Abstract
Background
The best surgical strategy for the management of perforated diverticulitis with generalized peritonitis of the sigmoid colon is not clearly defined. The aim of this retrospective cohort study was to evaluate the value of a damage control strategy.
Methods
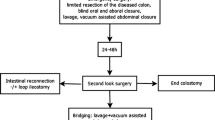
All patients who underwent emergency laparotomy for perforated diverticular disease of the sigmoid colon with generalized peritonitis between 2010 and 2015 were included. The damage control strategy (study group), included a two- stage procedure: limited resection of the diseased colonic segment, closure of proximal colon and distal stump, and application of an abdominal vacuum at the initial surgery followed by second-look laparotomy 24–48 h later At this point a choice was made between anastomosis and Hartmann’s procedure. The control group consisted of patients receiving definitive reconstruction (anastomosis or Hartmann’s procedure) at the initial operation.
Results
Thirty-seven patients were included in the study. Damage control strategy was applied in 19 patients and the control group consisted of 18 patients. Both groups were comparable in terms of demographics, severity of peritonitis, and comorbidities. The overall postoperative mortality was 11 % (n = 4). There were no statistically significant differences between both groups regarding postoperative morbidity and mortality; however, a significantly higher proportion of patients in the control group had a stoma after the initial hospital stay (83 vs. 47 %, p = 0.038). This difference was still significant after adjustment for sex, age, Mannheim Peritonitis Index, American Society of Anesthesiologists class and presence of septic shock at presentation. At the end of the follow-up period, 15 of 17 survivors in the study group and 13 of 16 survivors in the control group had their intestinal continuity restored (p = 0.66).
Conclusions
Damage control strategy in patients with generalized peritonitis due to perforated diverticulitis leads to a significantly reduced stoma rate after the initial hospital stay without an increased risk of postoperative morbidity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The majority of patients with acute diverticular disease are treated conservatively. However, perforated diverticular disease complicated by generalized peritonitis remains a challenging and life-threatening condition leading to emergency surgery in most cases. Unfortunately, there is no clearly established treatment algorithm for this patient population. Resection of the affected bowel segment followed by an end colostomy (Hartmann’s procedure) and primary anastomosis with/without defunctioning ileostomy are the strategies still used by most surgeons [1]. However, reversal of the end colostomy is accompanied by a significant risk of postoperative adverse events [2, 3]. Intestinal continuity can only be restored in 50–70 % of patients treated with Hartmann’s procedure [2]. Nevertheless, Hartmann’s procedure remains the most common surgical treatment for patients with acute perforated diverticulitis, particularly in cases of fecal peritonitis [3–5]. Several studies have analyzed the role of primary anastomosis versus Hartmann’s procedure. No significant differences regarding postoperative morbidity and mortality were shown in the majority of these trials. However, patients with primary anastomosis were significantly younger, Hinchey scores and peritonitis stages were lower and the operation was done by experienced surgeons [1] in some of these studies. The only prospective randomized trial, conducted by Oberkofler et al. [4] compared Hartmann’s procedure to primary anastomosis with defunctioning ileostomy. The authors demonstrated that primary anastomosis was not inferior to Hartmann’s procedure; yet, all patients received a stoma using either one of these approaches. A damage control strategy was recommended by some authors for patients with perforated diverticular disease complicated by generalized peritonitis [6–9]. A detailed analysis was reported by a study group from Innsbruck, Austria [6, 7]. Damage control strategy is a two-step approach consisting of limited resection of the perforated bowel segment, blind closure of the proximal colonic segment and distal stump and temporary abdominal closure followed by definitive reconstruction 24–48 h later. During the second operation, Hartmann’s procedure or an anastomosis is performed depending on patients’ overall and local conditions. Intestinal continuity was restored in 76 % of patients at as part of the initial treatment and in 93 % of patients by the end of the follow-up in the study from Innsbruck, and the mortality rate was 9.8 % [6, 7]. Thus, the proportion of patients with restored intestinal continuity after their initial hospital stay seems to be considerably higher after damage control surgery than in most studies on patients with diverticulitis with generalized peritonitis treated with colonic resection. The damage control approach was introduced in our department in 2010; however, some surgeons still adhered to primary definitive surgery during the study period. Therefore, we were able to compare damage control to the conventional strategy in the present retrospective analysis.
Materials and methods
Thirty-seven consecutive patients who underwent emergency laparotomy for perforated sigmoid diverticulitis with generalized peritonitis between January 1, 2010, and April 30, 2015, were included in the present study.
The severity of peritonitis was measured using the Mannheim Peritonitis Index (MPI). The MPI is a specific scoring system for assessment of peritonitis prognosis including age, gender, organ failure, presence of cancer, duration of peritonitis, involvement of colon, extent of spread and character of the peritoneal fluid. The MPI has been shown to be highly accurate in predicting individual outcome and is superior to the APPACHE II-Score [10–12].
The term “postoperative intraabdominal septic complications” was used for anastomotic leak, intraabdominal abscess, intestinal fistula. “Wound complications” was used for abdominal wound dehiscence and superficial wound infection. Postoperative complications were graded according to the Clavien–Dindo classification [13]. Postoperative complication rate, length of hospital stay and number of procedures performed were calculated both for the initial hospital stay and as a sum of two hospital stays when patients were discharged with a stoma (ileostomy or colostomy) and underwent restoration of intestinal continuity later.
Surgical technique
The respective surgical procedure was determined by the treating surgeon. The damage control strategy (study group) included a two-stage procedure: limited resection of the perforated colonic segment, closure of the colon proximally and distally, peritoneal lavage, and temporary abdominal closure by negative pressure wound therapy in the course of initial surgery, followed by planned relaparotomy 24–48 h later. At that point the decision about reconstruction (anastomosis ∓ ileostomy or Hartmann’s procedure) was made according to the patient’s general condition and persistence of peritonitis. A primary anastomosis without defunctioning ileostomy was the preferred method of reconstruction; however, in case of persistent severe peritonitis and/or septic shock, Hartmann’s procedure was performed. For negative pressure wound therapy, a vacuum-assisted closure system was used (VAC, KCI, San Antonio, Texas, USA). After covering all intraabdominal structures with the greater omentum as far as possible, the intraabdominal part of the VAC-system was placed in the abdominal cavity. It was covered by a fenestrated non-adhesive interface layer to prevent intraabdominal damage. A non-covered foam was placed subcutaneously as a second layer and sealed by an adhesive film (Figs. 1, 2). After completion of the dressing, continuous negative pressure (−125 mmHg) was applied. In the control group, a definitive reconstruction, Hartmann’s procedure or primary anastomosis (with/without defunctioning ileostomy), was performed during initial surgery. Laparotomy was used in all cases. Laparoscopic lavage was not performed during the study period. The splenic flexure was not routinely mobilized. In case of primary anastomosis, no washout was performed and an end-to-end double-stapled colorectal anastomosis was fashioned in all patients.
Statistical analysis
The Mann–Whitney U test was applied in the case of non-normal distribution for continuous variables. Comparisons between patient groups were made using Fisher’s exact test for categorical variables. Factors potentially affecting the risk of hospital discharge with a stoma (ileostomy or colostomy) were included in a multivariate analysis using a stepwise logistic regression model. The Kaplan–Meier method was used for survival analysis.
Results
Between February 2010 and April 2015, 37 patients (M:F 15:22) underwent emergency sigmoid resection for generalized peritonitis due to perforated diverticular disease. Mean age at the time of surgery was 67.4 years (range 33–85 years). There were 7 patients with fecal peritonitis and 30 patients with purulent peritonitis. The MPI average was 18.6 (range 6–35). The anesthesiological risk was classified as American Society of Anesthesiologists (ASA) I in 2 patients, as ASA II in 8 patients, as ASA III in 23 patients and as ASA IV in 4 patients. Nine patients presented with signs of septic shock at the time of surgery. Overall postoperative mortality was 11 % (n = 4).
Nineteen patients (study group) underwent damage control surgery and primary definite surgery was performed in 18 patients (control group). There were no statistically significant differences between both groups at baseline (Table 1). The procedures were performed by 16 different surgeons: 10 surgeons, accounting for 14 operations, performed only one of the two procedures and 6 surgeons accounting for 23 operations performed both procedures. An end colostomy was formed at planned relaparotomy in 4 of 19 patients (two of whom died), and an anastomosis was created in the 15 remaining study group patients (Fig. 3). A defunctioning ileostomy was formed in 4 patients and a further patient received a defunctioning ileostomy because of an anastomotic leakage. A Hartmann’s procedure was performed in 14 of 18 patients in the control group (two of whom died); a primary anastomosis was created in 4 patients. A defunctioning ileostomy was formed in 1 of the latter. Table 2 demonstrates outcome differences between both groups during the initial hospital stay. There were no statistically significant differences between both groups regarding postoperative morbidity and mortality; however, a significantly higher proportion of patients in the control group had a stoma (83 vs. 47 %, p = 0.038) at the end of the initial hospital stay. This difference was still significant after adjustment for potential confounding factors (sex, MPI, ASA class and presence of septic shock at presentation).
All 33 survivors were followed up (Fig. 4). In the study group, stoma reversal was performed in 5 of 7 patients discharged from the hospital with a stoma. Two patients did not undergo stoma reversal due to significant comorbidities. Stoma reversal was performed in 11 of 13 patients discharged from hospital with a stoma in the control group. However, one of the latter patients received a stoma again to treat a postoperative anastomotic leak. In 2 patients, the end colostomy was not reversed due to significant comorbidities. There were no additional deaths in either group during the follow-up period. Table 3 shows differences between the groups at the end of the follow-up period. There were no statistically significant differences between the groups regarding combined postoperative morbidity, length of hospital stay and proportion of patients with a stoma at the end of the follow-up period. However, the interval between stoma formation and stoma reversal was significantly shorter in the study group than in the control group (median 1.9 months vs. median 4.5 months, p = 0.013).
Discussion
We adopted the damage control strategy in 2010 after a thorough description of the technique was published by Perathoner et al. [7]. Between 2010 and 2015, 37 patients underwent surgery for perforated diverticulitis with generalized peritonitis. Nineteen of were treated using damage control strategy. Significantly more patients were discharged from the hospital with restored intestinal continuity, and the interval between stoma formation and stoma reversal was significantly shorter in the study group than in the control group. When the damage control strategy was used the overall number of operations, the length of hospital stay, the overall complication rate, the rate of abdominal wall complications and mortality did not increase.
Despite its frequency, the surgical therapy for perforated diverticulitis complicated by generalized peritonitis has not yet been standardized. Surgical options are Hartmann’s procedure and primary anastomosis with or without defunctioning ileostomy. Numerous retrospective studies have demonstrated non-homogeneous results. Data from a prospective randomized trial by Oberkofler et al. [4] showed that primary anastomosis with defunctioning ileostomy was superior to Hartmann’s procedure regarding the rate of final restoration of intestinal continuity, perioperative complications, overall length of hospital stay and costs. On the other hand, a randomized trial conducted by Binda et al. [14] resulted in similar stoma closure rates in the Hartmann’s and defunctioning ileostomy groups. However, suggested advantages of primary anastomosis should be interpreted carefully due to selection bias in some studies [15].
During the past two decades, laparoscopic lavage has gained increasing popularity [16, 17] in the treatment of patients with perforated diverticular disease. A systematic review by Toorenvliet et al. [18] showed successful treatment by laparoscopic lavage in 95.7 % of 231 patients (Hinchey II: n = 44, Hinchey III: n = 178, Hinchey IV: n = 8). The mortality rate was low : 1.7 %. An elective resection of the sigmoid colon was performed later in 87 % of patients. Angenete et al. [19] first demonstrated the feasibility of laparoscopic lavage for patients with purulent peritonitis in a prospective randomized trial (DILALA-Trial). Morbidity and mortality were the same as in patients undergoing Hartmann’s procedure. Recently, results of 2 prospectively randomized trials were published. The Scandiv Trial and the LOLA-section of LADIES trial compared laparoscopic lavage with sigmoidectomy in patients with purulent perforated diverticulitis. Both trials demonstrated non-superiority of laparoscopic lavage. The reoperation rate was significantly increased in the laparoscopic lavage group in the Scandiv Trial, whereas the mortality and high morbidity rate did not decrease. Also, the LOLA-arm was closed by the safety monitoring board due to an increased event rate in the lavage group. The analysis of included patients showed a significant increase of reinterventions, both in short- and long-term follow-up [20–22]. However, most failures in the lavage group were due to a false differentiation between purulent and feculent peritonitis and due to missed colorectal cancer. Thus, there still might be an indication for laparoscopic lavage in carefully selected patients with Hinchey III stage diverticular disease. Also, combination of both procedures, laparoscopic limited resection and lavage followed by laparoscopic formation of an anastomosis 24–48 h after initial surgery, might be a promising alternative for some patients.
Damage control strategy combines all requirements of the emergency setting: short operation time, clearance of the primary septic focus, improved selection of patients able to undergo a primary anastomosis. Kafka-Ritsch et al. [6] reported a restoration of the intestinal continuity in 76 % of their cases which is similar to our findings. Eighty-eight percent (15 of 17) of survivors in the damage control surgery group had their intestinal continuity restored at the end of the follow-up in the present study. Taking the severity of the disease into account, these are favorable results.
Key limitations of the present study are its retrospective character and the small number of patients. Both groups were comparable in terms of demographics and disease severity; however, a selection bias cannot be definitely ruled out. There were a larger number of patients with a fecal peritonitis in the control group although statistical significance was not reached. Only further studies comparing the damage control surgery and conventional surgery could show whether our findings and the findings of Innsbruck group [6, 7] would be confirmed in a prospective randomized trial.
Conclusions
Damage control strategy is a promising concept for patients in need of primary resection for perforated diverticulitis complicated by generalized peritonitis. It provides fast clearance of the septic focus during the initial operation and can be applied to patients with fecal peritonitis.
References
Holmer C, Kreis ME (2014) Diverticular disease—choice of surgical procedure. Chirurg 85:308–313
Aydin HN, Remzi FH, Tekkis PP, Fazio VW (2005) Hartmann’s reversal is associated with high postoperative adverse events. Dis Colon Rectum 48:2117–2126
Vermeulen J, Coene PPLO, Van Hout NM et al (2009) Restoration of bowel continuity after surgery for acute perforated diverticulitis: should Hartmann’s procedure be considered a one-stage procedure? Colorectal Dis 11:619–624
Oberkofler CE, Rickenbacher A, Raptis DA et al (2012) A multicenter randomized clinical trial of primary anastomosis or Hartmann’s procedure for perforated left colonic diverticulitis with purulent or fecal peritonitis. Ann Surg 256:819–826 (discussion 826–827)
Vermeulen J, Lange JF (2010) Treatment of perforated diverticulitis with generalized peritonitis: past, present, and future. World J Surg 34:587–593
Kafka-Ritsch R, Birkfellner F, Perathoner A et al (2012) Damage control surgery with abdominal vacuum and delayed bowel reconstruction in patients with perforated diverticulitis Hinchey III/IV. J Gastrointest Surg 16:1915–1922
Perathoner A, Klaus A, Mühlmann G, Oberwalder M, Margreiter R, Kafka-Ritsch R (2010) Damage control with abdominal vacuum therapy (VAC) to manage perforated diverticulitis with advanced generalized peritonitis—a proof of concept. Int J Colorectal Dis 25:767–774
Cirocchi R, Arezzo A, Vettoretto N et al (2014) Role of damage control surgery in the treatment of Hinchey III and IV sigmoid diverticulitis: a tailored strategy. Medicine 93:e184
Langenfeld SJ (2013) Mandatory exploration is not necessary for patients with acute diverticulitis and free intraperitoneal air. J Trauma Acute Care Surg 74:1376
Rogy M, Függer R, Schemper M, Koss G, Schulz F (1990) The value of 2 distinct prognosis scores in patients with peritonitis. The Mannheim Peritonitis Index versus the Apache II score. Chirurg 61:297–300
Függer R, Rogy M, Herbst F, Schemper M, Schulz F (1988) Validation study of the Mannheim Peritonitis Index. Chirurg 59:598–601
Demmel N, Muth G, Maag K, Osterholzer G (1994) Prognostic scores in peritonitis: the Mannheim Peritonitis Index or APACHE II? Langenbecks Arch Chir 379:347–352
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Binda GA, Karas JR, Serventi A, Study Group on Diverticulitis et al (2012) Primary anastomosis vs nonrestorative resection for perforated diverticulitis with peritonitis: a prematurely terminated randomized controlled trial. Colorectal Dis 14:1403–1410
Cirocchi R, Trastulli S, Desiderio J et al (2013) Treatment of Hinchey stage III-IV diverticulitis: a systematic review and meta-analysis. Int J Colorectal Dis 28:447–457
O’Sullivan GC, Murphy D, O’Brien MG, Ireland A (1996) Laparoscopic management of generalized peritonitis due to perforated colonic diverticula. Am J Surg 171:432–434
Myers E, Hurley M, O’Sullivan GC, Kavanagh D, Wilson I, Winter DC (2008) Laparoscopic peritoneal lavage for generalized peritonitis due to perforated diverticulitis. Br J Surg 95:97–101
Toorenvliet BR, Swank H, Schoones JW, Hamming JF, Bemelman WA (2010) Laparoscopic peritoneal lavage for perforated colonic diverticulitis: a systematic review. Colorectal Dis 12:862–867
Angenete E, Thornell A, Burcharth J et al (2014) Laparoscopic lavage is feasible and safe for the treatment of perforated diverticulitis with purulent peritonitis: the first results from the randomized controlled trial DILALA. Ann Surg 263:117–122
Swank HA, Vermeulen J, Lange JF, Dutch Diverticular Disease (3D) Collaborative Study Group et al (2010) The ladies trial: laparoscopic peritoneal lavage or resection for purulent peritonitis and Hartmann’s procedure or resection with primary anastomosis for purulent or faecal peritonitis in perforated diverticulitis (NTR2037). BMC Surg 10:29
Vennix S, Musters GD, Mulder IM, Ladies trial colloborators et al (2015) Laparoscopic peritoneal lavage or sigmoidectomy for perforated diverticulitis with purulent peritonitis: a multicentre, parallel-group, randomised, open-label trial. Lancet 386:1269–1277
Schultz JK, Yaqub S, Wallon C, SCANDIV Study Group et al (2015) Laparoscopic lavage vs primary resection for acute perforated diverticulitis: the SCANDIV randomized clinical trial. JAMA 314:1364–1375
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was conducted according to ethical standards.
Informed consent
Due to the retrospective character, informed consent was not accomplished.
Rights and permissions
About this article
Cite this article
Sohn, M., Agha, A., Heitland, W. et al. Damage control strategy for the treatment of perforated diverticulitis with generalized peritonitis. Tech Coloproctol 20, 577–583 (2016). https://doi.org/10.1007/s10151-016-1506-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10151-016-1506-7