Abstract
Sphenoid wing meningiomas are generally considered as skull base meningiomas (SBMs). However, given their surgical similarities with non-skull base meningiomas (NSBMs), we hypothesized that lateral sphenoid wing meningiomas (LSWMs) without bone invasion (BI) should be considered as NSBMs. N = 65 LSWMs without BI operated between 1990 to 2010 at a single-center were compared to N = 352 NSBMs, represented by convexity meningiomas (CMs), and to N = 23 SBMs, represented by spheno-orbital meningiomas (SOMs), with respect to baseline demographics, clinical presentations, Simpson grades, complications, adjuvant therapies, as well as overall survival (OS) and progression-free survival (PFS). Only WHO grade I meningiomas were included. No significant differences in baseline demographics, clinical presentation, or pre-operative KPS were found between the three groups. Simpson grade 1–3 was achieved in 90.1% of LSWMs, 97.1% in CMs (p = 0.05), and 82.6% in SOMs (p = 0.23). There were no significant differences in postoperative infection, hematoma, neurological worsening, 30-day mortality, or OS between the three groups. Lower re-treatment rates were observed in LSWMs and CMs compared to SOMs (p = 0.06). With respect to PFS, there was no significant difference between LSWMs and CMs (89.1% and 88.5% at 5 years, respectively), whereas PFS was significantly higher in LSWMs than in SOMs (79% at 5 years) (p = 0.05). LSWMs without BI should be considered as an intermediate entity between NSBMs and SBMs. LSWMs are similar to SOMs with respect to extent of resection, but more similar to CMs with respect to re-treatment rates and PFS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intracranial meningiomas are mostly benign lesions, representing 30 to 40% of all operated primary intracranial tumors [17, 23, 32]. In patients with growing and/or symptomatic intracranial meningiomas, surgical excision of the tumor and its dural attachment is often recommended [7, 13, 20].
Sphenoid wing meningiomas (SWMs) are the most frequent tumors of the sphenoid ridge and constitute 15–20% of all intracranial meningiomas, representing the third most common group of intracranial meningiomas [1, 12, 24]. A classification scheme that divided the sphenoid wing into thirds along the medio-lateral axis was developed by Cushing and Eisenhardt [5], and SWMs were categorized into medial, middle, and lateral. Lastly, lateral sphenoid wing meningiomas (LSWMs) with hyperostosis, bone infiltration (BI), and extension to the orbit are called spheno-orbital meningiomas (SOMs) [22, 25, 29]. These sub-classes may have different clinical presentations, risks of surgery, as well as prognosis [4, 31].
In the anatomical sub-division of meningiomas provided by Al-Mefty et al. [8], SWMs are considered as middle skull base meningiomas (SBM) (Table 1). However, LSWMs without BI are technically more straightforward and less demanding than SOMs [9, 27] and could be considered as non-skull base meningioma (NSBM). To test this hypothesis, we compared LSWMs without BI to NSBMs, represented by convexity meningiomas (CMs), and SBMs, represented by spheno-orbital meningiomas (SOMs), with respect to their baseline characteristics, clinical presentation, Simpson grades, complications, adjuvant therapies, as well as overall survival (OS) and progression-free survival (PFS).
Materials and methods
Patient cohort
From a database including a total of N = 1469 intracranial meningiomas operated between 1990 and 2010 at Oslo University Hospital (OUH), all LSWMs, CMs, and SOMs were identified. Data were acquired retrospectively from 1990 to 2002 and prospectively from 2003 to 2010. Meningioma locations were defined according to Al-Mefty classification [8]. LSWMs with BI, hyperostosis, and extension to the orbit were classified as SOMs [3, 22, 25, 27, 29]. WHO grade II and III lesions were excluded from the analysis as they would distort the overall analysis of PFS and OS because of uneven distributions between the three groups.
A sub-cohort of n = 65 LSWMs without BI (4.4% of the total cohort), n = 23 (1.6%) SOMs, and n = 352 (24%) CMs was identified (Table 2). Preoperative imaging studies (or reports for older cases) were reviewed to confirm tumor location, contrast enhancement, and bone invasion.
Baseline demographic data, pre-operative Karnofsky Performance Status (KPS), presenting symptoms (seizures, raised intracranial pressure (ICP), onset of new neurological deficit or no symptoms), Simpson grades, post-operative complications (hematoma, infection, neurological worsening), 30-day mortality, and adjuvant therapies (second surgery, conventional radiotherapy, stereotactic radiosurgery) were retrieved.
The extent of resection (EOR) was assessed using the Simpson grading system [30], based on the surgical report in conjunction with postoperative imaging. Gross total resection (GTR) was defined as Simpson grade I, II, or III resections, according to the criteria of the European Association of Neuro-Oncology [10].
The post-operative image surveillance was left open to the treating physician’s discretion and has not followed a rigorous scheme. Tumor progression was defined as any retreatment for tumor recurrence by means of surgery, conventional fractionated radiotherapy, or stereotactic radio-surgery. PFS was calculated from time of surgery to time of retreatment or censoring. OS was defined as the period between index surgery and all-cause mortality, or otherwise, date of the last follow-up.
Ethics
The study was regulated by the Personal Data Act/Personal Health Data Filing System Act and approved by the Data Protection Official at OUH (2017/5204). Informed consent was not required according to the Personal Data Act/Personal Health Data Filing System Act.
Statement of human and animal rights
All the procedures followed were in accordance with ethical standards on human experimentation.
Statistical analysis
Statistical analysis was performed using R v3.5.1 (https://www.r-project.org). The significant p value was defined at 0.05. For the three groups, mean age and KPS upon admission were calculated (mean and standard deviation). The comparisons of the populations’ characteristics were performed using the Kruskal-Wallis test, a non-parametric equivalent of the ANOVA, and the post-hoc analysis was performed using the Mann-Whitney U test. A pairwise comparison was also performed between the three groups to compare OS and PFS using the log-rank test.
Results
Patients demographic data
Baseline demographics and symptoms upon admission are summarized in Table 2. With respect to mean age at surgery, female-to-male ratio, and pre-operative KPS, patients with LSWMs without BI were similar to patients with CMs and SOMs: mean age upon admission was 56.7 ± 12.9 years, and the female-to-male ratio was 2.8.
Presenting symptoms upon admission
Regarding the presenting symptoms of patients with LSWMs without BI, n = 26 patients (40%) presented with neurological deficit upon admission; n = 22 patients (33.8%) had seizures prior to admission, n = 20 (30.8%) patients presented with symptoms of raised intracranial pressure, and 9 (13.8%) were asymptomatic. With respect to the neurological deficit upon admission, the presence of raised intracranial pressure, and the number of asymptomatic patients, no statistically significant difference was found between the three groups. The incidence of seizure was significantly higher in patients with CMs (Kruskal-Wallis; p < 0.01); it was similar in LSWMs and CMs (33.8% and 43.2%, respectively), whereas a non-significant trend towards an increased rate of seizures in the LSWM group was found (p = 0.08) when compared to the SOMs group (8.7%). Finally, CMs had significantly more seizures than SOMs (p < 0.01; Table 2).
Tumor characteristics and EOR
Rates of GTR are summarized in Table 2. GTR was achieved in n = 59 (90.1%) of patients with LSWMs without BI and was significantly less frequent than in CMs (97.1%; p = 0.05). Rates of GTR were similar between LSWMs without BI and SOMs (82.6%).
Post-operative complications
Post-operative complications and their distributions are listed in Table 2. Results concerning post-operative complications were similar between LSWMs without BI, CMs, and SOMs, pertaining to post-operative infection, hematoma, neurological worsening, and 30-day mortality (Table 2).
Adjuvant therapy
A summary of adjuvant therapies is provided in Table 2. In the LSWMs without BI group, 4.6% of the patients had second operation and 1.5% had post-operative radiosurgery. No patient had conventional radiotherapy. In the CM group, 6% of patients had a second surgery, 1.7% had conventional radiotherapy, and 1.1% had radiosurgery. In the SOM group, 17% of the patients had second surgery, 4.3% had conventional radiotherapy, and 4.3% underwent radiosurgery. Trends towards increased rate of re-operation and post-operative radiosurgery were observed in patients with SOMs, but the differences did not reach statistical significance.
OS and PFS
The OS and PFS curves are shown in Figs. 1 and 2. LSWMs without BI showed similar OS to CMs and SOMs. In pairwise Kaplan-Meier analysis, the 5-year PFS was significantly higher in LSWMs without BI when compared to SOMs (89.1% versus 79%, p = 0.05). CMs (88.5%) had also superior PFS compared to SOMs (p = 0.03). With respect to the 10-year PFS, similar results were found: there was no significant difference between LSWMs without BI and CMs (89.1% and 85.7%, respectively), whereas PFS was significantly higher in LSWMs without BI than in SOMs (64.6%, p = 0.05; Fig. 2).
Progression-free survival curves of patients with lateral sphenoid wing meningiomas, convexity meningiomas, and spheno-orbital meningiomas. Lateral sphenoid wing meningiomas had similar progression-free survival rates than convexity meningiomas and significantly higher progression-free survival than spheno-orbital meningiomas. There is a significant difference in progression-free survival rates between spheno-orbital meningiomas and the two other groups. LSWMs lateral sphenoid wing meningiomas, CMs convexity meningiomas, SOMs spheno-orbital meningiomas
Discussion
LSWMs: At the interface between SBMs and NSBMs
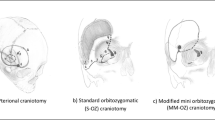
According to Al-Mefty’s original definition, LSWMs without BI are considered as SBMs (Table 1) [8]. In contrast to SOMs, which generally require extensive surgical exposure and often have sub-total resections [9, 27], surgery of LSWMs without BI is more similar to a CM and often straightforward due to the absence of critical neurovascular structure around the lateral third of the sphenoid wing. We therefore hypothesized that LSWMs BI invasion should be considered as NSBMs rather than SBMs. In order to test this hypothesis, a monocentric cohort of n = 65 patients with WHO grade I LSWMs without BI was compared to NSBMs (represented by n = 352 WHO grade 1 CMs) and to SBMs (represented by n = 23 WHO grade 1 SOMs).
With respect to the extent of resection, GTR was achieved in 90.1% of patients with LSWMs, which was significantly less frequent than in CMs (97.1%; p = 0.05). Overall, the rates of GTR were similar between LSWMs without BI and SOMs (82.6%; see Table 2). Despite slightly inferior rates of GTR, patients with LSWMs had similar PFS rates to patients with CMs, but higher PFS than the SOMs (Fig. 1). LSWMs without BI and CMs had similar re-treatment rates (3.4% and 3.1%, respectively), whereas patients with SOMs showed a trend towards higher re-treatment rates. Although the difference did not reach statistical significance (p = 0.06), it should be kept in mind that this trend was observed on a limited number of patients and should be studied in a larger cohort. Altogether, LSWMs were similar to SOMs in terms of surgery but were similar to CMs in terms of short- and long-term outcomes. Consequently, we can consider LSWMs without BI as an intermediate entity between SBMs and NSBMs, as their current classification into SBMs does not reflect the reality in terms of outcomes, which is of paramount importance in term of patient care and prognosis, when confronted to LSWMs without BI. Our findings are in line with Guduk et al. [12], who recently reported their surgical outcomes in a series of 141 SWMs and concluded that SWM constitute a heterogeneous group of lesions, with characteristics of both SBMs and CMs.
LSWMs without BI and SOMs: differences in terms of PFS and re-treatment rates
In addition to their particular clinical presentation, surgical management and outcomes when compared to NSBMS [4, 6, 7, 11, 14,15,16, 18, 19], SBMs have higher re-treatment rates and shorter PFS [19]. The need for an accurate classification of meningiomas into SBMs and NSBMs appears therefore of paramount importance. Our data show that in terms of re-treatment rates and PFS, LSWMs without BI have the characteristics of NSBMs, despite showing rates of GTR similar to SOMs. This is interesting because while the mainstay of treatment of LSWMs without BI is tumor resection through a pterional craniotomy [12], the resection of SOMs often requires a larger fronto-temporal craniotomy, followed by an extra-dural removal of the tumor-infiltrated bone [9, 27]. The degree of surgical difficulty may vary according to the consistency and the adhesiveness to neurovascular structure, which may prevent aggressive and radical surgery, whereas in the case of LSWMs, especially if the arachnoid plane is preserved and the lesion is soft, none of the principles of skull base surgery apply.
In their surgical series of 63 patients with SOMs, Ringel et al. report that 76% of patients had tumor remnants, of which 39% were progressive. Freeman et al. reported similar results in 25 cases [9]. In their series on SWMs, Guduk et al. report tumor recurrence in 27% (38/141) of cases, of which 40% (15/38) were SOMs and 18% (7/38) were LSWMs. Altogether, these series show that SOMs are more difficult to control despite accurate surgery, unlike LSWMs without BI. In our series, rates of GTR were surprisingly high in SOMs, and similar to those achieved in LSWMs (Table 2).
From a surgical point of view, the radical resection of LSWMs without BI is safe and feasible, as our results show a high GTR and very low complications and re-treatment rates. Guduk et al. [12] achieved GTR in 100% of LSWMs without BI; in their report, GTR was considered as Simpson 1 and 2, contrary to the current guidelines of EANO [10]. In our cohort, Simpson 1 and 2 resections were achieved in 89.3% of LSWMs (Table 2). This may be due to a higher proportion of orbital invasion of the LSWMs without BI in our series.
With respect to SOMs, Guduk et al. [12] achieved GTR in only 12.9% of SOMs, whereas in our cohort, Simpson 1 and 2 resections were achieved in 56.5% and 26.1% of cases, respectively. Our results contrast those previously reported in the literature [9, 12, 27], testifying of a certain heterogeneity among SOMs. Overall, it appears that SOMs have less favorable outcomes when it comes to PFS, despite rather more favorable surgical results than previously reported in the literature. Despite not being significant, the higher re-treatment rate in SOMs has to be considered, since previously reported data corroborate this finding [19].
The role of an accurate classification
Some authors question the pertinence of the classical three-tier classification of SWMs, advocating for more straightforward dichotomization into middle SWMs and LSWMs, since this would reflect the clinical reality more accurately [21, 31]. On the basis of our data and in the perspective with previously reported series [9, 12, 27], we believe that a simplification of the current classification is misleading. The location of the lesion is only one of multiple variables in meningioma surgery: the presence of BI, the clinical presentation, as well as genetic alterations and inherent biology [26, 28] are to be considered. Altogether, this may favor a more detailed classification of SWMs [21]. Moreover, our findings allow to preemptively decide whether or not tumors in the sphenoid wing could be allocated to a skull base team or not.
Limitations
The retrospective data collection before 2003 is a limitation. Also, due to the long period of data collecting starting in 1990, tumor size and molecular biomarkers such as Mib-1 or Ki67 were not available for all patients, despite being known factors influencing OS/PFS [2]. Furthermore, surgical and monitoring techniques evolved dramatically during the study period. Regarding mortality, no disease-specific mortality was registered.
Conclusion
LSWMs without BI should be considered as a mixed entity between NSBMs and SBMs. Their surgical outcome, pertaining to the EOR, is similar to SBMs, whereas their re-treatment rates and PFS are more in line with NSBMs.
References
Aziz KM, Froelich SC, Cohen PL, Sanan A, Keller JT, van Loveren HR (2002) The one-piece orbitozygomatic approach: the MacCarty burr hole and the inferior orbital fissure as keys to technique and application. Acta Neurochir 144:15–24. https://doi.org/10.1007/s007010200001
Barrett OC, Hackney JR, McDonald AM, Willey CD, Bredel M, Fiveash JB (2019) Pathologic predictors of local recurrence in atypical meningiomas following gross total resection. Int J Radiat Oncol Biol Phys 103:453–459. https://doi.org/10.1016/j.ijrobp.2018.09.019
Basso A, Carrizo A, Kreutel A, Martino A, Cerisola J, Torrieri A, Amezua L (1978) Surgical treatment of the spheno-orbital tumors (author’s transl). Neurochirurgie 24:71–82
Champagne PO, Lemoine E, Bojanowski MW (2018) Surgical management of giant sphenoid wing meningiomas encasing major cerebral arteries. Neurosurg Focus 44:E12. https://doi.org/10.3171/2018.1.FOCUS17718
Cushing WH, Eisenhardt L (1962) Meningiomas. Their classification, regional behaviour, life history, and surgical end results, vol 1. New York
da Silva CE, de Freitas PE (2015) Large and giant skull base meningiomas: the role of radical surgical removal. Surg Neurol Int 6:113. https://doi.org/10.4103/2152-7806.159489
da Silva CE, Peixoto de Freitas PE (2016) Recurrence of skull base meningiomas: the role of aggressive removal in surgical treatment. J Neurol Surg B Skull Base 77:219–225. https://doi.org/10.1055/s-0035-1566251
DeMonte F, McDermott MW, Al-Mefty O (2011) Al-Mefty’s meningiomas, 2nd New edn. Thieme Medical Publishers Inc, New York ISBN 9781604060539
Freeman JL, Davern MS, Oushy S, Sillau S, Ormond DR, Youssef AS, Lillehei KO (2017) Spheno-orbital meningiomas: a 16-year surgical experience. World Neurosurg 99:369–380. https://doi.org/10.1016/j.wneu.2016.12.063
Goldbrunner R, Minniti G, Preusser M, Jenkinson MD, Sallabanda K, Houdart E, von Deimling A, Stavrinou P, Lefranc F, Lund-Johansen M, Moyal EC, Brandsma D, Henriksson R, Soffietti R, Weller M (2016) EANO guidelines for the diagnosis and treatment of meningiomas. Lancet Oncol 17:e383–e391. https://doi.org/10.1016/S1470-2045(16)30321-7
Gonen L, Nov E, Shimony N, Shofty B, Margalit N (2018) Sphenoorbital meningioma: surgical series and design of an intraoperative management algorithm. Neurosurg Rev 41:291–301. https://doi.org/10.1007/s10143-017-0855-7
Guduk M, Ozduman K, Pamir MN (2019) Sphenoid wing meningiomas: surgical outcomes in a series of 141 cases and proposal of a scoring system predicting extent of resection. World Neurosurg. https://doi.org/10.1016/j.wneu.2018.12.175
Hasseleid BF, Meling TR, Ronning P, Scheie D, Helseth E (2012) Surgery for convexity meningioma: Simpson grade I resection as the goal: clinical article. J Neurosurg 117:999–1006. https://doi.org/10.3171/2012.9.JNS12294
Jorgensen M, Heegaard S (2018) A review of nasal, paranasal, and skull base tumors invading the orbit. Surv Ophthalmol 63:389–405. https://doi.org/10.1016/j.survophthal.2017.07.001
Konglund A, Rogne SG, Helseth E, Meling TR (2013) Meningioma surgery in the very old-validating prognostic scoring systems. Acta Neurochir 155:2263–2271; discussion 2271. https://doi.org/10.1007/s00701-013-1872-0
Konglund A, Rogne SG, Lund-Johansen M, Scheie D, Helseth E, Meling TR (2013) Outcome following surgery for intracranial meningiomas in the aging. Acta Neurol Scand 127:161–169. https://doi.org/10.1111/j.1600-0404.2012.01692.x
Lassen B, Helseth E, Ronning P, Scheie D, Johannesen TB, Maehlen J, Langmoen IA, Meling TR (2011) Surgical mortality at 30 days and complications leading to recraniotomy in 2630 consecutive craniotomies for intracranial tumors. Neurosurgery 68:1259–1268; discussion 1268-1259. https://doi.org/10.1227/NEU.0b013e31820c0441
Meling TR, Da Broi M, Scheie D, Helseth E (2018) Skull base versus non-skull base meningioma surgery in the elderly. Neurosurg Rev. https://doi.org/10.1007/s10143-018-1005-6
Meling TR, Da Broi M, Scheie D, Helseth E (2019) Meningiomas: skull base versus non-skull base. Neurosurg Rev 42:163–173. https://doi.org/10.1007/s10143-018-0976-7
Meling TR, Da Broi M, Scheie D, Helseth E, Smoll NR (2019) Meningioma surgery-are we making Progress? World Neurosurg. https://doi.org/10.1016/j.wneu.2019.01.042
Morcos JJ (2013) Sphenoid wing meningiomas. J Neurosurg 119:82–84. https://doi.org/10.3171/2012.6.JNS112303
Mourits MP, van der Sprenkel JW (2001) Orbital meningioma, the Utrecht experience. Orbit 20:25–33
Ostrom QT, Gittleman H, Xu J, Kromer C, Wolinsky Y, Kruchko C, Barnholtz-Sloan JS (2016) CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2009-2013. Neuro-Oncology 18:v1–v75. https://doi.org/10.1093/neuonc/now207
Pieper DR, Al-Mefty O, Hanada Y, Buechner D (1999) Hyperostosis associated with meningioma of the cranial base: secondary changes or tumor invasion. Neurosurgery 44:742–746 discussion 746-747
Pompili A, Derome PJ, Visot A, Guiot G (1982) Hyperostosing meningiomas of the sphenoid ridge--clinical features, surgical therapy, and long-term observations: review of 49 cases. Surg Neurol 17:411–416
Preusser M, Brastianos PK, Mawrin C (2018) Advances in meningioma genetics: novel therapeutic opportunities. Nat Rev Neurol 14:106–115. https://doi.org/10.1038/nrneurol.2017.168
Ringel F, Cedzich C, Schramm J (2007) Microsurgical technique and results of a series of 63 spheno-orbital meningiomas. Neurosurgery 60:214–221; discussion 221-212. https://doi.org/10.1227/01.NEU.0000255415.47937.1A
Sahm F, Schrimpf D, Stichel D, Jones DTW, Hielscher T, Schefzyk S, Okonechnikov K, Koelsche C, Reuss DE, Capper D, Sturm D, Wirsching HG, Berghoff AS, Baumgarten P, Kratz A, Huang K, Wefers AK, Hovestadt V, Sill M, Ellis HP, Kurian KM, Okuducu AF, Jungk C, Drueschler K, Schick M, Bewerunge-Hudler M, Mawrin C, Seiz-Rosenhagen M, Ketter R, Simon M, Westphal M, Lamszus K, Becker A, Koch A, Schittenhelm J, Rushing EJ, Collins VP, Brehmer S, Chavez L, Platten M, Hanggi D, Unterberg A, Paulus W, Wick W, Pfister SM, Mittelbronn M, Preusser M, Herold-Mende C, Weller M, von Deimling A (2017) DNA methylation-based classification and grading system for meningioma: a multicentre, retrospective analysis. Lancet Oncol 18:682–694. https://doi.org/10.1016/S1470-2045(17)30155-9
Shrivastava RK, Sen C, Costantino PD, Della Rocca R (2005) Sphenoorbital meningiomas: surgical limitations and lessons learned in their long-term management. J Neurosurg 103:491–497. https://doi.org/10.3171/jns.2005.103.3.0491
Simpson D (1957) The recurrence of intracranial meningiomas after surgical treatment. J Neurol Neurosurg Psychiatry 20:22–39. https://doi.org/10.1136/jnnp.20.1.22
Sughrue ME, Rutkowski MJ, Chen CJ, Shangari G, Kane AJ, Parsa AT, Berger MS, McDermott MW (2013) Modern surgical outcomes following surgery for sphenoid wing meningiomas. J Neurosurg 119:86–93. https://doi.org/10.3171/2012.12.JNS11539
Wiemels J, Wrensch M, Claus EB (2010) Epidemiology and etiology of meningioma. J Neuro-Oncol 99:307–314. https://doi.org/10.1007/s11060-010-0386-3
Acknowledgments
The authors would like to thank Eirik Helseth MD, PhD; David Scheie MD, PhD; Bernt Filip Hasseleid MD; Andreas Mathisen MD; Andreas Hessen Schei MD; and Kristina M. Ødegaard MD for their valuable contributions in collecting data for this manuscript and the neurosurgeons of Rikshospitalet and Ullevaal for their dedicated patient care.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was regulated by the Personal Data Act/Personal Health Data Filing System Act and approved by the Data Protection Official at OUH (2017/5204). Informed consent was not required according to the Personal Data Act/Personal Health Data Filing System Act. All the procedures followed were in accordance with ethical standards on human experimentation.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Corniola, M.V., Lemée, JM., Schaller, K. et al. Lateral sphenoid wing meningiomas without bone invasion—still skull base surgery?. Neurosurg Rev 43, 1547–1553 (2020). https://doi.org/10.1007/s10143-019-01181-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-019-01181-6