Abstract
Objective
To assess the efficacy, outcome and complications of post-chemotherapy laparoscopic retroperitoneal lymph node dissection (L-RPLND) for stage IIA/B testicular germ cell tumor (GCT) patients in comparison with open RPLND (O-RPLND).
Methods
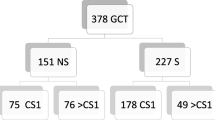
L-RPLND was performed in 14 patients with stage IIA/B non-seminoma GCTs among 154 non-seminoma patients who received RPLND after completion of chemotherapy with tumor marker normalization at our institution between 1998 and 2013. Their outcomes were compared with those of 14 patients with stage IIA/B non-seminoma GCTs treated with O-RPLND during the same period. Clinical parameters were compared between L-RPLND and O-RPLND.
Results
There were no significant differences in the background characteristics of the two groups except for follow-up duration (36 months for L-RPLND, 70 months for O-RPLND; p = 0.02). Blood loss during surgery was significantly less for the L-RPLND group than for the O-RPLND group (155 mL for L-RPLND, 700 mL for O-RPLND; p < 0.001). Parameters related to post-operative recovery were significantly better for the L-RPLND group than for the O-RPLND group. Histopathological examination showed no difference between the two groups. Neither group had disease recurrence.
Conclusion
Post-chemotherapy L-RPLND with a bilateral template and nerve-sparing method was safe, effective, and showed a high preservation rate of antegrade ejaculation with no deterioration of outcomes compared to O-RPLND.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Post-chemotherapy retroperitoneal lymph node dissection (PC-RPLND) is an essential part of the management of advanced testicular germ cell tumors (GCTs), especially non-seminoma GCTs. Open RPLND (O-RPLND) is recommended in the current guidelines [1, 2]. After the first report of laparoscopic RPLND (L-RPLND) by Rukstalis, its indication has been extended from staging to therapy [3–7]. We have also reported our preliminary experience [8]. The technical feasibility of the approach has been reported by several investigators [6, 9]; however, most reports, including ours, showed the feasibility of L-RPLND without comparing it to their own O-RPLND series.
In this study, intra- and post-operative clinical parameters of our consecutive series of L-RPLND and O-RPLND were compared.
Patients and methods
Patients
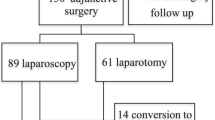
Between June 1998 and December 2013, 175 post-chemotherapy RPLNDs were performed at our institution. L-RPLND was started in September 2009. Before starting L-RPLND for stage IIA/B GCTs (stage IIC excluded), O-RPLND was a standard procedure at our institution. Laparoscopic or open RPLND was chosen by patient preference after starting L-RPLND. Of 154 non-seminoma patients with RPLND, L-RPLND and O-RPLND were performed in 14 patients each with stage IIA/B during this period. All patients had induction or salvage chemotherapy and obtained serum tumor marker (STM) normalization. Patients who obtained true complete response were excluded from this study. Patient characteristics are summarized in Table 1. A significant difference was observed only in median follow-up (36 months for L-RPLND vs 70 months for O-RPLND; p = 0.02). The Institutional Review Board approved the performance of L-RPLND, and written, informed consent was obtained from all patients.
Surgical procedure
O-RPLND was performed with a full bilateral template with a nerve-sparing technique according to our previous report [10]. L-RPLND was performed bilaterally using the extra-peritoneal approach, as in our report [8]. Briefly, the patient was placed in the supine position. We developed the retroperitoneal space between the psoas muscle and the ureter until the ipsilateral great vessel could be seen. A prospective identification and sparing of the sympathetic nerves was carried out in all patients. The first two patients who underwent L-RPLND had a unilateral modified template to ensure the safety of L-RPLND. Drainage tubes were removed after the amount of discharge decreased to <100 ml in the L-RPLND and 500 ml in the O-RPLND group.
Follow-up strategy
The usual follow-up strategy was as follows—baseline serum tumor marker (STM) levels and computed tomography (CT) were carried out 4 weeks after the last surgery. Blood tests including STM levels, CT (chest to pelvis), and physical examinations were then repeated every 3 months in the first year. In viable cancer cases, STM levels were measured every month in the first year. In the 2nd and 3rd years, STM levels and CT were checked every 3 or 4 months. In the 4th and 5th years, STM levels and CT were checked every 6 months. After 5 years, medical check-ups were performed annually.
Statistical analysis and assessment of complications
Statistical differences between the two groups were analyzed by the chi-squared test for dependent variables, Fisher’s exact probability test, and Mann–Whitney’s U test, as appropriate.
Intra- and post-operative complications were categorized using the Clavien−Dindo classification (10). We defined antegrade ejaculation as any discharge of seminal fluid at ejaculation by patient report.
Results
Peri-operative variables (Table 2)
Median tumor sizes before treatment and before RPLND were not significantly different between L-RPLND and O-RPLND groups. Blood loss during surgery was significantly less in the L-RPLND group than in the O-RPLND group. Operation time showed no significant difference between L-RPLND and O-RPLND groups because a bilateral template was used for L-RPLND. No conversion to open surgery was required with L-RPLND.
Times to initiation of oral intake and walking after surgery were significantly shorter in the L-RPLND group than in the O-RPLND group. The median period to permission of discharge was also significantly shorter in the L-RPLND group than in the O-RPLND group.
On histopathological examination, both types of RPLND showed similar proportions of necrosis, teratoma, and viable cancer. Three viable cancer cases were found in the O-RPLND group and the histology of those cases were embryonal carcinoma, seminoma and unknown malignancy, respectively. Two of three patients who only underwent induction chemotherapy had 2 cycles of adjuvant chemotherapy.
With regard to antegrade ejaculation, uni-or bilateral nerve-sparing RPLND was performed in all patients. All but one patient in each group had antegrade ejaculation.
Complications
Peri-operative complications are summarized in Table 3.
Intervention by percutaneous puncture was required in one case with a lymphatic cyst. In this case, inflammation of the lymphatic cyst occurred on the 10th day after surgery. Chyle leaks were more often observed in the L-RPLND group; however, the chyle leaks disappeared within a few days by taking a conventional low-fat diet. On the other hand, surgical site infections, including fat necrosis, were observed only in the O-RPLND group.
One O-RPLND case developed acute respiratory distress syndrome 2 days after surgery and required intensive care unit treatment.
Clinical outcomes
There was no disease recurrence in either group at the time of writing, with a median follow-up of 36 months for the L-RPLND group and 70 months for the O-RPLND group.
Discussion
For residual mass resection after chemotherapy for advanced testicular GCTs, post-chemotherapy RPLND is mandatory [1, 2]. O-RPLND with a full template is standard procedure. Nerve sparing is often not possible because of encasement of the residual mass to be excised. In addition, post-chemotherapy RPLND is a complex, high-risk procedure that sometimes requires vascular procedures and often requires complete mobilization of the vena cava and aorta. Because of these morbidity and complicated surgical procedures of O-RPLND, less invasive surgery that does not adversely affect disease-free survival is required, especially in early stage GCTs.
L-RPLND began as a diagnostic tool for stage I GCTs [3, 4]. After confirmation of its safety, its indication has been extended to include therapy [5, 6, 9]. A unilateral, modified template with an intra-abdominal approach has been a common procedure in most reports of L-RPLND. In the early L-RPLND era, a modified template was not part of standard post-chemotherapy RPLND. Therefore, bilateral L-RPLND using a nerve-sparing technique was developed [11]. The accumulated experience of bilateral L-RPLND with nerve sparing [12–14] has shown that its safety and oncologic outcomes have been comparable to those of O-RPLND.
Although the follow-up duration was significantly shorter for L-RPLND than for O-RPLND, the major complications and oncologic outcomes were comparable. In fact, no recurrence has occurred to date. Our extra-retroperitoneal surgical procedure with nerve sparing [8] was based on the reports of Arai et al. [7], LeBlanc et al. [11] and Steiner et al. [12]. No open conversions or major complications occurred. Times to initiation of oral intake and walking after surgery were significantly shorter for L-RPLND than for O-RPLND patients. In addition, the length of hospital stay was also significantly shorter for L-RPLND patients.
Kenney and Tuerk reviewed the reported complications [15]. Their report showed that vascular injury and hemorrhage were the most common complications of post-chemotherapy L-RPLND. With regard to post-operative complications, no ileus and no wound infections were observed, but retrograde ejaculation was reported. Winter et al. reported that tumor size and International Germ Cell Consensus Classification (IGCCC) [16] predicted additional vascular procedures such as inferior vena cava interventions or aortic prosthesis [17]. Intermediate or poor prognosis showed a significantly higher risk of intervention than good prognosis, and residual tumor size >5 cm had a higher risk. In the present study, neither ileus nor bowel injury were observed. In this study, chyle leak was the most frequent complication and observed more in the L-RPLND group than in the O-RPLND group. In the L-RPLND group, the drainage tube was usually removed after starting a normal diet. On the other hand, patients in the O-RPLND group usually start meals three or four days after surgery and the drainage tube is usually removed before starting meals due decreased lymphatic fluid. Since chyle leaks are usually observed more frequently after starting a meal, early timing of starting a diet in L-RPLND patients might be associated with the frequent occurrence of chyle leaks. This may be preventable by using a clip to seal the lymphatic vessels and by a low fat diet. In fact, when we started to use clips, the number of chyle leaks decreased. Steiner et al. reported that chyle leaks were not observed after introducing a low fat diet [18]. In addition, no additional vascular procedure and nephrectomy were observed with our strict indication of post-chemotherapy L-RPLND. In this study, we performed L-RPLND in patients with stage IIA/IIB and mainly IGCCC good prognosis (93.3 %). Therefore, this indication would be acceptable for post-chemotherapy L-RPLND. With regard to ejaculation, there was no significant difference between L-RPLND and O-RPLND (p = 0.89). This means that bilateral nerve-sparing post-chemotherapy L-RPLND is feasible for the management of advanced GCTs. The reported retrograde ejaculation rate was 0–5 % with primary RPLND for stage I non-seminoma and up to 12.5 % with post-chemotherapy L-RPLND [15]. The present result with post-chemotherapy L-RPLND with a bilateral template, which is a more complicated procedure, was identical to that with primary L-RPLND.
Bilateral RPLND using a laparoscopic or open method is the standard method for the management of post-chemotherapy residual masses. However, the extent of RPLND has been discussed [19–21]. Recently, Vallier et al. reported a validation study to determine whether a modified template would be reasonable [22]. They wrote that a unilateral modified template resection during post-chemotherapy RPLND was suitable if pre-chemotherapy metastases were found in the primary landing zone of the tumor-bearing testicle and if the post-chemotherapy residual masses were <5 cm in diameter. Otherwise, a full bilateral template RPLND is recommended. With the accumulated evidence, the modified template is now suitable for well-defined small residual masses after chemotherapy. In this situation, less invasive surgery was desired for low-stage GCT patients for resection of post-chemotherapy residual masses. Although bilateral L-RPLND was recognized to be safe and effective, modified unilateral L-RPLND might be a standard of care for stage IIA/B, low-volume, well-defined residual masses after chemotherapy. Our first case of L-RPLND with a modified template has not developed recurrence or metastasis to date.
In recent advances in L-RPLND, patients with more advanced GCTs such as infiltrating great vessels could be candidates for L-RPLND. Aufderklamm et al. showed bilateral L-RPLND in patients with vascular infiltration is feasible and reproducible when laparoscopic vascular surgery can be reliably handled [23].
From these findings, less invasive L-RPLND with a modified template would be a feasible procedure for stage IIA/IIB GCTs and bilateral L-RPLND with more complicated cases with infiltrating great vessels might be one of the treatment options.
Despite the limitations of this study, i.e., the small number of patients and short follow-up, the safety and comparable oncologic outcomes of L-RPLND and O-RPLND seem clear.
In conclusion, post-chemotherapy L-RPLND with a bilateral template and nerve-sparing method was safe, effective, and showed a high preservation rate of antegrade ejaculation without adversely affecting the outcome compared to O-RPLND. Modified L-RPLND might become a standard method for post-chemotherapy management of patients with stage IIA/B non-seminoma GCTs.
References
Guidelines on Testicular Cancer, European Association of Urology, 2012
NCCN Clinical Practice Guidelines in Oncology, Testicular Cancer, 2012
Klotz L (1994) Laparoscopic retroperitoneal lymphadenectomy for high-risk stage I nonseminomatous germ cell tumor: report of four cases. Urology 43:752–756
Janetschek G, Reissigl A, Peschel R et al (1994) Laparoscopic retroperitoneal lymph node dissection for clinical stage I nonseminomatous testicular tumor. Urology 44:382–391
Palese MA, Su LM, Kavoussi LR (2002) Laparoscopic retroperitoneal lymph node dissection after chemotherapy. Urology 60:130–134
Maldonado-Valadez R, Schilling D, Anastasiadis AG et al (2007) Post-chemotherapy laparoscopic retroperitoneal lymph-node dissection in testis cancer patients. J Endourol 21:1501–1504
Arai Y, Kaiho Y, Yamada S, Saito H et al (2012) Extraperitoneal laparoscopic retroperitoneal lymph node dissection after chemotherapy for nonseminomatous testicular germ-cell tumor: surgical and oncological outcomes. Int Urol Nephrol 44:1389–1395
Kimura Y, Nakamura T, Kawauchi A et al (2013) Post-chemotherapy nerve-sparing laparoscopic retroperitoneal lymph node dissection in stage IIB testicular cancer. Int J Urol 20:837–841
Calestroupat JP, Sanchez-Salas R, Cathelineau X et al (2009) Postchemotherapy laparoscopic retroperitoneal lymph node dissection in nonseminomatous germ-cell tumor. J Endourol 23:645–650
Miki T, Mizutani Y, Nakamura T et al (2009) Post-chemotherapy nerve-sparing retroperitoneal lymph node dissection for advanced germ cell tumor. Int J Urol 16:379–382
LeBlanc E, Caty A, Dargent D et al (2001) Extraperitoneal laparoscopic para-aortic lymph node dissection for early stage nonseminomatous germ cell tumors of the testis with introduction of a nerve sparing technique: description results. J Urol 165:89–92
Steiner H, Zangerl F, Sto¨hr B et al (2008) Results of bilateral nerve sparing laparoscopic retroperitoneal lymph node dissection for testicular cancer. J Urol 180:1348–1352
Steiner H, Leonhartsberger N, Stoehr B et al (2013) Postchemotherapy laparoscopic retroperitoneal lymph node dissection for low-volume, stage II, nonseminomatous germ cell tumor: first 100 patients. Eur Urol 63:1013–1017
Aufderklamm S, Todenhöfer T, Hennenlotter J et al (2013) Bilateral laparoscopic postchemotherapy retroperitoneal lymph-node dissection in nonseminomatous germ cell tumors—a comparison to template dissection. J Endourol 27:856–861
Kenney PA, Tuerk IA (2008) Complications of laparoscopic retroperitoneal lymph node dissection in testicular cancer. World J Urol 26:561–569
International Germ Cell Cancer Collaborative Group (1997) International germ cell consensus classification: a prognostic factor-based staging system for metastatic germ cell cancers. J Clin Oncol 15:594–603
Winter C, Pfister D, Busch J et al (2012) Residual tumor size and IGCCCG risk classification predict additional vascular procedures in patients with germ cell tumors and residual tumor resection: a multicenter analysis of the German Testicular Cancer Study Group. Eur Urol 61:403–409
Steiner H, Peschel R, Janetschek G et al (2004) Long-term results of laparoscopic retroperinonael lymphnode dissection: a single-center 10-year experience. Urology 63:550–555
Carver BS, Shayegan B, Eggener S et al (2007) Incidence of metastatic nonseminomatous germ cell tumor outside the boundaries of a modified postchemotherapy retroperitoneal lymph node dissection. J Clin Oncol 25:4365–4369
Beck SD, Foster RS, Bihrle R et al (2007) Is full bilateral retroperitoneal lymph node dissection always necessary for postchemotherapy residual tumor? Cancer 110:1235–1240
Heidenreich A, Pfister D, Witthuhn R et al (2009) Postchemotherapy retroperitoneal lymph node dissection in advanced testicular cancer:radical or modified template resection. Eur Urol 55:217–226
Vallier C, Savoie PH, Delpero JR et al (2014) External validation of the Heidenreich criteria for patient selection for unilateral or bilateral retroperitoneal lymph node dissection for post-chemotherapy residual masses of testicular cancer. World J Urol 32:1573–1578
Aufderklamm S, Todenhöfer T, Hennenlotter J et al. (2014) Postchemotherapy laparoscopic retroperitoneal lymph node dissection for nonseminomatous germ cell tumors infiltrating the great vessels. J Endourol 28:668–674
Acknowledgments
The authors would like to express their great appreciation to Dr. Gaku Kawabata, Head of the Division of Urology of Kansai Rosai Hospital, Hyogo, Japan, for initiating and supporting extraperitoneal laparoscopic RPLND at our institution.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We have no financial disclosures or conflicts of interest and no funding sources.
About this article
Cite this article
Nakamura, T., Kawauchi, A., Oishi, M. et al. Post-chemotherapy laparoscopic retroperitoneal lymph node dissection is feasible for stage IIA/B non-seminoma germ cell tumors. Int J Clin Oncol 21, 791–795 (2016). https://doi.org/10.1007/s10147-015-0934-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-015-0934-0