Abstract
Cerebral hyperperfusion (HP) complicates the postoperative course of patients with moyamoya disease (MMD) after direct revascularization surgery. Crossed cerebellar diaschisis (CCD) has been considered to be rarely associated with HP after revascularization surgery. This study aimed to describe the clinical features and factors associated with CCD secondary to cerebral HP after revascularization surgery for MMD. We analyzed 150 consecutive hemispheres including 101 in adults and 49 in pediatric patients who underwent combined direct and indirect bypass for MMD. Using single-photon emission computed tomography (SPECT), serial cerebral blood flow (CBF) was measured immediately after the surgery and on postoperative days 2 and 7. Pre- and postoperative voxel-based analysis of SPECT findings was performed to compare the changes in regional CBF. Multivariate logistic regression analysis was performed to test the effect of multiple variables on CCD. Asymptomatic and symptomatic HP was observed in 41.3% (62/150) and 16.7% (25/150) of the operated hemispheres, respectively. CCD was observed in 18.4% (16/87) of these hemispheres with radiological HP. Multivariate analysis revealed that the occurrence of CCD was significantly associated with symptomatic HP (p = 0.0015). Voxel-based analysis showed that the CBF increase in the operated frontal cortex, and the CBF reduction in the contralateral cerebellar hemisphere on day 7 were significantly larger in symptomatic HP than in asymptomatic HP (median 11.3% vs 7.5%; − 6.0% vs − 1.7%, respectively). CCD secondary to postoperative HP is more common than anticipated in MMD. CCD could potentially be used as an indicator of severe postoperative HP in patients with MMD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Moyamoya disease (MMD) is a steno-occlusive cerebrovascular disorder characterized by progressive occlusion of the supraclinoid segment of the internal carotid artery that results in the formation of an abnormal vascular network [33]. Direct bypass procedures effectively improve cerebral hemodynamics immediately after surgery in patients with MMD [1]. However, cerebral hyperperfusion (HP) complicates the postoperative course after direct revascularization surgery [10, 22, 37, 39]. Among such cases, HP-related transient neurological deficits (TNDs) occur in approximately 30% of adult and 5% of pediatric MMD patients who underwent direct bypass procedures [10, 11, 37].
A phenomenon that was termed crossed cerebellar diaschisis (CCD) was first described in the 1980s [3]. CCD is defined as a matched reduction in metabolism and blood flow in the cerebellar hemisphere contralateral to the supratentorial lesions. It is frequently observed after ischemic or hemorrhagic stroke affecting the cortico-ponto-cerebellar pathway [8, 17, 31]. Such interruption is usually secondary to decreased cerebral perfusion; however, it may also be associated with supratentorial HP after revascularization procedures [15, 28]. Several studies have shown that CCD correlates with stroke severity and infarct hypoperfusion volume, although very few have reported that CCD is associated with HP after revascularization surgery in MMD [15, 16, 19, 25]. We hypothesized that CCD is indicative of the severity of postoperative HP. Through serial single-photon emission computed tomography (SPECT), this study aimed to investigate the overall prevalence of HP-related CCD after bypass surgery for MMD and to determine the factors associated with the occurrence of HP-related CCD.
Methods
Patients and surgical procedures
This study was approved by the Research Ethics Committee of Hokkaido University Hospital (#015-0086). One hundred fifty consecutive revascularization surgeries were performed in 67 adult and 32 pediatric patients (< 18 years) who underwent combined direct and indirect revascularization for MMD at our hospital between 2006 and 2016. Surgical revascularization was considered for patients with hemodynamic compromise in the presence or absence of ischemic symptoms including headache. It was also considered for patients with hemorrhagic presentation. The surgical procedure has been described previously [16, 25]. Direct bypass procedures including superficial temporal artery–middle cerebral artery (MCA) anastomosis were performed in all hemispheres. In addition, indirect bypass procedures were performed, such as an encephalo-duro-arterio-myo-synangiosis procedure.
Perioperative management and radiological examinations
Presurgical regional cerebral blood flow (CBF) was quantitatively measured using 123I N-isopropyl-p-iodoamphetamine SPECT. Postoperative CBF measurements were performed immediately after surgery and at postsurgical days 2 and 7. Postoperative HP was defined as a focal and intense increase in CBF followed by its normalization in subsequent SPECT studies [37, 39]. A CBF value in the operated MCA territory that was greater than 150% of that in the ipsilateral cerebellum was considered as a reference when radiological HP was evaluated [36,37,38]. HP-related CCD was qualitatively evaluated when the CBF was reduced in the cerebellar hemisphere contralateral to the operated cerebral hemisphere associated with HP compared with the ipsilateral cerebellar hemisphere or preoperative images. The reduction of CBF in the cerebellar hemisphere was confirmed in multiple slices of cerebellum on the SPECT study. The resolution of CCD was confirmed by the subsequent SPECT studies. The evaluation of SPECT was performed by two authors (HU and KK), and a consensus was reached. Postsurgical systolic blood pressure was maintained at 100 to 140 mmHg with strict blood pressure monitoring and control using intravenous antihypertensive drug, if necessary. When TND was observed, MRI and MRA were performed to check the patency of direct bypass and the existence of any fresh ischemic lesions. Then, when SPECT showed HP in the area that corresponded to TND without new ischemic lesions, it was considered as symptomatic HP.
Voxel-based perfusion analysis
Voxel-based analyses were performed to visualize changes in regional CBF after surgery with the use of SPECT on preoperative day and postoperative day 7. The analysis included all 105 hemispheres from which raw quantitative SPECT data could be extracted. These included 63 of 87 hemispheres with radiological HP (symptomatic, 17; asymptomatic, 46), which involved 12 of 16 hemispheres with CCD (Table 1). The details of image acquisition and processing of image data are described elsewhere [19, 21]. Briefly, the SPECT images were spatially normalized to the standard SPECT template in Statistical Parametric Mapping version 8 (SPM8; Wellcome Department of Cognitive Neurology, University College London, London, UK; http://www.fil.ion.ucl.ac.uk/spm/). Alterations in regional CBF were compared voxel by voxel with each patient’s corresponding preoperative images using the paired t test model in SPM8. To explore the differences, statistical significance was set at p < 0.001 without correction for family-wise error. Clusters showing statistical significance and containing more than 50 voxels were considered as areas with significant changes. Images obtained in patients with left revascularization were flipped to the right side, and the hemisphere with revascularization was treated as the right hemisphere. Thus, the perfusion mapping image was produced by raw quantitative SPECT data. A region of interest (ROI)-based analysis was also conducted to compare the changes in regional CBF of the operated frontal hemisphere and contralateral cerebellar hemisphere. Spherical ROIs (15 mm diameter) were defined using MarsBaR available as an SPM plug-in, which encircles the center of the largest cluster showing significant changes in the voxel-based analysis (http://marsbar.sourceforge.net).
Data analyses
The relationships between the occurrence of CCD and several clinical factors, including age, sex, operated side, disease onset type, and HP-related TNDs were evaluated. We compared the data between the groups using the χ2 test, the unpaired t test, or the Mann-Whitney test, as appropriate. A multivariate logistic regression analysis was conducted to determine the effects of the multiple variables on HP-related CCD. The multivariate analysis was performed using the parameters that achieved significance levels of p < 0.05 in the univariate analysis. The level of significance was set at p < 0.05 for the rest of the analyses described above.
Results
Demographic data
In the present study, 101 hemispheres in 67 adults and 49 hemispheres in 32 children were revascularized. The mean ages of the children and adults were 8.7 and 42.3 years, respectively, and the patients’ ages ranged from 1 to 71 years. The patients’ clinical diagnoses were associated with ischemic symptoms, including headaches, transient ischemic attacks and infarctions in 125 hemispheres, intracranial bleeding in 19 hemispheres, and other symptoms in six hemispheres (Table 1).
Incidence of HP-related CCD
Postoperative HP, including symptomatic and asymptomatic occurrences, was detected in 13 (26.5%) of 49 pediatric hemispheres and 74 (73.3%) of 101 adult hemispheres (Table 1). Of these, HP-related CCD was detected in 16 of 150 (10.7%) total hemispheres and 16/87 (18.4%) of hemispheres presented with HP (Table 2). HP-related CCD was observed most often on postoperative day 7 among the three timepoints after surgery (Fig. 1). In two of the cases (3 and 8; Table 2), the detection of CCD preceded the onset of HP-related TNDs. Voxel-based perfusion mapping visualized a significant increase of CBF in the operated frontal cortex and a reduction in the contralateral cerebellar hemisphere on postoperative day 7 (Fig. 2).
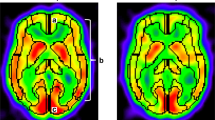
Perioperative serial single-photon emission computed tomography of a 52-year-old woman who underwent left-sided surgery (case 1; Table 2). She had developed the neurological deficits motor aphasia and facial palsy on postoperative day (POD) 2. The images demonstrate a focal increase in cerebral blood flow in the left frontal lobe and a reduction in the right cerebellar hemisphere (arrow head) on POD 7. Two months after surgery, the reduction in cerebral blood flow observed in the right cerebellar hemisphere had resolved. PreOp, preoperative
Voxel-based perfusion mapping demonstrating a significant increase in cerebral blood flow (CBF) in the operated frontal cortex and a reduction in CBF in the contralateral cerebellar hemisphere on postoperative day 7 compared with the preoperative state. The revascularized hemisphere is set to the right side. Voxels with significant increases in CBF are shown in red, whereas voxels with decreases in CBF are shown in blue. Color bars represent T values. Lower-threshold T values correspond to a statistical significance of p < 0.001 with uncorrected family-wise error
Correlations between HP-related CCD and TNDs
HP-related TNDs were observed in one of 49 (2.0%) pediatric and 24 of 101 (23.8%) adult hemispheres (Table 1). CCD was more frequently observed in symptomatic HP than in asymptomatic HP (9/25 (36.0%) vs 7/62 (11.3%), p = 0.017; Fig. 3). The effects of various factors on HP-related CCD are shown in Table 3. Logistic regression analysis demonstrated a correlation between the occurrence of HP-related CCD and TNDs (OR, 6.6; 95% CI, 2.1–21.2; p = 0.0015). Univariate analysis showed that HP-related CCD was observed more significantly in left-sided surgery. Gender and disease onset type were not correlated with HP-related CCD. Voxel-based ROI analysis showed that CBF changes in the operated frontal lobe and the contralateral cerebellum were significantly larger in symptomatic HP than in asymptomatic HP (median 11.3% vs 7.5%, p = 0.035; − 6.0% vs − 1.7%, p = 0.020, respectively; Fig. 4).
Bar graph from region of interest–based analysis illustrating the cerebral blood flow (CBF) changes in the operated frontal cortex and contralateral cerebellar hemisphere on postoperative day (POD) 7 in cases with symptomatic and asymptomatic hyperperfusion (HP). The columns and bars indicate the median and interquartile range, respectively. *p < 0.05
Discussion
While postoperative HP is now a well-recognized phenomenon, especially in adult patients with MMD [10, 13, 29, 30, 36, 37, 39, 40], HP-related CCD has not been recognized as frequently and it has been investigated in only a few studies. There are case reports on HP-related CCD after bypass surgery for MMD [15] and after carotid endarterectomy [28]. This study reports that the incidence of CCD among MMD patients presenting with HP was 18.4%, which is more frequent than anticipated and within the published range of CCD after supratentorial strokes (15.6–46.2%) [6, 7, 23, 24, 26, 27, 32].
This study showed that CCD was more commonly observed in symptomatic HP than in asymptomatic HP. The degree of the postoperative CBF increase is considered as one of the major factors associated with the occurrence of HP-related TNDs. We have previously reported the critical threshold by which a CBF increase is associated with HP-related TNDs [21]. Thus, symptomatic HP is considered a more severe form of HP compared with asymptomatic HP in terms of the degree of CBF increase. In fact, in the present study, the voxel-based ROI analysis demonstrated that the CBF increase within the operated frontal cortex was larger in symptomatic HP than in asymptomatic HP.
Previous SPECT studies have demonstrated that the degree of supratentorial hypoperfusion is correlated with the occurrence of CCD in stroke patients. Sommer et al. reported that the occurrence of CCD was significantly influenced by the degree of the supratentorial CBF reduction in patients with acute MCA infarction [32]. Nocun et al. reported that the degree of cerebral hypoperfusion, in the chronic stage of stroke, was a determinant of CCD and correlated with the degree of cerebellar hypoperfusion [27]. These are respectively representing the number of impaired neurons in the cortico-ponto-cerebellar pathway and so the severity of the functional damage. Similarly, the degree of HP was associated with the occurrence as well as the degree of CCD among cases with symptomatic and asymptomatic HP in the present study. Thus, conversely, the occurrence of contralesional cerebellar CBF reduction can be considered as an indicator of symptomatic HP in MMD. CCD would be an additional radiological marker of severe neuronal damage due to HP besides the focal intense CBF increase in the anastomosed hemisphere. Of the 16 hemispheres with HP-related CCD, seven (43.8%) were still asymptomatic. These cases may potentially be at risk of becoming symptomatic, which implies that meticulous postoperative management with regard to blood pressure control and radiological follow-up may be necessary [9, 12, 35, 37, 39]. In contrast, the detection of CCD did not always precede the onset of HP-related TNDs. This may limit the value of CCD as a predictor of symptomatic HP.
In our study, a significantly higher proportion of CCD occurred following the presentation of left-sided HP. The higher incidence of CCD after left-sided MCA infarctions has also been reported in a large cohort [32]. Previous voxel-based diffusion tensor imaging analysis demonstrated a number of fractional anisotropy asymmetries including leftward asymmetry of corticospinal tract and the cerebellum in healthy individuals [34]. This data suggests that the amount of interconnection between the frontal cortex and the cerebellar hemisphere is more intense on the left. Furthermore, the left side is said to be associated with speech, complex language processing, and verbal working memory [2, 4, 5, 20, 32]. Symptomatic HP is also reported to occur more frequently after left-sided surgery in MMD [37]. The higher incidences of CCD and symptomatic HP may be attributed to the vulnerability of left side of the cerebral hemisphere.
This study had some limitations. First, the small number of CCD cases was retrospectively investigated in a single institute. Second, the incidence of HP-related CCD was evaluated by CBF on SPECT. CCD can be detected by other hemodynamic/metabolic parameters, such as by cerebral blood volume, mean transit time, and fluorodeoxyglucose metabolism [18, 31, 32]. Therefore, the incidence may vary depending on the parameters analyzed or imaging modalities. Furthermore, it is hard in some cases to conclude that CCD is exclusively secondary to HP because HP can be associated with simultaneous ischemia at surrounding areas [14]. Finally the long-term effect of HP-related CCD on the clinical outcome such as cognitive function is unclear.
Conclusions
This perioperative serial SPECT study demonstrated that transient CCD secondary to postoperative HP is not a rare phenomenon in MMD. This finding has clinical applications, as CCD could potentially be a radiological indicator associated with symptomatic HP or severe neuronal damage due to HP in MMD. Although postoperative HP and CCD are considered a temporal phenomenon in MMD, the long-term effects of intense HP as well as CCD should be investigated in the future.
References
Arikan F, Vilalta J, Torne R, Noguer M, Lorenzo-Bosquet C, Sahuquillo J (2015) Rapid resolution of brain ischemic hypoxia after cerebral revascularization in moyamoya disease. Neurosurgery 76:302–312; discussion 312. https://doi.org/10.1227/NEU.0000000000000609
Barbas H, Garcia-Cabezas MA, Zikopoulos B (2013) Frontal-thalamic circuits associated with language. Brain Lang 126:49–61. https://doi.org/10.1016/j.bandl.2012.10.001
Baron JC, Bousser MG, Comar D, Castaigne P (1981) "Crossed cerebellar diaschisis" in human supratentorial brain infarction. Trans Am Neurol Assoc 105:459–461
Binkofski F, Buccino G (2006) The role of ventral premotor cortex in action execution and action understanding. J Physiol Paris 99:396–405. https://doi.org/10.1016/j.jphysparis.2006.03.005
Chen SH, Desmond JE (2005) Temporal dynamics of cerebro-cerebellar network recruitment during a cognitive task. Neuropsychologia 43:1227–1237. https://doi.org/10.1016/j.neuropsychologia.2004.12.015
De Reuck J, Decoo D, Lemahieu I, Strijckmans K, Goethals P, Van Maele G (1997) Crossed cerebellar diaschisis after middle cerebral artery infarction. Clin Neurol Neurosurg 99:11–16. https://doi.org/10.1016/s0303-8467(96)00561-6
Forster A, Kerl HU, Goerlitz J, Wenz H, Groden C (2014) Crossed cerebellar diaschisis in acute isolated thalamic infarction detected by dynamic susceptibility contrast perfusion MRI. PLoS One 9:e88044. https://doi.org/10.1371/journal.pone.0088044
Fu J, Chen WJ, Wu GY, Cheng JL, Wang MH, Zhuge Q, Li JC, Zhang Q, Zhang Y, Xia NZ, Yang YJ (2015) Whole-brain 320-detector row dynamic volume CT perfusion detected crossed cerebellar diaschisis after spontaneous intracerebral hemorrhage. Neuroradiology 57:179–187. https://doi.org/10.1007/s00234-014-1456-6
Fujimura M, Inoue T, Shimizu H, Saito A, Mugikura S, Tominaga T (2012) Efficacy of prophylactic blood pressure lowering according to a standardized postoperative management protocol to prevent symptomatic cerebral hyperperfusion after direct revascularization surgery for moyamoya disease. Cerebrovasc Dis 33:436–445. https://doi.org/10.1159/000336765
Fujimura M, Kaneta T, Mugikura S, Shimizu H, Tominaga T (2007) Temporary neurologic deterioration due to cerebral hyperperfusion after superficial temporal artery-middle cerebral artery anastomosis in patients with adult-onset moyamoya disease. Surg Neurol 67:273–282. https://doi.org/10.1016/j.surneu.2006.07.017
Fujimura M, Mugikura S, Kaneta T, Shimizu H, Tominaga T (2009) Incidence and risk factors for symptomatic cerebral hyperperfusion after superficial temporal artery-middle cerebral artery anastomosis in patients with moyamoya disease. Surg Neurol 71:442–447. https://doi.org/10.1016/j.surneu.2008.02.031
Fujimura M, Niizuma K, Inoue T, Sato K, Endo H, Shimizu H, Tominaga T (2014) Minocycline prevents focal neurological deterioration due to cerebral hyperperfusion after extracranial-intracranial bypass for moyamoya disease. Neurosurgery 74:163–170; discussion 170. https://doi.org/10.1227/NEU.0000000000000238
Furuya K, Kawahara N, Morita A, Momose T, Aoki S, Kirino T (2004) Focal hyperperfusion after superficial temporal artery-middle cerebral artery anastomosis in a patient with moyamoya disease. Case report. J Neurosurg 100:128–132. https://doi.org/10.3171/jns.2004.100.1.0128
Hayashi T, Shirane R, Fujimura M, Tominaga T (2010) Postoperative neurological deterioration in pediatric moyamoya disease: watershed shift and hyperperfusion. J Neurosurg Pediatr 6:73–81. https://doi.org/10.3171/2010.4.PEDS09478
Hokari M, Kuroda S, Simoda Y, Uchino H, Hirata K, Shiga T, Nakayama N, Houkin K, Tamaki N (2012) Transient crossed cerebellar diaschisis due to cerebral hyperperfusion following surgical revascularization for moyamoya disease: case report. Neurol Med Chir (Tokyo) 52:350–353. https://doi.org/10.2176/nmc.52.350
Houkin K, Kamiyama H, Takahashi A, Kuroda S, Abe H (1997) Combined revascularization surgery for childhood moyamoya disease: STA-MCA and encephalo-duro-arterio-myo-synangiosis. Childs Nerv Syst 13:24–29. https://doi.org/10.1007/s003810050034
Infeld B, Davis SM, Lichtenstein M, Mitchell PJ, Hopper JL (1995) Crossed cerebellar diaschisis and brain recovery after stroke. Stroke 26:90–95. https://doi.org/10.1161/01.str.26.1.90
Kajimoto K, Oku N, Kimura Y, Kato H, Tanaka MR, Kanai Y, Kitagawa K, Maruno M, Yoshimine T, Hori M, Hatazawa J (2007) Crossed cerebellar diaschisis: a positron emission tomography study with L-[methyl-11C] methionine and 2-deoxy-2-[18F]fluoro-D-glucose. Ann Nucl Med 21:109–113. https://doi.org/10.1007/bf03033988
Kazumata K, Tha KK, Uchino H, Shiga T, Shichinohe H, Ito M, Nakayama N, Abumiya T (2017) Topographic changes in cerebral blood flow and reduced white matter integrity in the first 2 weeks following revascularization surgery in adult moyamoya disease. J Neurosurg 127:260–269. https://doi.org/10.3171/2016.6.JNS16653
Kazumata K, Tokairin K, Sugiyama T, Ito M, Uchino H, Osanai T, Kawabori M, Nakayama N, Houkin K (2019) Association of cognitive function with cerebral blood flow in children with moyamoya disease. J Neurosurg Pediatr:1–7. https://doi.org/10.3171/2019.7.PEDS19312
Kazumata K, Uchino H, Tokairin K, Ito M, Shiga T, Osanai T, Kawabori M (2018) Cerebral hyperperfusion syndrome after revascularization surgery in moyamoya disease: region-symptom mapping and estimating a critical threshold. World Neurosurg 114:e388–e395. https://doi.org/10.1016/j.wneu.2018.02.190
Kim JE, Oh CW, Kwon OK, Park SQ, Kim SE, Kim YK (2008) Transient hyperperfusion after superficial temporal artery/middle cerebral artery bypass surgery as a possible cause of postoperative transient neurological deterioration. Cerebrovasc Dis 25:580–586. https://doi.org/10.1159/000132205
Kim SE, Choi CW, Yoon BW, Chung JK, Roh JH, Lee MC, Koh CS (1997) Crossed-cerebellar diaschisis in cerebral infarction: technetium-99m-HMPAO SPECT and MRI. J Nucl Med 38:14–19
Komaba Y, Mishina M, Utsumi K, Katayama Y, Kobayashi S, Mori O (2004) Crossed cerebellar diaschisis in patients with cortical infarction: logistic regression analysis to control for confounding effects. Stroke 35:472–476. https://doi.org/10.1161/01.STR.0000109771.56160.F5
Kuroda S, Houkin K, Ishikawa T, Nakayama N, Iwasaki Y (2010) Novel bypass surgery for moyamoya disease using pericranial flap: its impacts on cerebral hemodynamics and long-term outcome. Neurosurgery 66:1093–1101; discussion 1101. https://doi.org/10.1227/01.NEU.0000369606.00861.91
Lin DD, Kleinman JT, Wityk RJ, Gottesman RF, Hillis AE, Lee AW, Barker PB (2009) Crossed cerebellar diaschisis in acute stroke detected by dynamic susceptibility contrast MR perfusion imaging. AJNR Am J Neuroradiol 30:710–715. https://doi.org/10.3174/ajnr.A1435
Nocun A, Wojczal J, Szczepanska-Szerej H, Wilczynski M, Chrapko B (2013) Quantitative evaluation of crossed cerebellar diaschisis, using voxel-based analysis of Tc-99m ECD brain SPECT. Nucl Med Rev Cent East Eur 16:31–34. https://doi.org/10.5603/NMR.2013.0006
Ogasawara K, Kobayashi M, Komoribayashi N, Fukuda T, Inoue T, Terasaki K, Ogawa A (2005) Transient crossed cerebellar diaschisis secondary to cerebral hyperperfusion following carotid endarterectomy. Ann Nucl Med 19:321–324. https://doi.org/10.1007/bf02984626
Ogasawara K, Komoribayashi N, Kobayashi M, Fukuda T, Inoue T, Yamadate K, Ogawa A (2005) Neural damage caused by cerebral hyperperfusion after arterial bypass surgery in a patient with moyamoya disease: case report. Neurosurgery 56:E1380; discussion E1380. https://doi.org/10.1227/01.neu.0000159719.47528.2e
Ohue S, Kumon Y, Kohno K, Watanabe H, Iwata S, Ohnishi T (2008) Postoperative temporary neurological deficits in adults with moyamoya disease. Surg Neurol 69:281–286; discussion 286-287. https://doi.org/10.1016/j.surneu.2007.01.047
Sobesky J, Thiel A, Ghaemi M, Hilker RH, Rudolf J, Jacobs AH, Herholz K, Heiss WD (2005) Crossed cerebellar diaschisis in acute human stroke: a PET study of serial changes and response to supratentorial reperfusion. J Cereb Blood Flow Metab 25:1685–1691. https://doi.org/10.1038/sj.jcbfm.9600162
Sommer WH, Bollwein C, Thierfelder KM, Baumann A, Janssen H, Ertl-Wagner B, Reiser MF, Plate A, Straube A, von Baumgarten L (2016) Crossed cerebellar diaschisis in patients with acute middle cerebral artery infarction: occurrence and perfusion characteristics. J Cereb Blood Flow Metab 36:743–754. https://doi.org/10.1177/0271678X15617953
Suzuki J, Takaku A (1969) Cerebrovascular "moyamoya" disease. Disease showing abnormal net-like vessels in base of brain. Arch Neurol 20:288–299
Takao H, Abe O, Yamasue H, Aoki S, Sasaki H, Kasai K, Yoshioka N, Ohtomo K (2011) Gray and white matter asymmetries in healthy individuals aged 21-29 years: a voxel-based morphometry and diffusion tensor imaging study. Hum Brain Mapp 32:1762–1773. https://doi.org/10.1002/hbm.21145
Uchino H, Ito M, Fujima N, Kazumata K, Yamazaki K, Nakayama N, Kuroda S, Houkin K (2015) A novel application of four-dimensional magnetic resonance angiography using an arterial spin labeling technique for noninvasive diagnosis of moyamoya disease. Clin Neurol Neurosurg 137:105–111. https://doi.org/10.1016/j.clineuro.2015.07.003
Uchino H, Kazumata K, Ito M, Nakayama N, Kuroda S, Houkin K (2014) Intraoperative assessment of cortical perfusion by indocyanine green videoangiography in surgical revascularization for moyamoya disease. Acta Neurochir 156:1753–1760. https://doi.org/10.1007/s00701-014-2161-2
Uchino H, Kuroda S, Hirata K, Shiga T, Houkin K, Tamaki N (2012) Predictors and clinical features of postoperative hyperperfusion after surgical revascularization for moyamoya disease: a serial single photon emission CT/positron emission tomography study. Stroke 43:2610–2616. https://doi.org/10.1161/STROKEAHA.112.654723
Uchino H, Nakamura T, Houkin K, Murata J, Saito H, Kuroda S (2013) Semiquantitative analysis of indocyanine green videoangiography for cortical perfusion assessment in superficial temporal artery to middle cerebral artery anastomosis. Acta Neurochir 155:599–605. https://doi.org/10.1007/s00701-012-1575-y
Uchino H, Nakayama N, Kazumata K, Kuroda S, Houkin K (2016) Edaravone reduces Hyperperfusion-related neurological deficits in adult moyamoya disease: historical control study. Stroke 47:1930–1932. https://doi.org/10.1161/STROKEAHA.116.013304
Uno M, Nakajima N, Nishi K, Shinno K, Nagahiro S (1998) Hyperperfusion syndrome after extracranial-intracranial bypass in a patient with moyamoya disease—case report. Neurol Med Chir (Tokyo) 38:420–424. https://doi.org/10.2176/nmc.38.420
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Hokkaido University Hospital ethical review board approval (IRB#015-0086) was received before the study. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Uchino, H., Kazumata, K., Ito, M. et al. Crossed cerebellar diaschisis as an indicator of severe cerebral hyperperfusion after direct bypass for moyamoya disease. Neurosurg Rev 44, 599–605 (2021). https://doi.org/10.1007/s10143-020-01265-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-020-01265-8