a lumen varies by its location within a cylinder. For this reason, gas flowing at the periphery of the taTME accesses channel flows at a slower velocity than CO2 at the center (white arrows), thereby creating a velocity gradient. ∆P, pressure differential; P 1, pressure at start of access channel; P 2, pressure at end of access channel; μ, dynamic (shear) viscosity; L, length of cylinder (taTME access channel/trocar); Q, volumetric flow rate; R, radius of cylinder (access channel/trocar); A 1, trocar cross-sectional surface area; A 2, taTME access platform cross-sectional surface area. V 1, velocity of CO2 within trocar; V 2, velocity of CO2 within taTME access channel
Cyclic billowing
In the past 7 years, an important technical limitation has been overcome by use of alternate insufflation systems in conjunction with a transanal apparatus. Essentially, these systems solve the problem of pneumatic instability that created the well-known phenomena of cyclic billowing and smoke accumulation, reported to be the commonest technical complication during taTME in the largest registry data series published to date [12].
Cyclic billowing is defined as the sudden and repetitive collapse of the operative work space caused by pneumatic instability. It occurs because of the cyclic nature of gas delivery utilized by rudimentary insufflators that periodically sample pressure and deliver CO2 via a pump mechanism in response to pressure fluctuations. As these adjustments are made in a cyclic fashion, the small working space of the taTME surgeon cyclically contracts and expands. Although insufflation with standard insufflators produces a negligible effect within the abdominal cavity, it often results in loss of a stable, CO2-created operative field during taTME dissection. Thus, cyclic billowing is primarily related to the mode of insufflation and the continuum mechanics of gas delivery.
Today, many taTME surgeons utilize a non-valve trocar system which maintains insufflation by a pressured gas barrier of insufflated CO2 (AirSEAL®, ConMed, Inc., Utica, NY, USA) [13,14,15], although other devices are also available and/or under development by industry. An important component of system design is that CO2 pressure, smoke evacuation, and CO2 fresh gas delivery are all separately controlled via a composite disposable tubing which contains three separate lumens [16, 17]. The ability to constantly monitor and separately regulate CO2 pressure provides a more stable pneumatic working space that minimizes cyclic billowing. This non-valve trocar insufflation system is erroneously described as a ‘high flow’ system by many surgeons. However, the flow in most instances is relatively low and, for a typical taTME operation, the pressure is set to 8–12 mmHg and flow rates are typically 8 L/min. However, when an increased rate of delivery is required (such as during suctioning of smoke plumes and blood), such systems are designed to compensate immediately by increasing flow rates to up to 40 L/min in real time, such that the working space does not collapse [16]. The end result is a clear operative field, with minimal billowing [14]. This type of insufflation delivery represents the most significant solution to the purely technical limitations of taTME, which previously utilized conventional laparoscopic insufflators as described with the original transanal minimally invasive surgery (TAMIS) technique [18], or insufflators designed for intraluminal surgery, such as with the transanal endoscopic microsurgery (TEM) apparatus.
Operative vectors and hyper-TME
This effect relates to the directionality of insufflation. Recall that a vector, in physics and mathematics, is defined as a force (magnitude) together with a direction in three-dimensional space. Thus, because the insufflation with taTME is delivered transanally rather than transabdominally, the insufflation vectors are completely opposed to those encountered in the abdominal approach (Fig. 2). Furthermore, when discussing direction, up to down versus down to up, one can also consider this as a type of polarity with respect to the surgical field. In this regard, the reverse approach to TME, or up to down, can be described as reverse polarity. The alternate viewpoint of taTME, coupled with the unique magnitude and direction of pneumatic insufflation utilized by this technique and reverse polarity, can more globally be considered as an operative vector which reshapes the surgeon’s impression of the surgical field in a very specific manner.
Relationship of insufflation vectors is shown, with a comparison between taTME and abdominal approaches. Insufflation vectors are of no consequence with abdominal surgery, but with taTME they exhibit important effects, including the creation of anatomic distortion during dissection of the subperitoneal pelvis. It is important to realize that the pelvic cavity is fully occupied by viscera and only becomes an empty cavity after rectal extirpation
The reason for this has to do with the directionality of the pelvic fascial planes themselves. Because of the layering effect of extra-mesenteric fascial planes, the operative vectors (imparted by taTME’s insufflation direction) expose extra-mesenteric planes that otherwise would not be exposed, as CO2 insufflation from the abdomen does not normally result in air dissection of these planes [6]. This often leads to surgical specimens which contain ‘extra’ tissue, including fragments of non-mesenteric adipose tissue and juxtaposed fascia that lie beyond the Heald envelope, resulting in a ‘hyper’-mesorectal excision (hyper-dissection) or hyper-TME (Fig. 3).
Hyper-TME defines a dissection that is inclusive of planes beyond the mesorectal envelope. In this schematic diagram, it is demonstrated how taTME can lead to deep entry into extra-mesorectal planes, especially posteriorly. From above (green arrow), the arrangement of anatomic planes and the pathway of dissection keep the surgeon’s dissection along the Heald envelope. In contrast, from below (red arrow), the surgeon’s dissection can be inadvertently directed posteriorly, deep to the endopelvic fascia resulting in a hyper-mesocolic excision with resultant risk of damage to the pelvic autonomic and vascular plexus
Anatomic distortion
With conventional, up-to-down minimally invasive surgery (MIS) laparoscopic and robotic techniques, abdominal cavity insufflation does not exhibit an appreciable distortive effect on native visceral anatomy, probably because the volume insufflated is several-fold more than the volume of the actualized (potential) space created with taTME beneath the peritoneum, within the dissected work space of the subperitoneal pelvis (Fig. 4). Additionally, most pressure from laparoscopic, abdominal cavity insufflation results in force vectors that are distributed anteriorly in a uniform manner over a relatively large surface area (i.e., the average peritoneal lining in adults measures ~ 1.5 m2 [19]), thus causing doming of the anterior abdominal wall. It is important to note that visceral organ deformity is negligible if not completely nonexistent with conventional laparoscopy.
Shown is the taTME surgeon’s view of the pelvic cavity after extirpation of the rectum and its mesentery. It is only after the rectum and the mesentery are subtracted from the posterior pelvic compartment that one can appreciated the subperitoneal pelvis as a true cavity, with an approximate volume of 70–150 cm3 depending on pelvic geometry, gender and overall patient size, and importantly the degree of completed dissection. CO2 dissection of this potential space created during taTME results in special challenges not encountered with standard laparoscopy
In contradistinction, with taTME, the ultra-low volume insufflated of the actualized pelvic space exhibits significant forward pressure on the mobilized viscera (especially the rectum and its mesentery), resulting in anatomic distortion, in essence, a marked contour deformity of rectum and its mesentery (Fig. 5a, b). The more mobilized the rectum becomes, the more the anatomic distortion becomes evident, as the anatomy is effectively ‘compressed’ anteriorly and distracted cephalad opposite the direction of CO2 inflow. This global effect results in deformity of the native shape of the rectum and its mesentery (Fig. 6a–c). Because of the Hagen–Poiseuille law and the velocity gradient established though the cylindrical taTME access channel, the effect is likely most pronounced centrally. Thus, during the posterior dissection, there appears to be a mesenteric concavity which resembles a ‘cobra’ (Figs. 5b, 6c).
Anatomic distortion resultant from the unique effect of insufflation of an actualized, potential space is illustrated. a Dissection with CO2 distorts the rectum, as if to ‘press’ it anteriorly and cephalad, b the mesorectal envelope and its cheveux d’ange are shown, note the concavity of the envelope, a direct result of CO2 centrally pressing the mesentery forward. This is an effect that can likely be explained by the Hagen–Poiseuille law as a velocity gradient is created such that the flow velocity is highest centrally and lowest at the periphery of the taTME operative workspace
An artist’s rendition of the distortive effects of CO2 insufflation is illustrated, a anatomy as it may appear without insufflation, b anatomic distortion with insufflation in midsagittal view, and c the effect of anatomic distortion from the taTME’s surgeon’s point of view with cobra like concavity of the midmesorectum resultant from the effects of the velocity gradient established by the Hagen–Poiseuille law
It is known that surgical approaches to taTME vary widely, with some surgeons preferring a posterior first approach, while others prefer an anterior first approach, and still others prefer a circumferential approach to dissection. In the circumferential approach, the distortion results in en face flattening of the rectum and mesorectum (Fig. 7a, b). Regardless of approach, the imposed anatomic distortion is still observed (Fig. 8a, b). It is imperative that taTME surgeons recognize the challenge posed by distortion, particularly as the degree of distortion tends to become more prominent with progression of the caudocranial dissection. Such anatomic distortion is unfamiliar to non-taTME minimally invasive rectal surgeons. Simply stated, anatomic distortion observed with taTME is not realized with abdominal approaches, because anatomic planes do not become aberrant with MIS approaches and traditional pneumoperitoneum.
It should also be appreciated that this anatomic distortion is not present with the predecessor to taTME, TATA [11], because with the latter, the subperitoneal pelvic dissection (according to the original description and prior to later modifications) is completed in a traditional manner, without a transanal access channel and thus without pneumopelvis, underscoring a crucial difference between taTME and (the original) TATA, as they are not the same operation due principally to this point of distinction (Table 4).
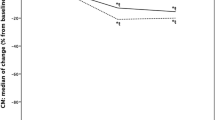
Change in workspace volume (dv/dt)
It is crucial to understand that the working space created by taTME undergoes dramatic changes during the process of dissection. Initially, the extra-luminal component of this workspace (after the purse string has been secured) starts just as a ~ 1 cm3 area. It then gradually expends with taTME dissection (Fig. 9). The workspace eventually increases and approaches (approximately) the volume of a tennis ball or ~ 150 cm3, which is markedly smaller than laparoscopic operative fields. This underscores another point of distinction between taTME and conventional, up-to-down MIS laparoscopic and robotic techniques, as laparoscopic workspaces do not appreciably change with dissection time, while for taTME the volume of workspace (v) increases as a function of dissection time (t) and can mathematically be expressed as a differential, dv/dt.
Relationship between extra-luminal workspace volume and taTME dissection time (dv/dt) is shown. During the first portion of the taTME operation, A the purse string is applied and there is not yet any extra-luminal workspace; as the rectal wall is circumferentially divided, B there is a gradual increase in the workspace volume, and after complete division, the workspace rapidly increases, C before it begins to plateau just prior to peritoneal entry. Upon entry, the pelvic workspace communicates with the abdominal cavity, and at this point of union, the operation is reduced to a single field
Triangles and O’s
Because of the small operative field volume with taTME, as well as the unique methodology of insufflation used by transanal access platforms, specific CO2 effects are often pronounced. These have been previously described in detail as ‘triangles’ and ‘O’s’ [20] as this pertains to the exaggerated effect of CO2 on fascial planes as observed with taTME. Specifically, violation of fascia planes can simulate a halo or ‘O’ sign and this is created as gas evenly distributes force at the point of violation, causing it to expand uniformly in a symmetric circle or ellipse. Tethering points that have not been released during dissection often leave a triangular shape due to unidirectional traction without counter-traction (Fig. 10). It is important to understand that ‘triangles’ and ‘Os’ are created by the combination of (a) CO2 insufflation and (b) operative dissection. These local effects are not unique to taTME and can be observed with all insufflation-based MIS surgeries; nevertheless, this local insufflation effect seen during plane dissection is probably more prominent with taTME and, when properly understood, can improve the taTME surgeon’s operative frame of reference. The visualization of a ‘halo’ should lead to careful plane reassessment.
Triangles and O’s, as described by Bernardi et al. [20]. a A schematic diagram of the rectum, mesorectum, and fascia planes is illustrated. Posteriorly, a tethering point of the endopelvic fascia has not been released. b The taTME insufflation vector and relative focused view of a small operative field create the appearance of a triangle. The correct plane of dissection thus releases the tethering point of the endopelvic fascia (green line) and does not violate this plane (red line). c In any plane, a violation of a correct or incorrect fascial plane results in evenly distributed pressure at the point of violation creating a circle or ellipse. This is commonly described as the halo sign or ‘O’ sign. While these findings are observed during standard laparoscopy, they become more exaggerated within the small workspace created during taTME
CO2 entrainment and embolization
Case descriptions and anecdotal reports suggest that, although small, a real risk of CO2 embolization via the venous system can occur during taTME [21, 22]. This may be related to a combination of factors including (a) high flow insufflation systems with gas delivery into a finite workspace, (b) inadvertent violation of the pelvic sidewall veins, prostatic venous plexus, and sacral venous plexus, particularly the latter, when it involves Batson’s plexus [23] and its tributaries since these veins have no valves, (c) operating at a pressure in excess of systemic venous pressure (creating a pressure gradient with resultant gas entrainment), and (d) patient position in steep Trendelenburg, which places the operative field above the level of the heart, where venous pressure is often below atmospheric pressure.
CO2 venous embolism can lead to decreased pulmonary capillary gas exchange, cardiac arrhythmia, pulmonary hypertension, and right ventricular strain (via a ‘gas lock’ effect which results in right ventricular outflow obstruction), cardiovascular collapse, as well as death [24]. Thus, taTME surgeons should be extremely knowledgeable about diagnosis and rapid treatment.
Diagnosis can be made through recognition of acute onset physiologic changes including hemodynamic instability, decreased PaO2 and PaCO2, metabolic acidosis, increased right ventricular afterload, with decreased left ventricular filling. Transesophageal echocardiogram is the most sensitive diagnostic test, as it can detect air emboli ≥ 0.1 mg/kg [25]. However, the test is technically complex, requires a skilled operator, and is not likely to be immediately available in most operating theaters. Therefore, treatment should be initiated once the diagnosis is suspected based on clinical criteria.
A sudden and otherwise unexplained decrease in end-tidal CO2(EtCO2) should immediately alert the taTME surgeon to the possibility of CO2 embolization, as this is often the first clinical parameter to be detected [21]. However, an increase in EtCO2 has also been observed, and thus, surgeons and anesthesiologists must interpret this finding the context of the overall clinical picture [24, 26]. Treatment requires immediate cessation of gas delivery and surgery, placement in the left lateral decubitus position (i.e., left side down tilt, with immediate leveling of the operating table’s head down tilt in the case of taTME), fluid resuscitation, increased ventilation, and delivery of 100% O2 [26]. Increasing positive end expiratory pressure (PEEP) may decrease the pressure gradient between the lacerated venous vessels and the heart, thereby decreasing the probability of further gas entrainment. It is also beneficial to flood the operative field with sterile saline to prevent further entrainment of gas.
CO2 embolization of bacteria and tumor (theoretical)
Some aspects of taTME are unique to the procedure, such as closure of the lumen with division of the rectum distal to the tumor or, in the case of very low cancers, with intersphincteric dissection. Purse string formation is often imperfect, and small defects are frequently observed, sometimes leading to inadvertent dilation of the entire large bowel. Hypothetically, imperfect purse string closure of the rectum could lead to bacterial contamination within the resection bed as organisms become aerosolized. Thus, the combination of insufflation vectors directed en face toward the rectum and a purse string defect may result in colonization of microorganisms within the pelvis, leading to post-surgical infection. Whether or not attributed to taTME CO2 insufflation vectors, contamination of the sterile field has been documented with TAMIS-TME [27].
Because live neoplastic cells can also exfoliate and implant at other locales [28,29,30,31,32,33,34], the taTME surgeon must be aware of the potential risk of insufflation-induced tumor cell implantation, particularly when the purse string contains one or more defects [6]. Except for instances in which the bowel lumen is grossly perforated, it is improbable that insufflator-induced aerosolization of tumor cells or bacterial organisms is associated with non-taTME approaches to rectal cancer extirpation, and therefore, this potential morbidity is unique to taTME. The clinical importance of this remains unknown.
Discussion
Despite decades of global experience with laparoscopy using gas insufflation systems, little research has been done relating to gas kinetics, fluid dynamics of CO2, and the effect the gas itself imparts on the operative field. The general principles of physics can be applied to gas delivery via the taTME apparatus.
Today, there are three distinct oncologic approaches to rectal resection which require an up-to-down component (reverse polarity) in operative extirpation; they are: (a) the abdominoperineal resection (APR), (b) the original TATA operation, and (c) taTME (including taTME for APR [35]). For all of these reverse polarity operations, only taTME delivers subperitoneal CO2 to create the pelvic workspace utilizing commercially available access channels and devices. This is a fundamental difference and crucial distinction that separates taTME from other perineal approaches to curative intent resection. In this respect, taTME even differs from local excision of rectal neoplasia via TAMIS/TEM because although the techniques share identical operative vectors, the insufflated gas is distributed throughout a much larger volume when intraluminal surgery is performed.
While operative vectors are of no consequence in standard laparoscopy and other abdominal minimally invasive techniques, they have marked and important effects during taTME. CO2 insufflation vectors and the effect they impart on the subperitoneal pelvis impose gross anatomic distortion, a concept that has not been previously described with taTME. Additionally, the velocity gradient defined by fluid dynamics and the Hagen–Poiseuille law likely effects the appearance and shape of the deformity, with the classic concavity of the posterior mesorectum; essentially, this law of fluid dynamics states that velocity is greatest centrally and least at the periphery as it is transmitted through a cylinder, so the central part of the mesentery is thus most compressed by this (relatively) higher velocity gas flow, leading to the appearance of a concave distortion of the mesentery (Fig. 5).
Transanal TME surgeons must appreciate the dynamics of the workspace and the fact that the subperitoneal pelvis represents a potential (micro) space and not a true cavity and it is only by forced insufflation that this potential space can be created and observed to significantly increase in volume as a function of taTME dissection time. It is only with complete extirpation of the rectum and its mesentery that the posterior compartment can be realized as a true cavity.
taTME is a relatively new operative approach that is still in the process of being investigated and learnt. By examining the effects of pneumatic insufflation on anatomy, we have shown how this can increase the complexity of taTME dissection–factors which help explain why this new technique has proved difficult to master, even for skilled surgeons.
The fluid dynamics of CO2 and its effect upon anatomic planes with resultant gross anatomic distortion shift the contour of native viscera. As a result, surgeon perception itself shifts, potentially leading to a loss of the operative frame of reference and an increase in surgeon misperception. While new techniques for structure identification are being currently explored [36, 37], it is still imperative for taTME instructors and trainees to understand the unique effects of CO2 on the operative field as it increases case complexity and is likely one of the key reasons there is a steep learning curve for taTME. Collectively, the effects of insufflation and operative vectors represent one of the most important challenges for taTME surgeons as they are still poorly understood and have not yet been described although seven years have passed since the inception of the technique [38]. This highlights a global deficiency in the fundamental elements necessary to master taTME.
Conclusions
Although CO2 is an invisible gas, CO2 insufflation with taTME produces quite visible effects. It is important for the practicing surgeon to understand these aspects as they represent a crucial distinction between taTME and all other approaches to TME. The effect imposed by insufflation with taTME, although valuable in facilitating dissection, can result in anatomic distortion and misperception of operative planes.
References
Cahill RA, Hompes R (2015) Transanal total mesorectal excision. Br J Surg 102(13):1591–1593. doi:10.1002/bjs.9933
Rullier E (2015) Transanal mesorectal excision: the new challenge in rectal cancer. Dis Colon Rectum 58(7):621–622. doi:10.1097/DCR.0000000000000395
Atallah S (2015) Transanal total mesorectal excision: full steam ahead. Tech Coloproctol 19:57–61
Heald RJ (2013) A new solution to some old problems: transanal TME. Tech Coloproctol 17(3):257–258. doi:10.1007/s10151-013-0984-0
Atallah SB, Dubose AC, Burke JP, Nassif G, deBeche-Adams T, Frering T, Albert MR, Monson JRT (2017) Uptake of transanal total mesorectal excision in North America: initial assessment of a structured training program and the experience of delegate surgeons. Dis Colon Rectum. doi:10.1097/DCR.00000000000000823
Atallah S, Albert M, Monson JR (2016) Critical concepts and important anatomic landmarks encountered during transanal total mesorectal excision (taTME): toward the mastery of a new operation for rectal cancer surgery. Tech Coloproctol 20(7):483–494
Knol J, Chadi SA (2016) Transanal total mesorectal excision: technical aspects of approaching the mesorectal plane from below. Minim Invasive Ther Allied Technol 25(5):257–270. doi:10.1080/13645706.2016.1206572
Knol JJ, D’Hondt M, Souverijns G, Heald B, Vangertruyden G (2015) Transanal endoscopic total mesorectal excision: technical aspects of approaching the mesorectal plane from below—a preliminary report. Tech Coloproctol 19(4):221–229. doi:10.1007/s10151-015-1275-8 (Epub 2015 Feb 22)
Kneist W, Hanke L, Kauff DW, Lang H (2016) Surgeons’ assessment of internal anal sphincter nerve supply during TaTME—inbetween expectations and reality. Minim Invasive Ther Allied Technol 25(5):241–246. doi:10.1080/13645706.2016.1197269 (Epub 2016 Jun 22)
Kneist W, Rink AD, Kauff DW, Konerding MA, Lang H (2015) Topography of the extrinsic internal anal sphincter nerve supply during laparoscopic-assisted TAMIS TME: five key zones of risk from the surgeons’ view. Tech Coloproctol 19:751–752
Marks G, Mohiuddin M, Rakinic J (1991) New hope and promise for sphincter preservation in the management of cancer of the rectum. Semin Oncol 18:388–398
Penna M, Hompes R, Arnold S, Wynn G, Austin R, Warusavitarne J, Moran B, Hanna GB, Mortensen NJ, Tekkis PP, Collaborative TaTME Registry (2017) Transanal total mesorectal excision: international registry results of the first 720 cases. Ann Surg 266(1):111–117. doi:10.1097/SLA.0000000000001948
Nepple KG, Kallogjeri D, Bhayani SB (2013) Benchtop evaluation of pressure barrier insufflator and standard insufflator systems. Surg Endosc 27(1):333–338. doi:10.1007/s00464-012-2434-x Epub 2012 Jul 26
Nicholson G, Knol J, Houben B, Cunningham C, Ashraf S, Hompes R (2015) Optimal dissection for transanal total mesorectal excision using modified CO2 insufflation and smoke extraction. Colorectal Dis 17(11):O265–O267. doi:10.1111/codi.13074
Bislenghi G, Wolthuis AM, de Buck van Overstraeten A, D’Hoore A (2015) AirSeal system insufflator to maintain a stable pneumorectum during TAMIS. Tech Coloproctol 19:43–45
Horstmann M, Horton K, Kurz M, Padevit C, John H (2013) Prospective comparison between the AirSeal® System valve-less Trocar and a standard Versaport™ Plus V2 Trocar in robotic-assisted radical prostatectomy. J Endourol 27(5):579–582. doi:10.1089/end.2012.0632 (Epub 2013 Feb 5)
Luketina RR, Knauer M, Köhler G, Koch OO, Strasser K, Egger M, Emmanuel K (2014) Comparison of a standard CO 2 pressure pneumoperitoneum insufflator versus AirSeal™: study protocol of a randomized controlled trial. Trials 15(1):239
Atallah S (2014) Transanal minimally invasive surgery for total mesorectal excision. Minim Invasive Ther Allied Technol 23:10–16
Albanese AM, Albanese EF, Miño JH, Gómez E, Gómez M, Zandomeni M, Merlo AB (2009) Peritoneal surface area: measurements of 40 structures covered by peritoneum: correlation between total peritoneal surface area and the surface calculated by formulas. Surg Radiol Anat 31(5):369–377. doi:10.1007/s00276-008-0456-9 (Epub 2009 Jan 14)
Bernardi MP, Bloemendaal AL, Albert M, Whiteford M, Stevenson AR, Hompes R (2016) Transanal total mesorectal excision: dissection tips using ‘O’s and ‘triangles’. Tech Coloproctol 20(11):775–778 (Epub 2016 Oct 1)
Ratcliffe F, Hogan AM, Hompes R (2017) CO2 embolus: an important complication of TaTME surgery. Tech Coloproctol 21(1):61–62. doi:10.1007/s10151-016-1565-9 (Epub 2016 Dec 7)
Rouanet P, Mourregot A, Azar CC et al (2013) Transanal endoscopic proctectomy: an innovative procedure for difficult resection of rectal tumors in men with narrow pelvis. Dis Colon Rectum 56:408–415
Nathoo N, Caris EC, Wiener JA, Mendel E (2011) History of the vertebral venous plexus and the significant contributions of Breschet and Batson. Neurosurgery 69(5):1007–1014
van Hulst RA, Klein J, Lachmann B (2003) Gas embolism: pathophysiology and treatment. Clin Physiol Funct Imaging 23(5):237–246
Martineau A, Arcand G, Couture P, Babin D, Perreault LP, Denault A (2003) Transesophageal echocardiographic diagnosis of carbon dioxide embolism during minimally invasive saphenous vein harvesting and treatment with inhaled epoprostenol. Anesth Analg 96(4):962–964 (table of contents)
Park EY, Kwon JY, Kim KJ (2012) Carbon dioxide embolism during laparoscopic surgery. Yonsei Med J 53(3):459–466
Velthuis S, Veltcamp Helbach M, Tuynman JB, Le TN, Bonjer HJ, Sietses C (2015) Intra-abdominal bacterial contamination in TAMIS total mesorectal excision for rectal carcinoma: a prospective study. Surg Endosc 29:3319–3323
Rygick AN, Fain SN, Pestovskaja GN (1969) Viability of cancer cells penetrating tissues during operation for cancer of the rectum. Dis Colon Rectum 12:351–356
Skipper D, Cooper AJ, Marson JE, Taylor I (1987) Exfoliated cells and in vitro growth in colorectal cancer. Br J Surg 74:1049–1052
Umpleby HC, Fermor B, Symes MO, Williamson RC (1984) Viability of exfoliated colorectal carcinoma cells. Br J Surg 71:659–663
Yu S-K, Cohn I (1968) Tumor implantation on colon mucosa. Arch Surg 96:956–958
Tsunoda A, Shibusawa M, Kamiyama G, Takata M, Yokoyama N, Kusano M (1996) Viability of free malignant cells in specimens removed because of colorectal carcinoma. Jpn J Gastroenterol Surg 29:1022–1027
Keynes WM (1961) Implantation from the bowel lumen in cancer of the large intestine. Ann Surg 153:357–364
McGrew EA, Laws JF, Cole WH (1954) Free malignant cells in relation to recurrence of carcinoma of the colon. JAMA 154:1251–1254
Atallah S, Albert M, DeBeche-Adams T, Nassif G, Polavarapu H, Larach S (2013) Transanal minimally invasive surgery for total mesorectal excision (TAMIS-TME): a stepwise description of the surgical technique with video demonstration. Tech Coloproctol 17(3):321–325. doi:10.1007/s10151-012-0971-x (Epub 2013 Feb 2)
Atallah S, Mabardy A, Volpato AP, Chin T, Sneider J, Monson JRT (2017) Surgery beyond the visible light spectrum: theoretical and applied methods for localization of the male urethra during transanal total mesorectal excision. Tech Coloproctol 21(6):413–424. doi:10.1007/s10151-017-1641-9 (Epub 2017 Jun 6)
Barnes TG, Penna M, Hompes R, Cunningham C (2017) Fluorescence to highlight the urethra: a human cadaveric study. Tech Coloproctol 21(6):439–444. doi:10.1007/s10151-017-1615-y (Epub 2017 May 30)
Sylla P, Rattner DW, Delgado S, Lacy AM (2010) NOTES transanal rectal cancer resection using transanal endoscopic microsurgery and laparoscopic assistance. Surg Endosc 24(5):1205–1210. doi:10.1007/s00464-010-0965-6 (Epub 2010 Feb 26)
Acknowledgements
The authors appreciate the invaluable assistance of Stephanie Philippaerts and the iLappSurgery Foundation in development of medical illustrations contained herein.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. S. Atallah is a consultant for ConMed, Inc, Ethicon, Applied Medical, Inc, MedRobotics, Medicaroid, Inc and THD America. Dr. J. Knol, R. Hompes, P. Gonzalez and S. Chadi have no financial disclosures. Design of some illustrations included in this manuscript was funded by iLappSurgery Foundation.
Ethical approval
This research was performed in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained in accordance with the standards set forth by hospital regulations.
Rights and permissions
About this article
Cite this article
Atallah, S., Gonzalez, P., Chadi, S. et al. Operative vectors, anatomic distortion, fluid dynamics and the inherent effects of pneumatic insufflation encountered during transanal total mesorectal excision. Tech Coloproctol 21, 783–794 (2017). https://doi.org/10.1007/s10151-017-1693-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10151-017-1693-x