Abstract
Background
Acute myeloid leukemia (AML) is a heterogeneous blood disease with poor treatment effect and high recurrence rate. With the deepening of non-coding RNA research, more and more miRNAs have been found to participate in various physiological processes of tumors. In this study, we tried to find the miRNA related to the prognosis of AML.
Methods
Collect gene expression data and clinical information of AML patients in the Cancer Genome Atlas database for statistical analysis. The expression level of miR-195 of each patient was standardized by logCPM and then produced as a box plot according to subtype classification. TargetScan was used to predict the target genes of miR-195, and these genes were subjected to GO pathway enrichment analysis by Metascape. Differential miRNAs were screened through the DESeq2 package in the R language. Survival rates were estimated using the Kaplan–Meier method and the log-rank test. The multivariate Cox proportional hazard models of EFS and OS were established.
Results
We found that the expression of miR-195 was the lowest in cytogenetically normal (CN-) AML, and high expression of miR-195 only promoted the prognosis of chemotherapy-only CN-AML patients (EFS: P = 0.016; OS: P = 0.035). Multivariate analysis showed that miR-195high was a favorable and independent factor for CN-AML (both P < 0.05). Further analysis showed that miR-195 may affect signal transduction through ANHAK2 in AML.
Conclusion
We found that high expression of miR-195 can increase prognosis time of chemotherapy-only CN-AML patients, providing a new possibility for treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute myeloid leukemia (AML) is a genetically heterogeneous disorder [1], its incidence is three to four per 100,000 per year with a median age of 65–71 years of diagnosis [2,3,4]. AML is still a highly resistant disease of conventional chemotherapy, molecular profiling and next-generation sequencing by PCR has revealed a variety of recurrent gene mutations, such as FLT3, DNMT3A, IDH [5]. These mutations result in resistance to conventional chemotherapy. The total cure rate of AML is only 30–40% [6]. Cytogenetically normal acute myeloid leukemia (CN-AML) is the most common karyotype and usually fall into intermediate-risk cytogenetics. Gene mutations also have been found to be associated with CN-AML. It is necessary to find new factors for the prognosis of AML. Aberrantly expressed genes are likely to be useful for defining molecular risk in CN-AML.
MicroRNA (miRNA) is a small non-coding 21–23 nucleotide RNA which can regulate gene expression by binding to the untranslated region of the target mRNA molecule, thereby inhibiting transcription or inducing mRNA degradation [7, 8]. MiRNAs have been shown to play important roles in a variety of tumor activities, such as proliferation, angiogenesis, apoptosis, invasion and metastasis [9,10,11,12].
MiR-195 shows different expression patterns and functions in various cancers [13,14,15,16]. It has been reported that miR-195 can inhibit the proliferation of liver cancer cells by targeting Cyclin D1, CDK6 and E2F3 [16], and promote the apoptosis of colorectal cancer cells by inhibiting Bcl-2 [17]. However, the biological function of miR-195 in AML is not clear. In this study, we explored the impact of miR-195 on the prognosis of patients with AML who have been diagnosed and treated, especially in normal karyotypes.
Methods
Patients
A total of 188 adult de novo AML patients with complete clinical data and miR-195 expression information from The Cancer Genome Atlas (TCGA, https://tcga-data.nci.nih.gov/tcga) database were included in the study. Ninety patients were treated with chemotherapy only, including 40 CN-AML patients. Among 40 chemotherapy-only CN-AML patients, there were 36 patients with gene mRNA expression data. In addition, there were 35 CN-AML patients who had received allogeneic hematopoietic stem cell transplantation (allo-HSCT). A total of 75 CN-AML patients were involved in this study. Chemotherapy-only CN-AML patients age ranged from 35 to 88. Clinical features at diagnosis were described, including peripheral blood (PB) white blood cell counts (WBC), blast percentages in PB and bone marrow (BM), French–American–British (FAB) subtypes, and the frequencies of known recurrent genetic mutations. Detailed clinical and molecular characteristics could be found on the TCGA website. Event-free survival (EFS) and overall survival (OS) were the primary endpoints of this study. EFS was defined as the time from diagnosis to withdrawal of the study due to lack of complete remission, relapse, or death, or was censored at the last follow-up. OS was defined as the time from diagnosis to death or was censored at the last follow-up. Informed consent was provided to all patients and the study protocol was approved by the University of Washington Human Research Committee.
Analysis of the miR-195 expression
To analyze the expression level of each AML subtype patient, we normalized the expression level of miR-195 by logCPM (count-per-millon), and then made a box plot. In addition, after subtracting the median expression level from the expression level of each patient, we made a histogram to show the expression level distribution of miR-195 in 40 chemotherapy-only CN-AML patients.
Bioinformatics analysis
We entered miR-195 in the TargetScan website (http://www.targetscan.org/vert_72/) to predict its potential target genes and downloaded the result list. Then we used Metascape (http://metascape.org/) to carry out the GO (Gene Ontology) gene pathway enrichment analysis of target genes. According to the expression of miRNAs in the miR-195high group and the miR-195low group, the DESeq2 package in the R language screen the differential miRNAs [18]. The threshold for screening differential genes was set at fold change > 1 and false discovery rate < 5%.
Statistical analysis
The clinical and molecular characteristics of the patients were summarized using descriptive statistical methods. Data sets were described by median and/or range. The Mann–Whitney U test was used as appropriate to compare numerical comparison and chi-square test for comparison of categorical data. Survival rates were estimated using the Kaplan–Meier method and the log-rank test. The multivariate Cox proportional hazard models of EFS and OS were established using a limited backward elimination process. The statistical significance level was 0.05 for a two-tailed test. All statistical analyses were performed using SPSS software 20.0 and GraphPad Prism software 7.0.
Results
Prognostic significance of miR-195 in CN-AML
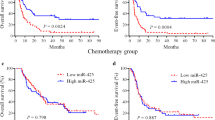
According to the median expression levels of the miR-195 in chemotherapy-only group, all patients were divided into two groups. Kaplan–Meier analysis demonstrated that the chemotherapy patients with high expressions of miR-195 had longer EFS than those with low expressions (P = 0.009, Fig. 1A), while OS had no difference (Fig. 1B). Then, among all patients included in the TCGA database, we analyzed the expression of miR-195 in each subtype and found that the CN-AML group had the lowest expression, which attracted our attention (Fig. 1C). Similarly, after using the median of expression to divide CN-AML patients into two groups, there were both longer EFS and OS in the miR-195high group (both P < 0.05, Fig. 1D, E; Supplementary Fig. 1). Meanwhile in allo-HSCT CN-AML patients, it had no difference about EFS and OS in two different expression groups (Fig. 1F, G).
Kaplan–Meier curves of EFS and OS in different expression levels of miR-195. A, B EFS and OS results in all chemotherapy patients (n = 90); C The expression level of miR-195 in each AML subtype; D, E EFS and OS results in chemotherapy-only CN-AML patients (n = 40); F, G EFS and OS results in allo-HSCT patients (n = 35). EFS event-free survival, OS overall survival
Clinical and molecular characteristics of the CN-AML patients in chemotherapy-only group
All normal karyotype chemotherapy patients (n = 40) were divided by miR-195 median expression levels, respectively (Table 1). Comparing to the miR-195low group, the miR-195gigh group had less WBC count (P = 0.011), BM blasts (P = 0.038) and PB blasts (P = 0.009), and fewer patients with FLT3-ITD (P = 0.019). No significant differences were found in age, gender distribution, FAB subtypes, and frequency of other recurrent genetic mutations (NPM1, DNMT3A, IDH1/IDH2, RUNX1, NRAS/KRAS, TET2 and TP53) between the two groups.
Multivariate analyses of EFS and OS in CN-AML patients
To assess the prognostic significance of the aforementioned clinical and molecular characteristics in the CN-AML chemotherapy patients, we chose the expression levels of miR-195 (high vs. low), age (≥ 60 vs. < 60), WBC count (≥ 15 vs. < 15 × 109/L), BM blasts (≥ 70 vs. < 70%), FLT3-ITD (positive vs. negative), and other common genetic mutations (NPM1, RUNX1, TET2 and DNMT3A; mutated vs. wild) to construct multivariate analyses (Table 2). High miR-195 expression and age ≥ 60 were identified as independent risk factors for EFS (both P < 0.05), while high miR-195 expression and TET2 mutation were independent risk factors for OS (both P < 0.05).
Potential mechanism of miR-195 in chemotherapy-only CN-AML group
To explore the mechanism of miR-195 in chemotherapy-only CN-AML, we first used TargentScan to predict the potential target genes of miR-195. In the end, we got 82 related target genes with expression data in the TCGA database (Supplementary Table 1). After GO enrichment of these target genes, it was found that they were mainly concentrated in some pathways, such as protein localization to membrane, stress-activated protein kinase signaling cascade, mitogen-activated protein kinase (MAPK) signaling pathway, and so on (Fig. 2A). Then we analyzed the prognostic effects of 82 target genes in chemotherapy-only CN-AML group. And based on miRNA's main inhibitory effect on target genes, we screened out the most likely downstream target of miR-195, AHNAK nucleoprotein 2 (AHNAK2). Among the 36 CN-AML chemotherapy patients, patients with high expression of AHNAK2 had shorter OS, which was the opposite of the results of miR-195 (P = 0.0492, Fig. 2B). Then we also screened the differential miRNAs between two groups, and 18 miRNAs were down-expression in miR-195low group (Fig. 2C, 2D).
Potential mechanism of miR-195 in chemotherapy-only CN-AML group. A The GO enrichment results of 82 target genes of miR-195; B chemotherapy-only CN-AML patients with high expression of AHNAK2 had shorter OS; C, D Results of miR-195 target miRNA by unsupervised clustering heat map and volcano map analysis. GO gene ontology, AHNAK2 AHNAK nucleoprotein 2, OS overall survival
Discussion
In this study, we found in CN-AML, the expression of miR-195 was the lowest. And high expression of miR-195 was only beneficial to the prognosis in chemotherapy CN-AML patients, while no difference in common karyotypes. But these prognostic effects were not observed in CN-AML patients with allo-HSCT. This may be because allo-HSCT is currently the only treatment that may cure AML. The treatment is more intense and can overcome some problems that chemotherapy cannot handle.
The prognostic effect of MiR-195 had been found in some tumors. In pediatric AML, the expression of miR-195 was reduced in bone marrow or serum, and patients with low expression of miR-195 also had worse survival [19]. In bladder cancer and gastric cancer, low expression of miR-195 was also a poor prognostic factor [20, 21]. According to our analysis, highly expressed miR-195 is an independent prognostic factor for CN-AML, representing longer OS and EFS. This is most likely due to the fact that the miR-195high patient group has fewer WBC count, BM blasts, PM blasts and lower frequency FLT3-ITD mutations.
The pathway enrichment of miR-195 target genes showed that the main concentrated pathways were protein localization to membrane, stress-activated protein kinase signaling cascade, and MAPK signaling pathway. These three pathways were closely related to the extra- and intracellular signal transduction. Previous studies had found that in bladder cancer and cervical cancer, miR-195 can affect the MAPK pathway to regulate tumor cell proliferation and apoptosis [20, 22]. In addition, we screened out the potential target gene of miR-195 in AML, ANHAK2, which not only had a negative prognostic effect due to its increased expression in a variety of tumors [23,24,25], but also participated in cell signal transduction. In lung adenocarcinoma, down-regulating the expression of ANHAK2 can reduce the activity of the MAPK pathway to inhibit cell proliferation [26]. In this study, we guessed that in chemotherapy-only CN-AML, miR-195 was likely to play a role in cell signal transduction through ANHAK2. Of course, these needed follow-up experiments to confirm. Meanwhile, according to the differentially expressed miRNAs obtained, miR-195 may also have an effect on other miRNAs in chemotherapy-only CN-AML.
According to the results of OS in Table 2, the TET2 mutation is also an independent prognostic factor for CN-AML, in addition to miR-195. TET2 mutations are present in multiple lymphoid and myeloid lines as well as CD34+ progenitor cells, suggesting early clonal mutations in the stem cell compartment [27]. TET2 mutations are present in 32% of patients in secondary acute myeloid leukemia [28]. The TET2 mutation status has been found to be an uncertain prognostic indicator. Some studies have shown that TET2 is significantly associated with a poor prognosis in AML, in contrast, others have found that it is associated with a good prognosis in MDS [29, 30]. The role of TET2 in CN-AML needs further exploration.
In conclusion, our analysis finds that high expression of miR-195 is an independent and favorable prognostic factor in AML normal karyotype chemotherapy patients. But their effects can be eliminated by allo-HSCT. The results of the study are limited by the small sample size and further studies of larger samples are needed.
References
Papaemmanuil E, Döhner HPJC (2016) Genomic classification in acute myeloid leukemia. N Engl J Med 375(9):900–901
Dinmohamed AG, Visser O, Posthuma EFM et al (2016) MDS classification is improving in an era of the WHO 2016 criteria of MDS: a population-based analysis among 9159 MDS patients diagnosed in the Netherlands. Cancer Epidemiol 50:137–140
Juliusson G, Lazarevic V, Hörstedt AS et al (2012) Acute myeloid leukemia in the real world: why population-based registries are needed. Blood 119:3890–3899
Nagel G, Weber D, Fromm E et al (2017) Epidemiological, genetic, and clinical characterization by age of newly diagnosed acute myeloid leukemia based on an academic population-based registry study (AMLSG BiO). Ann Hematol 96:1993–2003
Hackl H, Astanina KRW (2017) Molecular and genetic alterations associated with therapy resistance and relapse of acute myeloid leukemia. J Hematol Oncol 10:51
Rowe JM (2010) The evolving paradigm of prognostic factors in AML: introduction to the Acute Leukemia Forum 2010. Best Pract Res Clin Haematol 23:453–456
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Holland EC (2001) Gliomagenesis: genetic alterations and mouse models. Nat Rev Genet 2:120–129
Reardon DA, Rich JN, Friedman HS et al (2006) Recent advances in the treatment of malignant astrocytoma. J Clin Oncol 24(8):1253–1265
Maher EA, Furnari FB, Bachoo RM et al (2001) Malignant glioma: genetics and biology of a grave matter. Genes Dev 15(11):1311–1333
Sanai N, Alvarez-Buylla A, Berger MS (2005) Neural stem cells and the origin of gliomas. N Engl J Med 353(8):811–822
Zhu Y, Parada LF (2002) The molecular and genetic basis of neurological tumours. Nat Rev Cancer 2(8):616–626
Porkka KP, Pfeiffer MJ, Waltering KK et al (2007) MicroRNA expression profiling in prostate cancer. Cancer Res 67(13):6130–6135
Zanette DL, Rivadavia F, Molfetta GA et al (2007) MiRNA expression profiles in chronic lymphocytic and acute lymphocytic leukemia. Braz J Med Biol Res 40(11):1435–1440
Wong T-S, Liu X-B, Wong B-H et al (2008) Mature miR-184 as potential oncogenic microRNA of squamous cell carcinoma of tongue. Clin Cancer Res 14(9):2588–2592
Teng X, Ying Z, Yujuan X et al (2009) MicroRNA-195 suppresses tumorigenicity and regulates G1/S transition of human hepatocellular carcinoma cells. Hepatology 50(1):113–121
Liu L, Chen L, Yingxin Xu et al (2010) microRNA-195 promotes apoptosis and suppresses tumorigenicity of human colorectal cancer cells. Biochem Biophys Res Commun 400(2):236–240
Juliana C-S, Douglas D, Lopes FM (2017) RNA-Seq differential expression analysis: An extended review and a software tool. PLoS ONE 12(12):e0190152
Hong Z, Zhang R, Qi H (2018) Diagnostic and prognostic relevance of serum miR-195 in pediatric acute myeloid leukemia. Cancer Biomark 21(2):269–275
Yang K, Tang H, Ding M et al (2019) Expression of miR-195 and MEK1 in patients with bladder cancer and their relationship to prognosis. Int J Clin Exp Pathol 12(3):843–850
Zou J, Liao X, Zhang J et al (2018) Dysregulation of miR-195-5p/-218-5p/BIRC5 axis predicts a poor prognosis in patients with gastric cancer. J Biol Regul Homeost Agents 33(5):1377–1385
Zhang J, Jiang P, Wang S et al (2021) LncRNA LIPE-AS1 predicts poor survival of cervical cancer and promotes its proliferation and migration via modulating miR-195–5p/MAPK pathway. Front Oncol 11:639980
Wang M, Li X, Zhang J et al (2017) AHNAK2 is a novel prognostic marker and oncogenic protein for clear cell renal cell carcinoma. Theranostics 7(5):1100–1113
Zheng L, Li S, Zheng X et al (2021) AHNAK2 is a novel prognostic marker and correlates with immune infiltration in papillary thyroid cancer: Evidence from integrated analysis. Int Immunopharmacol 90:107185
Zhang S, Lu Y, Qi L et al. (2020) AHNAK2 is associated with poor prognosis and cell migration in lung adenocarcinoma. Biomed Res Int 8571932
Wang D-W, Zheng H-Z, Cha Na et al (2020) Down-regulation of AHNAK2 inhibits cell proliferation, migration and invasion through inactivating the MAPK pathway in lung adenocarcinoma. Technol Cancer Res Treat 19:1533033820957006
Alexander ES, Azim MM, Austin K et al (2010) Next-generation sequencing of the TET2 gene in 355 MDS and CMML patients reveals low-abundance mutant clones with early origins, but indicates no definite prognostic value. Blood 116(19):3923–3932
Jankowska AM, Szpurka H, Tiu RV et al (2009) Loss of heterozygosity 4q24 and TET2 mutations associated with myelodysplastic/myeloproliferative neoplasms. Blood 113(25):6403–6410
Omar A-W, Ann M, Cyrus H et al (2009) Genetic characterization of TET1, TET2, and TET3 alterations in myeloid malignancies. Blood 114(1):144–147
Olivier K, Véronique G-B, Meyling C et al (2009) TET2 mutation is an independent favorable prognostic factor in myelodysplastic syndromes (MDSs). Blood 114(15):3285–3291
Acknowledgements
This work was supported by grants from Xinjiang Joint Fund of National Natural Science Foundation of China (U1903117).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary Fig 1
The expression level of miR-195 of each chemotherapy-only CN-AML patient (n=40). Blue represented the miR-195low group; red represented the miR-195high group. (TIF 1557 KB)
About this article
Cite this article
Cui, L., Zeng, T., Zhang, L. et al. High expression of miR-195 is related to favorable prognosis in cytogenetically normal acute myeloid leukemia. Int J Clin Oncol 26, 1986–1993 (2021). https://doi.org/10.1007/s10147-021-01999-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-021-01999-5