Abstract
Microvascular decompression (MVD) is considered an effective treatment for trigeminal neuralgia (TN). However, the anatomical and clinical variables associated with a better outcome are not fully examined. The authors performed a systematic review and meta-analysis of the literature investigating the immediate and long-term clinical results of MVD for TN, and the impact of the anatomical features of the neurovascular conflict on the outcome. The systematic search of three databases was performed for studies published between January 1990 and November 2021. PRISMA guidelines were followed. Random-effects meta-analysis was used to pool the analyzed outcomes, and random-effect meta-regression was used to examine the association between the effect size and potential confounders. A funnel plot followed by Egger’s linear regression was used to test publication bias. A total of 9 studies were included in this analysis, including 2102 patients with trigeminal neuralgia. The immediate post-operative rate of BNI I was 82.9%, whereas surgical failure (BNI IV-V) was reported in approximately 2.6% of patients. CSF leak was the most common postoperative complication (2.4%). The rate of BNI I at last follow up was 64.7% (p < 0.01), showing a significant negative correlation after multiple meta-regression with the rate of patients with isolated venous conflict (p < 0.01). On the other hand, the evidence of an arterial conflict proved is positive association with a favorable outcome (p < 0.01). At the last follow-up, BNI IV-V was reported in 19.2% (95% CI 8.9–29.5%, p < 0.01, I2 = 97.3%). This meta-analysis confirms the safety and efficacy of MVD for TN. The occurrence of serious postoperative complications is very low. The long-term outcome is associated with the type of vascular structure involved, being pure venous conflict associated with a higher risk of surgical failure. These findings should be considered when planning surgery for patients with TN.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atreus of Cappadocia first described unilateral facial pain causing spams in the second century C.E. [30], and in 1756, Nicholas André coined the French term “tic douloureux” [4]. Nowadays, the so-called “classic” trigeminal neuralgia (TN) has been characterized as agonizing, paroxysmal, perceived in one or more divisions of the trigeminal nerve, with repetitive bursts of few seconds, exacerbated by cutaneous stimuli [5]. Subsequently, the hypothesis of a causal relationship between the TN and vascular compression of the trigeminal root entry zone was first released by Dandy [10], and then Gardner and Miklos [16], and later by Jannetta and colleagues [19], standardizing a surgical technique to separate and solve the vascular conflict, with surprising clinical results. The microvascular decompression (MVD), as stated by Burchiel in 2016, still represents a “major bright spot in the otherwise difficult world of pain surgery” [6]. Currently, although the specific mechanism of pain genesis is unknown [6], it is accepted worldwide that, when contact between an artery and the trigeminal nerve can be proven, the surgical treatment is advised and associated with good outcome in”classical TN” [6, 8, 19, 40]. Nonetheless, consensus is not achieved in relation to the long-term rate of a pain-free/medication-free patient after MVD for TN. The aim of our study is to examine the clinical results of this surgical intervention assessing the rates of patients that are pain-free at the last follow-up. Furthermore, we will try to differentiate the result according to the Barrow Neurological Institute (BNI) pain intensity score and analyze the impact on the outcome of the anatomical features of the neurovascular conflict.
Methods
Literature search
A comprehensive literature search of PubMed, Embase, and Ovid MEDLINE was conducted for studies published from January 1990 to November 2021. PRISMA guidelines (preferred reporting items for systematic reviews and meta-analysis [28]) were followed. Our systematic review was registered and accepted in the PROSPERO database with the following ID: CRD42021292153. The keywords, the detailed search strategy, and the inclusion criteria are reported in Table 1. In cases of overlapping patient populations, only the series with the largest number of patients or most detailed data were included. Two independent readers (D.D.C. and N.B.) screened articles in their entirety to determine eligibility for inclusion. A third author solved discrepancies (P.P.).
Data collection
From each study, we extracted the following: (1) demographic data, (2) mean follow-up, (3) trigeminal neuralgia characteristics and pain classification system, (4) radiological characteristics, (5) previous treatment, (6) surgical technique, (7) intraoperative findings, (8) post-operative complications, and (9) clinical outcome (immediately after surgery and at the last follow-up). Classical trigeminal neuralgia, as defined by Cruccu and colleagues [8], was divided in two groups considering the presence or the absence of continuous pain [8, 40]. When reported as typical/atypical or TN 1/2 [5], we collected data with the current classification. The post-operative outcome was classified according to the BNI pain intensity score, as follows: BNI I (no pain, no medications), BNI II-III (occasional pain but in control with medications, if required), BNI IV-V (no control with medication, no pain relief). The early post-operative deficit was defined as the onset of new deficit or the worsening of a preoperative clinical condition after the surgical treatment. Permanent post-operative deficit was defined as the persistence of post-operative acquired deficit at the last follow-up.
Outcomes
The primary objective of this systematic review and meta-analysis was to determine the long-term outcome of TN after MVD, in terms of the rate of BNI I. The secondary objective was to examine the impact of potential confounders on the analyzed outcome for the following variables: mean age, rate of female patients, preoperative mean symptoms duration, rate of typical trigeminal neuralgia, rate of patients with no evidence of neurovascular conflict during surgery, rate of patients with only venous conflict, rate of patients with superior cerebellar artery (SCA) conflict, and the study publication year.
Quality scoring
A modified version of the Newcastle–Ottawa Scale [36] was used for the quality assessment of the.
included studies. The quality assessment was performed by two authors independently (D.D.C and N.B.), and the senior author solved discrepancies.
Statistical analysis
Inter-observer agreement was tested with Cohen’s kappa coefficient (k). The Wald method was used to calculate confidence intervals (CI) for event rates. In order to assess the heterogeneity of the data, the Higgins index (I2) [17] was used, in which I2 > 50% suggests substantial heterogeneity. DerSimonian and Laird random-effects models were subsequently applied [11]. Predictors of outcome were analyzed with random meta-regression. The graphical representation of the meta-analysis was performed by forest plot. Heterogeneity (I2) > 50% was studied and analyzed with a sensitivity analysis (leave-one-out meta-analysis) Funnel plot followed by Egger’s linear regression test was used for bias assessment [32]. Differences were considered significant at p < 0.05. Statistical analyses were performed with SPSS version 23 (SPSS Inc. SPSS® Chicago, IL, USA), with ProMeta version3 (Internovi, Cesena, Italy) and OpenMeta[Analyst] (http://www.cebm. brown.edu/openmeta/).
Results
Literature review
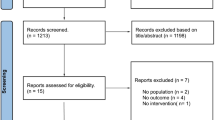
Studies included in our systematic review are summarized in Table 2. Intra-observer agreement was 0.82. The search flow diagram is shown in Fig. 1. Nine studies) and 2102 patients with trigeminal neuralgia that underwent MVD were analyzed in this review.
Quality of studies
Three articles presented a prospective design, and six studies were retrospective single-center analyses. All studies were rated “high quality” (detailed data online resource 1). Intra-observer agreement was 0.88.
Demographic data and clinical characteristics
Overall, the mean age of the included patients ranged between 53 and 65 years old, and the proportion of female patients was 61.6% (95% CI 59.5–63.6%). The left side was less commonly involved (39.6%, 95% CI 37.4–41.8%) and the mean symptoms duration before surgery ranged between 5 and 7.5 years. Symptoms were more commonly reported together in the second (V2) and the third division (V3) of the trigeminal nerve (33.5%, 95% CI 31.2–35.8%), and the pain was classified as typical in 92.7% of cases (95% CI 91.5–93.8%). Only three studies reported the rate of previous treatment before MVD (25.4%, 95% CI 23.2–27.6%), although no adequate data were provided to differentiate the result for intervention type (Table 3).
Surgical technique and intraoperative findings
The retrosigmoid approach was the surgical route in all cases. The superior cerebellar artery (SCA) was found to be the offending vessel in 74.7% of patients (95% CI 72.4–76.4%). Furthermore, only venous contact and no vascular conflict were observed in 11.1% (95% CI 9.6–12.5%) and 6.1% (95% CI 4.1–8%), respectively. Only two studies described in detail the degree of compression [14, 29]. The most common post-operative complications were CSF leak and facial numbness (2.4%, 95% CI 1.7–3.1% and 2.1%, 95% CI 1.5–2.8%, respectively). Moreover, hearing impairment was reported in approximately 1% of patients (detailed data are shown in Table 4).
Clinical outcome
Overall, immediately after surgery, 82.9% (95% CI 74.9–88.9%, p < 0.01, I2 = 93.4%) of patients reported a complete resolution of preoperative symptoms (BNI I), whereas 2.6% (95% 1–4.1%, p < 0.01, I2 = 68.6%) were classified as “surgical failures” (BNI IV-V). Clinical follow-up ranged between 3 and 10 years, and the long-term outcome analysis demonstrated that 64.7% of patients were classified as BNI I (95% CI 56.9–72.4%, p < 0.01, I2 = 88.1%) (forest plot is provided in Fig. 2). Furthermore, BNI III-IV and BNI IV-V were reported in 17% (95% CI 11.2–22.8%, p < 0.01. I2 = 89.6%) and 19.2% (95% CI 8.9–29.5%, p < 0.01, I2 = 97.3%) (forest plots in online resource 2). A summary of the long-term post-operative outcome is provided in Table 5.
Forest plot detailing the pooled rate and 95% confidence intervals for the rate of long-term BNI I after MVD for TN. The overall rate of BNI I (no pain, no medication) was 64.7% (95% CI 56.9–72.4%, p < 0,01, I2 = 88.1%). b The leave-one-out meta-analysis showed that no individual study independently influenced the analyzed outcome
Heterogeneity, confounder analysis, and publication bias
Higgins’ index was above 50% for all analyses, showing substantial heterogeneity. Accordingly, a sensitivity analysis was performed demonstrating that no individual study significantly influenced the analyzed outcomes (Fig. 2, online resource 2). Furthermore, the funnel plots followed by Egger’s linear regression excluded publication bias for all analyses (Fig. 3, Table 5, online resource 2). Nonetheless, the rate of BNI IV-V at the last follow-up demonstrated asymmetry on the funnel plot. Accordingly, we decided to perform a meta-regression for follow-up length that showed a significant negative association between the mean age and the final rate of BNI IV-V. Nevertheless, multiple meta-regression was not significant for any of the confounders (online resource 3). The rate of long-term BNI I was investigated with meta-regression to assess the impact of covariates for the analyzed outcome. Accordingly, simple meta-regression demonstrated that the outcome significantly varied in relation to the vascular structure involved (arterial, venous or no conflict) (Table 6). The multiple meta-regression confirmed the result, showing that the presence of an isolated venous conflict independently decreased the rate of BNI I patients at last follow-up (p < 0.01) (Table 6) (scatter plot in Fig. 4).
Scatter plot of the association between the rates of isolated venous compression and the long-term rate of BNI I among MVD for trigeminal neuralgia population (p < 0.01). Meta-regression was performed using random effect meta regression model, and the covariate was proven to influence the outcome in both uni- and multiple meta-regression
Discussion
Our systematic review and meta-analysis of approximately 2700 MVDs for TN demonstrated several interesting findings. Overall, the immediate post-operative rate of BNI I was 82.9% (p < 0.01), whereas surgical failure (BNI IV-V) was reported in approximately 2.6% of patients. Our most important result is that the rate of BNI I at last follow up is 64.7% (p < 0.01), showing a significant negative correlation with the rate of patients with evidence of pure venous conflict during surgery (Fig. 4, p < 0.01). Furthermore, although the mean follow up ranged between 1.8 and 10 years among studies (Table 2), multiple meta-regression did not prove any significant impact on the analyzed outcome (p = 0.83).
Demographic data and clinical characteristics
The incidence of TN is 3 to 5 per 100 thousand persons per year [20, 23]. Since the seminal work of Jannetta [19], MVD for TN has been increasingly accepted as an effective treatment for pain control in patients with vascular conflict with the trigeminal nerve within the cerebellopontine angle [9, 33, 37]. In current literature, MVD for TN is associated with an overall rate of post-operative pain relief of 90% that decreases to approximately 80% 1 year after surgery [38]. Moreover, the average TN recurrence rate is 4% per year, and a successful outcome at 15 years is reported in 73.3%, confirming the long-term good outcome of this population [7, 38]. Our study showed that, immediately after surgery, 82.9% of patients were classified as BNI I, and 2.6% as BNI IV-V. Since the first reports, it has been suggested that the female population is often associated with a lower rate of success among MVD for TN [2]. Nonetheless, although we reported a higher rate of women (61.6%) patients, we did not find a significant association between gender and long-term outcome (p = 0.22) [31]. Similarly, as previously reported by several studies, the duration of symptoms before the surgery seems not to predict the outcome (p = 0.40) [31]. In the last decade, it has been largely debated if there is any significant association between the clinical features of TN and the surgical outcome of MVD [22, 34]. Accordingly, the distinction between the so-called “typical” TN and its counterpart, the “atypical” TN, has been extensively investigated. Burchiel and colleagues underlined that the major difference between the two clinical presentations is the proportional presence of episodic (TN 1) versus constant pain (TN 2) [5]. Accordingly, they suggested that the two entities should be more duly considered as two different clinical representation of the same continuous spectrum of trigeminal nerve neuropathy. The terms typical an atypical should not be used to subgroup TN, as suggested by the latest classification and guidelines [8, 40]. Nonetheless, in surgical practice, the presence of continuous pain was frequently associated with a long-term unfavorable outcome [26]. Interestingly, our results did not show any significant association between the rate of classical TN without continuous pain and the long-term outcome (Table 6, online resource 3). It is worth noting that the definition of classical TN is not homogeneous among studies. Furthermore, it has been reported by Sindou and colleagues that, when patients with “atypical facial pain” are excluded from the analysis, the long-term outcome is similar between the previously defined TN 1 and 2 [31]. Accordingly, due to selection bias and the reduced predictive value, TN classification in “typical” or “atypical” should not be considered as a reliable prognostic factor among MVD for TN population.
TN characteristics and vascular conflict
The presence of a NVC that involved the SCA was significantly associated with a better outcome in our univariate analysis, and the absence of any vascular conflict, or the presence of only a venous contact, showed a negative correlation with the outcome (Table 6, Fig. 4). These findings sustain the theory advanced by Jannetta et al. that the TN should be considered as an “entrapment syndrome” in which the mechanical separation between the vessel and the nerve can lead to symptom control [2] (Fig. 5A). On the other hand, since the first observation made by Dandy, it was observed that TN could occur without NVC [10]. Indeed, our study demonstrated that approximately 5% of patients had no NVC. It has been proposed that, due to demographic and histological differences, patients with TN and no evidence of NVC should be considered as a separate entity [21]. Classic demyelination of the trigeminal nerve close to the compression cannot be observed in these patients, and an aberrant activity within the trigeminal ganglion or an abnormal efferent reflex activity within the brainstem has been proposed as the pathological mechanism behind the symptoms [12, 13].
Accordingly, it is conceivable that this subgroup of patients has a separate clinical history and a higher chance of symptoms recurrency that need repeated ablative procedures [21].
TN due to venous conflict is widely accepted at the current state of knowledge [3, 15, 25] (Fig. 5B). Our study demonstrated that the presence of a pure venous conflict independently affects the outcome with a negative correlation in multiple meta-regression (p < 0.01). Sindou and colleagues analyzed a series of 55 patients with TN and venous conflict demonstrating that at 10 years, 70.6% of patients were reported as BNI I [15], showing no correlation between the degree of compression and the clinical presentation. Furthermore, they did not find a significant difference in long-term outcomes between arterial and venous conflicts [31]. On the other hand, Barker and colleagues identified the presence of a venous conflict as one of the predictors of pain recurrency [2]. Even if beyond the aim of our study, nowadays there is no consensus on the surgical management of venous conflicts. Sacrificing or preserving the venous structures is constantly debated in the current literature [15, 18]. Furthermore, it is worth noting that a pure venous conflict is a rare entity (11% in our review), and, as underlined by Dumot et al., it is not always possible to differentiate other anatomical features that can affect the outcome, such as nerve root atrophy and arachnoiditis [15]. Nonetheless, our study showed that the long-term outcome is independently affected by the presence of an isolated venous conflict (p < 0.01).
Strength and limitations
Our study has limitations. All series but three have a retrospective design. The main limitation of our work is that, due to the quality of the data itself, we could not compare outcomes with the Kaplan–Meier estimator or hazard ratios, and we decided to analyze the outcome at the “last follow-up”. Furthermore, many series did not report data on previous and additional treatments. Similarly, we could not perform a distinct pooled analysis according to the type of conflict (arterial, venous, no vascular contact), and the degree of arterial conflict. Furthermore, the definition of typical and atypical trigeminal neuralgia is not standardized among the included studies, increasing the risk of selection bias for the analyzed outcomes. Furthermore, the meta-regression to assess the impact of confounders on the long-term outcome was performed on less than ten studies. Nonetheless, the analysis showed significant results in both simple and multiple meta-regression, increasing the weight of the association. Accordingly, although the risk of bias is non-negligible due to the quality of the data, the sensitivity analysis and the funnel plot followed by Egger’s linear regression excluded publication bias, strengthening our findings. In addition, to be consistent with our inclusion criteria, we decided to exclude some important papers dealing with TN from the analysis, decreasing the selection bias.
Conclusions
This systematic review and meta-analysis of more than 2000 patients demonstrated that at the last follow-up 64.7% (p < 0.01) is classified as BNI I, confirming the long-term efficacy of MVD for TN, in particular when an arterial conflict can be demonstrated (p < 0.01). Neurosurgeons should be aware that the effectiveness of surgery decreases when a vascular conflict cannot be found during posterior fossa exploration (p < 0.01), strengthening the idea that this subgroup of patients should be considered as a separate population. Furthermore, the presence of an isolated venous conflict independently affected the long-term outcome (p < 0.01), suggesting a higher risk of TN recurrency for this subgroup of patients.
Data Availability
Not applicable.
Code availability
Not applicable.
References
Amaya Pascasio L, De La Casa-Fages B, Esteban de Antonio E, Grandas F, García-Leal R, Ruiz Juretschke F (2021) Microvascular decompression for trigeminal neuralgia: a retrospective analysis of long-term outcomes and prognostic factors. Neurologia. https://doi.org/10.1016/j.nrl.2021.03.009
Barker FG 2nd, Jannetta PJ, Bissonette DJ, Larkins MV, Jho HD, Barker FG II, Jannetta PJ, Bissonette DJ, Larkins MV, Jho HD (1996) The long-term outcome of microvascular decompression for trigeminal neuralgia. N Engl J Med 334:1077–1083. https://doi.org/10.1056/NEJM199604253341701
Barker FG II, Jannetta PJ, Babu RP, Pomonis S, Bissonette DJ, Jho HD (1996) Long-term outcome after operation for trigeminal neuralgia in patients with posterior fossa tumors. J Neurosurg 84:818–825. https://doi.org/10.3171/jns.1996.84.5.0818
Brown JA, Coursaget C, Preul MC, Sangvai D (1999) Mercury water and cauterizing stones: Nicolas Andre and tic douloureux. J Neurosurg 90:977–981. https://doi.org/10.3171/jns.1999.90.5.0977
Burchiel KJ (2003) A new classification for facial pain. Neurosurgery 53:1164–1166; discussion 1166–1167. https://doi.org/10.1227/01.NEU.0000088806.11659.D8
Burchiel KJ (2016) Trigeminal neuralgia: new evidence for origins and surgical treatment. Neurosurgery 63:52–55. https://doi.org/10.1227/NEU.0000000000001276
Burchiel KJ, Clarke H, Haglund M, Loeser JD (1988) Long-term efficacy of microvascular decompression in trigeminal neuralgia. J Neurosurg 69:35–38. https://doi.org/10.3171/jns.1988.69.1.0035
Cruccu G, Finnerup NB, Jensen TS, Scholz J, Sindou M, Svensson P, Treede R-D, Zakrzewska JM, Nurmikko T (2016) Trigeminal neuralgia: new classification and diagnostic grading for practice and research. Neurology 87:220–228. https://doi.org/10.1212/WNL.0000000000002840
Cruccu G, Gronseth G, Alksne J, Argoff C, Brainin M, Burchiel K, Nurmikko T, Zakrzewska JM (2008) AAN-EFNS guidelines on trigeminal neuralgia management. Eur J Neurol 15:1013–1028. https://doi.org/10.1111/j.1468-1331.2008.02185.x
Dandy WE (1934) Concerning the cause of trigeminal neuralgia. Am J Surg 24:447–455. https://doi.org/10.1016/S0002-9610(34)90403-7
DerSimonian R, Laird N (2015) Meta-analysis in clinical trials revisited. Contemp Clin Trials. https://doi.org/10.1016/j.cct.2015.09.002
Devor M, Amir R, Rappaport ZH (2002) Pathophysiology of trigeminal neuralgia: the ignition hypothesis. Clin J Pain 18:4–13
Devor M, Govrin-Lippmann R, Rappaport ZH (2002) Mechanism of trigeminal neuralgia: an ultrastructural analysis of trigeminal root specimens obtained during microvascular decompression surgery. J Neurosurg 96:532–543
Duan Y, Sweet J, Munyon C, Miller J (2015) Degree of distal trigeminal nerve atrophy predicts outcome after microvascular decompression for Type 1a trigeminal neuralgia. J Neurosurg 123:1512–1518. https://doi.org/10.3171/2014.12.JNS142086
Dumot C, Brinzeu A, Berthiller J, Sindou M (2017) Trigeminal neuralgia due to venous neurovascular conflicts: outcome microvascular decompression in a series of 55 consecutive patients. Acta Neurochir (Wien) 159:237–249. https://doi.org/10.1007/s00701-016-2994-y
Gardner WJ, Miklos MV (1959) Response of trigeminal neuralgia to decompression of senso1. Hong JM1. Hong JM, Chung C-S, Bang OY, Yong SW, Joo IS, Huh K (2009). J Am Med Assoc 170:1773–1776. https://doi.org/10.1001/jama.1959.03010150017004
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ Br Med J 327:557–560. https://doi.org/10.1136/bmj.327.7414.557
Inoue T, Shitara S, Goto Y, Prasetya M, Fukushima T (2021) Petrosal vein involvement in neurovascular conflict in trigeminal neuralgia: surgical technique and clinical outcomes. Oper Neurosurg (Hagerstown, Md) 20:E264–E271. https://doi.org/10.1093/ons/opaa422
Jannetta PJ (1976) Microsurgical approach to the trigeminal nerve for tic douloureux. In: Pain-Its Neurosurgical Management. Karger Publishers, pp 180–200
Kitt CA, Gruber K, Davis M, Woolf CJ, Levine JD (2000) Trigeminal neuralgia: opportunities for research and treatment. Pain 85:3–7
Ko AL, Lee A, Raslan AM, Ozpinar A, McCartney S, Burchiel KJ (2015) Trigeminal neuralgia without neurovascular compression presents earlier than trigeminal neuralgia with neurovascular compression. J Neurosurg 123:1519–1527
Li ST, Pan Q, Liu N, Shen F, Liu Z, Guan Y, Sindou M, Burchiel KJ (2004) Trigeminal neuralgia: what are the important factors for good operative outcomes with microvascular decompression. Surg Neurol 62:400–405. https://doi.org/10.1016/j.surneu.2004.02.028
Love S, Coakham HB (2001) Trigeminal neuralgia: pathology and pathogenesis. Brain 124:2347–2360
Masuoka J, Matsushima T, Inoue K, Nakahara Y, Takase Y, Kawashima M (2015) Outcome of microvascular decompression for trigeminal neuralgia treated with the stitched sling retraction technique. Neurosurg Rev 38:361–5; discussion 365. https://doi.org/10.1007/s10143-015-0607-5
Matsushima T, Huynh-Le P, Miyazono M (2004) Trigeminal neuralgia caused by venous compression. Neurosurgery 55:334–339. https://doi.org/10.1227/01.neu.0000129552.87291.87
Miller JP, Magill ST, Acar F, Burchiel KJ (2009) Predictors of long-term success after microvascular decompression for trigeminal neuralgia: clinical article. J Neurosurg 110:620–626. https://doi.org/10.3171/2008.9.17660
Mizobuchi Y, Nagahiro S, Kondo A, Arita K, Date I, Fujii Y, Fujimaki T, Hanaya R, Hasegawa M, Hatayama T, Inoue T, Kasuya H, Kobayashi M, Kohmura E, Matsushima T, Masuoka J, Morita A, Nishizawa S, Okayama Y, Shigeno T, Shimano H, Takeshima H, Yamakami I (2021) Microvascular decompression for trigeminal neuralgia: a prospective, multicenter study. Neurosurgery 89:557–564. https://doi.org/10.1093/neuros/nyab229
Moher D, Liberati A, Tetzlaff J, Altman DG, Grp P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement (reprinted from annals of internal medicine). Phys Ther 89:873–880. https://doi.org/10.1371/journal.pmed.1000097
Nunta-Aree S, Patiwech K, Sitthinamsuwan B (2018) Microvascular decompression for treatment of trigeminal neuralgia: factors that predict complete pain relief and study of efficacy and safety in older patients. World Neurosurg 110:e979–e988. https://doi.org/10.1016/j.wneu.2017.11.147
Rose FC (1999) Trigeminal neuralgia. Arch Neurol 56:1163–1164. https://doi.org/10.1001/archneur.56.9.1163
Sindou M, Leston J, Decullier E, Chapuis F (2007) Microvascular decompression for primary trigeminal neuralgia: long-term effectiveness and prognostic factors in a series of 362 consecutive patients with clear-cut neurovascular conflicts who underwent pure decompression. J Neurosurg 107:1144–1153. https://doi.org/10.3171/JNS-07/12/1144
Sterne JAC, Harbord RM (2004) Funnel plots in meta-analysis stata J 4:127–141
Szapiro J Jr, Sindou M (1986) Micro-surgical treatment of trigeminal neuralgia. Results and prognostic factors of the microvascular decompression. Neurochirurgie 32:103–121
Tyler-Kabara EC, Kassam AB, Horowitz MH, Urgo L, Hadjipanayis C, Levy EI, Chang Y-F (2002) Predictors of outcome in surgically managed patients with typical and atypical trigeminal neuralgia: comparison of results following microvascular decompression. J Neurosurg 96:527–531. https://doi.org/10.3171/jns.2002.96.3.0527
Wang DD, Raygor KP, Cage TA, Ward MM, Barbaro NM, Chang EF, Westcott S, Barbaro NM, Chang EF (2018) Prospective comparison of long-term pain relief rates after frst-time microvascular decompression and stereotactic radiosurgery for trigeminal neuralgia. J Neurosurg 128:68–77. https://doi.org/10.3171/2016.9.JNS16149
Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, Tugwell P (2013) The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. Ottawa Hosp Res Inst 1–4. https://doi.org/10.2307/632432
Zakrzewska JM, Akram H (2011) Neurosurgical interventions for the treatment of classical trigeminal neuralgia. Cochrane Database Syst Rev 9
Zakrzewska JM, Sindou M, Leston J, Howeidy T, Decullier E, Chapuis F (2006) Micro-vascular decompression for primary trigeminal neuralgia (typical or atypical) Long-term effectiveness on pain; prospective study with survival analysis in a consecutive series of 362 patients - commentary. Acta Neurochir (Wien) 148:1245. https://doi.org/10.1007/s00701-006-0809-2
Zhang H, Lei D, You C, Mao B-Y, Wu B, Fang Y (2013) The long-term outcome predictors of pure microvascular decompression for primary trigeminal neuralgia. World Neurosurg 79:756–762. https://doi.org/10.1016/j.wneu.2012.01.040
The International Classification of Headache Disorders, 3rd edition Copyright. https://doi.org/10.1177/0333102417738202
Acknowledgements
We thank Prof. Beth De Felici for the English revision.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
DTDC and PP contributed to the study conception and design. DTDC and NB performed a literature search, collection, screening, and analysis. DTDC and NB interpreted the results and assessed the risk of bias. DTDC wrote the draft of the manuscript. All authors critically reviewed, commented, and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
For this type of study, formal consent is not required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Human and animal ethics
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Di Carlo, D., Benedetto, N. & Perrini, P. Clinical outcome after microvascular decompression for trigeminal neuralgia: a systematic review and meta-analysis. Neurosurg Rev 46, 8 (2023). https://doi.org/10.1007/s10143-022-01922-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10143-022-01922-0